Abstract
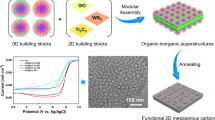
Intermetallic nanoparticles (iNPs) exhibit ordered superlattice structures characterized by unique properties, for example, long-range ordering, robust metallic bonding and site-isolation effects. Multicomponent (>2) iNPs are particularly interesting for the development of advanced metallic catalysts for electrochemical applications. Integration of iNPs within mesoporous carbon nanostructures enhances mass and electron transfer during electrolysis and provides a protective mesoporous confinement that prevents iNP sintering and loss during operation. Here we describe a generalized two-step strategy to integrate iNPs with up to eight metal components into mesoporous carbon nanostructures that allows control over the ordering degree, phases and morphology. Ligand-assisted interfacial assembly of monomicelles on diverse metal substrates (using a laboratory-made amphiphilic copolymer as a structure-directing agent, with dopamine acting as both carbon precursor and metal-coordinating ligand) results in mesostructured metal–organic superstructures. All of the examples described have at least one noble metal (Pt or Pd) combined with transition metal elements (for example, Fe, Co, among others). Thermal processing of these metal–organic superstructures in an ammonia (NH3) atmosphere induces the formation of chemically ordered iNPs while simultaneously creating the mesoporous structure. The Protocol also includes procedures for two example electrochemical applications: the oxygen reduction reaction and nitrate reduction reaction for NH3 production. The entire synthetic procedure takes ~5 d, while physical characterization via electron microscopy, X-ray diffraction and nitrogen sorption isotherms require ~2 d. Investigating the catalytic mechanisms, utilizing in situ Fourier-transform infrared spectroscopy and online differential electrochemical mass analysis typically take 4–6 h for electrocatalytic reactions.
Key points
-
Multisite catalysts are important in driving complex electrochemical processes. This Protocol describes a general approach to incorporate intermetallic nanoparticles into mesoporous carbon nanostructures with different ordering degree, crystalline phases and morphology.
-
The approach starts with the preparation of mesostructured metal–organic superstructures via a ligand-assisted interfacial monomicelle assembly process. Then, annealing metal–organic superstructures in NH3 facilitates the formation of a mesoporous structure and the crystallization of metals into intermetallic nanoparticles.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout












Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. Source data are provided with this paper.
References
Sakamoto, N. et al. Dinuclear Cu(I) molecular electrocatalyst for CO2-to-C3 product conversion. Nat. Catal. 7, 574–584 (2024).
Wang, X. et al. Developing a class of dual atom materials for multifunctional catalytic reactions. Nat. Commun. 14, 7210 (2023).
Yoon, A. et al. Revealing catalyst restructuring and composition during nitrate electroreduction through correlated operando microscopy and spectroscopy. Nat. Mater. https://doi.org/10.1038/s41563-024-02084-8 (2025).
Hou, L. et al. Rationally designed Mo/Ru-based multi-site heterogeneous electrocatalyst for accelerated alkaline hydrogen evolution reaction. Adv. Mater. 36, 2410039 (2024).
Zhou, D. et al. Cooperation of different active sites to promote CO2 electroreduction to multi-carbon products at ampere-level. Angew. Chem. Int. Ed. 63, e202400439 (2024).
Li, J. et al. Molecular-scale CO spillover on a dual-site electrocatalyst enhances methanol production from CO2 reduction. Nat. Nanotechnol. https://doi.org/10.1038/s41565-025-01866-8 (2025).
Nakaya, Y. & Furukawa, S. Catalysis of alloys: classification, principles, and design for a variety of materials and reactions. Chem. Rev. 123, 5859–5947 (2023).
Lin, F., Li, M., Zeng, L., Luo, M. & Guo, S. Intermetallic nanocrystals for fuel-cells-based electrocatalysis. Chem. Rev. 123, 12507–12593 (2023).
Yoo, T. Y. et al. Direct synthesis of intermetallic platinum–alloy nanoparticles highly loaded on carbon supports for efficient electrocatalysis. J. Am. Chem. Soc. 142, 14190–14200 (2020).
Yao, Y. et al. High-entropy nanoparticles: synthesis-structure-property relationships and data-driven discovery. Science 376, eabn3103 (2022).
Li, M. et al. High-entropy alloy electrocatalysts go to (sub-)nanoscale. Sci. Adv. 10, eadn2877 (2024).
