Abstract
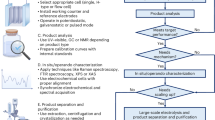
Decentralized, sustainable ammonia production could have an immense global impact. Here we describe an electrolytic approach to synthesizing ammonia directly from air and water under ambient conditions, which could be developed and optimized toward this goal. The system integrates a gliding arc discharge plasma reactor for generating \({\rm{NO}}_{\rm{x}}\) from air with a membrane electrode assembly reactor for the electrochemical reduction of \({\rm{NO}}_{\rm{x}}^{-}\) to ammonia, enhancing both the efficiency and scalability of the process. Furthermore, the plasma-generated \({\rm{NO}}_{\rm{x}}\) feedstock can be substituted with \({\rm{NO}}_{\rm{x}}\) derived from industrial waste, further extending the potential of this system. In this Protocol, we describe the fundamental principles of this plasma-electrochemical nitrogen reduction reaction (PE-N2RR) system and provide advice for experimental standardization, operational mechanisms and data analysis methods. The procedure starts with the synthesis of the catalyst—a La1.5Sr0.5Ni0.5Fe0.5O4 perovskite oxide—at either laboratory or industrial scale. This catalyst is sufficiently stable to enable the \({\rm{NO}}_{\rm{x}}^{-}\) RR to continuously work under strongly acidic conditions. We highlight the key operating parameters that are necessary for plasma-based \({\rm{NO}}_{\rm{x}}\) production and electrochemical \({\rm{NO}}_{\rm{x}}^{-}\) reduction reaction systems. This information and framework can be used to optimize and streamline the entire PE-N2RR system. A moderate level of expertise in electrochemistry, plasma systems and catalyst synthesis is recommended to ensure successful execution. The setup of the entire PE-N2RR system, from catalyst synthesis to the configuration of plasma and electrochemical, is estimated to take 72 h. The full reaction operation test requires 200 h, whereas in situ electrochemical characterizations take 3 h.
Key points
-
This Protocol describes a plasma-coupled electrochemical approach for ammonia synthesis using air and water as feedstocks. The system integrates a gliding arc discharge plasma system for generating \({\rm{NO}}_{\rm{x}}\) and a membrane electrode assembly electrochemical system to reduce \({\rm{NO}}_{\rm{x}}^{-}\) into ammonia.
-
This integrated strategy overcomes the low efficiency limitations of conventional electrochemical nitrogen reduction reaction while retaining broad adaptability across diverse application scenarios.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout













Similar content being viewed by others
Data availability
All data generated or analyzed during this study are available from the corresponding author upon reasonable request. All the data that support the plots within this paper have been published previously44,45,46. Source data are provided with this paper.
References
Gong, Z. et al. Modulating metal-nitrogen coupling in anti-perovskite nitride via cation doping for efficient reduction of nitrate to ammonia. Angew. Chem. Int. Ed. 62, e202308775 (2023).
Li, L. et al. Efficient nitrogen fixation to ammonia through integration of plasma oxidation with electrocatalytic reduction. Angew. Chem. Int. Ed. 60, 14131–14137 (2021).
Chen, F. et al. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. 7, 1032–1043 (2024).
Chen, F. Y. et al. Efficient conversion of low-concentration nitrate sources into ammonia on a Ru-dispersed Cu nanowire electrocatalyst. Nat. Nanotechnol. 17, 759–767 (2022).
Dong, K. et al. H2O2-mediated electrosynthesis of nitrate from air. Nat. Synth. 3, 763–773 (2024).
Bu, Y. et al. Electrical pulse‐driven periodic self‐repair of Cu–Ni tandem catalyst for efficient ammonia synthesis from nitrate. Angew. Chem. Int. Ed. 62, e202217337 (2023).
Jiang, H. F. et al. Enabled efficient ammonia synthesis and energy supply in a zinc–nitrate battery system by separating nitrate reduction process into two stages. Angew. Chem. Int. Ed. 62, e202218717 (2023).
Zhang, G. et al. Tandem electrocatalytic nitrate reduction to ammonia on MBenes. Angew. Chem. Int. Ed. 62, e202300054 (2023).
Xu, J. W. et al. Breaking local charge symmetry of iron single atoms for efficient electrocatalytic nitrate reduction to ammonia. Angew. Chem. Int. Ed. 62, e202308044 (2023).
