Abstract
A Hepatitis C (HCV) e-Consult Direct-To-Treatment (DTT) program managed by midlevel providers was developed at the Veteran Affairs Greater Los Angeles Healthcare System (VAGLAHS) which provided remote referral and, in some, remote management of HCV. DTT patients were more likely to be initiated on HCV treatment compared to standard of care (SOC), lending support for similar programs of remote engagement in HCV care.
Similar content being viewed by others
Introduction
An estimated 2.4 million people in the United States have chronic hepatitis C virus (HCV) infection1, with Veterans having a higher rate (5.4%) than the general U.S. population (1.8%)2. Treatment is recommended for all persons living with HCV, with life expectancy > 12 months3. The Department of Veteran Affairs (VA) has been a leader in hepatitis treatment, particularly with the advent of Direct-Acting Antivirals (DAAs) in 20144. The pillars of the VA’s elimination initiative are novel HCV screening and treatment programs, including mid-level provider care management and telehealth4,5,6. Furthermore, the COVID-19 pandemic has impacted HCV screening and treatment, emphasizing the increased need for telehealth interventions for HCV and generally7. Hepatitis C virus antibody testing decreased by 59% in April 2020 and hepatitis C virus RNA-positive results fell by 62% in March 2020, with continued reductions by 39% in July 2020, while hepatitis C virus treatment prescriptions decreased by 43% in May, 37% in June, and 38% in July compared to corresponding months in 2018 and 2019, highlighting the importance of novel methods of screening, linkage to care, and treatment8.
Indeed, one of the barriers to HCV evaluation and treatment may be timely access to HCV providers. Studies have outlined difficulty with specialist referrals and long specialist wait times as barriers to HCV treatment9,10,11. Quickly identifying the appropriate HCV provider and limiting confusion for the person living with HCV is an important component of effective treatment9. At VAGLAHS, a hepatitis C e-Consult Direct-To-Treatment (DTT) program was established from March 2016 to November 2017 to expedite treatment evaluation. In this pilot program, primary care providers were offered an option to select an “e-consult” for HCV management. A midlevel practitioner reviewed the electronic medical record to determine treatment eligibility within 3 business days and used an algorithm to determine HCV treatment eligibility. If deemed eligible, the midlevel practitioner contacted the Veteran and directly initiated HCV therapy. If deemed ineligible, the Veteran was directed to evaluation by the infectious diseases/hepatology service. Standard of care was defined as treatment evaluation through the usual referral pathway, which required an initial face to face visit in the infectious diseases/hepatology clinic. This program operated from March 2016–November 2017.
As we evaluate ways to improve HCV care delivery in the DAA and pandemic era, we sought to characterize this historical pilot program of Veterans that were referred to the HCV e-consult DTT program, and to compare the health care utilization and clinical outcomes of patients who initiated HCV treatment via DTT versus referral via SOC practices.
Methods
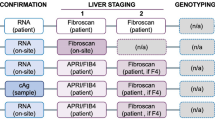
This retrospective chart review study was conducted at the VAGLAHS to evaluate the HCV e-consult mechanism (Fig. 1). Adults 18 years or older who had an HCV e-consult submitted between March 1, 2016 and November 13, 2017 were included.
HCV e-consult process.
Data was collected via chart review from VAGLAHS using the Computerized Patient Record System. Demographic information (age, gender, race, ethnicity, social history and medical history) was collected at the time of HCV e-consult evaluation. Treatment eligibility criteria are highlighted in Supplemental Table 1. Briefly, patients without clinical cirrhosis, fibrosis, substance abuse disorder, alcohol use disorder, malignancy, mental health disorders, chronic obstructive pulmonary disease, or heart disease were eligible for inclusion. Endpoints included HCV treatment initiation (yes/no), HCV treatment completion (yes/no), SVR12 (defined by ≥ 10 weeks post-treatment), and the number of HCV treatment-related clinic visits. Outcomes were compared between Veterans who initiated treatment via DTT and patients who were ineligible for DTT and referred to standard of care. Descriptive statistics, t-tests, and chi-squared tests were used.
Achievement of SVR12 was based upon patients who completed SVR12 labs. Of those who completed treatment but did not reach SVR12, one patient had virologic failure, while 24 missed their lab collection. Adverse events were reported common events, evaluated based on the patients’ HCV clinical treatment notes. These included headache, nausea, fatigue, diarrhea, weight loss, insomnia, suppressed appetite, and itchy redness and burning on lower extremities. Only one person discontinued HCV treatment due to adverse events.
Ethics approval and consent to participate
The study was approved by the local institutional review board (IRB) at VAGLAHS, and all study activities were carried out in accordance with the relevant guidelines. Informed consent was waived by the VAGLAHS IRB due to the retrospective nature of this investigation.
