Abstract
This study examines the effect of phycoerythrin (PE) from a cyanobacterial Nostoc strain encapsulated with alginate as a potential prebiotic to produce synbiotic ice cream products with Lactobacillus casei. It was found that the addition of the encapsulated PE affected, mostly favourably, the physicochemical properties, antioxidant activity, probiotic survival, volatile compound contents, and sensory acceptability of the synbiotic ice cream samples before and after aging at the freezing periods of one day to eight weeks. Thus, it confirms the prebiotic potential of PE for synbiotic ice creams with L. casei.
Similar content being viewed by others
Introduction
Phycoerythrin (PE) is a red pigment that is sensitive to light and belongs to the phycobiliprotein family and is mostly found in cyanobacterial species. Moreover, Phycoerythrin has a high degree of sensitivity towards fluctuations in pH, salt content, temperature deviations, desiccation, and light-induced stress1,2. On an equivalent molar basis, PE absorbs lighter than allophycocyanin (APC) and phycocyanin (PC) because it contains additional phycobilin. Only phycoerythrobilin (PEB) chromophores are commonly carried by PEs from freshwater and soil-dwelling cyanobacteria, and these PEs have absorbance spectra with maximum values of approximately 565 nm3. PEs generate fluorescence between 575 and 580 nm and substantially absorb light between 480 and 570 nm. PEs are divided into three primary kinds depending on phycobilin types and their spectral properties: (1) B-phycoerythrin (B-PE) (~ max ~ 540–560 nm, shoulder ~ 495 nm), (2) R-phycoerythrin (R-PE) (~ max ~ 565, 545 and 495 nm), and (3) C-phycoerythrin (C-PE) (~ max ~ 563, 543 and ~ 492 nm). Due to its flavour features, cooling impact, and beneficial nutritional characteristics, ice cream has emerged as one of the most extensively eaten dairy products across all age groups on a global scale4. The quality of the final product is often determined by the combination of milk, emulsifiers, stabilizers, sweeteners, and flavourings in the ice cream sample. The manufacturing process also impacts on the final quality of ice cream, where aging is essential to achieving the ideal flavour of ice cream5. The inclusion of fruits, spices, and ingredients high in protein, probiotics, and prebiotics may result in an ice cream that is more nutritional and healthier as a functional food. The positive functions of milk proteins, lipids, lactose, and other ingredients further contribute to the functionality of ice cream as a probiotic carrier food.
Probiotics are living bacteria that benefit consumers when taken in sufficient quantities5. Microbes must exhibit tolerance to gastrointestinal tract conditions, the capacity to auto-aggregate, behave normally, sustain flora, and be non-pathogenic. Probiotic products must comprise at least 107 colony-forming unit CFU mL-1 of the probiotic strain6. Ice cream may be a perfect carrier for probiotic microbes because it combines milk proteins, lipids, lactose, and various components. The production of probiotic ice cream using different formulations has been the subject of a limited number of studies7,8,9,10,11. The most popular probiotic bacteria in probiotic dairy products are lactobacilli, and to a lesser extent, Bifidobacteria microbial strains6.
Prebiotics consist of indigestible carbohydrates, mostly oligosaccharides, which promote the development or activity of certain probiotic bacteria in the colon, therefore benefiting the host organism. Microalgae are well recognized as a significant reservoir of sulfated polysaccharides, with the specific composition of these polysaccharides exhibiting variation across different taxonomic groups. The primary role of these polysaccharides with relatively high molecular weight is their abundance of hydroxyl (OH) groups, which renders them hydrophilic. These entities have been seen to form intra-chain hydrogen bond networks, resulting in increased stiffness, rigidity, and suitability as thickening agents. The consistent arrangement of their structures also facilitates their engagement with external ions and inter-chain hydrogen bonding, such as the process of gelation. The food and pharmaceutical sectors often use various types of carbohydrates, such as ulvans, carrageenan, and alginates, derived from macroalgae as functional additives12. Prebiotic substances may be utilized in product compositions to stimulate the development and viability of probiotics. The research on ice cream formulations has mostly included dietary fibers and oligosaccharides such as inulin as prebiotic ingredients6,13,14. Further, algal biomass and its derivatives, such as PE, may be used to make synbiotic ice cream products because of the abundance of colours and other substances that provide stabilizing functions as a sustainable alternative to artificial food dyes4,15,16.
Alginate, a biodegradable polymer, frequently serves as an encapsulating material due to its ability to form gel beads in a solution at ambient temperature without the need for organic solvents. Since it is non-toxic and often utilized as a suitable cross-formation with polymer alginate microspheres. The encapsulation of the PE and bacteria by alginate is an innovative method to protect their integrity5,11 in harsh processing conditions9. For example, De Oliveira et al.17 encapsulated a Spirulina sp. cyanobacterial strain with maltodextrin and soy lecithin, compared to alginates in chocolate milk samples, while Tiepo et al.18. encapsulated S. platensis with maltodextrin and Arabic gum in ice cream18. Ganesan and Shanmugam (2020) pioneered the use of algal PE in ice cream production. Their study employed PE extracted from the macroalgal strain, Kappaphycus alvarezii, biomass19. They also encapsulated it with a macroalgal derivative, kappa-carrageenan, as well as guar gum to protect the integrity of the PE during the processing. The results showed that ice cream enriched with the encapsulated PE had better rheology, and the intensity of the pink colour increased during 90 days of storage. Further, this sample had better antioxidant activity, and the viability of this PE was confirmed. Further, Ganesan et al.20 reviewed the extraction, encapsulation, and application of the PE from macroalgae in foods20. The focus was on the macroalgae-based PEs and their extraction and encapsulation since it is necessary to protect the integrity of the PE in the harsh ice cream processing conditions. In addition, Garcia et al.2 used a B-PE extract from the microalgal Porphyridium cruentum strain as a natural food colorant in milk-based products2. They focused on the colour stability of the PE in the milk products.
The cyanobacterium Nostoc sp. is a photoautotrophic bacteria that is found in abundance in nature. It has been used as a dietary supplement by humans for millennia due to its well-known nutritional benefits. The food and medicinal sectors have used several algae species, including Spirulina platensis, Chlorella spp, Tetraselmis sp. and Dunaliella salina. However, there is a scarcity of studies evaluating Nostoc sp. as a prebiotic bacterium.
To our knowledge, this is the first-ever study to examine the potential of PE from a cyanobacterial Nostoc strain encapsulated with alginate to protect its integrity in harsh processing environments in ice cream with the L. casei strain.
Therefore, bacterial strains and alginate-encapsulated PEs were introduced either alone or in a mixed culture to the control sample before or after aging to create probiotic and synbiotic ice cream in a range of five ice cream samples: control sample (C), probiotic sample before aging (LBA), synbiotic sample before aging (PLBA), probiotic sample after aging (LAA), and synbiotic sample after aging (PLAA). Moreover, the physicochemical properties, antioxidant activity, probiotic survival, volatile compound (VC) contents, and sensory acceptability of the probiotic and synbiotic ice cream samples before and after aging at the freezing periods of one day to eight weeks were studied in detail.
This study investigated the development of synbiotic ice cream. We incorporated Lactobacillus casei and alginate-encapsulated phycoerythrin from Nostoc sp. ASN_M into the ice cream mix, evaluating the impact on probiotic viability during storage (eight weeks). We assessed the effects of these additions on various technological properties, including physicochemical characteristics, overrun, color, melting rate, rheology, volatile compound formation, antioxidant activity, and sensory attributes. Our findings are presented and discussed in relation to existing literature. Additionally, we propose directions for future research in this field.
