Abstract
In this paper, a new type of magnetic mesoporous material (Fe3O4@SiO2@KIT-6@2-ATP@CuI) was designed and synthesized and its application in the synthesis of amides and anilines was investigated. The structure of Fe3O4@SiO2@KIT-6@2-ATP@CuI was characterized and identified using FTIR, SEM, XRD, TGA, BET, VSM, and ICP techniques. An external magnet can easily remove the synthesized catalyst from the reaction medium, and be reused in several consequence runs.
Similar content being viewed by others
Introduction
Functional anilines are versatile intermediates for the preparation of agricultural chemicals, pigments, pharmaceuticals, and dyes1,2,3,4,5,6,7,8. Because of their importance, many methods have been developed for the reduction of nitroarenes to produce corresponding anilines. Generally, the methods can be classified into two types. In the common procedure, the stoichiometric reduction of the corresponding nitroarenes occurred using an appropriate reducing agent such as Na2S2O4, Fe, Sn, or Zn; this method often reasons environmental problems such as large amounts of waste acids and residues produced during the reaction. In the second procedure, the hydrogenation of nitro compounds is performed by metal catalysts in the presence of an appropriate catalyst9,10,11.
Amides are important raw materials for the production of detergents, lubricants, drug stabilizers, and mediators in peptide and protein synthesis12,13,14,15,16,17,18. For preparing amides from nitriles different methods have been reported in the literature, hydration of nitriles to the corresponding amides is one of the extensively studied procedures19,20,21,22,23,24.
Today, the use of magnetic nanoparticles (MNPs) in catalytic reactions is wildly studied. Magnetic mesoporous silica (MMS) nanoparticles due to their many important properties such as excellent stability (thermal and chemical), high surface area, simple and easy separation from the reaction medium, and recyclability, show excellent catalytic performance in a wide range of organic reactions25,26,27,28. In this research project, we have synthesized a new and efficient catalyst (Fe3O4@SiO2@KIT-6@2-ATP@CuI) that has the advantage of both magnetic nanoparticles and mesoporous materials. In this research, the catalytic aspects of Fe3O4@SiO2@KIT-6@2-ATP@CuI have been examined for hydrating nitriles and reducing nitroarenes.
Result and discussion
Preparation and characterization of Fe3O4@SiO2@KIT-6@2-ATP@CuI
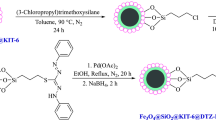
The Fe3O4@SiO2@KIT-6 was prepared as mentioned procedure in our newly published work29. Subsequently, the prepared nanoparticles were first functionalized by (3-chloropropyl) trimethoxysilane and then reacted with 2-amino thiophenol. Finally, Cu(I) was coordinated with Fe3O4@SiO2@KIT-6@2-ATP (Fig. 1).
Stepwise preparation of Fe3O4@SiO2@KIT-6@2-ATP@CuI.
After designing and fabricating Fe3O4@SiO2@KIT-6@2-ATP@CuI, the synthesized magnetic mesoporous structure was characterized by different techniques.
Infrared spectroscopy is one of the most widely used analyses for the identification of different functional groups of organic compounds. Various devices have been developed for infrared spectroscopy, the most widely used of which are Fourier transform devices. Therefore, Fourier transforms infrared spectroscopy (FT-IR) was used to identify the synthesized catalyst30. In Fig. 2, the synthetic steps of magnetic mesoporous catalyst have been studied by FT-IR analysis. Peaks appearing in 459 cm−1, 457 cm−1, 462 cm−1, 460 cm−1, 640 cm−1, 635 cm–1, and 634 cm−1 in the spectra of Fe3O4@SiO2@KIT-6 (Fig. 2a), Fe3O4@SiO2@KIT-6@CPTMS (Fig. 2b), Fe3O4@SiO2@KIT-6@2-ATP (Fig. 2c) and Fe3O4@SiO2@KIT-6@2-ATP@CuI (Fig. 2d) is related to the stretching vibration of the Fe–O bond. Also, the stretching vibration of the Si–O–Si bond in the region of 1077–1083 cm−1 appears in Fig. 2a–d. In the Fe3O4@SiO2@KIT-6@2-ATP spectrum (Fig. 2c), the peak is shown in 3513 cm−1 and 3429 cm−1 corresponding to the NH stretching vibration. In the spectrum, Fe3O4@SiO2@KIT-6@2-ATP@CuI (Fig. 2d) the peak that appears at 3444 cm−1 is belong to the N–H stretching vibration.
