Abstract
The occurrence of cisplatin (CDDP)-induced nephrotoxicity (CIN) has decreased with advancements in supportive care. In contrast, we reported that baseline diabetes mellitus (DM) complications significantly worsen CIN. This study aimed to determine further risk factors associated with CIN development in DM patients. Patients with thoracic cancer requiring DM pharmacotherapy, who received CDDP (≥ 60 mg/m2)-containing regimens using the short hydration method (n = 140), were enrolled in this retrospective multicenter observational study. The primary endpoint of the present study was the elucidation of risk factors (patient factors, DM medication influence, and treatment-related factors) associated with CIN development in patients with DM. Cisplatin-induced nephrotoxicity occurred in 22.1% of patients with DM. The median worst variation of serum creatinine levels and creatinine clearance (worst level − baseline level) was 0.16 mg/dL (range: − 0.12–1.41 mg/dL) and − 15.9 mL/min (− 85.5–24.3 mL/min), respectively. Multivariate logistic regression analyses identified female sex as the singular risk factor for CIN development in the DM population (adjusted odds ratio; 2.87, 95% confidence interval; 1.08–7.67, P = 0.04). Diabetes mellitus medication and treatment-related factors did not affect CIN development. In conclusion, our study revealed that female sex is significantly associated with CIN development in patients with DM and thoracic cancer.
Similar content being viewed by others
Introduction
Chemotherapy is the main treatment for advanced cancer, and the management of chemotherapy-induced adverse effects is one of the most important factors in providing more effective treatment and maintaining the quality of life of patients1.
Cisplatin (CDDP) is a key chemotherapeutic agent used to treat lung, head and neck, ovarian, esophageal, and urological malignancies2. Cisplatin-induced nephrotoxicity (CIN) is known to be its dose-limiting toxicity2, and reportedly occurs in 5–40% of the patients2,3,4. It is usually reversible, but becomes unreversible in cases of severe symptoms, and symptomatic treatment is limited2,3,4. Oxidative stress, DNA damage, mitochondrial dysfunction, inhibition of protein synthesis, involvement of the tumor necrosis factor family, and decreased autophagy are all associated with CIN development3,4,5,6,7,8.
Magnesium supplementation, quality antiemetic therapy, and appropriate diuretic and hydration administration are the most important CIN prophylaxes and contribute to the reduction of CIN development to 0–10%3,4. Many reports evaluated CIN risk factors; however, most of the reports were not performed following the most recent CDDP administration methods with sufficient CIN prophylaxis described above4. It has been reported that the co-administration of non-steroidal anti-inflammatory drugs (NSAIDs) and baseline comorbidities of diabetes mellitus (DM) are risk factors for CIN development in the short hydration method, which is the most advanced method for CDDP administration3. Non-steroidal anti-inflammatory drugs have already been reported to worsen CIN9,10,11. In addition, it has been shown that baseline DM complications significantly increase the CIN development rate in the method4. The prevalence of DM and prediabetes is growing12. Therefore, opportunities for administering CDDP to patients with DM are expected to increase.
This study aimed to determine the risk factors associated with CIN development in a short hydration method in patients with DM for early treatment.
Ethics approval and consent to participate
All procedures performed in this study were conducted in accordance with the ethical standards of the institutional and national research committee and the 1964 Helsinki Declaration and its later amendments, or comparable ethical standards. This study was approved by the institutional review board of each participating institution (in case of Hokkaido University Hospital, approval number: 020–0366). The requirement for formal consent for this study was waived by the Ethical Review Board for Life Science and Medical Research of Hokkaido University Hospital and the Institutional Review Boards of KKR Sapporo Medical Center, Sapporo Medical University, Teine Keijinkai Hospital, and NTT Medical Center Sapporo.