George, E. P., Raabe, D. & Ritchie, R. O. High-entropy alloys. Nat. Rev. Mater. 4, 515–534 (2019).
Fu, H. et al. High-entropy rare earth materials: synthesis, application and outlook. Chem. Soc. Rev. 53, 2211–2247 (2024).
Liang, J. et al. Atomic arrangement engineering of metallic nanocrystals for energy-conversion electrocatalysis. Joule 3, 956–991 (2019).
Liu, H. et al. Unveiling atomic-scaled local chemical order of high-entropy intermetallic catalyst for alkyl-substitution-dependent alkyne semihydrogenation. J. Am. Chem. Soc. 146, 20193–20204 (2024).
Feng, G. et al. Atomically ordered non-precious Co3Ta intermetallic nanoparticles as high-performance catalysts for hydrazine electrooxidation. Nat. Commun. 10, 4514 (2019).
Gong, T., Rudman, K. K., Guo, B. & Hall, A. S. Electrochemical synthesis of nanostructured ordered intermetallic materials under ambient conditions. Acc. Chem. Res. 56, 1373–1383 (2023).
Song, R. et al. Ultrafine nanoporous intermetallic catalysts by high-temperature liquid metal dealloying for electrochemical hydrogen production. Nat. Commun. 13, 5157 (2022).
Xiong, Y. et al. Revealing the atomic ordering of binary intermetallics using in situ heating techniques at multilength scales. Proc. Natl Acad. Sci. USA 116, 1974–1983 (2019).
Jia, Z. et al. A Novel multinary intermetallic as an active electrocatalyst for hydrogen evolution. Adv. Mater. 32, 2000385 (2020).
Gamler, J. T. L., Ashberry, H. M., Skrabalak, S. E. & Koczkur, K. M. Random alloyed versus intermetallic nanoparticles: a comparison of electrocatalytic performance. Adv. Mater. 30, 1801563 (2018).
Zeng, W.-J. et al. Phase diagrams guide synthesis of highly ordered intermetallic electrocatalysts: separating alloying and ordering stages. Nat. Commun. 13, 7654 (2022).
Wang, Y. et al. Ordering-dependent hydrogen evolution and oxygen reduction electrocatalysis of high-entropy intermetallic Pt4FeCoCuNi. Adv. Mater. 35, 2302067 (2023).
Feng, G. et al. Engineering structurally ordered high-entropy intermetallic nanoparticles with hHigh-activity facets for oxygen reduction in practical fuel cells. J. Am. Chem. Soc. 145, 11140–11150 (2023).
Yan, Y. et al. Intermetallic nanocrystals: syntheses and catalytic applications. Adv. Mater. 29, 1605997 (2017).
Chen, W. et al. High-entropy intermetallic PtRhBiSnSb nanoplates for highly efficient alcohol oxidation electrocatalysis. Adv. Mater. 34, 2206276 (2022).
Li, X. et al. Atomic structure evolution of Pt–Co binary catalysts: single metal sites versus intermetallic nanocrystals. Adv. Mater. 33, 2106371 (2021).
Bueno, S. L. A., Ashberry, H. M., Shafei, I. & Skrabalak, S. E. Building durable multimetallic electrocatalysts from intermetallic seeds. Acc. Chem. Res. 54, 1662–1672 (2021).
Wang, Y. et al. Rapid room-temperature synthesis of a metastable ordered intermetallic electrocatalyst. J. Am. Chem. Soc. 141, 2342–2347 (2019).
Gong, T. et al. Atomic ordering-induced ensemble variation in alloys governs electrocatalyst on/off states. J. Am. Chem. Soc. 147, 510–518 (2025).
Gao, Q. et al. Breaking adsorption-energy scaling limitations of electrocatalytic nitrate reduction on intermetallic CuPd nanocubes by machine-learned insights. Nat. Commun. 13, 2338 (2022).
Guo, W. et al. General synthesis and atomic arrangement identification of ordered Bi–Pd intermetallics with tunable electrocatalytic CO2 reduction selectivity. Nat. Commun. 15, 1573 (2024).
Chen, H.-S. et al. Preserving the exposed facets of Pt3Sn intermetallic nanocubes during an order to disorder transition allows the elucidation of the effect of the degree of alloy ordering on electrocatalysis. J. Am. Chem. Soc. 142, 3231–3239 (2020).
