Abstract
The number of Methicillin-resistant Staphylococcus aureus (MRSA) cases in communities and hospitals is on the rise worldwide. In this work, a nonlinear deterministic model for the dynamics of MRSA infection in society was developed to visualize the significance of awareness in interventions that could be applied in the prevention of transmission with and without optimal control. Positivity and uniqueness were verified for the proposed corruption model to identify the level of resolution of infection factors in society. Furthermore, how various parameters affect the reproductive number \({R}_{0}\) and sensitivity analysis of the proposed model was explored through mathematical techniques and figures. The global stability of model equilibria analysis was established by using Lyapunov functions with the first derivative test. A total of seven years of data gathered from a private hospital consisting of inpatients and outpatients of MRSA were used in this model for numerical simulations and for observing the dynamics of infection by using a non-standard finite difference (NSFD) scheme. When optimal control was applied as a second model, it was determined that increasing awareness of hand hygiene and wearing a mask were the key controlling measures to prevent the spread of community-acquired MRSA (CA-MRSA) and hospital-acquired MRSA (HA-MRSA). Lastly, it was concluded that both CA-MRSA and HA-MRSA cases are on the rise in the community, and increasing awareness concerning transmission is extremely significant in preventing further spread.
Similar content being viewed by others
Introduction
Staphylococcus genus is a common inhabitant of the skin and mucous membranes, and under certain circumstances can cause different types of diseases on a variety of species. Staphylococcus aureus (S. aureus) is the most important species in this genus which can cause serious infections in various tissues and organs with its many pathogenic factors1. S. aureus is one of the leading causes of both hospital-acquired and community-acquired infections, such as septic arthritis, osteomyelitis, bacteremia, and especially wound and soft tissue infections which are among the first nosocomial infections2,3,4. In 1960, methicillin—a semi-synthetic derivative of penicillin—was developed as a beta-lactam group of antibiotics named penicillinase-resistant penicillin, which achieved great success in the treatment of staphylococcal infections. However, soon later in the late 1970s and early 1980s methicillin-resistant Staphylococcus aureus (MRSA) strains began to emerge hindering the treatment by the use of methicillin caused by these infections. The mecA gene located in a 21–67 kb DNA region called Staphylococcal cassette chromosome mec (SCCmec) is responsible for methicillin resistance in staphylococci5,6. These strains are increasingly becoming a serious health problem as a result of multidrug resistance development and the availability of limited treatment options7,8. Hence, awareness of MRSA as a public health burden should arise since it can no longer be tackled only with hospital infection prevention and control measures9. On the other hand, innovative approaches such as the use of mathematical models in forecasting infectious diseases play significant roles in public health. The use of the models is enhanced by constructing compartments that divide the studied population according to the designed model allowing researchers to identify the structure of the disease, predict the future of the disease, and introduce necessary control strategies whether applicable. These models can be generated for almost every field of health sciences including infectious diseases, cancer, tumors, etc.10,11,12. As an example, in the study13, extended-spectrum beta-lactamases (ESBL) resistance in Escherichia coli (E. coli) isolates was evaluated via mathematical modeling13. In other studies, it was shown that with optimal control theory that can be applied to control bacterial growth14, antibiotic resistance can be evaluated via sensitivity analysis of a deterministic mathematical model as in Ref.15. Mathematical modeling is an extremely effective and popular method not only in bacterial infections but also in viral and parasitic infections. In literature, there exist many studies in the area that made contributions to public health16,17,18,19,20,21,22.
To study transmission dynamics, assess various infection control interventions, assess the burden of infection, and promote further understanding of LTCF epidemiology, models that explicitly represent the relationship between the risk of infection and the current population of infectious individuals are helpful23. Several mathematical models have been utilized to illustrate MRSA transmission in hospital environments24,25. However, the findings and public health consequences derived from these investigations might not be relevant in non-hospital contexts26. With this presented study, a cross-infection model with diffusive bacteria in the environment is provided to examine the part that ambient bacteria play in the dynamics of hospital infections27. MRSA poses a serious threat to both public and clinical health. Limited information exists on local lineage profiles. This study provides data on the incidence of cases that are acquired in hospitals and the community28. Although it is yet unclear how much of an impact each intervention has, infection control programs and antimicrobial stewardship are beneficial in lowering the burden of diseases caused by multidrug-resistant organisms. The presented model describes the impact of interventions at the ward level to achieve this goal.
In the study29, the authors modified the Ross-Macdonald model for explaining the cross-transmission dynamics of carbapenem-resistant Klebsiella pneumoniae (CRKP) in hospitals. This model takes into account healthcare staff as the vectors that transmit both susceptible and resistant bacteria among hospitalized patients29. The effectiveness of therapies for infections linked to healthcare is evaluated using mathematical models. These models, like any analytical technique, involve a lot of assumptions. These assumptions lack a biological foundation and may inadvertently affect the projected effects of interventions. Consequently, to specifically assess the implications of these hypotheses, we created a model of MRSA transmission30. A mathematical model of a few common bacterial illnesses in our culture, including strep throat, TB, fever, and S. aureus, was examined in30,31,32,33,34, demonstrating the critical role that mathematics plays in helping to understand behavior, control methods, and presentation techniques.