Infrared spectra of Fe3O4@SiO2@KIT-6 (a), Fe3O4@SiO2@KIT-6@CPTMS (b), Fe3O4@SiO2@KIT-6@2-ATP (c), Fe3O4@SiO2@KIT-6@2-ATP@CuI (d).
Scanning electron microscope (SEM) is one of the most common tools used in nanotechnology to analyze the morphology of nanostructural materials. The bombardment of the sample causes electrons to be released from the sample towards the positively charged plate, where these electrons become signals. The movement of the beam on the sample provides a set of signals based on which the microscope can display an image of the sample surface on the computer screen. So, in general, it is possible to obtain information including topography, components, and morphology of the sample31.
To consider the morphology and particle shape of the magnetic mesoporous catalyst, the SEM image of Fe3O4@SiO2@KIT-6 (a), Fe3O4@SiO2@KIT-6@CPTMS (b), Fe3O4@SiO2@KIT-6@2-ATP (c), Fe3O4@SiO2@KIT-6@2-ATP@CuI (d) has been prepared, which has been brought in Fig. 3. The SEM images confirm the mesoporous catalyst formed in spherical shapes.
SEM image of Fe3O4@SiO2@KIT-6 (a), Fe3O4@SiO2@KIT-6@CPTMS (b), Fe3O4@SiO2@KIT-6@2-ATP (c), Fe3O4@SiO2@KIT-6@2-ATP@CuI (d).
Thermogravimetric analysis (TGA), using a specific heating program and under a controlled atmosphere, measures weight changes in air or an inert atmosphere and records the mass reduction as a function of increasing temperature. Based on the results of thermal gravimetric analysis, it is possible to calculate the amount of combustible or vaporizable materials, including water and organic materials of the sample32.
Figure 4 shows the TGA diagram of a catalyst activated with 2-amino thiophenol. According to the diagram, the first weight loss (under 250 °C, about 3%) is related to the evaporation of adsorbed solvents. The second weight loss, which is about 12% and occurred at temperatures between 250 to 650 °C, is related to the removal of immobilized organic compounds, indicating that 2-amino thiophenol was successfully immobilized into KIT-6 magnetic channels.
TGA diagram of Fe3O4@SiO2@KIT-6@2-ATP@CuI.
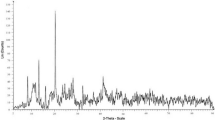
The X-ray diffraction pattern for the Fe3O4@SiO2@KIT-6@2-ATP@CuI catalyst is shown in Figs. 5 and 6, (low and wide angle respectively). The low-angle XRD spectrum shows in Fig. 5. In the high-angle XRD spectrum (Fig. 6), the peaks appearing at 43.79°, 50.54°, and 73.24° correspond to the copper metal-stabilized into the channels of the catalyst, and a broad peak of 20–30 is related to the amorph silica layer33,34.
XRD patterns of Fe3O4@SiO2@KIT-6@2-ATP@CuI (low angle XRD).
XRD patterns of Fe3O4@SiO2@KIT-6@2-ATP@CuI (wide angle XRD).
Figure 7 shows the nitrogen adsorption/desorption isotherm of Fe3O4@SiO2@KIT-6@2-ATP@CuI. The isothermal adsorption–desorption curve for Fe3O4@SiO2@KIT-6@2-ATP@CuI shows type IV of IUPAC isotherms, indicating the magnetic material form in a mesoporous structure. The N2 adsorption–desorption isotherm had a sharp bend at P/P0, indicating capillary density in uniform mesopores.
N2 adsorption–desorption isotherm of Fe3O4@SiO2@KIT-6@2-ATP@CuI.