Guan, J., Zhang, J., Wang, X., Zhang, Z. & Wang, F. Synthesis of L10-iron triad (Fe, Co, Ni)/Pt intermetallic electrocatalysts via a phosphide-induced structural phase transition. Adv. Mater. 35, 2207995 (2023).
Shao, R.-Y. et al. Promoting ordering degree of intermetallic fuel cell catalysts by low-melting-point metal doping. Nat. Commun. 14, 5896 (2023).
Liang, J. et al. Metal bond strength regulation enables large-scale synthesis of intermetallic nanocrystals for practical fuel cells. Nat. Mater. 23, 1259–1267 (2024).
Yang, C.-L. et al. Sulfur-anchoring synthesis of platinum intermetallic nanoparticle catalysts for fuel cells. Science 374, 459–464 (2021).
Yao, Y. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
Ma, X. et al. Heterophase intermetallic compounds for electrocatalytic hydrogen production at industrial-scale current densities. J. Am. Chem. Soc. 146, 20594–20603 (2024).
Peng, L. et al. One-dimensionally oriented self-assembly of ordered mesoporous nanofibers featuring tailorable mesophases via kinetic control. Nat. Commun. 14, 8148 (2023).
Liu, Y. et al. Molecular-level modulation of N, S-co-doped mesoporous carbon nanospheres for selective aqueous catalytic oxidation of ethylbenzene. Angew. Chem. Int. Ed. 64, e202419438 (2025).
Gao, T.-N. et al. Solvent-induced self-assembly strategy to synthesize well-defined hierarchically porous polymers. Adv. Mater. 31, 1806254 (2019).
Zhang, W. et al. Two-electron redox chemistry via single-atom catalyst for reversible zinc–air batteries. Nat. Sustain. 7, 463–473 (2024).
Velty, A. & Corma, A. Advanced zeolite and ordered mesoporous silica-based catalysts for the conversion of CO2 to chemicals and fuels. Chem. Soc. Rev. 52, 1773-1946, A (2023).
Zhao, W. et al. Mesoporous carbon confined intermetallic nanoparticles as highly durable electrocatalysts for the oxygen reduction reaction. J. Mater. Chem. A 8, 15822–15828 (2020).
Wang, Y. et al. A universal solid-phase synthetic strategy for ultrafine intermetallic libraries confined in ordered mesoporous carbon. Adv. Mater. 37, 2416111 (2025).
Wang, K. et al. Kinetic diffusion-controlled synthesis of twinned intermetallic nanocrystals for CO-resistant catalysis. Sci. Adv. 8, eabo4599 (2022).
Qi, Z. et al. Sub-4 nm PtZn intermetallic nanoparticles for enhanced mass and specific activities in catalytic electrooxidation reaction. J. Am. Chem. Soc. 139, 4762–4768 (2017).
Xie, M. et al. Periodic adjacent Pd-Fe pair sites for enhanced nitrate electroreduction to ammonia via accelerating proton relay. Adv. Energy Mater. 14, 2401717 (2024).
Zhu, G. et al. Constructing structurally ordered high-entropy alloy nanoparticles on nitrogen-rich mesoporous carbon nanosheets for high-performance oxygen reduction. Adv. Mater. 34, 2110128 (2022).
Zhu, G. et al. Accelerating tandem electroreduction of nitrate to ammonia via multi-site synergy in mesoporous carbon-supported high-entropy intermetallics. Adv. Mater. 37, 2413560, 2 (2025).
Tsai, A. P., Kameoka, S., Nozawa, K., Shimoda, M. & Ishii, Y. Intermetallic: a pseudoelement for catalysis. Acc. Chem. Res. 50, 2879–2885, 6 (2017).
Zhou, M., Li, C. & Fang, J. Noble-metal based random alloy and intermetallic nanocrystals: syntheses and applications. Chem. Rev. 121, 736–795 (2021).
Liu, X. et al. Inducing covalent atomic interaction in intermetallic Pt alloy nanocatalysts for high-performance fuel cells. Angew. Chem. Int. Ed. 62, e202302134 (2023).
Peng, Z. & Sun, K. Intermetallic interphases in lithium metal and lithium-ion batteries. InfoMat 3, 1083–1109 (2021).
Ye, Y. et al. Solid-solution or intermetallic compounds: phase dependence of the Li-alloying reactions for Li-metal batteries. J. Am. Chem. Soc. 145, 24775–24784 (2023).