Materials and methods
Cyanobacterial biomass isolation and growth
Cyanobacterial Nostoc sp. ASN_M strain was obtained from paddy fields in the Golestan region of Iran (36.54.41 N, 54.47.25 W). Soil samples were distributed in sterile Petri dishes comprising liquid BG-11 medium. To obtain a cyanobacterial monoculture, the medium was made nitrogen-free with a pH of 7.1 and incubated for two weeks at 28 ± 1 °C under constant artificial illumination of 1500–2000 lx. Selected colonies were moved to a new solid medium after 14 days of development. For bacterial-free cultures, colonies were examined for bacterial contamination on caseinate-glucose agar and dextrose-peptone broth. On various agar slants, the chosen bacterial-free colony was kept alive. After 20 days, the isolate was given a last wash in sterile deionizer water and put into 1 L of newly made liquid BG110 medium.
Chemicals and alginates
Indicators of protein molecular weight and other analytical components were purchased from Hi-Media, Merck, and Sigma, respectively. Double-distilled water was used in the production of all reagents and buffers. The alginate and the CaCl2 were obtained from a local grocery store and were safe for human consumption.
Extraction and purification of phycoerythrin
To extract phycoerythrin (PE), approximately 500 mL of a homogenized, 14-day-old log-phase culture was centrifuged at 4000 rpm after resuspending the pellet in 100 mL of 20 mM acetate buffer (pH 5.1). The cell suspension was subjected to repeated cycles of freezing (− 20 °C) and thawing at room temperature over four days, until the supernatant turned dark purple. Following centrifugation at 5,000 rpm for 10 min, the resulting crude extract was further purified according to the method described by Afreen and Fatma21. The crude extract was gradually added to a constant stream of solid ammonium sulphate until it reached 65% saturation. The solution was centrifuged at 4500×g for 10 min after standing for 12 h in a cool environment. The pellets were dialyzed overnight after being re-suspended in a tiny amount of 50 mM acetic acid-sodium acetate buffer (pH 7.1). A 0.45 μm filter removed the extract from the dialysis membrane22. The Specord 200 spectrophotometer (Analytik Jena, Germany) was used to scan the sample in the 300–750 nm wavelength range and to acquire the absorption spectra.
Absorbance at 565, 620, and 650 nm was used to determine the concentrations of PE, PC, and APC in various extracts and PBPs containing solutions following De Oliveira et al.17. The purity ratio (A562/A280) was used to evaluate PE purity at each step, and the absorbance at 562 nm was used to quantify PE concentration.
Toxicity assays of PE
Wild-type C. elegans var. Bristol-N2 was gifted from the Pasteur Institute of Tehran, Iran. Nematode growth agar was used to culture C. elegans. Eggs were deposited on agar plates containing nematode growth agar (NGA, Biomol, USA) for 48 h, and young larvae were harvested and suspended in M9 worm buffer. The toxicity test of the PE was evaluated at concentrations of 0, 0.125, 0.2, 0.5, and 1 mg/ml. The nematode without PE was used as a control. Quadruplicate tests were carried out for 24 h at 18 °C23.
Encapsulation of phycoerythrin by alginate
The alginate solution was made at a 3% (w/v) concentration. Alginate and PE were combined in a 1:1 (w/w) ratio, and the mixture was then thoroughly combined by implementing a magnetic stirrer. The homogenized suspension was loaded into the syringe, injected into a 2.5% (w/v) CaCl2 solution with a 23G needle, and agitated with a magnetic stirrer. The beads were collected and rinsed with distilled water to eliminate Cl ions. To compute the encapsulation efficacy, CaCl2 and distilled water were gathered, and the beads were refrigerated. Before and after the aging stages, 0.4 g of encapsulated PE was added to 150 g of pasteurized ice cream mixture at 37 °C. The encapsulated PE beads were not dehydrated before being added to the ice cream mix.
Probiotic strain cultivation and preparation
L. casei microbial strain was cultivated overnight at 30 °C to produce 108 CFU mL−1 of bacterial cells in MRS broth (Merck, Germany). The inoculated broth was centrifuged at a speed of 4500×g for 30 min, at 4 °C to separate the bacteria from the MRS broth. The broth was completely removed by centrifuging the bacterial pellet with peptone water (0.1%, w/v) at 4500×g for 10 min at 4 °C. This technique was carried out three times. Peptone water and the bacterial pellet devoid of broth were combined. Before and after aging, a mixture comprising 108 CFU mL−1 of bacterial cells was added (1%, v/v) to a mixture of pasteurized ice cream at 37 °C.
Production of ice cream
Figures 1 and 2 shows the production process. First, the raw milk was pre-heated to 40 °C and then pasteurizing in 75 °C for 15 min. Then, after the cooling process to about 37 °C, the incubation and heating of L. casei and PE-encapsulation was performed at 4 °C for 24 h. The ice cream ingredients, such as sugar, sahlep, and emulsifier, were annexed in accordance with the recipe given in the scheme, and sahlep was utilized as a stabilizer. The ice cream products were produced in 500 mL of ice cream mix using a Delonghi II Gelataio ICK5000 ice cream machine (China), and the method was adapted previously described by Durmaz et al.4. The ice cream production facility conducted probiotic and synbiotic ice cream trials, adding probiotic cultures to the ice cream mixture and agitating it for 30 min at room temperature before using the ice cream machine. Furthermore, samples of ice cream made with prebiotics were incubated for 24 h at 4 °C.
Production flow chart of probiotic ice cream samples. Five different ice cream samples were produced in total: C control sample, LBA probiotic sample before aging, LPBA synbiotic sample before aging, LAA probiotic sample after aging, LPAA synbiotic sample after aging.
Phycoerythrin production process and adding the probiotic L. casei strain before or after aging steps.
Determination of physicochemical properties
On the first day, ice cream samples were subjected to physicochemical analyses, including titratable acidity, pH, protein, fat, ash, and overrun, employing AOAC techniques 947.05, 981.12, 930.33, 952.06, 930.30, and 968.14, respectively24.
The melting properties of ice cream products were characterized on day one using a TA Instrument Q100 (New Castle, USA) and the DSC technique employing ice cream samples. In hermetically sealed aluminium containers, the thermal performance of 10–15 mg of ice cream samples was assessed between 30 and 20 °C25. The total sugar content was calculated by the Fehling technique following Iranian Standard 2450.
Each sample of probiotic and synbiotic ice cream was assessed for its viscosity by applying a viscometer (Brookfield RH, USA) after it had settled at 25 ± °C for 20 h. The dry matter percentage of ice cream samples was obtained utilizing the AOAC techniques )941.08( and the moisture content was measured using the AOAC techniques (at one day) and after 2, 4, 6, and 8 weeks24.
Assessment of DPPH radical scavenging activity
The method by Sagdic et al.26 was adapted to produce the extracted materials. In this study, 5 g of each sample was mixed with 25 mL of a 75% methanol solution and then stored at 25 °C for 24 h. A pure extract for spectrophotometric analysis was obtained by filtering the mixture through a membrane filter with a diameter of 0.45 μm. Then, extracts were kept at 4 °C while their total phenolic contents (TPC) and antioxidant activities were tested. The Folin–Ciocalteu technique was used with a few modifications to determine the amount of TPC in sample extracts27. Ice cream extract in 0.1 mL was combined with Folin-Ciocalteu reagent (Merck, Germany), 20% sodium carbonate (w/v), 1.5 mL, and 7.9 mL of distilled water. Two hours were spent maintaining the mélange in the dark at 25 °C. The absorbance at 750 nm was checked after incubation. Using the gallic acid (anhydrous form, Merck, Germany) curve, the findings were expressed in milligrams (gallic acid equivalents, GAE) per 100 g of ice cream samples.
All samples were subject to a TPC examination. The 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) experiment was carried out using a modified version of the technique to assess the antioxidant potential of ice cream extracts28. The free radical form of DPPH (90% purity) was obtained from Sigma-Aldrich (USA). We employed a DPPH radical scavenging assay to determine the extract concentration required for 50% inhibition (EC50 value). This assay involved plotting the percentage of DPPH inhibition against various extract concentrations. Changes in antioxidant activity were evaluated after one day, two weeks, four weeks, six weeks, and eight weeks of frozen storage at – 20 °C.