The pore and surface properties of Fe3O4@SiO2@KIT-6@2-ATP@CuI were calculated and considered using a Brunauer-Emmet-Teller (BET) and Barret-Joyner-Halenda (BJH) methods (Tables 1 and 2).
As shown in Fig. 8, the magnetic property of Fe3O4@SiO2@KIT-6@2-ATP@CuI (1.38 emu/g) shows a significant decrease compared to Fe3O4@SiO2@KIT-6 nanoparticles (3.30 emu/g). The magnetic property of the mesoporous catalyst reflects the fact that the surface of the nanoparticles is coated with SiO2 and organic groups.
The magnetic curve of Fe3O4@SiO2@KIT-6 (A) and Fe3O4@SiO2@KIT-6@2-ATP@CuI (B).
ICP analysis was used to determine the exact amount of loaded Cu on the magnetic mesoporous catalyst and showed a value of 1.11 × 10–3 mol/g.
Catalytic studies
Hydration of benzonitriles to amides
After the structure characterized the magnetic mesoporous catalyst, its catalytic activity in the synthesis of amides was investigated. The reaction of 4-chlorobenzonitrile as model substrate was investigated in the presence of potassium hydroxide, various solvents such as water, ethanol, methanol, tetrahydrofuran, and 1-propanol, variable amounts of catalyst, and different temperature conditions. In protic polar solvents, coordination between the solvent and benzonitrile with the catalyst activates the cyano group in the nitrile substrate. Among protic polar solvents, 1-propanol led to more amide formation due to its coordination with the substrate33,35. Finally, 1-propanol, 40 mg of catalyst, 70 °C temperature, and 2 mmol of potassium hydroxide were selected as optimal conditions (Table 3).
After obtaining the reaction conditions, the reaction of different benzonitriles was performed under optimal conditions and a variety of amides were synthesized (Fig. 9). The results including reaction times and yields are reported in Table 4.
Hydration of benzonitriles to amides.
The hydration mechanism in the presence of Fe3O4@SiO2@KIT-6@2-ATP@CuI is proposed in Fig. 10. Initially, the coordination of benzonitrile with the copper atom from the catalyst may lead to an increase in the electrophilicity of the nitrile carbon (intermediate I), which by the addition of HO- ion leads to producing intermediate (III). Finally, the tautomerism of coordinated imines leads to an amide (IV)36.
The suggested mechanism of amide synthesis in the presence of Fe3O4@SiO2@KIT-6@2-ATP@CuI as a catalyst.
Reduction of nitroarenes to anilines
In another catalytic study, the activity of Fe3O4@SiO2@KIT-6@2-ATP@CuI in the reduction of nitroarenes to aromatic amines was investigated (Table 5). After considering the effect of different solvents, temperature conditions, and different amounts of catalyst; water as the solvent, and room temperature were selected as the optimal conditions for the preparation of anilines from nitroarenes.
The effect of solvent on the reduction of nitroarenes was analyzed through articles and the results show that protic polar solvents are more suitable solvents for the reduction of nitroarenes than aprotic polar solvents5.
After obtaining the optimal conditions, the reduction of different derivatives of nitroarenes to aromatic amines was performed and the results can be seen in Table 6 (Fig. 11).
Reduction of nitroarenes to anilines.
A proposed mechanism for the reduction of nitro compounds in the presence of Fe3O4@SiO2@KIT-6@2-ATP@CuI is provided in Fig. 1237.
A suggested mechanism for the reduction of nitro compounds by NaBH4 in the presence of a catalytic amount of Fe3O4@SiO2@KIT-6@2-ATP@CuI.
Reusability of the catalyst
To investigate the recovery of described catalyst, the reduction reaction of 1-chloro-4-nitrobenzene was selected as the sample reaction. The reaction was selected using 1-chloro-4-nitrobenzene, sodium borohydride, and water as the solvent in the presence of Fe3O4@SiO2@KIT-6@2-ATP@CuI. After the reaction was complete, it was separated by an external magnetic field, washed with ethanol and water, and then used in the next run. This cycle was repeated up to four times (Fig. 13).
Catalyst recovery study.