Results
One hundred and ninety-four (194) HCV e-consults were completed during the study timeframe. Seven patients with undetectable viral loads and 2 duplicate e-consults were excluded. Of 186 e-consults, 85 patients (46%) were eligible to initiate HCV treatment via DTT. Veterans were predominantly male (98%) with an average age of 61 ± 8 years and 63 ± 9 years for eligible and non-eligible patients, respectively (p = 0.066) (Table 1). Compared to those not eligible for DTT, DTT eligible patients had lower rates of recent drug use (9% vs 19%) and history of drug use (26% vs 31%) (p = 0.090). Similarly, eligible patients had lower rates of recent polysubstance use (1% vs. 8%, p = 0.085). Baseline comorbidities were similar among both groups, except for malignancy, which was lower in DTT eligible patients (4% vs 12%, p = 0.037). HCV genotype was similarly distributed between the two groups, apart from genotype 3 which was less frequent in DTT eligible patients (4% vs. 13%). Eligible patients also had lower FIB-4 (1.44 ± 0.42 vs. 2.62 ± 1.83, p < 0.001) and APRI scores (0.41 ± 0.19 vs. 0.79 ± 0.72, p < 0.001), and none of the eligible patients had advanced fibrosis as compared to non-eligible patients (0% vs 21%, p < 0.001).
More Veterans received treatment when initiated through the DTT program as opposed to those who received SOC (71% vs 55%, p = 0.034) and the DTT group also had fewer mean face-to-face clinic visits during HCV treatment (3.0 ± 0.7 vs 4.2 ± 1.3, p < 0.001). Of patients starting treatment (DTT vs SOC), the mean time to HCV treatment initiation (90.4 ± 66.9 vs 106.4 ± 69.4 days, p = 0.210) and treatment completion (75% vs 77%, p = 0.822) were similar. Importantly SVR12 rates were similar (95% vs 94%, p = 0.939). Rates of adverse events were also comparable for DTT vs SOC (17% vs 11%, p = 0.353) (Table 1).
Discussion
This retrospective chart review evaluated a historical HCV e-consult DTT program with the aim of comparing clinical outcomes of patients who initiated HCV treatment via DTT versus SOC practices. Veterans eligible for DTT were more likely to initiate DAA therapy than those who were not. There was also a trend towards shorter time to HCV treatment initiation in patients who initiated via DTT as compared to ineligible patients who were referred to specialists. HCV treatment completion and SVR12 rates were similar for both groups, but the DTT group had fewer face-to-face clinic visits during HCV treatment.
To our knowledge, this is the first evaluation of a program which referred patients directly to a remote online evaluation system (HCV e-consult DTT program) with subsequent successful HCV treatment initiation and completion by midlevel providers, highlighting a potential model for other integrated healthcare systems. There exist other telehealth programs that have been demonstrated to be effective for HCV treatment12,13,14,15. Of particular note, at the VA, one study found that sites that offered video conferencing between primary care providers and people living with HCV as part of the HCV VA-ECHO program had higher treatment initiation rates than sites that did not offer this16. However, this program and others did not utilize the same e-consult DTT model reported here. The novelty of the e-consult DTT program lies in the potential for reduced time between HCV diagnosis and treatment. Furthermore, this DTT program has implications for pandemic-era losses in screening, linkage to care, and treatment initiation. The COVID-19 pandemic saw reduced HCV screening and treatment across all populations; telehealth interventions can close this gap by offering safer, more convenient healthcare alternatives for people affected by the pandemic.
We report SVR12 rates of 95% with this program. Other programs that have implemented remote linkage to care and/or treatment have found similar success, with SVR rates between 93.3–98.5%17,18,19. However, these programs differed from the model described here in a few important ways. Linkage to care and treatment were either not managed or not exclusively managed by midlevel practitioners17,18,19, they did not take place in integrated healthcare systems18,19, and some exclusively evaluated people who inject drugs (PWID)18,19.
This cohort was evaluated 7 years ago; as such, there have been advances in telehealth outreach and technology since. Moving forward, these data are important due to their demonstration of the effectiveness of telehealth and DTT interventions in a time when these programs were less prevalent. The success of the program in linking Veterans to care and achieving SVR underlines the utility of telehealth programs with midlevel providers, particularly as specialists are often less accessible. Additionally, it is important to note that, now that telehealth programs are more established, those with substance abuse and alcohol abuse disorders are more likely to be given the opportunity to be treated via electronic means.
Limitations include a limited sample size and short study timeframe. Second, this study was completed at a single healthcare facility with Veterans; thus, results may not be applicable to other populations. The study also did not evaluate patient acceptance of the program or include any quality of life indices. Those who received treatment via SOC were likely a disparate population compared to those referred through DTT. With regards to the higher proportion of DTT patients initiating therapy, it is possible that those referred to standard of care had more comorbidities, requiring more diagnostic evaluation that delayed care. Future research could evaluate clinical outcomes in patients with similar baseline characteristics who initiated treatment via DTT versus SOC.
As compared to SOC, the HCV e-consult DTT program managed by midlevel practitioners required fewer visits for patients to achieve similar SVR12 rates, results which may have important cost-saving and post-pandemic-era safety ramifications. Overall, our findings lend support for this e-consult program in other integrated healthcare systems.
Availability of data and materials
The data analyzed during this study are not available due to the sensitive nature of health information.