Assessment of the probiotic survival in ice creams
After one day, two weeks, four weeks, six weeks, and eight weeks of freezing at − 20 °C viable bacterial counts were determined for five distinct ice cream samples of C, LBA, LPBA, LAA, and LPAA. L. casei was enumerated using MRS agar (Merck, Germany) supplemented with 0.05% vancomycin (Sigma-Aldrich, United States). At 30 °C plates were incubated for two days in anaerobic conditions6. To compute colony counts, log CFU g−1 was converted.
Consumer sensory evaluation
Following Lawless and Heymann29, all ice cream samples were subjected to sensory assessment after a week of storage. Samples of ice cream were examined by employing a nine-point hedonic scale, with values varying from 1 (extremely dislike) to 5 (extremely like). Eleven panellists with limited to no prior knowledge had attended training sessions at Selcuk University's Food Engineering Department, where specific feature explanations were presented and sensory acceptability testing was performed.
Ice cream product volatile compound (VC) analysis
The volatile compounds (VCs) present in the control (C) and synbiotic LPBA ice cream samples were profiled after one week of freezing using gas chromatography-mass spectrometry (GC–MS). This technique separates and identifies the individual VCs within a sample. An Agilent 7000 Quadrupole GC–MS system with electron impact ionization was employed for analysis. By comparing the obtained mass spectra to databases like the NIST library (containing over 62,000 patterns) and the Fiehn Mass Spectra Libraries, the identities, molecular weights, and structures of the VCs were determined.
Statistical analysis of data
Using Excel and SPSS software (version 24), statistical analysis of the data gathered from every experiment was conducted. All data were generated by experimenting three times. Significant discrepancies between the components were discovered using a one-way analysis of variance with a 95% confidence interval (< 0.05). Subsequently, the Tukey test was used to compare the means, and then Excel was used to present the results of our analyses graphically.
Statement
All procedures were carried out following applicable rules and guidelines. The authors did not conduct human experiments or use human tissue samples.
Results
Extraction, purification, and characterization of phycoerythrin
The PE content and purity of the ice cream samples were examined at each stage of purification, as indicated in Table 1. The PE purity ratio rose from 0.797 to 3.20, while the PE content increased from 0.108 to 0.193 μg/mL during the purification processes. The reddish-pink-coloured protein precipitated in the buffer displayed absorption maxima at 562 nm with a shoulder peak at 617, a characteristic spectrum for PEB containing PE (Fig. 3).
Spectra of PE extracted and purified from Nostoc sp. ASN_M, spectra show absorption maxima at 562 nm.
Toxicity of PE against C. elegans
C. elegans survival rates were not affected by PE, with 100% to 96.1% survival at all concentrations used (Table 2).
Physicochemical qualities of ice cream samples
Table 3 displays the physicochemical characteristics of five ice cream samples. This table shows that protein content (%), fat content (%), ash (%), overrun (%), total sugar (%), melting (g/min), and viscosity (cP) did not differ significantly between samples (P > 0.05).
However, the titratable acidity and pH values of the C sample were substantially distinct (P < 0.05) from those of the other samples because of the fermentation method.
The C and synbiotic LPBA samples had the highest and lowest pH values, respectively, while the pH values of the other samples varied from 6.66 ± 0.00 to 6.59 ± 0.01 (Table 3).
Further, the overall solid content increased slightly in the probiotic and synbiotic ice cream samples for all the freezing periods, from one day to eight weeks (Table 4). However, the moisture contents decreased in the synbiotic ice cream samples throughout all the freezing periods (Table 5).
Evaluation of changes in antioxidant activity
Table 5 illustrates the EC50 values for the DPPH radical-scavenging abilities of the ice cream samples. It was determined that symbiotic LPBA samples had the highest radical scavenging activity and the lowest EC50 value across the freezing periods, followed by the probiotic ice cream samples and the synbiotic LPAA sample. On the contrary, at week eight, the C sample had the lowest radical scavenging activity and the maximum EC50 value (P < 0.05; Table 6).
Evaluation of probiotic survival and prebiotic potential
Table 7 lists the bacterial counts found in five samples of ice cream. After eight weeks of freezing, the bacterial quantities in all samples decreased compared to the other freezing periods (P < 0.05). On the other hand, the probiotic and synbiotic samples had significantly higher bacterial counts for all the freezing periods, while the synbiotic LPBA samples had the highest bacterial counts for all the freezing periods. In other words, the synbiotic samples (LPBA and LPAA) had higher bacterial counts compared to the probiotic samples (LBA and LAA), respectively. Further, the after-aging LAA and LPAA samples had the lowest bacterial counts compared to the before-aging LBA and LPBA samples, respectively, for all the freezing periods. In other words, aging resulted in the lowest bacterial counts.
Sensory acceptability of ice cream samples
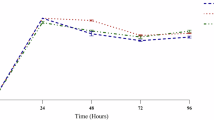
The sensory qualities of ice cream products in terms of flavour, colour, texture, and general acceptance are illustrated in detail in Fig. 4. The appearance, crystalline structure, and dissolving characteristics of the probiotic LBA and LAA samples were comparable to those of the C sample. However, the aroma, flavour, colour, texture, or total acceptance ratings of the ice cream samples varied from one another (P > 0.05). The probiotic LBA sample was the one with the best flavour and aroma. Nevertheless, compared to the C sample, the synbiotic LPBA and LPAA samples had poorer ratings for aroma, flavour, colour, texture, and overall acceptability attributes (P ≤ 0.05). However, the symbiotic LPBA sample had a higher general acceptance score than the C sample.
Sensory scores of ice cream products produced with PE and L. casei according to the sensory acceptability test before and after aging.
Volatile compound (VC) profiles of synbiotic ice cream samples
The C sample comprised 25 VCs (aldehyde, ketone, ester, and alcohol), according to the results of the GC–MS characterization of the VCs in ice cream samples. In comparison, the synbiotic LPBA sample contained 27 VCs (aldehyde, ketone, ester, and alcohol). The most common VCs found in the C sample were acetone (23.55%), ethanol (15.47%), 3_methyl butanal (4.95%), 2_3_butanedione (5.69%), 3-methyl-1-butanol (5.24%), and 1-hexene (4.82%). The VCs found in the synbiotic LPBA sample, on the other hand, were mostly acetone (17.97%), ethanol (9.76%), acetaldehyde (7.48%), isobutyraldehyde (5.89%), and 3-methyl-1-butanol (4.81%). In general, the symbiotic LPBA sample had more or less VCs compared to the C sample, as shown in Table 8.
Discussion
Ice cream is a popular dairy product enjoyed by people of all ages around the globe. It is appreciated for its delicious taste, refreshing qualities, and nutritional benefits. The food and beverage industry is currently exploring natural dyes as alternatives to synthetic dyes due to concerns regarding their toxicity, carcinogenicity, and safety when added to food products at certain concentrations. The increasing demand for certain food products is driven by evolving consumer preferences. Nowadays, consumers are seeking out natural foods that undergo minimal processing and offer health benefits through bioactive properties, such as antioxidant effects. Nevertheless, the application of natural dyes in food is constrained by the characteristics of the food matrix in which the pigment is distributed, as well as its interactions with other components like proteins, polysaccharides, lipids, sugars, salts, and more30,31. The primary aim of this study is to develop an ice cream that is enriched with cyanobacterial pigment. The ice cream subjected to microbial, physiochemical, and antioxidant analysis. Additionally, the ice cream evaluated for its synbiotic properties with Lactobacillus casei.
The Extraction and purification results of our study showed that both PE content and purity of the ice cream samples increased to almost four times from crude extract to purified PE, illustrating the process's effectiveness in producing high-purity PE. In comparison to our study, in the previously article by Nowruzi et al.22, the purified C-phycoerythrin resulted in the purification of 65.04 µg mL−1 via acetate buffer (pH 5.1). In another study by the same author, the concentration and purity of extracted PE from Nostoc sp. were increased after each purification step by almost four times (from 1.5 to 6.22)32.