Conclusions
In this paper, the Fe3O4@SiO2@KIT-6@2-ATP@CuI as a magnetic mesoporous catalyst was designed and synthesized through the combination of Fe3O4 and KIT-6 nanoparticles. The catalytic ability of this mesoporous magnetic material was studied for the preparation of benzamides and anilines. The reported procedure in this research project offers the advantages of reasonable yields and green reaction medium, versatile catalyst preparation procedure, short reaction times of catalytic reactions, easy separation (it could be easily separated and recovered due to its magnetic properties), catalyst recyclability, and high catalyst chemical stability.
In Table 7, the Fe3O4@SiO2@KIT-6@2-ATP@CuI magnetic catalyst was compared with other previously reported catalysts for the reduction of nitrobenzene. As is evidenced in this table, the obtained results by the described catalyst in this research are comparable with other reductive systems.
Experimental
Synthesis of Fe3O4@SiO2@KIT-6@2-ATP
Fe3O4@SiO2@KIT-6 nanoparticles were synthesized using a method previously reported in the literature38. In a 50 mL balloon, Fe3O4@SiO2@KIT-6 (1 g) was sonicated for 30 min in toluene (25 mL), then, 1.5 mL of (3-chloropropyl) trimethoxysilane (CPTMS) was added and the resulting mixture was stirred for 24 h at 90 °C under nitrogen atmosphere. The obtained solid was washed with dichloromethane (50 mL) and dried in an oven. In a round bottom balloon, a mixture of Fe3O4@SiO2@KIT-6@CPTMS (1 g), and one gram of 2-amino thiophenol (2-ATP) was refluxed in the presence of triethylamine (2 mL) in ethanol for 20 h. After separating the obtained precipitate and washing it with ethanol, it was dried at 50 °C39.
Synthesis of Fe3O4@SiO2@KIT-6@2-ATP@CuI
The solid precipitate that was obtained in the previous step, was dissolved in ethanol and 2 mmol of CuCl was added and refluxed for 20 h. After the end of the reaction, the Fe3O4@SiO2@KIT-6@2-ATP@CuI catalyst was separated and washed several times with ethanol40.
The general method for the hydration of benzonitriles to amides
To perform hydration of benzonitrile, in a 5 mL round bottom flask, benzonitrile (1 mmol), potassium hydroxide (2 mmol), and 40 mg of Fe3O4@SiO2@KIT-6@2-ATP@CuI were added to 1-propanol and the mixture was stirred at 70 °C. The progress of the reaction was followed by TLC. After the completion reaction, the catalyst was separated by an external magnet and the corresponding product was extracted.
4-Cyanobenzamide
1HNMR (300 MHz, DMSO-d6) δ 8.01 (2H, d, J = 8 Hz), 7.95 (2H, d, J = 8 Hz), 7.66 (s, 2H).
4-Nitrobenzamide
1HNMR (300 MHz, DMSO-d6) δ 8.28 (2H, d, J = 8.1), 8.08 (2H, d, J = 8.1), 7.71 (s, 2H).
The general method for the reduction of nitroarenes to anilines
To prepare anilines from nitroarenes, a mixture of nitroarene, NaBH4 (5 mmol), and 20 mg catalyst was stirred at room temperature. TLC was used to monitor the progress of the reaction and the product was obtained in high yield after catalyst isolation.
4-Bromoaniline
1HNMR (300 MHz, CDCl3): δ 7.23 (2H, d, J = 7 Hz), 6.57 (2H, d, J = 7 Hz), 3.53 (s, 2H).
2-Aminobenzyl alcohol
1HNMR (300 MHz, CDCl3): δ 6.93–7.05 (m, 2H), 6.49–6.62 (m, 2H), 4.95 (s, 1H), 4.87 (s, 2H), 4.38 (s, 2 H) (Supplementary Information).
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Shi, Y. et al. Pt decorated hierarchical Sb2WO6 microspheres as a surface functionalized photocatalyst for the visible-light-driven reduction of nitrobenzene to aniline. J. Mater. Chem. A 8, 18755–18766 (2020).