References
Hofmeister, M. G. et al. Estimating prevalence of hepatitis C virus infection in the United States, 2013–2016. Hepatology (Baltimore, MD) 69, 1020–1031. https://doi.org/10.1002/hep.30297 (2019).
Dominitz, J. A. et al. Elevated prevalence of hepatitis C infection in users of United States veterans medical centers. Hepatology (Baltimore, MD) 41, 88–96. https://doi.org/10.1002/hep.20502 (2005).
Hepatitis, C. guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology (Baltimore, MD) 62, 932–954. https://doi.org/10.1002/hep.27950 (2015).
Gonzalez, R. et al. HCV elimination in the US Department of Veterans Affairs. Clin. Liver Dis. 18, 1–6. https://doi.org/10.1002/cld.1150 (2021).
Yakovchenko, V. et al. Mapping the road to elimination: A 5-year evaluation of implementation strategies associated with hepatitis C treatment in the veterans health administration. BMC Health Serv. Res. 21, 1348. https://doi.org/10.1186/s12913-021-07312-4 (2021).
Belperio, P. S., Chartier, M., Ross, D. B., Alaigh, P. & Shulkin, D. Curing hepatitis C virus infection: Best practices from the U.S. Department of Veterans Affairs. Ann. Intern. Med. 167, 499–504. https://doi.org/10.7326/m17-1073 (2017).
Hoyt, K. & Reynolds, A. The patient perspective on telemedicine. Clin. Liver Dis. 19, 167–170. https://doi.org/10.1002/cld.1188 (2022).
Kaufman, H. W. et al. Decreases in hepatitis C testing and treatment during the COVID-19 pandemic. Am. J. Prev. Med. 61, 369–376. https://doi.org/10.1016/j.amepre.2021.03.011 (2021).
Amoako, A., Ortiz-Paredes, D., Engler, K., Lebouché, B. & Klein, M. B. Patient and provider perceived barriers and facilitators to direct acting antiviral hepatitis C treatment among priority populations in high income countries: A knowledge synthesis. Int. J. Drug Policy 96, 103247. https://doi.org/10.1016/j.drugpo.2021.103247 (2021).
McGowan, C. E. & Fried, M. W. Barriers to hepatitis C treatment. Liver Int. 32(Suppl 1), 151–156. https://doi.org/10.1111/j.1478-3231.2011.02706.x (2012).
Morrill, J. A., Shrestha, M. & Grant, R. W. Barriers to the treatment of hepatitis C. Patient, provider, and system factors. J. Gen. Intern. Med. 20, 754–758. https://doi.org/10.1111/j.1525-1497.2005.0161.x (2005).
Hartl, L. et al. HCV hotline facilitates hepatitis C elimination during the COVID-19 pandemic. J. Viral Hepatitis 29, 1062–1072. https://doi.org/10.1111/jvh.13746 (2022).
Case, L., Wright, J. & Ryan, Y. Comparison of hepatitis C treatment outcomes between telehepatology and specialty care clinics in the era of direct-acting antivirals. J. Telemed. Telecare 27, 463–468. https://doi.org/10.1177/1357633x19885750 (2021).
Stephens, D. et al. An evaluation of hepatitis C virus telehealth services serving tribal communities: Patterns of usage, evolving needs, and barriers. J. Public Health Manag. Pract. JPHMP https://doi.org/10.1097/phh.0000000000001061 (2019).
Syed, T. A., Cherian, R., Lewis, S. & Sterling, R. K. Telemedicine HCV treatment in department of corrections results in high SVR in era of direct-acting antivirals. J. Viral Hepatitis 28, 209–212. https://doi.org/10.1111/jvh.13392 (2021).
Beste, L. A. et al. Telemedicine specialty support promotes hepatitis C treatment by primary care providers in the Department of Veterans Affairs. Am. J. Med. 130, 432–8.e3. https://doi.org/10.1016/j.amjmed.2016.11.019 (2017).
Cooper, C. L. et al. Direct-acting antiviral therapy outcomes in Canadian chronic hepatitis C telemedicine patients. Ann. Hepatol. 16, 874–880. https://doi.org/10.5604/01.3001.0010.5277 (2017).
Rosato, V. et al. Telemedicine improves HCV elimination among Italian People who use drugs: An innovative therapeutic model to increase the adherence to treatment into addiction care centers evaluated before and during the COVID-19 pandemic. Biology https://doi.org/10.3390/biology11060800 (2022).
Talal, A. H. et al. Integrated, co-located, telemedicine-based treatment approaches for hepatitis C virus management in opioid use disorder patients on methadone. Clin. Infect. Dis. 69, 323–331. https://doi.org/10.1093/cid/ciy899 (2018).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
D.B. and C.T. developed the concept for this manuscript. C.T. performed chart review and data analyses. N.M. and C.T. wrote the manuscript. All authors reviewed and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohtashemi, N.Z., Teng, C.Y., Benhammou, J. et al. Evaluation of and implications for a novel hepatitis C e-consult direct-to-treatment pilot program. Sci Rep 13, 17241 (2023). https://doi.org/10.1038/s41598-023-43052-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-43052-7