Adding algal biomass and their derivatives such as PE to ice cream changes its physicochemical properties such as ice cream mix, particularly pH and acidity as they raise the proportion of solids-non-fat in the combination. During storage, lactic acid is produced by lactic acid bacteria and causes the consumption of existing lactose and the production of hydrogen. The decrease in pH occurs due to the production of acid by lactic acid bacteria and the increase in acidity. During their numerous studies, researchers have stated that the activity of starter bacteria causes a significant drop in pH during the storage period. The pH scale is displayed as the negative logarithm of the molar concentration of hydrogen ions, and as the strength of the acid or hydrogen ions increases, the pH scale decreases and determines the concentration of hydrogen ions or ionized hydrogen in the sample. For example, in our study the LBA and LPBA samples had lower pH values compared to the C and LAA and LPAA samples. Our findings were consistent with the related studies, which indicated that probiotic ice cream samples had lower pH values (Alamprese et al.8; Da Silva et al.9). Similarly, the probiotic LBA, LAA and synbiotic LAA and LPAA samples had higher acidity values compared to the C samples with the highest value for the LPBA sample.
Tiepo et al.18 observed that cyanobacterial Spirulina platensis increased acidity and lowered pH of the ice cream samples confirming our results on the effect of the alginate-encapsulated PE addition18. Additionally, Sagdic et al.11. found that L. casei lowered pH and raised acidity of the ice cream samples during fermentation, confirming our results for the probiotic LBA sample. Further, the fermented ice cream's ability to retain flavour was adversely affected by the pH levels of the ice cream samples11. It is stated that by increasing the number of microalgae and probiotics, the amount of acid production increased, which was due to the high growth of probiotics in high concentrations of microalgae. Atik et al.33 fortified the vegan kefir with Spirulina platensis and showed that increasing the concentration of Spirulina platensis decreased the pH values of kefir samples33. In the study by Agustini et al.34 in evaluation of physicochemical, microbiological and sensory characteristics of yogurts enriched with Spirulina platensis, a decrease in pH concentration was also observed with an increase in the number of Spirulina platensis bacteria34. The same results were also seen in the study by Mocanu et al.35.
Ice cream is a semi-solid food that is made by freezing milk, fat, and sugar mixture with or without food additives. Commercial ice creams are deficient in protein and have antioxidant properties, are easy to melt, and are high in fat. Therefore, it is necessary to improve the quality of ice cream. Therefore, by adding nutrients with high nutritional value, such as microalgae, the nutrients of ice cream a can be provided. Since ice cream may be a major part of the diet, especially for children; the use of eatable algae paves the way to produce food rich in essential nutrients and increase their quality36,37. The high protein content of microalgae is one of the main reasons for considering them as the main source of protein. In our study, Protein content (%), did not differ significantly between samples (P > 0.05), confirming the findings of Da Silva et al.9 who found that probiotic Bifidobacterium animalis subsp. lactis BLC1 had no impact on protein level of the ice cream sample and contradicting those of Pandiyan et al.13 who found that adding L. acidophilus and S. boulardii to ice cream samples increased their protein content. Compared to our result, Shafaei Bajestani et al.38 showed that ice cream enriched with Spirulina sp. had higher protein content38. Morover in a study by Verga et al.39, the benefits of Spirulina biomass-enriched milk were reported. The results showed that the addition of Spirulina biomass increased the content of amino acids, vitamins, and fatty acids. This may be because the presence and abundance of bioactive substances in the biomass of Spirulina are of great nutritional importance for the production of functional dairy foods39. Akalin et al.40 in the study of the effect of Spirulina biomass on the microbiological activity of traditional yogurts and probiotics during refrigerated storage showed that the protein content of algae-containing samples increased significantly compared to the control sample40.
Microalgae are known for their high fat content, comparable to vegetable oils. While some species can accumulate up to 85% fat under specific conditions, the typical range falls between 20 and 40% of their dry weight. These fats are primarily triglycerides composed of fatty acids with chain lengths between 14 and 22 carbons. Notably, Spirulina, a commonly studied microalgae, boasts 5–7% lipids rich in essential fatty acids like linoleic acid and gamma-linolenic acid41. Nostoc sp. is cholesterol-free and rich in unsaturated fatty acids, which makes it suitable for treating and preventing atherosclerosis, obesity, and high blood pressure. Due to the direct effects of gamma-linolenic acid on the immune system and the treatment of many diseases, therefore, there has always been great interest in producing high concentrations of gamma-linolenic acid42. In the study of Bosnia et al.43 on the effect of concentrations of 0.25, 0.5, and 1 g/kg S. platensis 500 powder on traditional Greek cheese, it was shown that increasing the concentration of Spirulina, the amount of fat and moisture increase and decrease, respectively43, however, the Augustini results in 2016 showed that the application of Spirulina platensis powder in concentrations of 0%, 1% and 1.5% on ice cream and soft cheese did not change the fat content but reduced the moisture content15. The increased fat content of Spirulina-enriched cheese was also observed in studies by Beheshtipour et al.43. Tiepo et al.18 found that in comparison with control samples, adding 1% pure or microencapsulated S. platensis to ice cream increased its protein and fat content. This was expected, given the high protein and lipid content of S. platensis biomass. Similarly, Agustini et al.15 found that the addition of this cyanobacterial strain resulted in a significant effect for protein, total solid, fat and total sugar, overrun, melting point and sensory properties of ice cream samples. De Oliveira et al.17 found that a Spirulina sp. strain microencapsulated with maltodextrin and soy lecithin increased the protein content, DPPH and phenolic compounds and reduced total lipids of the chocolate milk samples.
As the consumers currently desire low-fat products, probiotic inoculation may be helpful when making low-fat ice cream. Similarly, Pandiyan et al.13 found that adding L. acidophilus and S. boulardii to ice cream samples lowered their fat content. However, in our study, adding encapsulated PE and bacteria had no significant deference in the fat content (%) (P > 0.05) of the ice cream samples both before and after aging.
The results of ash in our study showed a gradual increase. Compared to our study, the amount of cheese ash in the study of Beheshtipour et al.43, as well as Shafaei Bajestani et al.38 research, had increased between 10 and 11%. This increase in ash is due to the addition of PE of this microalgae due to the high percentage of micoalgal ash content. Moreover, the overrun, also known as the rise in ice cream volume, brought on by adding air, was crucial to the ice cream's quality. In our study, overrun (%), did not differ significantly between samples (P > 0.05). On the contrary, Agustini et al.15 found that S. Platensis-enriched samples had higher overrun values compared to control samples. Similarly, Da Silva et al.9 found that the probiotic ice cream had no change in overrun compared to C samples. The addition of extra air may have increased the mixture's volume. According to Malik et al.16, the ice cream prepared with the Spirulina biomass exhibited an increase in the overrun behaviour confirming the findings of Agustini et al.15.
The presence of L. rhamnosus, however, did not seem to have any significant impact on the overrun levels of the ice cream samples according to Alamprese et al.8. It is suggested that Ice cream with higher overrun would have fewer ice crystals and increases the volume of air increases after addition of solid-lipid microparticles in the ice cream. Further, it is reported that fat and protein content relatively influence ice cream overrun and contribute air interfaces stabilization44.
The melting characteristics of ice cream are crucial to its long-term stability and how consumers experience it in terms of texture and flavour release4. Melting rate is a useful indicator of structural development and resistance to collapse in ice cream as a time against average dripped volume45. Melting point property is attributed to water holding capacity, which refers to the interaction between protein, product, and water that results in some water remaining with the product. In a study by Priyanka Malik et al.16, the addition of Spirulina to ice cream increased the melting resistance. This may be due to the protein and fat in ice cream fortified with Spirulina biomass46. Rasouli et al.47 in another study optimized the formulation of traditional Iranian ice cream containing Spirulina microalgae47.