Wang, Z. et al. High-entropy alloys to activate the sulfur cathode for lithium–sulfur batteries. Energy Envion. Sci. 6, e12358 (2023).
Moskovits, M., Srnová-Šloufová, I. & Vlčková, B. Bimetallic Ag–Au nanoparticles: extracting meaningful optical constants from the surface-plasmon extinction spectrum. J. Chem. Phys. 116, 10435–10446 (2002).
Liu, K. et al. Porous Au–Ag nanospheres with high-density and highly accessible hotspots for SERS analysis. Nano Lett. 16, 3675–3681 (2016).
Cortie, M. B. & McDonagh, A. M. Synthesis and optical properties of hybrid and alloy plasmonic nanoparticles. Chem. Rev. 111, 3713–3735 (2011).
Tong, L. et al. Bright three-photon luminescence from gold/silver alloyed nanostructures for bioimaging with negligible photothermal toxicity. Angew. Chem. Int. Ed. 49, 3485–3488 (2010).
Guo, J. et al. Modular assembly of superstructures from polyphenol-functionalized building blocks. Nat. Nanotechnol. 11, 1105–1111 (2016).
Guo, Y., Sun, Q., Wu, F.-G., Dai, Y. & Chen, X. Polyphenol-containing nanoparticles: Synthesis, properties, and therapeutic delivery. Adv. Mater. 33, 2007356 (2021).
Lim, H. et al. A universal approach for the synthesis of mesoporous gold, palladium and platinum films for applications in electrocatalysis. Nat. Protoc. 15, 2980–3008 (2020).
Peng, L., Peng, H., Li, W. & Zhao, D. Monomicellar assembly to synthesize structured and functional mesoporous carbonaceous nanomaterials. Nat. Protoc. 18, 1155–1178 (2023).
Wei, F. et al. Solution-based self-assembly synthesis of two-dimensional-ordered mesoporous conducting polymer nanosheets with versatile properties. Nat. Protoc. 18, 2459–2484 (2023).
Qiu, P. et al. Modular assembly of metal nanoparticles/mesoporous carbon two-dimensional nanosheets. NPG Asia Mater. 15, 35 (2023).
Cui, M. et al. Multi-principal elemental intermetallic nanoparticles synthesized via a disorder-to-order transition. Sci. Adv. 8, eabm4322 (2022).
Wang, Y. et al. Synthesis of high-etropy-alloy nanoparticles by a step-alloying strategy as a superior multifunctional electrocatalyst. Adv. Mater. 35, 2302499 (2023).
Cao, G. et al. Liquid metal for high-entropy alloy nanoparticles synthesis. Nature 619, 73–77 (2023).
Hafner, J. Ab-initio simulations of materials using VASP: density-functional theory and beyond. J. Comput. Chem 29, 2044–2078 (2008).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Zhao, Z. et al. Versatile synthesis of uniform mesoporous superparticles from stable monomicelle units. Nat. Protoc. 18, 1155–1178 (2024).
Luo, W. et al. A micelle fusion–aggregation assembly approach to mesoporous carbon materials with rich active sites for ultrasensitive ammonia sensing. J. Am. Chem. Soc. 138, 12586–12595 (2016).
Angot, S., Taton, D. & Gnanou, Y. Amphiphilic stars and dendrimer-like architectures based on poly(ethylene oxide) and polystyrene. Macromolecules 33, 5418–5426 (2000).
Ton-That, C., Shard, A. G., Daley, R. & Bradley, R. H. Effects of annealing on the surface composition and morphology of PS/PMMA blend. Macromolecules 33, 8453–8459 (2000).
Zhao, Z. et al. General synthesis of ultrafine monodispersed hybrid nanoparticles from highly stable monomicelles. Adv. Mater. 33, 2100820 (2021).
Zhao, Y. L. et al. Superior high-temperature properties and deformation-induced planar faults in a novel L12-strengthened high-entropy alloy. Acta Mater. 8, 517–527 (2020).
Shen, B. et al. Crystal structure engineering in multimetallic high-index facet nanocatalysts. Proc. Natl Acad. Sci. USA 118, e2105722118 (2021).
Lan, K. et al. Precisely designed mesoscopic titania for high-volumetric-density pseudocapacitance. J. Am. Chem. Soc. 143, 14097–14105 (2021).