Wu, A. et al. Direct ammonia synthesis from the air via gliding arc plasma integrated with single atom electrocatalysis. Appl. Catal. B 299, 120667 (2021).
Kim, K.-H. et al. Energy-efficient electrochemical ammonia production from dilute nitrate solution. Energy Environ. Sci. 16, 663–672 (2023).
Sousa, J. et al. Techno-economic study of an electrolysis-based green ammonia production plant. Ind. Eng. Chem. Res. 61, 14515–14530 (2022).
Nami, H. et al. Green ammonia production using current and emerging electrolysis technologies. Renew. Sust. Energ. Rev. 199, 114517 (2024).
Li, S. F. et al. Long-term continuous ammonia electrosynthesis. Nature 629, 92–97 (2024).
Fu, X. et al. Continuous-flow electrosynthesis of ammonia by nitrogen reduction and hydrogen oxidation. Science 379, 707–712 (2023).
Zhang, H. et al. Unveiling cutting-edge developments in electrocatalytic nitrate-to-ammonia conversion. Adv. Mater. 36, 2312746 (2024).
Zhang, R. et al. Electrochemical nitrate reduction in acid enables high-efficiency ammonia synthesis and high-voltage pollutes-based fuel cells. Nat. Commun. 14, 8036 (2023).
Chen, G. et al. Electrochemical reduction of nitrate to ammonia via direct eight-electron transfer using a copper–molecular solid catalyst. Nat. Energy 5, 605–613 (2020).
Zhang, K. et al. Energy-efficient and cost-effective ammonia electrolysis for converting ammonia to green hydrogen. Cell Rep. Phys. Sci. 5, 102171 (2024).
Wang, L. et al. Greening ammonia toward the solar ammonia refinery. Joule 2, 1055–1074 (2018).
Collado, L. et al. Light-driven nitrogen fixation routes for green ammonia production. Chem. Soc. Rev. 53, 11334–11389 (2024).
Jinuk, C. et al. Recent advances in 2D structured materials with defect-exploiting design strategies for electrocatalysis of nitrate to ammonia. Energy Mater. 4, 400020 (2024).
Li, L. Q. et al. Electrocatalytic nitrogen cycle: mechanism, materials, and momentum. Energy Environ. Sci. 9027−9050 (2024).
Han, B. et al. Recent advances in metal-organic framework-based nanomaterials for electrocatalytic nitrogen reduction. Small Methods 7, 2300277 (2023).
Mushtaq, M. A. et al. A metal coordination number determined catalytic performance in manganese borides for ambient electrolysis of nitrogen to ammonia. Adv. Mater. 36, 2313086 (2024).
Wang, D. et al. Oxygen-bridged copper–iron atomic pair as dual-metal active sites for boosting electrocatalytic NO reduction. Adv. Mater. 35, 2304646 (2023).
Yu, Y.-Z. et al. Advanced ruthenium-based electrocatalysts for NO reduction to ammonia. Adv. Mater. 37, 2412363 (2025).
Liang, J. et al. Advances in ammonia electrosynthesis from ambient nitrate/nitrite reduction. Chem 9, 1768–1827 (2023).
Soloveichik, G. Electrochemical synthesis of ammonia as a potential alternative to the Haber–Bosch process. Nat. Catal. 2, 377–380 (2019).
Liang, W. P. et al. Cascade N2 reduction process with DBD plasma oxidation and electrocatalytic reduction for continuous ammonia synthesis. Environ. Sci. Technol. 57, 14558–14568 (2023).
Liu, W. et al. Efficient ammonia synthesis from the air using tandem non-thermal plasma and electrocatalysis at ambient conditions. Nat. Commun. 15, 3524 (2024).
Eyde, S. Oxidation of atmospheric nitrogen and development of resulting industries in norway. J. Ind. Eng. Chem. 4, 771–774 (1912).
Bian, W. et al. Nitrogen fixed into HNO3 by pulsed high voltage discharge. J. Electrostat. 70, 317–326 (2012).
Patil, B. S. et al. Plasma assisted nitrogen oxide production from air: using pulsed powered gliding arc reactor for a containerized plant. Aiche J. 64, 526–537 (2018).
