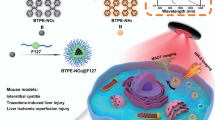
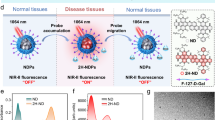
Abstract
Light-excited dual-mode imaging that integrates multispectral optoacoustic tomography (MSOT) and near-infrared region II fluorescence (NIR-II FL) imaging allows complementary deep-tissue visualization with high anatomical resolution and molecular sensitivity, thereby enhancing the accuracy of biomedical diagnostics. Activatable probes further advance this approach by producing signals upon recognition of disease-related biomarkers, which reduces background interference and improves imaging specificity. BTPE-NO2@F127, a benzothiadiazole-based nanoprobe, exhibits selective activation of both optoacoustic and NIR-II FL signals in response to hydrogen peroxide (H2O2), a crucial early stage biomarker of liver injury, thus permitting cross-validated detection of hepatic damage in vivo with excellent signal-to-background ratio contrast. Here we present detailed procedures for preparation of the BTPE-NO2@F127 nanoprobe and its applications in MSOT/NIR-II FL dual-mode imaging of trazodone- or ischemia–reperfusion-induced liver injury in mice. Compared to conventional liver injury diagnostic methods, such as invasive tissue biopsy, ex vivo blood analysis and previously reported fluorescent/optoacoustic probes, BTPE-NO2@F127 offers real-time, in situ monitoring with high sensitivity and signal-to-background ratio, as well as mutually corroborating signals for increased reliability. The fabrication of BTPE-NO2@F127, including the chemical synthesis and characterization, requires ~17 d, while the in vitro validation of its H2O2 responsiveness takes ~5 d. Notably, the complete workflow of data acquisition and analysis for MSOT/NIR-II FL dual-mode imaging of liver injury in mice using BTPE-NO2@F127 can be accomplished within 10 h. The protocol is easy to follow and suitable for clinicians and researchers with a basic understanding of chemistry and bioimaging techniques.
Key points
-
This is a protocol for the preparation and characterization of the activatable BTPE-NO2@F127 nanoprobe and its applications in multispectral optoacoustic and near-infrared region II fluorescence dual-mode imaging of liver injury models in mice.
-
Compared to conventional liver injury diagnostic methods, such as invasive tissue biopsy and ex vivo blood analysis, this approach offers real-time, in situ monitoring with high sensitivity and low noise, as well as mutually corroborating signals for increased reliability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data discussed in this protocol are available in the supporting primary research paper41. The raw datasets for Figs. 4 and 5 are provided in the Source Data file, and for Supplementary Figs. 18 and 20–25, they are provided in Supplementary Data 1. All other data are available for research purposes from the corresponding authors upon reasonable request. Source data are provided with this paper.
References
Yankeelov, T. E., Abramson, R. G. & Quarles, C. C. Quantitative multimodality imaging in cancer research and therapy. Nat. Rev. Clin. Oncol. 11, 670–680 (2014).
He, S., Song, J., Qu, J. & Cheng, Z. Crucial breakthrough of second near-infrared biological window fluorophores: design and synthesis toward multimodal imaging and theranostics. Chem. Soc. Rev. 47, 4258–4278 (2018).
Wang, F. et al. In vivo NIR-II fluorescence imaging for biology and medicine. Nat. Photon. 18, 535–547 (2024).
Schmidt, E. L. et al. Near-infrared II fluorescence imaging. Nat. Rev. Methods Primers 4, 23 (2024).
Ouyang, J. et al. Nanoaggregate probe for breast cancer metastasis through multispectral optoacoustic tomography and aggregation-induced NIR-I/II fluorescence imaging. Angew. Chem. Int. Ed. 59, 10111–10121 (2020).
Chen, Y., Yang, Y. & Zhang, F. Noninvasive in vivo microscopy of single neutrophils in the mouse brain via NIR-II fluorescent nanomaterials. Nat. Protoc. 19, 2386–2407 (2024).
Wang, L. V. & Hu, S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science 335, 1458–1462 (2012).
Wu, Y. et al. Cucurbit[8]uril-based water-dispersible assemblies with enhanced optoacoustic performance for multispectral optoacoustic imaging. Nat. Commun. 14, 3918 (2023).
Taruttis, A. & Ntziachristos, V. Advances in real-time multispectral optoacoustic imaging and its applications. Nat. Photon. 9, 219–227 (2015).
Wu, Y. et al. A nanoprobe for diagnosing and mapping lymphatic metastasis of tumor using 3D multispectral optoacoustic tomography owing to aggregation/deaggregation induced spectral change. Adv. Funct. Mater. 29, 1807960 (2019).