Babel, V. & Hiran, B. L. Heterogeneous AgPd alloy nanocPt decorated hierarchical Sb2WO6 microspheres as a surface functionalized photocatalyst for the visible-light-driven reduction of nitrobenzene to anilineatalyst for selective reduction of aromatic nitro compounds using formic. Catal. Lett. 150, 1865–1869 (2020).
Singh, P., Halder, M., Ray, S., Bandyopadhyay, B. & Sen, K. Biomolecule-mediated generation of Ru nanocatalyst for sustainable reduction of nitrobenzene. ACS Omega 4, 21267–21278 (2019).
Romanazzi, G. et al. Polymer supported nickel nanoparticles as recyclable catalyst for the reduction of nitroarenes to anilines in aqueous medium. Mol. Catal. 446, 31–38 (2018).
Niakan, M. & Asadi, Z. Selective reduction of nitroarenes catalyzed by sustainable and reusable DNA-supported nickel nanoparticles in water at room temperature. Catal. Lett. https://doi.org/10.1007/s10562-019-02741-7 (2019).
Zhang, Q. et al. Highly efficient hydrogenation of nitrobenzene to aniline over Pt/CeO2 catalysts: The shape effect of the support and key role of additional Ce3+ sites. ACS Catal. 10, 10350–10363 (2020).
Krogul-Sobczak, A., Cedrowski, J., Kasperska, P. & Litwinienko, G. Reduction of nitrobenzene to aniline by Co/H2O in the presence of palladium nanoparticles. Catalysts 9, 404 (2019).
Wang, H., Liu, X., Xu, G., Guo, Z. & Zhang, Y. In situ synthesis of Fe–N–C catalysts from cellulose for hydrogenation of nitrobenzene to aniline. Chinese J. Catal. 40, 1557–1565 (2019).
Bao, L. et al. Palladium supported on metal–organic framework as a catalyst for the hydrogenation of nitroarenes under mild conditions. Appl. Organomet. Chem. 34, 1–8 (2020).
Zubar, V., Dewanji, A. & Rueping, M. Chemoselective hydrogenation of nitroarenes using an air-stable base-metal catalyst. Org. Lett. 23, 2742–2747 (2021).
Manrique, E. et al. A heterogeneous ruthenium dmso complex supported onto silica particles as a recyclable catalyst for the efficient hydration of nitriles in aqueous medium. Inorg. Chem. 58, 8460–8470 (2019).
Kumar Reddy, N. N., Nageswara Rao, S. D., Patil, R. & Adimurthy, S. Transition metal-free hydration of nitriles to amides mediated by NaOH. Adv. Mater. Sci. 3, 1–7 (2018).
Xing, X. et al. Highly active platinum catalysts for nitrile and cyanohydrin hydration: Catalyst design and ligand screening via high-throughput techniques. J. Am. Chem. Soc. 140, 17782–17789 (2018).
Xu, D. P., Xiong, M. & Kazemnejadi, M. Efficient reduction of nitro compounds and domino preparation of 1-substituted-1H-1,2,3,4-tetrazoles by Pd(II)-poly salophen coated magnetite NPs as a robust versatile nanocomposite. RSC Adv. 11, 12484–12499 (2021).
Sanz Sharley, D. D. & Williams, J. M. J. A selective hydration of nitriles catalysed by a Pd(OAc)2-based system in water. Tetrahedron Lett. 58, 4090–4093 (2017).
Sultana, S., Borah, G. & Gogoi, P. K. Ru (III) Schiff-base complex anchored on nanosilica as an efficient and retrievable catalyst for hydration of nitriles. Appl. Organomet. Chem. 33, 1–11 (2019).
Guo, B. & De Vries, J. G. Hydration of nitriles using a metal-ligand cooperative ruthenium pincer catalyst. Chem. Sci. 10, 10647–10652 (2019).
Kuwabara, J., Sawada, Y. & Yoshimatsu, M. Nitrile hydration reaction using copper iodide/cesium carbonate/DBU in nitromethane-water. Synletter 29, 2061–2065 (2018).
Wang, H. et al. Nanorod manganese oxide as an efficient heterogeneous catalyst for hydration of nitriles into amides. Ind. Eng. Chem. Res. 58, 17319–17324 (2019).
