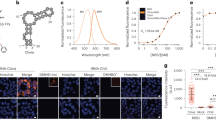
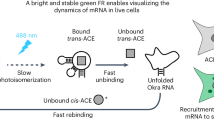
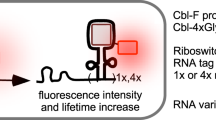
Abstract
RNAs exhibit complex dynamics in cells, including expression, splicing, localization, translation and degradation, and these processes are highly coordinated and tightly regulated both spatially and temporally. To better understand the biological function of diverse RNAs, approaches that allow monitoring of RNA with high spatiotemporal resolution are essential. Fluorescent RNAs (FRs), fluorescent protein–like entities consisting of RNA aptamers and their cognate fluorogenic dyes, have emerged as a promising approach for imaging RNA dynamics in live cells. We recently reported the development of several high-performance FRs, named Pepper, Clivia and Okra, that show advantageous properties, including high cellular brightness and photostability, low ion dependence and/or large Stokes shifts, and have been used to image diverse RNA species in live cells. In this protocol, we provide easy, efficient and generalizable strategies for using FRs to visualize different RNA species in bacteria and mammalian cells by expressing the RNA of interest tagged with one or more copies of the aptamer. We also provide a detailed procedure for multiplexed RNA imaging using orthogonal FRs and the steps to perform super-resolution live imaging of RNAs. The protocol typically takes 5–7 d, including cloning, transfection of mammalian cells or transformation of bacteria, live imaging and results analysis. This protocol is applicable to the real-time monitoring of the localization and dynamics of RNAs of interest in live cells.
Key points
-
Single or tandem copies of the RNA aptamers Pepper, Okra or Clivia are fused to a target RNA expressed exogenously in bacteria or mammalian cells. The aptamer binds its cognate fluorogenic dye, rendering the RNA fluorescent for live imaging.
-
Because of their small size and high brightness and stability, fluorescent RNA tags permit live imaging of diverse RNAs, including small noncoding and messenger RNAs, while the orthogonality of the tags allows them to be multiplexed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
References
Cech, T. R. & Steitz, J. A. The noncoding RNA revolution—trashing old rules to forge new ones. Cell 157, 77–94 (2014).
Battich, N., Stoeger, T. & Pelkmans, L. Image-based transcriptomics in thousands of single human cells at single-molecule resolution. Nat. Methods 10, 1127–1133 (2013).
Wang, K. C. & Chang, H. Y. Molecular mechanisms of long noncoding RNAs. Mol. Cell 43, 904–914 (2011).
Buxbaum, A. R., Haimovich, G. & Singer, R. H. In the right place at the right time: visualizing and understanding mRNA localization. Nat. Rev. Mol. Cell Biol. 16, 95–109 (2015).
Lu, X., Kong, K. Y. S. & Unrau, P. J. Harmonizing the growing fluorogenic RNA aptamer toolbox for RNA detection and imaging. Chem. Soc. Rev. 52, 4071–4098 (2023).
Huang, Z., Guo, X., Ma, X., Wang, F. & Jiang, J. H. Genetically encodable tagging and sensing systems for fluorescent RNA imaging. Biosens. Bioelectron. 219, 114769 (2023).
Truong, L. & Ferre-D’Amare, A. R. From fluorescent proteins to fluorogenic RNAs: tools for imaging cellular macromolecules. Protein Sci. 28, 1374–1386 (2019).
Fang, M., Jiang, Y., Chen, X. & Yang, Y. Capabilities and challenges for the use of fluorescent RNAs in RNA dynamics research. Trends Cell Biol. 36, 86–99 (2025).
Chen, X. et al. Visualizing RNA dynamics in live cells with bright and stable fluorescent RNAs. Nat. Biotechnol. 37, 1287–1293 (2019).
Huang, K. et al. Structure-based investigation of fluorogenic Pepper aptamer. Nat. Chem. Biol. 17, 1289–1295 (2021).