Razansky, D., Buehler, A. & Ntziachristos, V. Volumetric real-time multispectral optoacoustic tomography of biomarkers. Nat. Protoc. 6, 1121–1129 (2011).
Guo, B. et al. Precise deciphering of brain vasculatures and microscopic tumors with dual NIR-II fluorescence and photoacoustic imaging. Adv. Mater. 31, 1902504 (2019).
Hu, B. et al. NIR-II fluorescence/photoacoustic dual ratiometric probes with unique recognition site for quantitatively visualizing H2S2 in vivo. Angew. Chem. Int. Ed. 64, e202418378 (2025).
Liang, M. et al. Furan-modified thiadiazolo quinoxaline as an electron acceptor for constructing second near-infrared aggregation-induced emission fluorophores for beyond 1300 nm fluorescence/photoacoustic imaging and photothermal therapy. Aggregate 5, e458 (2024).
Yang, Q. et al. Donor engineering for NIR-II molecular fluorophores with enhanced fluorescent performance. J. Am. Chem. Soc. 140, 1715–1724 (2018).
Li, S. et al. In vivo real-time pharmaceutical evaluations of near-infrared II fluorescent nanomedicine bound polyethylene glycol ligands for tumor photothermal ablation. ACS Nano 14, 13681–13690 (2020).
Antaris, A. L. et al. A high quantum yield molecule-protein complex fluorophore for near-infrared II imaging. Nat. Commun. 8, 15269 (2017).
Qi, J. et al. Boosting fluorescence–photoacoustic–Raman properties in one fluorophore for precise cancer surgery. Chem 5, 2657–2677 (2019).
Qi, J. et al. Real-time and high-resolution bioimaging with bright aggregation-induced emission dots in short-wave infrared region. Adv. Mater. 30, 1706856 (2018).
Li, B. et al. Photothermal therapy of tuberculosis using targeting pre-activated macrophage membrane-coated nanoparticles. Nat. Nanotechnol. 19, 834–845 (2024).
Wu, Y. et al. Activatable probes for diagnosing and positioning liver injury and metastatic tumors by multispectral optoacoustic tomography. Nat. Commun. 9, 3983 (2018).
Hu, Z., Feng, L. & Yang, P. 2,1,3-Benzothiadiazole derivative small molecule fluorophores for NIR-II bioimaging. Adv. Funct. Mater. 34, 2310818 (2024).
Voican, C. S., Corruble, E., Naveau, S. & Perlemuter, G. Antidepressant-induced liver injury: a review for clinicians. Am. J. Psychiat. 171, 404–415 (2014).
Liu, Y. et al. Protective effects of hydrogen enriched saline on liver ischemia reperfusion injury by reducing oxidative stress and HMGB1 release. BMC Gastroenterol. 14, 12 (2014).
Carvalhana, S. et al. Acute liver failure due to trazodone and diazepam. GE Port. J. Gastroenterol. 24, 40–42 (2017).
Moon, K. H. et al. Oxidative inactivation of key mitochondrial proteins leads to dysfunction and injury in hepatic ischemia reperfusion. Gastroenterology 135, 1344–1357 (2008).
Li, Y. et al. Design of AIEgens for near-infrared IIb imaging through structural modulation at molecular and morphological levels. Nat. Commun. 11, 1255 (2020).
Gao, S. et al. Albumin tailoring fluorescence and photothermal conversion effect of near-infrared-II fluorophore with aggregation-induced emission characteristics. Nat. Commun. 10, 2206 (2019).
Mei, J., Leung, N. L. C., Kwok, R. T. K., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission: together we shine, united we soar!. Chem. Rev. 115, 11718–11940 (2015).
Guo, Z., Yan, C. & Zhu, W. H. High-performance quinoline-malononitrile core as a building block for the diversity-oriented synthesis of AIEgens. Angew. Chem. Int. Ed. 59, 9812–9825 (2020).
Mei, J. et al. Aggregation-induced emission: the whole is more brilliant than the parts. Adv. Mater. 26, 5429–5479 (2014).
Li, Y. et al. Novel NIR-II organic fluorophores for bioimaging beyond 1550 nm. Chem. Sci. 11, 2621–2626 (2020).
Ding, D. et al. Ultrabright organic dots with aggregation-induced emission characteristics for real-time two-photon intravital vasculature imaging. Adv. Mater. 25, 6083–6088 (2013).
Harmatys, K. M. et al. Multipronged biomimetic approach to create optically tunable nanoparticles. Angew. Chem. Int. Ed. 57, 8125–8129 (2018).