Results
Patient characteristics
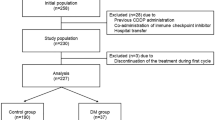
In total, 140 patients with thoracic cancer who underwent DM pharmacotherapy and received CDDP-containing treatment (≥ 60 mg/m2) were enrolled based on the eligibility criteria of this retrospective multicenter observational study (Fig. 1). Baseline patient characteristics are shown in Table 1. Approximately 84% of the participants were males, and the median age was 67 years (range 46–76 years). The percentage of patients with advanced cancer was 35.7% and 11.4% of those had undergone prior treatment. The proportion of patients who received chemoradiotherapy was 24.3%. Median baseline serum albumin level was 4.0 g/dL (2.2–5.0 g/dL), and that of HbA1c l was 7.0% (5.4–11.1%). Median baseline creatinine clearance was 93.7 mL/min (48.8–176.6 mL/min), and 2.9% of participants had less than 60 mL/min. The median CDDP dose in the first cycle was 80 mg/m2, and 2.9% of the participants received a dose reduction from treatment initiation. Patients who received co-administration of proton pump inhibitors (PPIs) or NSAIDs during treatment accounted for 36.4% and 20.7%, respectively. Dipeptidyl peptidase-4 (DPP-4) inhibitors or glucagon-like peptide-1 (GLP-1) analogs were administered to 75.7% of patients, followed by metformin (46.4%), sulfonylurea agents or glinides (32.9%), insulin (25.0%), sodium-glucose cotransporter 2 inhibitors (13.6%), and α-glucosidase inhibitors (10.0%). The total number of treatment cycles was 4 for 49.3%, one or two for 18.6%, 3 for 9.3%, and 5 or 6 for 2.1% each. In addition, 30% of the patients received a dose reduction during treatment because of gastrointestinal symptoms, neutropenia, febrile neutropenia, and fatigue; however, no patient received a reduction because of CIN.
Consort diagram of this study. CDDP cisplatin, ICI immune-checkpoint inhibitors, Mg magnesium.
Development of CIN and variation of serum creatinine levels and creatinine clearance
CIN was confirmed in 22.1% of patients. Median worst variation of serum creatinine (SCr) levels and creatinine clearance (worst level − baseline level) was 0.16 mg/dL (range: − 0.12–1.41 mg/dL) and − 15.9 mL/min (− 85.5–24.3 mL/min), respectively (Table 2). Most cases of CIN (80.6%) were confirmed during the early course of treatment, and no patient newly experienced CIN after the fifth cycle. The worst creatinine clearance was also confirmed during the early course of treatment. Moreover, CIN at the final evaluation (approximately 3 weeks after the last CDDP administration), which can be associated with unreversible CIN, was confirmed in 10.0% of the participants.
Risk factor analysis for CIN development
Univariate and multivariate logistic regression analyses were performed to identify the independent risk factors for CIN development in patients with DM (Table 3). Multivariate analysis identified that female sex was the singular independent patient’s risk factor (adjusted odds ratio; 2.87, 95% confidence interval; 1.08–7.67, P = 0.04). However, DM medication or CDDP-including treatment-related factors did not affect CIN development. Notably, the co-administration of NSAIDs, suggested factors in all patient populations in a meta-analysis11, was not suggested as a factor. The association between chemotherapy-induced gastrointestinal symptoms and CIN development was also assessed, as oral hydration plays an important role in the short hydration method. Patients exhibiting all-grade nausea, vomiting, and anorexia accounted for 58.6, 5.0, and 65.0% of the patients, respectively. Concerning problematic grade ≥ 2 symptoms that can affect oral hydration, the values were 20.7, 1.4, and 27.9%, respectively. However, its development was not associated with the development of CIN.
Discussion
We previously reported that patients with DM develop CIN at a significantly higher rate in a short hydration method4. Autophagy reportedly protects against CIN8,13,14, and its induction at the proximal tubule, where CIN mostly appears, is significantly suppressed in type 2 DM, which all participants met in previous and present studies4,15. Thus, we consider that the main mechanism of CIN degradation in type 2 DM is the reduction in renal autophagy. Owing to advances in supportive care, the development rate of CIN has significantly decreased; however, approximately 30% of patients with DM developed symptoms in our previous study4. Consequently, we aimed to identify further risk factors for the early detection and treatment of CIN in patients with DM.
The development rate of CIN was 22.1%, and female sex was identified as the single most significant risk factor for CIN development in patients with DM. Previous studies have reported that women are at a higher risk of developing CIN9,16,17,18. One possible hypothesis is sex differences in autophagy19,20. Tao et al. reported that lower visceral adiposity in female mice arises from more active estradiol-estrogen receptor α signaling, which regulates autophagy and adipogenesis20. However, sex was not associated with CIN development in the entire patient population in our previous studies3,4. In previous reports reporting the results, a short hydration method was not used, and the evaluated CDDP dosage was different from ours3,4,9,16,17,18, which might have induced the incongruence in the results. In addition, as autophagic activity at the proximal tubule is considered to be suppressed in patients with type 2 DM, further suppression in females may have affected the results. However, as the detailed mechanism for this finding remains elusive, further studies are required.