The results of their study showed that the enrichment of ice cream with Spirulina increases viscosity and melting resistance and reduces ice cream aeration. Based on the results of sensory evaluation, with increasing the number of Spirulina in the ice cream formulation, consumers' acceptance of the taste, color, texture, and overall acceptance of the product decreased. Finally, their conclusion showed that by enriching ice cream with Spirulina microalgae, a product with desirable physical and organoleptic properties can be achieved47.
It must be emphasized that in our study, the probiotic LBA and LAA and synbiotic LPBA and LPAA samples had no significant change in the melting behaviour confirming the findings of Da Silva et al.9. Total solid contents, ice crystal size, quantity and size of fat globules, stabilizers, emulsifiers, and storage are some factors that impact melting time. For instance, Augustini et al.15 found the increased melting durations for the prebiotic ice cream with S. platensis contradicting our findings. The greater fat content of the enriched samples may have hampered heat transfer and prolonged melting periods.
In our study, the probiotic LBA and synbiotic LPBA and LPAA samples had slightly higher viscosity values compared to the C and probiotic LAA samples. This finding shows the slight positive impact of the addition of the encapsulated PE to the synbiotic ice cream samples. In addition, there is no significant variation in the ash content of all samples (Table 2) confirming the findings of Da Silva et al.9. However, the moisture contents decreased across all samples (Table 4). As Pushpadass et al.48 showed, the moisture content of the ice cream products affects a range of physicochemical properties of the products48.
Regarding the overall solid content of ice cream products, Sarwar et al.14 found that S. boulardii samples had lowered overall solid content14. In our study, there was no significant change in the probiotic ice cream samples compared to the C samples at all freezing periods confirming Sarwar et al.14. However, the synbiotic LPBA and LPAA samples had higher total solid content compared to the C and probiotic LBA and LAA samples in all the freezing periods listed in Table 3. These findings show the positive slight impact of the addition of the encapsulated PE on the total solid content of the synbiotic ice cream samples.
The data presented here indicate that inoculating ice cream samples with the encapsulated PE might alter their physicochemical properties, mostly favourably. However, in comparison to the control sample, the detected modifications were not statistically significant.
Caenorhabditis elegans (C. elegans) is a favored model organism for environmental and developmental toxicology research due to several advantages. These include its abundance, ease of culture, and ability to thrive in both soil and aquatic environments. As a result, C. elegans provides valuable insights into the potential environmental impacts of pollutants. In this study, we employed C. elegans to assess the potential toxicity of PE. The results demonstrated no significant impact on C. elegans survival at any tested concentration (100% to 96.1% survival). This finding contrasts with the observations of Singh et al.49, who reported that phycocyanin (PC) extracted from Leptolyngbya sp. enhanced the activity of aged worms. These contrasting results highlight the need for further research to explore the potential benefits and risks associated with different types of phycobiliproteins.
Compounds containing antioxidant properties may not respond equally to different sources of free radicals. Therefore, the use of multiple assays to measure antioxidant capacity is necessary for appropriate assessments. It was shown that the reducing power of the FRAP method is dose-dependent and with increasing the concentration of phycocyanin, the antioxidant activity increases50. Similar results were observed in the study of the antioxidant and nutritional potential of cookies enriched with Biomass Spirulina platensis at concentrations of 2% and 5% by the FRAP method by Egea et al.51. Research conducted by Gabr et al.52 studies the antioxidant activity of phycocyanin extracted from S. platensis at concentrations of 5,10,15,20,25,30 µg/ml by the DPPH method showed that As the concentration of phycocyanin increases, its antioxidant activity increases. Shalaby et al.53 attributed the antioxidant activity of Spirulina extracts by DPPH to the presence of phenols with high levels of biologically active phytochemicals (sterols, flavonoids, reducing sugars, tannins, and anthraquinones)53. In addition, in a study on the evaluation of antioxidant activity by nitric oxide trapping method of S. platensis ethanolic extract in vitro, it was shown that its antioxidant activity is dose-dependent, and with increasing the concentration of the extract, the antioxidant activity increases54; Similar results were obtained in the study of extraction and purification of phycocyanin from the dry powder of Spirulina platensis and evaluation of its antioxidant, anticoagulant activity and prevention of DNA damage55.
The antioxidant properties of yogurt and cheese enriched with Spirulina were investigated by Barkala et al.56. In their study, after adding Spirulina to yogurt in different concentrations, they studied its effect on the fermentation process, texture, and nutritional and sensory properties of yogurt. The data showed that the addition of Spirulina biomass accelerates fermentation and preserves our tissue properties and sensory acceptance56.
According to the EC50 value, a sample's intensity should result in a 50% drop in the original DPPH concentration. While low EC50 values demonstrate considerable antioxidant activity, high EC50 values suggest poor antioxidant activity. Our findings suggested that antioxidant activity was considerably reduced to eight weeks of freezing at − 20 °C in all samples (P < 0.05) (Table 5). Similarly, Sagdic et al.26 found that after being frozen for 60 days, ice cream lost its ability to scavenge DPPH radicals confirming our findings. Further, the synbiotic LPBA samples had the highest antioxidant activity compared to C, LPA, LAA, and LPAA samples across all the freezing periods. In other words, the addition of the encapsulated PE resulted in the increasing antioxidant activity for the synbiotic ice cream samples both before and after aging. This finding justifies the use of the encapsulated PE in the ice cream products making them healthier for the consumers.
The most important finding in this respect is that both probiotic (LBA and LAA) and synbiotic (LPBA and LPAA) samples had more bacterial counts for all freezing periods from day one to two weeks. Secondly, the LAA and LPAA samples had less bacterial counts in relation to the respective LBA and LPBA samples. Thirdly, all samples had less bacterial counts after the six and eight weeks of freezing.
Fourthly, the use of the encapsulated PE resulted in the increased bacterial counts in the synbiotic LPBA and LPAA samples compared to probiotic samples, indicating the positive impact of the PE addition to the ice cream products. Similarly, the inulin and fructo-oligosaccharide prebiotics increased the viability of L. acidophilus and S. boulardii in ice cream samples13,14, confirming our findings in this respect.
The third finding implies that that viable bacterial populations could decrease by at least 1 log unit during eight weeks of freezing. Furthermore, storing at a low temperature (− 18 °C) might have contributed to the declining viable bacteria numbers during freezing.
It has also been recently documented that the viable counts of L. acidophilus, S. boulardii and L. rhamnosus decreased in frozen storage13,14,57 confirming our third finding. Bacterial numbers might have decreased because the mixture was stirred and had access to oxygen10. However, Abghari et al.7 showed that the viable count of L. rhamnosus did not change noticeably throughout the storage period of 12 weeks7.
As Mohammadi et al.5 suggests one way to preserve the bacterial populations after the eight weeks of freezing is the encapsulation of the bacteria as it was done in Li et al.58 following the case of the encapsulation of PE with alginate in this study and encapsulation of Spirulina biomass17,18. They encapsulated L. rhamnosus using pectin, glucose, and calcium chloride. They found that encapsulation increased tolerance of bacteria in the acid condition, protected them from protease digestion, and improved shelf time when stored at the ambient condition.
Similarly, the probiotic and synbiotic ice cream samples with L. casei and L. rhamnosus and inulin had bacterial populations above the minimal numbers required for a probiotic sample after eight weeks of freezing at − 20 °C6, confirming our findings. As predicted, in our sample the probiotic content of ice cream products decreased throughout storage, but more crucially, none had probiotics below 6.18 log CFU mL−1. The freezing process, which damaged cells and ultimately led to cell death, possibly caused the decline of probiotics after eight weeks of freezing5,11.