Jiang, L. et al. Large Stokes shift fluorescent RNAs for dual-emission fluorescence and bioluminescence imaging in live cells. Nat. Methods 20, 1563–1572 (2023).
Huang, K. et al. Structural basis of a small monomeric Clivia fluorogenic RNA with a large Stokes shift. Nat. Chem. Biol. 20, 1453–1460 (2024).
Zuo, F. et al. Imaging the dynamics of messenger RNA with a bright and stable green fluorescent RNA. Nat. Chem. Biol. 20, 1272–1281 (2024).
Jiang, L. et al. Bright and stable cyan fluorescent RNA enables multicolor RNA imaging in live Escherichia coli. Small 21, e2405165 (2025).
Schwanhausser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Singer, R. H. & Ward, D. C. Actin gene expression visualized in chicken muscle tissue culture by using in situ hybridization with a biotinated nucleotide analog. Proc. Natl Acad. Sci. USA 79, 7331–7335 (1982).
Femino, A. M., Fay, F. S., Fogarty, K. & Singer, R. H. Visualization of single RNA transcripts in situ. Science 280, 585–590 (1998).
Chen, K. H., Boettiger, A. N., Moffitt, J. R., Wang, S. & Zhuang, X. RNA imaging. Spatially resolved, highly multiplexed RNA profiling in single cells. Science 348, aaa6090 (2015).
Tyagi, S. Imaging intracellular RNA distribution and dynamics in living cells. Nat. Methods 6, 331–338 (2009).
Mao, S., Ying, Y., Wu, R. & Chen, A. K. Recent advances in the molecular beacon technology for live-cell single-molecule imaging. iScience 23, 101801 (2020).
Bertrand, E. et al. Localization of ASH1 mRNA particles in living yeast. Mol. Cell 2, 437–445 (1998).
Larson, D. R., Zenklusen, D., Wu, B., Chao, J. A. & Singer, R. H. Real-time observation of transcription initiation and elongation on an endogenous yeast gene. Science 332, 475–478 (2011).
Daigle, N. & Ellenberg, J. LambdaN-GFP: an RNA reporter system for live-cell imaging. Nat. Methods 4, 633–636 (2007).
Nelles, D. A. et al. Programmable RNA tracking in live cells with CRISPR/Cas9. Cell 165, 488–496 (2016).
Batra, R. et al. Elimination of toxic microsatellite repeat expansion RNA by RNA-targeting Cas9. Cell 170, 899–912.e10 (2017).
Yang, L. Z. et al. Dynamic imaging of RNA in living cells by CRISPR-Cas13 Systems. Mol. Cell 76, 981–997.e7 (2019).
Fusco, D. et al. Single mRNA molecules demonstrate probabilistic movement in living mammalian cells. Curr. Biol. 13, 161–167 (2003).
Tutucci, E. et al. An improved MS2 system for accurate reporting of the mRNA life cycle. Nat. Methods 15, 81–89 (2018).
Li, W. H., Maekiniemi, A., Sato, H., Osman, C. & Singer, R. H. An improved imaging system that corrects MS2-induced RNA destabilization. Nat. Methods 19, 1558–1562 (2022).
Wu, B. et al. Synonymous modification results in high-fidelity gene expression of repetitive protein and nucleotide sequences. Genes Dev. 29, 876–886 (2015).
Park, S. Y., Moon, H. C. & Park, H. Y. Live-cell imaging of single mRNA dynamics using split superfolder green fluorescent proteins with minimal background. RNA 26, 101–109 (2020).
Wu, B., Chen, J. & Singer, R. H. Background free imaging of single mRNAs in live cells using split fluorescent proteins. Sci. Rep. 4, 3615 (2014).
Wu, J. et al. Live imaging of mRNA using RNA-stabilized fluorogenic proteins. Nat. Methods 16, 862–865 (2019).
Kuffner, C. J., Marzilli, A. M. & Ngo, J. T. RNA-stabilized coat proteins for sensitive and simultaneous imaging of distinct single mRNAs in live cells. Nat. Methods 23, 153–164 (2026).