In contrast, the co-administration of NSAIDs was not associated with CIN development in patients with DM. In our previous studies evaluating the risk factors or meta-analyses of CIN in an all-patient population, NSAIDs co-administration was significantly associated with its development3,11. The main mechanism of NSAID-induced nephrotoxicity is a decrease in prostaglandin levels owing to cyclooxygenase-1 inhibition21. In contrast, prostaglandin E2 (PGE2) signaling may contribute to the progression and development of type 2 DM with insulin resistance, elevated fasting glucose, and glucose intolerance22. Fenske et al. reported that plasma PGE2 metabolite levels are twice as high in patients with type 2 DM than in controls22. In addition, we have reported that most NSAIDs do not affect renal autophagy in vitro14. Thus, we believe that the higher baseline PGE2 levels in patients with DM compensate for the renal influence of NSAIDs. However, it is necessary to compare the effects of concomitant NSAIDs use on CIN development between patients with and without DM complications, including PGE2 levels, to understand this phenomenon fully.
Autophagy is nephroprotective against CIN8,13,14, and autophagic activity is induced in type 1 DM and suppressed in type 2 DM15. Furthermore, renal damage caused by CDDP was reportedly reduced in streptozotocin-administered mice, which are recognized as models of type 1 DM23,24. The effect of autophagy can differ between DM types; therefore, it is necessary to evaluate the impact of the DM type on CIN clinically, focusing on autophagy.
Managing chemotherapy-induced nausea and vomiting (CINV) is important for sufficient oral hydration. Thus, adequate prevention and appropriate management of breakthrough gastrointestinal symptoms in accordance with the current guidelines are essential. In this study, adequate CINV management, considering CIN, was performed, resulting in no association between adverse gastrointestinal effects and CIN. Consequently, we should try to manage CINV with adequate understanding from the medical team.
This study had some limitations. First, this study was retrospectively conducted in a relatively small population, as patients with DM were limited to 14% of the total population. Second, as all patients in the present study had type 2 DM and renal pathology is different between type 1 and 2 DM15, the present results may differ from those of patients with type 1 DM. Third, we excluded patients receiving immune-checkpoint inhibitors (ICIs) as we tried to evaluate the direct association between the evaluated factors and CIN development. However, because combination treatment with ICIs is one of the main treatment strategies for several types of cancer, an assessment of this latter population is needed. Finally, we did not evaluate the genetic backgrounds or autophagy levels of the patients. Organic cation transporter 2 (OCT2), which transports CDDP to the proximal tubule, has single-nucleotide polymorphisms (SNPs)25. In addition, the 808G>T SNP in OCT2 was shown to ameliorate CIN without altering its disposition26. Therefore, the patients’ genetic backgrounds and/or other unknown factors may have affected the results. Furthermore, as we consider that autophagy plays an important role in the development of CIN in patients with DM, its assessment is required. Considering these limitations, our preliminary results should be validated by future studies.
In conclusion, our study revealed that female sex is significantly associated with CIN development in a short hydration method in patients with DM and thoracic cancer. Management of high-risk populations is crucial; therefore, further evaluation, particularly regarding prophylaxis, is necessary.
Methods
Patients
Patients with thoracic cancer who required DM pharmacotherapy and received CDDP-containing treatment (≥ 60 mg/m2) between December 2012 and September 2022 were enrolled. CDDP-including regimens were CDDP (75 mg/m2, day 1) + pemetrexed (500 mg/m2, day 1) ± bevacizumab (15 mg/kg, day 1), CDDP (80 mg/m2, day 1) + vinorelbine (20–25 mg/m2, days 1, 8) ± radiation, CDDP (80 mg/m2, day 1) + etoposide (100 mg/m2, days 1–3) ± radiation, CDDP (60 mg/m2, day 1) + irinotecan (60 mg/m2, days 1, 8, 15), and CDDP (80 mg/m2, day 1) + gemcitabine (1000 mg/m2, days 1 and 8).
All patients met the following baseline criteria: (1) age ≥ 20 years; (2) CDDP administration by a short hydration method; (3) 0 to 2 Eastern Cooperative Oncology Group performance status (ECOG-PS); (4) detailed patient information available from medical records; and (5) sufficient renal or liver function for treatment induction. Patients who received ICI co-administration, were transferred to the hospital during chemotherapy, discontinued the treatment during the first cycle, and whose dosage of magnesium premedication was outside 8 mEq were excluded. The primary endpoint of the present study was the elucidation of risk factors (patient factors, DM medication influence, and treatment-related factors) associated with CIN development in patients with DM. Based on our previous report that found CIN in 27% patients with DM4, we decided to include approximately 3–4 covariates in the multivariate analysis, resulting in the necessity of enrolling approximately 150 participants.