Wang, N., Ma, P., Xie, J. & Zhang, J. Transition metal-free NaOH-catalyzed hydration of nitriles to primary amides in NH3·H2O–DMSO mixture. Mol. Divers. 25, 1131–1136 (2021).
Hussain, M. A. et al. An efficient hydration of nitriles with ruthenium-supported heterogeneous catalyst in water under moderate conditions. J. Ind. Eng. Chem. 99, 187–195 (2021).
Czégéni, C.E., De, S., Udvardy, A., Derzsi, N.J., Papp, G. & Joó, F. Selective Hydration of Nitriles to Corresponding Amides in Air with Rh (I)-N-Heterocyclic Complex Catalysts. Catalysts. 10(1), 125 (2020).
Rahman, T., Borah, G. & Gogoi, P. K. Activated Mont K10-Carbon supported Fe2O3: A versatile catalyst for hydration of nitriles to amides and reduction of nitro compounds to amines in aqueous media. J. Chem. Sci. 133, 20–22 (2021).
KazemiMiraki, M., Arefi, M., Salamatmanesh, A., Yazdani, E. & Heydari, A. Magnetic nanoparticle-supported Cu–NHC complex as an efficient and recoverable catalyst for nitrile hydration. Catal. Lett. 148, 3378–3388 (2018).
Asgari, M., Soleymani, M., Miri, T. & Barati, A. A robust method for fabrication of monodisperse magnetic mesoporous silica nanoparticles with core-shell structure as anticancer drug carriers. J. Mol. Liq. 292, 111367 (2019).
Adam, A. et al. Orienting the pore morphology of core-shell magnetic mesoporous silica with the sol–gel temperature. Influence on MRI and magnetic hyperthermia properties. Molecules 26, 971 (2021).
He, H. et al. Thiol-ene click chemistry synthesis of a novel magnetic mesoporous silica/chitosan composite for selective Hg(II) capture and high catalytic activity of spent Hg(II) adsorbent. Chem. Eng. J. 405, 126743 (2021).
Abdolmohammadi, S., Shariati, S. & Mirza, B. Ultrasound promoted and Kit-6 mesoporous silica-supported Fe3O4 magnetic nanoparticles catalyzed cyclocondensation reaction of 4-hydroxycoumarin, 3,4-methylenedioxyphenol, and aromatic aldehydes. Appl. Organomet. Chem. 35, 1–10 (2021).
Guo, M. et al. Improving catalytic hydrogenation performance of Pd nanoparticles by electronic modulation using phosphine ligands. ACS Catal. 8, 6476–6485 (2018).
Undavalli, V. K., Ling, C., & Khandelwal, B. Impact of alternative fuels and properties on elastomer compatibility. in Aviation Fuels. 113–132 (Academic Press, 2021).
Nasrollahzadeh, M., Atarod, M., Sajjadi, M., Sajadi, S. M. & Issaabadi, Z. Plant-mediated green synthesis of nanostructures: Mechanisms, characterization, and applications. Interface Sci. Technol. 28, 199–322 (2019).
Ebnesajjad, S. Surface and material characterization techniques. in Handbook of Adhesives and Surface Preparation. 31–48 (Elsevier, 2011).
Sultana, S., Geetika, B. & Pradip, K. G. Ru (III) Schiff-base complex anchored on nanosilica as an efficient and retrievable catalyst for hydration of nitriles. Appl. Organomet. Chem. 33(1), e4595 (2019).
Amin, R., Khorshidi, A., Shojaei, A. F., Rezaei, S. & Faramarzi, M. A. Immobilization of laccase on modified Fe3O4@SiO2@Kit-6 magnetite nanoparticles for enhanced delignification of olive pomace bio-waste. Int. J. Biol. Macromol. 114, 106–113 (2018).
Yin, A. G. et al. Efficient bimetallic catalysis of nitrile hydration to amides by simple Pd(OAc)2/Lewis acid catalyst at ambient temperature. J. Organ. Chem. Eur. https://doi.org/10.1002/ejoc.201601495 (2016).