Lv, H. & Liu, B. Multidimensionally ordered mesoporous intermetallics: frontier nanoarchitectonics for advanced catalysis. Chem. Soc. Rev. 53, 11321–11333 (2024).
Mu, J. et al. Bimetallic PdFe3 nano-alloy with tunable electron configuration for boosting electrochemical nitrogen fixation. Adv. Energy Mater. 14, 2303558 (2024).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos. 52225204, W2511047, and 52572067), the Natural Science Foundation of Shanghai (grant no. 23ZR1479200), the Shanghai Scientific and Technological Innovation Project (grant no. 24520712800), Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (grant no. JYB2025XDXM402), the Innovation Program of Shanghai Municipal Education Commission (grant no. 2021-01-07-00-03-E00109), and AI-Enhanced Research Program of Shanghai Municipal Education Commission (grant no. AMEC-AI-DHUZ-04).
Author information
Authors and Affiliations
Contributions
P.Q. and W.L. developed the Protocol and guided the project. G.Z. and M.L. performed the experiments. W.B. and Y.J. analyzed the morphology. W.B. and W. L. contributed to the discussion and manuscript modification. All authors contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Yunqing Kang, Mohamed Barakat Zakaria and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Zhu, G. et al. Adv. Mater. 37, 2413560 (2025): https://doi.org/10.1002/adma.202413560
Xie, M. et al. Adv. Energy Mater. 14, 2401717 (2024): https://doi.org/10.1002/aenm.202401717
Qiu, P. et al. NPG Asia Mater. 15, 35 (2023): https://doi.org/10.1038/s41427-023-00482-z
Zhu, G. et al. Adv. Mater. 34, 2110128 (2022): https://doi.org/10.1002/adma.202110128
Extended data
Extended Data Fig. 1 Characterization of the various binary iNPs-mC.
(a) XRD patterns, (b) Dark field STEM image of PdMn iNPs-mC-CF and corresponding EDS maps, and (c) Dark field STEM image of PdZn iNPs-mC-CF and corresponding EDS maps. Panels b and c adapted from ref. 49; Wiley.
Extended Data Fig. 2 Atomic structure characterizations of binary PtFe iNPs-mC-GO and octonary PtPdFeCoNiCuMn iNPs-mC-GO.
(a1) Atomic HAADF-STEM image of PtFe iNPs-mC-GO and (a2) Enlarged HAADF-STEM image in white frame observed in figure a1; (a3) the corresponding FFT patterns; (a4) the simulative FFT pattern projected along the [101] direction and (a5) the simulative axial rotation atomic structure scheme. (b1) Atomic HAADF-STEM image of PtPdFeCoNiCuMn iNPs-mC-GO and (b2) Enlarged HAADF-STEM image in white frame observed in figure b1; (b3) the corresponding FFT patterns; (b4) the simulative FFT pattern projected along the [100] direction and (b5) the simulative axial rotation atomic structure scheme. Panels a and b reproduced from ref. 51; Wiley.
Extended Data Fig. 3 Atomic structure characterization of PdFeCoNiCu iNPs-mC-GO.
(a,b) Atom-scale HAADF-STEM image and corresponding FFT pattern acquired along the [100] zone axis, (c,d) Atom-scale HAADF-STEM image and corresponding FFT pattern acquired along [110] zone axis, (e) Atom-resolved HAADF-STEM image and corresponding EDS maps, (f) Schematic illustration of the elemental distribution across lattice sites, superposed on the HAADF image along the [100] zone axis, (g) Column intensities along the line in (e,h) Atomic fraction for each element. Reproduced from ref. 50; Wiley.
Extended Data Fig. 4 Characterization of the recycled PdFeCoNiCu iNPs-mC-GO catalyst after NO3−RR test.
(a) Dark field STEM image with corresponding EDS elemental mapping, (b) HAADF-STTEM image with its associated FFT pattern (c). Reproduced from ref. 50; Wiley.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–15 and Table 1.
Source data
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Fig. 10 (download XLSX )
Statistical source data.
Source Data Fig. 11 (download XLSX )
Statistical source data.
Source Data Fig. 12 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiu, P., Zhu, G., Li, M. et al. Ligand-assisted interfacial monomicelle assembly to incorporate intermetallic nanoparticles into mesoporous carbon nanostructures. Nat Protoc (2026). https://doi.org/10.1038/s41596-025-01326-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-025-01326-6