This study was approved by the Ethical Review Board for Life Science and Medical Research of Hokkaido University Hospital (Approval Number: 020-0515) and the Institutional Review Boards of KKR Sapporo Medical Center, Sapporo Medical University, Teine Keijinkai Hospital, and NTT Medical Center Sapporo, and was conducted in accordance with the Declaration of Helsinki and the STROBE statement. The requirement for formal consent for this study was waived by the Ethical Review Board for Life Science and Medical Research of Hokkaido University Hospital and the Institutional Review Boards of KKR Sapporo Medical Center, Sapporo Medical University, Teine Keijinkai Hospital, and NTT Medical Center Sapporo.
Treatment methods
The short hydration method has been described in detail in previous reports3,27,28. All regimens basically included antiemetic therapy according to the current national guidelines29; palonosetron 0.75 mg on day 1, oral aprepitant 125 mg on day 1 and 80 mg on days 2 and 3 or intravenous fosaprepitant 150 mg on day 1, and dexamethasone 9.9 mg infusion on day 1 and 8 mg orally on days 2–4. These prophylactic medications and other additional antiemetic drugs were administered at the physicians’ discretion.
Evaluation of CIN and other adverse effects
Toxicities in all subsequent treatment cycles were graded according to the Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. Renal function was evaluated based on SCr elevation measured using an enzymatic method. In this study, CIN was defined as a SCr elevation of ≥ 1.5 times or > 0.3 mg/dL from the baseline levels as referenced in previous reports4,9,10,30.
Statistical analysis
Univariate and multivariate logistic regression analyses were performed using the following possible covariates: sex, age, ECOG-PS, staging, body surface area (BSA), anemia, hypoalbuminemia, liver dysfunction (grade 1 or higher aspartate aminotransferase, alanine aminotransferase, total bilirubin elevation), renal dysfunction (creatinine clearance calculated by Cockroft–Gault formula of < 60 mL/min), concomitant hypertension, and co-administration of PPIs and NSAIDs as baseline patient factors; administration of DPP-4 inhibitors or GLP-1 analogs, metformin, sulfonylurea agents or glinides, insulin, sodium-glucose cotransporter 2 inhibitors, and α-glucosidase inhibitors as DM medication influence; and CDDP dosage, treatment courses, dose reduction during the treatment, radiation combination, and development of grade ≥ 2 nausea, vomiting, and anorexia for treatment-related factors according to previous reports3,4,9,11,16,17,18,30,31,32,33,34,35,36,37,38. Previously reported variables that demonstrated potential associations with CIN development in univariate logistic regression analysis (P < 0.20) were considered when building the multivariable model.
All analyses were performed using the JMP statistical software version 16.1 (SAS Institute Japan, Tokyo, Japan). P-values less than 0.05 were considered statistically significant.
Data availability
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
References
Jansman, F. G., Sleijfer, D. T., de Graaf, J. C., Coenen, J. L. & Brouwers, J. R. Management of chemotherapy-induced adverse effects in the treatment of colorectal cancer. Drug Saf. 24(5), 353–367. https://doi.org/10.2165/00002018-200124050-00002 (2001).
Go, R. S. & Adjei, A. A. Review of the comparative pharmacology and clinical activity of cisplatin and carboplatin. J. Clin. Oncol. 17(1), 409–422. https://doi.org/10.1200/JCO.1999.17.1.409 (1999).
Saito, Y. et al. Suitability of oral rehydration solution (ORS) for use in the cisplatin short hydration method. Anticancer Res. 42(6), 3185–3193. https://doi.org/10.21873/anticanres.15808 (2022).
Saito, Y. et al. Diabetes mellitus degenerates cisplatin-induced nephrotoxicity in short hydration method: A propensity score-matching analysis. Sci. Rep. 12(1), 21819. https://doi.org/10.1038/s41598-022-26454-x (2022).
Tsuruya, K. et al. Direct involvement of the receptor-mediated apoptotic pathways in cisplatin-induced renal tubular cell death. Kidney Int. 63(1), 72–82. https://doi.org/10.1046/j.1523-1755.2003.00709.x (2003).
Brady, H. R. et al. Mitochondrial injury: An early event in cisplatin toxicity to renal proximal tubules. Am. J. Physiol. 258(5 Pt 2), F1181-1187. https://doi.org/10.1152/ajprenal.1990.258.5.F1181 (1990).