Color in food has an important impact on consumers since it is one of the first characteristics we perceive from a product. Additionally, colored food is attractive, and color allows for better identification and selection among similar products. Coloration helps to relate water with food; for example, yogurt, animal or vegetal milk (e.g., soybean, coconut, or almonds) with fruits such as strawberries (reddish), blue berries (purple), and melon (green). Then, consumers would consider ingesting these types of food as beneficial to their well-being even though they may not have fruits59. Cyanobacterial PBPs are natural pigments used as colorants in some food products; for example, aqueous extracts of non-purified blue PC from Spirulina have been added in ice creams, yogurts, isotonic beverages, confectionery, and jellies60. Particularly, bright blue PC has been selected over others less-bright natural colorants such as gardenia blue and indigo in confectionery production61,62,63. Red PE has been mostly used as a fluorescent probe in biomedical studies, rather than in the food industry. The addition of microalgal biomass and their derivatives such as PE to ice cream, can change the sensory qualities such as colour, aroma, flavour, taste, and texture of the product. Sensory characteristics play an important role in the overall acceptance of food products by consumers. Therefore, consumers should avoid functional foods if the combined ingredients produce unpleasant tastes. The addition of microalgae creates an unpleasant odor similar to fish, which is due to its mineral content15. Researchers have studied methods to reduce the odor associated with Spirulina using activated charcoal adsorption, heating, enzymatic hydrolysis of lysozyme, the inclusion of beta-cyclodextrin, fermentation and solvent extraction, and fermentation and extraction of ethanol as the most effective method to reduce the odor associated with Spirulina has been also proved64.
According to Durmaz et al.4, adding P. cruentum enhanced the ice cream's aesthetics because of the pink pigments found in the microalgae. Nevertheless, the panellists felt that high levels of microalgae might harm sensory qualities. Since the panellists agreed that the bitter taste was stronger, adding more microalgae had a negative impact on the ice cream flavour. In comparison, the probiotic LBA sample was the one with the best flavour confirming the findings of Di Criscio et al.6. Further, compared to the C sample, the synbiotic LPBA and LPAA samples had poorer ratings for aroma, flavour, colour, texture, and overall acceptability attributes (P ≤ 0.05) confirming the findings of Di Criscio et al.6. However, there was a positive impact of the microencapsulation of S. platensis with Arabic gum or maltodextrin on the acceptability ratings18.
It can be stated that changes in texture is related to the fatty acid composition of the product, The products enriched with PE may have lowered the saturated fatty acids, such as C14:0 and increased the unsaturated fatty acids, such as C18:165. Overall, the sensory characteristics largely rely on the raw materials and production techniques used. Further investigation is required to evaluate the influence of diverse components, including VOCs, AAs, FAs, and peptides, on each sensory attribute. In another study by Lambiase et al.66, the sensory evaluation of cheese with Spirulina sp. Were performed. The results showed that Spirulina-mozzarella cheeses were perceived with a higher butter odor, a higher whey flavor, and a sweeter and bitterer taste than the samples from the control group. Furthermore, the panelists perceived higher oiliness and moisture intensities in the samples with Spirulina with a greater milk release when cutting and a tendentially higher tenderness than in the mozzarella cheeses from the control group, which, on the contrary, were perceived as being more grainy, cohesive, screechy, and having a greater consistency when cutting than the Spirulina-mozzarella cheeses.
Volatile organic compounds (VOCs) are chemical substances in nature and plant cultivation. Microalgae, a is one of the most VOC-rich substances. There are some different types of cyanobacteria VOCs components that have been discovered, and some of them are responsible for the authentic aroma such as β-ionone, heptadecane and dihydrvactinodiolide67. The environmental and taxonomical factors determine the composition of chemical content and VOC in microalgae biomass68. The environmental factor of cultivation such as, temperature, salinity, illumination, pH, nutrient and cultivation methods69. Composition of VOC in substances detection by Gas Chromatography (GC) separation techniques. Gas Chromatography (GC) is a technique for separating chemicals found in a sample. The retention time (RT) is the time it takes for a certain compound to transit the length of the column and is the defining property70.
When microalgal biomass is added to food products, its odor may influence the sensory properties of the final products. However, increasing the addition of microalgae in food products can improve the contain of bioactive compounds and antioxidant capacity but decrease the panelist acceptance51,71. The consumer acceptance and purchase intention of a food product will be primarily affected by odor and flavor. For example, The fortified dark chocolate with Spirulina has consumer preferences with lowest value (55.8%)72. The purchase intention consumers of the cookies fortified by Spirulina have lower values than formulation without auditioning Spirulina. The fishy odor of these volatile components reduces consumer acceptance and inhibits the development of edible algae73,74.
The inoculation stage, inoculation technique, co-inoculation of probiotics, and the range of volatile ingredients present in the synbiotic ice cream samples all influenced the volatile profile of ice cream products. Furthermore, given that the aging process is crucial for the sensory evaluation of ice cream products, these results implied that L. casei infection occurred before synbiotic ice cream production. Major aromatic components that significantly contribute to the aroma character of functional products include aldehydes, ketones, and esters14.
In this study, the VC profiles of C and synbiotic LPBA samples of ice cream were evaluated before the assessment of aging after one week of storage. The Table 7 shows that the major VCs were acetone and ethanol with over 23 and 15% of the C sample and the content of these VCs fell significantly in the PLBA sample compared to the C sample.
Conclusion
This is the first study, testing the usage of PE as a natural pigment extracted from Nostoc sp. together with a probiotic Lactobacillus sp. taking into account the aging process during probiotic ice cream production. In this perspective, PE from a Nostoc cyanobacterial strain encapsulated with alginate was introduced as potential prebiotics to produce synbiotic ice cream products with L. casei. The addition of the encapsulated PE affected, mostly favourable, the physicochemical properties, antioxidant activity, probiotic survival, VC contents, and sensory acceptability of the synbiotic ice cream samples before and after aging at the freezing periods from one day to eight weeks. The results of this study opens the ways for further research on using the PEs and other PBPs from other cyanobacterial and other algal strains encapsulated with alginate and other encapsulating materials to produce synbiotic ice cream and other dairy products using Lactobacilli and other microbial strains. As discussed in this paper, the research on the encapsulation of the bacteria could be integrated with such studies to protect the integrity of the bacteria in the harsh processing conditions of ice cream production and storage.
Data availability
The datasets used and/or analysed included in this published article [As supplemtray file, Supp. S1].
Abbreviations
- PE:
-
Phycoerythrin
- PBPs:
-
Phycobiliproteins
- B-PE:
-
B-phycoerythrin
- R-PE:
-
R-phycoerythrin
- C-PE:
-
C-phycoerythrin
- GIT:
-
Gastrointestinal tract
- IDF:
-
International dairy federation
- PE:
-
Phycoerythrin
- PC:
-
Phycocyanin
- APC:
-
Allophycocyanin
- PBPs:
-
Biliproteins
- DSC:
-
Differential scanning calorimetry
- GAE:
-
Gallic acid equivalents
- DPPH:
-
1,1-Diphenyl-2-picrylhydrazyl radical
- GC-MS:
-
Gas chromatography-mass spectrometry
- NIST:
-
National Institute Standard, and Technology
- WHO:
-
World Health Organization
References
Bhowmik, D., Dubey, J. & Mehra, S. Probiotic efficiency of Spirulina platensis-stimulating growth of lactic acid bacteria. World J. Dairy Food Sci. 4(2), 160–163 (2009).
García, A. B., Longo, E., Murillo, M. C. & Bermejo, R. Using a B-phycoerythrin extract as a natural colorant: Application in milk-based products. Molecules. 26(2), 297 (2021).
Chen, H., Qi, H. & Xiong, P. Phycobiliproteins: A family of algae-derived biliproteins: Productions, characterization and pharmaceutical potentials. Mar. Drugs. 20(7), 450 (2022).