Wang, W. et al. Nitrogen fixation by gliding arc plasma: better insight by chemical kinetics modelling. ChemSusChem 10, 2110–2110 (2017).
Rouwenhorst, K. H. R. et al. From the Birkeland–Eyde process towards energy-efficient plasma-based NOX synthesis: a techno-economic analysis. Energy Environ. Sci. 14, 2520–2534 (2021).
Van Alphen, S. et al. Effusion nozzle for energy-efficient \({\rm{NO}}_{\rm{x}}\) production in a rotating gliding arc plasma reactor. Chem. Eng. J. 443, 136529 (2022).
Wan, L. et al. Key components and design strategy of the membrane electrode assembly for alkaline water electrolysis. Energy Environ. Sci. 16, 1384–1430 (2023).
Huang, X.-J. et al. Spectroscopic study on rotational and vibrational temperature of N2 and \({\rm{N}}_{2}^{+}\) in dual-frequency capacitively coupled plasma. Phys. Plasmas. 15, 113504 (2008).
Huang, X. J. et al. Influence of exciting frequency on gas and ion rotational temperatures of nitrogen capacitively coupled plasma. Phys. Plasmas. 15, 073501 (2008).
Han, S. H. et al. Ultralow overpotential nitrate reduction to ammonia via a three-step relay mechanism. Nat. Catal. 6, 402–414 (2023).
He, W. et al. Splicing the active phases of copper/cobalt-based catalysts achieves high-rate tandem electroreduction of nitrate to ammonia. Nat. Commun. 13, 1129 (2022).
Sun, J. et al. Diatomic Pd−Cu metal‐phosphorus sites for complete N≡N bond formation in photoelectrochemical nitrate reduction. Angew. Chem. Int. Ed. 61, e202211373 (2022).
Guo, X. et al. Highly stable perovskite oxides for electrocatalytic acidic \({\rm{NO}}_{\rm{x}}^{-}\) reduction streamlining ammonia synthesis from air. Angew. Chem. Int. Ed. 63, e202410517 (2024).
Zhang, C. et al. Concentrated formic acid from CO2 electrolysis for directly driving fuel cell. Angew. Chem. Int. Ed. 63, e202317628 (2024).
Zhang, S. et al. Sustainable nitrogen fixation with nanosecond pulsed spark discharges: insights into free-radical-chain reactions. Green Chem. 24, 1534–1544 (2022).
Acknowledgements
We acknowledge financial support from the Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XDB0450401), the National Key R&D Program of China (grant nos. 2020YFA0406103 and 2022YFE0126500) and the National Natural Science Foundation of China (grant nos. 21725102, 51925703, 22232003, 22150610467 and 52261135635).
Author information
Authors and Affiliations
Contributions
J.L., T.S. and Y.X. supervised the project. X.G. and J.L. designed the experiments. X.G., Y.G., Z.C., S.Z., S.S. and J.M. carried out the experiments and analyzed the results. X.G., J.L., T.S. and Y.X. wrote the manuscript. D.M. contributed to manuscript modification. All authors read and approved the manuscript. X.G., Y. G. and C.Z. contributed equally.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Angela Daisley and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Guo, X. et. al. Angew. Chem. Int. Ed. 63, e202410517 (2024): https://doi.org/10.1002/anie.202410517
Zhang, C. et. al. Angew. Chem. Int. Ed. 63, e202317628 (2024): https://doi.org/10.1002/anie.202317628
Zhang, S. et. al. Green Chem. 24, 1534−1544 (2022): https://doi.org/10.1039/D1GC03859A
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–5.
Supplementary Data 1 (download XLSX )
Statistical source data.
Supplementary Data 2 (download XLSX )
Statistical source data.
Supplementary Data 3 (download XLSX )
Statistical source data.
Supplementary Data 4 (download XLSX )
Statistical source data.
Source data
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Fig. 9 (download XLSX )
Statistical source data.
Source Data Fig. 10 (download XLSX )
Statistical source data.
Source Data Fig. 11 (download XLSX )
Statistical source data.
Source Data Fig. 12 (download XLSX )
Statistical source data.
Source Data Fig. 13 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, X., Gao, Y., Zhang, C. et al. Plasma-coupled electrochemical ammonia synthesis from air and water under ambient conditions. Nat Protoc (2026). https://doi.org/10.1038/s41596-026-01332-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-026-01332-2