Park, M. S., De Leon, M. & Devarajan, P. Cisplatin induces apoptosis in LLC-PK1 cells via activation of mitochondrial pathways. J. Am. Soc. Nephrol. 13(4), 858–865. https://doi.org/10.1681/ASN.V134858 (2002).
Takahashi, A. et al. Autophagy guards against cisplatin-induced acute kidney injury. Am. J. Pathol. 180(2), 517–525. https://doi.org/10.1016/j.ajpath.2011.11.001 (2012).
Yoshida, T. et al. Protective effect of magnesium preloading on cisplatin-induced nephrotoxicity: A retrospective study. Jpn. J. Clin. Oncol. 44(4), 346–354. https://doi.org/10.1093/jjco/hyu004 (2014).
Kidera, Y. et al. Risk factors for cisplatin-induced nephrotoxicity and potential of magnesium supplementation for renal protection. PLoS One 9(7), e101902. https://doi.org/10.1371/journal.pone.0101902 (2014).
Okamoto, K. et al. Non-steroidal anti-inflammatory drugs are a risk factor for cisplatin-induced nephrotoxicity: A meta-analysis of retrospective studies. Anticancer Res. 40(3), 1747–1751. https://doi.org/10.21873/anticanres.14128 (2020).
Saeedi, P. et al. IDF Diabetes Atlas Committee Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 157, 107843. https://doi.org/10.1016/j.diabres.2019.107843 (2019).
Kaushal, G. P. & Shah, S. V. Autophagy in acute kidney injury. Kidney Int. 89(4), 779–791. https://doi.org/10.1016/j.kint.2015.11.021 (2016).
Okamoto, K. et al. Comparison of the nephroprotective effects of non-steroidal anti-inflammatory drugs on cisplatin-induced nephrotoxicity in vitro and in vivo. Eur. J. Pharmacol. 884, 173339. https://doi.org/10.1016/j.ejphar.2020.173339 (2020).
Sakai, S. et al. Proximal tubule autophagy differs in type 1 and 2 diabetes. J. Am. Soc. Nephrol. 30(6), 929–945. https://doi.org/10.1681/ASN.2018100983 (2019).
de Jongh, F. E. et al. Weekly high-dose cisplatin is a feasible treatment option: Analysis on prognostic factors for toxicity in 400 patients. Br. J. Cancer 88(8), 1199–1206. https://doi.org/10.1038/sj.bjc.6600884 (2003).
Sekine, I. et al. Innovator and generic cisplatin formulations: Comparison of renal toxicity. Cancer Sci. 102(1), 162–165. https://doi.org/10.1111/j.1349-7006.2010.01764.x (2011).
Miller, R. P., Tadagavadi, R. K., Ramesh, G. & Reeves, W. B. Mechanisms of Cisplatin nephrotoxicity. Toxins 2(11), 2490–2518. https://doi.org/10.3390/toxins2112490 (2010).
Shang, D., Wang, L., Klionsky, D. J., Cheng, H. & Zhou, R. Sex differences in autophagy-mediated diseases: Toward precision medicine. Autophagy 17(5), 1065–1076. https://doi.org/10.1080/15548627.2020.1752511 (2021).
Tao, Z. et al. Estradiol signaling mediates gender difference in visceral adiposity via autophagy. Cell Death Dis. 9(3), 309. https://doi.org/10.1038/s41419-018-0372-9 (2018).
Whelton, A. Nephrotoxicity of nonsteroidal anti-inflammatory drugs: Physiologic foundations and clinical implications. Am. J. Med. 106(5B), 13S-24S. https://doi.org/10.1016/s0002-9343(99)00113-8 (1999).
Fenske, R. J. et al. Plasma prostaglandin E2 metabolite levels predict type 2 diabetes status and one-year therapeutic response independent of clinical markers of inflammation. Metabolites 12(12), 1234. https://doi.org/10.3390/metabo12121234 (2022).
de Silva Faria, M. C. et al. Effect of diabetes on biodistribution, nephrotoxicity and antitumor activity of cisplatin in mice. Chem. Biol. Interact. 229, 119–131. https://doi.org/10.1016/j.cbi.2015.01.027 (2015).
Kitada, M., Ogura, Y. & Koya, D. Rodent models of diabetic nephropathy: Their utility and limitations. Int. J. Nephrol. Renovasc. Dis. 9, 279–290. https://doi.org/10.2147/IJNRD.S103784 (2016).