Zhou, W. J. et al. Fluorogenic interacting protein stabilization for orthogonal RNA imaging. Angew. Chem. Int. Ed. Engl. 64, e202502350 (2025).
Pham, T. G., Ajayi, O. & Wu, J. Orthogonal RNA-regulated destabilization domains for three-color RNA imaging with minimal RNA perturbation. Nat. Methods 23, 165–174 (2026).
Xia, C., Colognori, D., Jiang, X. S., Xu, K. & Doudna, J. A. Single-molecule live-cell RNA imaging with CRISPR-Csm. Nat. Biotechnol. 43, 2023–2030 (2025).
Englert, D. et al. Fast-exchanging spirocyclic rhodamine probes for aptamer-based super-resolution RNA imaging. Nat. Commun. 14, 3879 (2023).
Sunbul, M. et al. Super-resolution RNA imaging using a rhodamine-binding aptamer with fast exchange kinetics. Nat. Biotechnol. 39, 686–690 (2021).
Wirth, R., Gao, P., Nienhaus, G. U., Sunbul, M. & Jaschke, A. SiRA: a silicon rhodamine-binding aptamer for live-cell super-resolution RNA imaging. J. Am. Chem. Soc. 141, 7562–7571 (2019).
Li, X., Kim, H., Litke, J. L., Wu, J. & Jaffrey, S. R. Fluorophore-promoted RNA folding and photostability enables imaging of single Broccoli-tagged mRNAs in live mammalian cells. Angew. Chem. Int. Ed. Engl. 59, 4511–4518 (2020).
Cawte, A. D., Unrau, P. J. & Rueda, D. S. Live cell imaging of single RNA molecules with fluorogenic Mango II arrays. Nat. Commun. 11, 1283 (2020).
Buhler, B. et al. Avidity-based bright and photostable light-up aptamers for single-molecule mRNA imaging. Nat. Chem. Biol. 19, 478–487 (2023).
Zuo, F., Gao, Z., Chen, X. & Yang, Y. Fluorescent RNAs: new opportunities for drug discovery. Trends Pharmacol. Sci. 46, 1160–1176 (2025).
Wang, Z. et al. In situ spatial complementation of aptamer-mediated recognition enables live-cell imaging of native RNA transcripts in real time. Angew. Chem. Int. Ed. Engl. 57, 972–976 (2018).
Alam, K. K., Tawiah, K. D., Lichte, M. F., Porciani, D. & Burke, D. H. A fluorescent split aptamer for visualizing RNA-RNA assembly in vivo. ACS Synth. Biol. 6, 1710–1721 (2017).
Lescoute, A. & Westhof, E. Topology of three-way junctions in folded RNAs. RNA 12, 83–93 (2006).
Chandler, M. et al. Broccoli fluorets: split aptamers as a user-friendly fluorescent toolkit for dynamic RNA nanotechnology. Molecules 23, 3178 (2018).
Ong, W. Q., Citron, Y. R., Sekine, S. & Huang, B. Live cell imaging of endogenous mRNA using RNA-based fluorescence “turn-on” probe. ACS Chem. Biol. 12, 200–205 (2017).
Chen, Z. et al. Genetically encoded RNA-based sensors with Pepper fluorogenic aptamer. Nucleic Acids Res. 51, 8322–8336 (2023).
Sato, S. et al. Live-cell imaging of endogenous mRNAs with a small molecule. Angew. Chem. Int. Ed. Engl. 54, 1855–1858 (2015).
Zheng, H., Liu, X., Liu, L., Hu, J. & Chen, X. Imaging of endogenous RNA in live cells using sequence-activated fluorescent RNA probes. Nucleic Acids Res. 53, gkae1209 (2025).
Paige, J. S., Nguyen-Duc, T., Song, W. & Jaffrey, S. R. Fluorescence imaging of cellular metabolites with RNA. Science 335, 1194 (2012).