Wu, Y. et al. Unnatural triggers converted from tetrazine-attached sialic acid for activation of optoacoustic imaging-guided cancer theranostics. Angew. Chem. Int. Ed. 64, e202503850 (2025).
Pan, W. et al. Recent advances in NIR-II fluorescence/photoacoustic dual-modality imaging probes. Coord. Chem. Rev. 514, 215907 (2024).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Wang, Y., Hu, Y. & Ye, D. Activatable multimodal probes for in vivo imaging and theranostics. Angew. Chem. Int. Ed. 61, e202209512 (2022).
Lyu, Y. & Pu, K. Recent advances of activatable molecular probes based on semiconducting polymer nanoparticles in sensing and imaging. Adv. Sci. 4, 1600481 (2017).
Wu, Y., Zeng, F., Zhao, Y. & Wu, S. Emerging contrast agents for multispectral optoacoustic imaging and their biomedical applications. Chem. Soc. Rev. 50, 7924–7940 (2021).
Chen, J. et al. A H2O2-activatable nanoprobe for diagnosing interstitial cystitis and liver ischemia-reperfusion injury via multispectral optoacoustic tomography and NIR-II fluorescent imaging. Nat. Commun. 12, 6870 (2021).
Li, Z. et al. Bioinspired menstrual blood-derived stem cells-conditioned medium/polymersome nanoparticles for the treatment of carbon tetrachloride-induced acute liver failure in mice. Aggregate 5, e564 (2024).
Sanai, F. M. & Keeffe, E. B. Liver biopsy for histological assessment: the case against. Saudi J. Gastroenterol. 16, 124–132 (2010).
Salas, J. et al. Noninvasive imaging of drug-induced liver injury with 18F-DFA PET. J. Nucl. Med. 59, 1308–1315 (2018).
Hueper, K. et al. Assessment of liver ischemia reperfusion injury in mice using hepatic T2 mapping: comparison with histopathology. J. Magn. Reson. Imaging 48, 1586–1594 (2018).
Zhang, C. et al. Structurally modulated formation of cyanine J-aggregates with sharp and tunable spectra for multiplexed optoacoustic and fluorescence bioimaging. Angew. Chem. Int. Ed. 63, e202406694 (2024).
Zhu, H. et al. Synthesis of an ultrasensitive BODIPY-derived fluorescent probe for detecting HOCl in live cells. Nat. Protoc. 13, 2348–2361 (2018).
Muccelin, V. V. B. et al. Stability-focused nanoparticle development: applying particle size stability as a response factor in the design of experiments. ACS Omega 10, 26546–26550 (2025).
Acknowledgements
The work was supported by the National Research Foundation Singapore under its Competitive Research Programme (grant no. NRF-CRP26-2021-0002 to Y.Z.). This work was also supported by the National Natural Science Foundation of China (grant no. 52373209 to S.W. and 22274057 to F.Z.) and the Fund of Guangdong Provincial Key Laboratory of Luminescence from Molecular Aggregates (grant no. 2023B1212060003 to S.W.).
Author information
Authors and Affiliations
Contributions
Y.Z., S.W., F.Z., Y.W. and C.Z. conceived the idea and initiated the project. Y.W., C.Z. and J.C. contributed to the experimental work involved in this protocol. Y.W. and C.Z. wrote the protocol. Y.Z., S.W. and F.Z. supervised the study and the manuscript preparation. All authors contributed to the editing and reviewing of the draft and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Fabiao Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Wu, Y. et al. Angew. Chem. Int. Ed. 64, e202503850 (2025): https://doi.org/10.1002/anie.202503850
Zhang, C. et al. Angew. Chem. Int. Ed. 63, e202406694 (2024): https://doi.org/10.1002/anie.202406694
Chen, J. et al. Nat. Commun. 12, 6870 (2021): https://doi.org/10.1038/s41467-021-27233-4
Wu, Y. et al. Nat. Commun. 9, 3983 (2018): https://doi.org/10.1038/s41467-018-06499-1
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–25, Procedure and Tables 1–4.
Supplementary Data 1 (download XLSX )
Statistical source data for Supplementary Figs. 18 and 20–25.
Source data
Source Data Figs. 4 and 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, Y., Zhang, C., Chen, J. et al. Preparation of an activatable benzothiadiazole-based nanoprobe for multispectral optoacoustic and NIR-II fluorescence dual-mode imaging of liver injury. Nat Protoc (2026). https://doi.org/10.1038/s41596-026-01338-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-026-01338-w