Durmaz, Y. et al. Using spray-dried microalgae in ice cream formulation as a natural colorant: Effect on physicochemical and functional properties. Algal Res. 47, 101811 (2020).
Mohammadi, R., Mortazavian, A. M., Khosrokhavar, R. & da Cruz, A. G. Probiotic ice cream: Viability of probiotic bacteria and sensory properties. Ann. Microbiol. 61, 411–424 (2011).
Di Criscio, T. et al. Production of functional probiotic, prebiotic, and synbiotic ice creams. J. Dairy Sci. 93(10), 4555–4564 (2010).
Abghari, A., Sheikh-Zeinoddin, M. & Soleimanian-Zad, S. Nonfermented ice cream as a carrier for Lactobacillus acidophilus and Lactobacillus rhamnosus. Int. J. Food Sci. Technol. 46(1), 84–92 (2011).
Alamprese, C., Foschino, R., Rossi, M., Pompei, C. & Corti, S. Effects of Lactobacillus rhamnosus GG addition in ice cream. Int. J. Dairy Technol. 58(4), 200–206 (2005).
Da Silva, P. D. L., de Fátima, B. M., dos Santos, K. M. O. & Correia, R. T. P. Potentially probiotic ice cream from goat’s milk: Characterization and cell viability during processing, storage and simulated gastrointestinal conditions. LWT-Food Sci. Technol. 62(1), 452–457 (2015).
Hagen, M. & Narvhus, J. Production of Ice Cream Containing Probiotic Bacteria (Springer, 1999).
Homayouni, A., Azizi, A., Javadi, M., Mahdipour, S. & Ejtahed, H. Factors influencing probiotic survival in ice cream: A review. Int. J. Dairy Technol. 7(1), 1–10 (2012).
Gupta, S., Gupta, C., Garg, A. & Prakash, D. Prebiotic efficiency of blue green algae on probiotics microorganisms. J. Microbiol. Exp. 4(4), 4–7 (2017).
Pandiyan, C., Annal Villi, R., Kumaresan, G., Murugan, B. & Gopalakrishnamurthy, T. Development of synbiotic ice cream incorporating Lactobacillus acidophilus and Saccharomyces boulardii. Int. Food Res. J. 19(3), 1–10 (2012).
Sarwar, A. et al. Characterization of synbiotic ice cream made with probiotic yeast Saccharomyces boulardii CNCM I-745 in combination with inulin. LWT. 141, 110910 (2021).
Winarni Agustini, T. et al. Application of spirulina platensis on ice cream and soft cheese with respect to their nutritional and sensory perspectives. J. Teknol. 78(4–2), 245–251 (2016).
Malik, P., Kempanna, C. & Paul, A. Quality characteristics of ice cream enriched with Spirulina powder. Int. J. Food Nutr. Sci. 2(1), 44–50 (2013).
De Oliveira, T. T. B. et al. Microencapsulation of Spirulina sp. LEB-18 and its incorporation in chocolate milk: Properties and functional potential. LWT. 148, 111674 (2021).
Tiepo, C. B. V. et al. Addition of Spirulina platensis in handmade ice cream: Phisicochemical and sensory effects/Adição de Spirulina platensis em sorvete caseiro: Efeitos físico-químicos e sensoriais. Braz. J. Dev. 7, 88106–88123 (2021).
Ganesan, A. R. & Shanmugam, M. Isolation of phycoerythrin from Kappaphycus alvarezii: A potential natural colourant in ice cream. J. Appl. Phycol. 32, 4221–4233 (2020).
Ramu Ganesan, A. et al. Phycoerythrin: A pink pigment from red sources (rhodophyta) for a greener biorefining approach to food applications. Crit. Rev. Food Sci. Nutr. 63(31), 10928–10946 (2023).
Afreen, S. & Fatma, T. Extraction, purification and characterization of phycoerythrin from Michrochaete and its biological activities. Biocatal. Agric. Biotechnol. 13, 84–89 (2018).
Nowruzi, B., Fahimi, H. & Lorenzi, A. S. Recovery of pure C-phycoerythrin from a limestone drought tolerant cyanobacterium Nostoc sp. and evaluation of its biological activity. Anal. Biol. (2020).
Galetović, A. et al. Use of phycobiliproteins from atacama cyanobacteria as food colorants in a dairy beverage prototype. Foods. 9(2), 244 (2020).
Horwitz, W. & Latimer, G. W. Official Methods of Analysis of AOAC International, vol. 1 (AOAC International, 2000).
Molina, M. I., Petruccelli, S. & Añón, M. C. Effect of pH and ionic strength modifications on thermal denaturation of the 11S globulin of sunflower (Helianthus annuus). J. Agric. Food Chem. 52(19), 6023–6029 (2004).
Sagdic, O., Ozturk, I., Cankurt, H. & Tornuk, F. Interaction between some phenolic compounds and probiotic bacterium in functional ice cream production. Food Bioprocess Technol. 5, 2964–2971 (2012).
Čanadanović-Brunet, J. M. et al. Radical scavenging and antimicrobial activity of horsetail (Equisetum arvense L.) extracts. Int. J. Food Sci. Technol. 44(2), 269–278 (2009).
Kumar, T. & Jain, V. Appraisal of total phenol, flavonoid contents, and antioxidant potential of folkloric Lannea coromandelica using in vitro and in vivo assays. Scientifica. 2015, 1–10 (2015).
Lawless, H. T. & Heymann, H. Sensory Evaluation of Food: Principles and Practices (Springer, 2010).
Zheng, J. et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 70(4), 2782–2858 (2020).
Tan, H. T. et al. A review on a hidden gem: Phycoerythrin from blue-green algae. Mar. Drugs. 21(1), 28 (2022).
Nowruzi, B., Anvar, A. & Ahari, H. Extraction, purification and evaluation of antimicrobial and antioxidant properties of phycoerythrin from terrestrial cyanobacterium Nostoc sp. FA1. J. Microb. World. 13(2), 138–153 (2020).
Atik, D. S., Gürbüz, B., Bölük, E. & Palabıyık, İ. Development of vegan kefir fortified with Spirulina platensis. Food Biosci. 42, 101050 (2021).
Agustini, T., Soetrisnanto, D. & Ma’ruf, W. Study on chemical, physical, microbiological and sensory of yoghurt enriched by Spirulina platensis. Int. Food Res. J. 24(1), 1–10 (2017).
Mocanu, G., Botez, E., Nistor, O. V., Andronoiu, D. & Vlăsceanu, G. Influence of Spirulina platensis biomass over some starter culture of lactic bacteria. J. Agroaliment. Process. Technol. 19(4), 474–479 (2013).
Anvar, S. A. A., Nowruzi, B. & Afshari, G. A Review of the Application of Nanoparticles Biosynthesized by Microalgae and Cyanobacteria in Medical and Veterinary Sciences. (2023).
Pourbaba, H., Anvar, A. A., Pourahmad, R. & Ahari, H. Changes in acidity parameters and probiotic survival of the kefir using Lactobacillus acidophilus and Lactobacillus paracasei complementary probiotics during cold preservation. (2022).
Shafaei Bejestani, M., Anvar, A. A., Nowruzi, B. & Golestan, L. Production of cheese and ice cream enriched with biomass and supernatant of Spirulina platensis with emphasis on organoleptic and nutritional properties. Iran. J. Vet. Med. 18, 263–278 (2023).
Varga, L., Szigeti, J., Kovács, R., Földes, T. & Buti, S. Influence of a Spirulina platensis biomass on the microflora of fermented ABT milks during storage (R1). J. Dairy Sci. 85(5), 1031–1038 (2002).
Akalin, A., Unal, G. & Dalay, M. Influence of Spirulina platensis biomass on microbiological viability in traditional and probiotic yogurts during refrigerated storage. Italian J. Food Sci. 21(3), 357–364 (2009).