Fang, M. et al. Imaging intracellular metabolite and protein changes in live mammalian cells with bright fluorescent RNA-based genetically encoded sensors. Biosens. Bioelectron. 235, 115411 (2023).
DasGupta, S., Shelke, S. A., Li, N. S. & Piccirilli, J. A. Spinach RNA aptamer detects lead(II) with high selectivity. Chem. Commun. (Camb.) 51, 9034–9037 (2015).
Song, W., Strack, R. L. & Jaffrey, S. R. Imaging bacterial protein expression using genetically encoded RNA sensors. Nat. Methods 10, 873–875 (2013).
Kondo, Y., Oubridge, C., van Roon, A. M. M. & Nagai, K. Crystal structure of human U1 snRNP, a small nuclear ribonucleoprotein particle, reveals the mechanism of 5’ splice site recognition. eLife 4, e04986 (2015).
Pomeranz Krummel, D. A., Oubridge, C., Leung, A. K., Li, J. & Nagai, K. Crystal structure of human spliceosomal U1 snRNP at 5.5 Å resolution. Nature 458, 475–480 (2009).
Kim, G., Carroll, C. L., Wakefield, Z. P., Tuncay, M. & Fiszbein, A. U1 snRNP regulates alternative promoter activity by inhibiting premature polyadenylation. Mol. Cell 85, 1968–1981.e7 (2025).
Nevo-Dinur, K., Nussbaum-Shochat, A., Ben-Yehuda, S. & Amster-Choder, O. Translation-independent localization of mRNA in E. coli. Science 331, 1081–1084 (2011).
Acknowledgements
This work was financially supported by the National Key Research and Development Program of China (2022YFC3400100 to Y.Y. and X.C.; 2024YFA180006 to N.S.), NSFC (32121005, 92581203, 22437001 and 92357308 to Y.Y.; 32250009 to X.C.; 32501340 to F.Z.; 32401077 to X.X.), STI2030-Major Projects (2021ZD0202200 and 2021ZD0202203 to X.C.), the Shanghai Municipal Education Commission (2021 Sci & Tech 03-28), the Shanghai Science and Technology Commission (23J21900400 to X.C.), a grant from the Tianfu Jincheng Laboratory (TFJCPI20250016 to Y.Y.), the Shanghai Sailing Program (24YF2709300 to F.Z.), the Shanghai Municipal Health Commission (20254Y0019 to F.Z.), Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (JYB2025XDXM404), the State Key Laboratory of Bioreactor Engineering (to Y.Y. and X.C.) and the Fundamental Research Funds for the Central Universities (to Y.Y. and X.C.).
Author information
Authors and Affiliations
Contributions
Concepts were conceived by Y.Y. and X.C. Y.Y., X.C., F.Z., N.S. and X.X. designed the experiments and analyzed the data. F.Z. and X.X. performed plasmid construction. F.Z. and N.S. performed live-cell imaging and FISH experiments. M.F., L.J, Y.Z. and L.Z. gave technical support and conceptual advice. Y.Y., X.C., F.Z., N.S. and X.X. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Michael Ryckelynck and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key references
Chen, X. et al. Nat. Biotechnol. 37, 1287–1293 (2019): https://doi.org/10.1038/s41587-019-0249-1
Jiang, L. et al. Nat. Methods 20, 1563–1572 (2023): https://doi.org/10.1038/s41592-023-01997-7
Zuo, F. et al. Nat. Chem. Biol. 20, 1272–1281 (2024): https://doi.org/10.1038/s41589-024-01629-x
Supplementary information
Supplementary Information (download PDF )
Supplementary Figures 1–7, Supplementary Note 1
Supplementary Data 1 (download XLSX )
Source Data for Supplementary Figures
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zuo, F., Su, N., Xie, X. et al. Live-cell imaging of RNA dynamics using bright and stable fluorescent RNAs. Nat Protoc (2026). https://doi.org/10.1038/s41596-026-01343-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41596-026-01343-z