Fukushima-Uesaka, H. et al. Fourteen novel single nucleotide polymorphisms in the SLC22A2 gene encoding human organic cation transporter (OCT2). Drug Metab. Pharmacokinet. 19(3), 239–244. https://doi.org/10.2133/dmpk.19.239 (2004).
Iwata, K. et al. Effects of genetic variants in SLC22A2 organic cation transporter 2 and SLC47A1 multidrug and toxin extrusion 1 transporter on cisplatin-induced adverse events. Clin. Exp. Nephrol. 16(6), 843–851. https://doi.org/10.1007/s10157-012-0638-y (2012).
Horinouchi, H. et al. Short hydration in chemotherapy containing cisplatin (≥ 75 mg/m2) for patients with lung cancer: A prospective study. Jpn. J. Clin. Oncol. 43(11), 1105–1109. https://doi.org/10.1093/jjco/hyt122 (2013).
Sakaida, E. et al. Safety of a short hydration method for cisplatin administration in comparison with a conventional method: A retrospective study. Jpn. J. Clin. Oncol. 46(4), 370–377. https://doi.org/10.1093/jjco/hyv203 (2016).
Aogi, K. et al. Optimizing antiemetic treatment for chemotherapy-induced nausea and vomiting in Japan: Update summary of the 2015 Japan society of clinical oncology clinical practice guidelines for antiemesis. Int. J. Clin. Oncol. 26(1), 1–17. https://doi.org/10.1007/s10147-020-01818-3 (2021).
Sato, K. et al. Nephrotoxicity of cisplatin combination chemotherapy in thoracic malignancy patients with CKD risk factors. BMC Cancer 16, 222. https://doi.org/10.1186/s12885-016-2271-8 (2016).
Anand, A. J. & Bashey, B. Newer insights into cisplatin nephrotoxicity. Ann. Pharmacother. 27(12), 1519–1525. https://doi.org/10.1177/106002809302701219 (1993).
Madias, N. E. & Harrington, J. T. Platinum nephrotoxicity. Am. J. Med. 65(2), 307–314. https://doi.org/10.1016/0002-9343(78)90825-2 (1978).
Haas, A., Anderson, L. & Lad, T. The influence of aminoglycosides on the nephrotoxicity of cis-diamminedichloroplatinum in cancer patients. J. Infect. Dis. 147(2), 363. https://doi.org/10.1093/infdis/147.2.363 (1983).
Launay-Vacher, V., Rey, J. B., Isnard-Bagnis, C., Deray, G. & Daouphars, M. Prevention of cisplatin nephrotoxicity: State of the art and recommendations from the European society of clinical pharmacy special interest group on cancer care. Cancer Chemother. Pharmacol. 61(6), 903–909. https://doi.org/10.1007/s00280-008-0711-0 (2008).
Máthé, C. et al. Cisplatin nephrotoxicity aggravated by cardiovascular disease and diabetes in lung cancer patients. Eur. Respir. J. 37(4), 888–894. https://doi.org/10.1183/09031936.00055110 (2011).
Galfetti, E., Cerutti, A., Ghielmini, M., Zucca, E. & Wannesson, L. Risk factors for renal toxicity after inpatient cisplatin administration. BMC Pharmacol. Toxicol. 21(1), 19. https://doi.org/10.1186/s40360-020-0398-3 (2020).
Stewart, D. J. et al. Association of cisplatin nephrotoxicity with patient characteristics and cisplatin administration methods. Cancer Chemother. Pharmacol. 40(4), 293–308. https://doi.org/10.1007/s002800050661 (1997).
Miyoshi, T. et al. Preventive effect of 20 mEq and 8 mEq magnesium supplementation on cisplatin-induced nephrotoxicity: A propensity score-matched analysis. Support. Care Cancer 30(4), 3345–3351. https://doi.org/10.1007/s00520-021-06790-w (2022).
Funding
This work was funded by JSPS KAKENHI (Grant Number 22K15310).
Author information
Authors and Affiliations
Contributions
Designed the study: Y.S. and M.K. Performed the research: Y.S. Analyzed the data: Y.S. Contributed new methods or models: Y.S. Wrote the paper: Y.S. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saito, Y., Kobayashi, M., Tamaki, S. et al. Risk factor analysis for cisplatin-induced nephrotoxicity with the short hydration method in diabetic patients. Sci Rep 13, 17126 (2023). https://doi.org/10.1038/s41598-023-44477-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-44477-w