Ötleş, S. & Pire, R. Fatty acid composition of Chlorella and Spirulina microalgae species. J. AOAC Int. 84(6), 1708–1714 (2001).
Sajilata, M., Singhal, R. S. & Kamat, M. Y. Supercritical CO2 extraction of γ-linolenic acid (GLA) from Spirulina platensis ARM 740 using response surface methodology. J. Food Eng. 84(2), 321–326 (2008).
Bosnea, L. et al. Incorporation of Spirulina platensis on traditional greek soft cheese with respect to its nutritional and sensory perspectives. Proceedings (2020).
Flores, A. & Goff, H. Ice crystal size distributions in dynamically frozen model solutions and ice cream as affected by stabilizers. J. Dairy Sci. 82(7), 1399–1407 (1999).
Chen, W. et al. Effects of soy proteins and hydrolysates on fat globule coalescence and meltdown properties of ice cream. Food Hydrocoll. 94, 279–286 (2019).
Sofjan, R. P. & Hartel, R. W. Effects of overrun on structural and physical characteristics of ice cream. Int. Dairy 14(3), 255–262 (2004).
Rasoulu, F., Berenji, S. & Shahab, L. A. Optimization of Traditional Iranian Ice Cream Formulation Enriched with Spirulina Using Response Surface Methodology. (2017).
Pushpadass, H. A. et al. Physicochemical, thermal, and flow properties of ice cream powder as influenced by moisture content. J. Food Process. Preserv. 45(2), e15106 (2021).
Singh, N. K. et al. Phycocyanin moderates aging and proteotoxicity in Caenorhabditis elegans. J. Appl. Phycol. 28, 2407–2417 (2016).
Punampalam, R., Khoo, K. S. & Sit, N. W. Evaluation of antioxidant properties of phycobiliproteins and phenolic compounds extracted from Bangia atropurpurea. Malay. J. Fundam. Appl. Sci. 14(2), 289–297 (2018).
Egea, B., Campos, A., De Carvalho-Eliane, J. C. M. & Danesi, D. G. Antioxidant and nutritional potential of cookies enriched with Spirulina platensis and sources of fibre. J. Food Nutr. Res. 53(2), 171–179 (2014).
Gabr, G. A., El-Sayed, S. M. & Hikal, M. S. Antioxidant activities of phycocyanin: A bioactive compound from Spirulina platensis. J. Pharm. Res. Int. 32(2), 73–85 (2020).
Shalaby, E. A. & Shanab, S. M. Comparison of DPPH and ABTS Assays for Determining Antioxidant Potential of Water and Methanol Extracts of Spirulina platensis. (2013).
Anbarasan, V., Kumar, V. K., Kumar, P. S. & Venkatachalam, T. In vitro evaluation of antioxidant activity of blue green algae Spirulina platensis. Int. J. Pharm. Sci. Res. 2(10), 2616 (2011).
Kamble, S. P., Gaikar, R. B., Padalia, R. B. & Shinde, K. D. Extraction and purification of C-phycocyanin from dry Spirulina powder and evaluating its antioxidant, anticoagulation and prevention of DNA damage activity. J. Appl. Pharm. Sci. 3(8), 149–153 (2013).
Barkallah, M. et al. Effect of Spirulina platensis fortification on physicochemical, textural, antioxidant and sensory properties of yogurt during fermentation and storage. LWT. 84, 323–330 (2017).
Heenan, C., Adams, M., Hosken, R. & Fleet, G. Survival and sensory acceptability of probiotic microorganisms in a nonfermented frozen vegetarian dessert. LWT-Food Sci. Technol. 37(4), 461–466 (2004).
Li, R. et al. Preserving viability of Lactobacillus rhamnosus GG in vitro and in vivo by a new encapsulation system. J. Control. Release. 230, 79–87 (2016).
Griffiths, J. C. Coloring foods & beverages. Food Technol. 59(5), 1–10 (2005).
Food U, Administration D. Summary of Color Additives for Use in the United States in Foods, Drugs, Cosmetics, and Medical Devices. (US Department of Health and Human Services, 2017).
Jespersen, L., Strømdahl, L. D., Olsen, K. & Skibsted, L. H. Heat and light stability of three natural blue colorants for use in confectionery and beverages. Euro. Food Res. Technol. 220, 261–266 (2005).
Pandey, V., Pandey, A. & Sharma, V. Biotechnological applications of cyanobacterial phycobiliproteins. Int. J. Curr. Microbiol. Appl. Sci. 2(9), 89–97 (2013).
Sharma, N. K., Rai, A. K. & Stal, L. J. Cyanobacteria: An Economic Perspective (Wiley, 2013).
Bao, J., Zhang, X., Zheng, J.-H., Ren, D.-F. & Lu, J. Mixed fermentation of Spirulina platensis with Lactobacillus plantarum and Bacillus subtilis by random-centroid optimization. Food Chem. 264, 64–72 (2018).
Uzun, P. et al. The inclusion of fresh forage in the lactating buffalo diet affects fatty acid and sensory profile of mozzarella cheese. J. Dairy Sci. 101(8), 6752–6761 (2018).
Lambiase, C. et al. Use of cyanobacterium spirulina (Arthrospira platensis) in buffalo feeding: Effect on mozzarella cheese quality. Foods. 12(22), 4095 (2023).
Aguero, J. et al. Volatile components of a commercial sample of the blue-green algae Spirulina platensis. J. Essen. Oil Res. 15(2), 114–117 (2003).
Prasetiyo, H. et al. Off-odour identification from volatile organic compounds (VOCs) of Spirulina. in BIO Web of Conferences (2024).
Afriani, S., Uju, U. & Setyaningsih, I. Chemical composition of spirulina platensis which cultivated in photobioreactors with different photoperiodes. Jurnal Pengolahan Hasil Perikanan Indon. 21(3), 471–479 (2018).
Liu, L. et al. Economic analysis of volatile characteristics of Haematococcus pluvialis and the effect of roasting temperature on the profile of volatiles and chemical components. Algal Res. 64, 102690 (2022).
Boyanova, P. et al. Effect of Spirulina platensis on the quality and antioxidants characteristics of ice cream. in BIO Web of Conferences (2022).
Asti, G. K. & Ekantari, N. Consumer preferences for dark chocolate products fortified with Spirulina platensis using analytical hierarchy process method. in E3S Web Of Conferences (2020).
Hamdan, A. B., Riaty, C., Fitriya, W. & Ekantari, N. Effects of nanoencapsulated carotenoid of Spirulina platensis on the sensory profiles of dark and milk chocolate. in E3S Web Of Conferences (2020).
Wang, J., Zhang, M. & Fang, Z. Recent development in efficient processing technology for edible algae: A review. Trends Food Sci. Technol. 88, 251–259 (2019).
Author information
Authors and Affiliations
Contributions
Conceptualization, Bahareh Nowruzi, and Seyed Amir Ali Anvar. Methodology, Maryam Chamari, Rezvan Pourahmad, Shima yousefi. Software, Bahareh Nowruzi. Validation, Bahareh Nowruzi,. Formal analysis, Bahareh Nowruzi. Investigation, Maryam Chamari1. Resources, Bahareh Nowruzi. Data curation, Bahareh Nowruzi. Writing—original draft preparation, Bahareh Nowruzi. Writing—review and editing, Bahareh Nowruzi. Visualization, Bahareh Nowruzi. Supervision, Bahareh Nowruzi. Project administration, Bahareh Nowruzi. Revision and editing, Bahareh Nowruzi. All authors approved the manuscript’s published form once they had read it. Each Author attests that all procedures were followed in conformity with all applicable rules and regulations.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chamari, M., Anvar, S.A.A., Pourahmad, R. et al. Study of alginate-encapsulated phycoerythrin in promoting the biological activity of synbiotic ice cream with Lactobacillus casei. Sci Rep 14, 15471 (2024). https://doi.org/10.1038/s41598-024-66365-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-66365-7