Ghorbani-Choghamarani, A. & Taherinia, Z. Fe3O4@GlcA@Cu-MOF: A magnetic metal-organic framework as a recoverable catalyst for the hydration of nitriles and reduction of isothiocyanates, isocyanates, and isocyanides. ACS Comb. Sci. 22, 902–909 (2020).
El-Hout, S. I. et al. A green chemical route for synthesis of graphene supported palladium nanoparticles: A highly active and recyclable catalyst for reduction of nitrobenzene. in Applied Catalysis A: General. Vol. 503 (Elsevier B.V., 2015).
Moradi, Z. & Ghorbani-Choghamarani, A. Design and synthesis of Fe3O4@SiO2@KIT-6@DTZ-Pd0 as a new and efficient mesoporous magnetic catalyst in carbon–carbon cross-coupling reactions. Sci. Rep. 11, 1–12 (2021).
Mousavi, S. H., Manoochehri, M. & Afshar Taromi, F. Fabrication of a novel magnetic metal-organic framework functionalized with 2-amino thiophenol for preconcentration of trace silver amounts in water and wastewater. RSC Adv. 11, 13867–13875 (2021).
Ghorbani-Choghamarani, A., Sahraei, R., Taherinia, Z. & Mohammadi, M. Cu(I)@Isatin-glycine-boehmite nanoparticles: As novel heterogeneous catalyst for the synthesis and selective oxidation of sulfides. J. Iran. Chem. Soc. 18, 827–838 (2021).
Zeynizadeh, B. & Sepehraddin, F. Synthesis and characterization of magnetically nanoparticles of Fe3O4@APTMS@ZrCp2 as a novel and reusable catalyst for convenient reduction of nitro compounds with glycerol. J. Iran. Chem. Soc. 14, 2649–2657 (2017).
Chen, D. et al. Metal-free reduction of nitro aromatics to amines with B2(OH)4/H2O. Synletter 29, 1765–1768 (2018).
Manjunatha, K. et al. Magnetic nanoparticle-tethered Schiff base–palladium(II): Highly active and reusable heterogeneous catalyst for Suzuki-Miyaura cross-coupling and reduction of nitroarenes in aqueous medium at room temperature. Appl. Organomet. Chem. 32, 1–21 (2018).
Feng, X. P. et al. Immobilized β-cyclodextrin and palladium-pyridylaldehyde complex on silica nanoparticles as a highly active catalyst for Suzuki, reduction of nitroarenes and oxidative amination of aldehydes reactions in water. Mater. Today Commun. 26, 101909 (2021).
Dai, Y., Li, X., Wang, L. & Xu, X. Highly efficient hydrogenation reduction of aromatic nitro compounds using MOF derivative Co-N/C catalyst. New J. Chem. 45, 22908–22914 (2021).
Ioannou, D. I. et al. Selective reduction of nitroarenes to arylamines by the cooperative action of methylhydrazine and a tris(N-heterocyclic thioamidate) cobalt(III) complex. J. Org. Chem. 86, 2895–2906 (2021).
Acknowledgements
This work was supported by the research facilities of Ilam University and Bu-Ali Sina University.
Author information
Authors and Affiliations
Contributions
Z.M.: Methodology, Validation, Methodology, Investigation, Writing-original draft. A.G.-C.: Funding acquisition, Supervision, Conceptualization, Resources, Writing-review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moradi, Z., Ghorbani-Choghamarani, A. Fe3O4@SiO2@KIT-6@2-ATP@CuI as a catalyst for hydration of benzonitriles and reduction of nitroarenes. Sci Rep 13, 7645 (2023). https://doi.org/10.1038/s41598-023-34409-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-34409-z
This article is cited by
-
Efficient nitroarene reduction: innovative nickel immobilization on ZnFe₂O₄@EDTA nanocomposites
Monatshefte für Chemie - Chemical Monthly (2026)
-
Facile synthesis of PVA-supported ZnS nano-cottons as recyclable catalyst for the efficient reduction of p-nitrophenol and photodegradation of solochrome dark blue under the sunlight irradiation
Discover Catalysis (2025)