This study is proposed to determine the presence of CA-MRSA and HA-MRSA patients in the north side of Cyprus. In this article, Sect. "Introduction" is an introduction; developed model, biological feasibility such as positively invariant, equilibrium points, reproductive potential, sensitivity, and stability analysis are in Sects. "Formulation of mathematical model" and "Qualitative analysis of the model". The algorithm developed by using the NSFD method for simulation with analysis is discussed in Sect. “Numerical results with non-standard finite difference method”. In Sect. "Mathematical model with optimal control", we construct the optimal control of the system, and numerical results and discussion are considered in Sect. "Numerical simulations and discussion for the constructed mathematical model". The conclusion and the impact of the study are discussed in Sect. "Conclusion".
Formulation of mathematical model
In the presented paper, the model consists of eight compartments as follows: susceptible individuals \(\left(S\right)\), individuals who are infected with community-acquired S. aureus \(\left({C}_{C}\right)\), individuals who are infected with CA-MRSA \(\left({C}_{I}\right)\), individuals who are infected with hospital-acquired S. aureus \(\left({H}_{C}\right)\), individuals who are infected with HA-MRSA \(\left({H}_{I}\right)\), antibiotic group oxazolidinones \(\left(O\right)\), antibiotic group glycopeptides \(\left(G\right)\), antibiotic group trimethoprim-derivatives \(\left(T\right)\). In this model the compartment \(S\) consists of individuals who are susceptible to the S. aureus bacteria and who are capable of being infected with it. As authors, it is confirmed that all methods were performed in accordance with the relevant guidelines and regulations. The model is constructed by using ordinary differential equations at the time \(t\) and it is given below.
with initial conditions.
The details of the compartments and parameters are given in Tables 1 and 2.
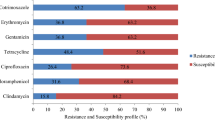
In this study, the target is to determine the effects of antibiotic groups oxazolidinones, glycopeptides, and trimethoprim-derivatives since they were the three leading antibiotic groups that the infected patients’ samples were obtained to be the most sensitive. The sensitivity percentages of samples to these antibiotic groups are illustrated in Fig. 1.
The sensitivity of Methicillin-resistant Staphylococcus aureus bacteria in patients.
Positively invariant region
An in-depth explanation of the circumstances in which the solutions of the suggested model hold positivity is the goal of the analysis. We have,
Since \(O\left(t\right)>0 \forall t\ge 0. \text{Hence}\),
Likewise,
Now, we establish the norm:
where \({D}_{\varphi }\) is the domain of \(\varphi .\)
We have,
This implies that,
Similarly, we find,
Theorem 2.1:
The specified model (1) and initial conditions (2) have a unique and bounded solution in \({R}_{+}^{8}.\)
Proof:
From model (1)\(,\) we have,
This suggests that the proposed system has a unique solution on \(\left(0,\infty \right).\)
If \((S\left(0\right), {C}_{C}\left(0\right),{C}_{I}\left(0\right), {H}_{C}\left(0\right),{H}_{I}\left(0\right), O\left(0\right), G\left(0\right),T(0))\in {R}_{+}^{8},\) then according to the Eq. (3), the solution cannot omit hyperplanes. Additionally, bounding the non-negative orthant on each hyperplane, the vector field points into \({R}_{+}^{8}\), i.e. the domain \({R}_{+}^{8}\) is a positively invariant set.
Theorem 2.2
Assume that \(\left(S, {C}_{C}, {C}_{I}, {H}_{C}, {H}_{I}, O, G, T\right)\) is one of the solutions of the proposed system with the following initial conditions:
Then, the set \(W\) below is biologically feasible and all of the solutions with non-negative initial conditions in \({\mathbb{R}}_{+}^{8}\) stay in \(\Lambda \) concerning the proposed system12.
Proof:
Let \(N\) denote the whole population. That is, \(N=S+{C}_{C}+{C}_{I}+{H}_{C}+{H}_{I}+O+G+T\). The addition of all of the terms that are on the right side of the system gives,
Since, all the state variables are positive, therefore, above equality makes it clear that,
Applying integration to both sides with respect to \(t\) yields,
For some arbitrary constant \(p\). With the use of Theorem 2.1, and Rota and Birkhoff for the above differential inequality, it can be obtained that as \(t\) tends to infinity, \(\infty \), \(0\le N\le\Lambda \). Consequently, the solutions of the given system enter the region \(\Lambda .\) Thus, it is certain that the model is feasible by means of biology and it is enough to consider the dynamics of the model in \(\Lambda \).
Qualitative analysis of the model
Equilibrium points
For the constructed model, there exists a disease-free equilibrium point, denoted by \({E}_{0}\). At this point, the disease is expected to die out in the population. In this case, \({E}_{0}\) is the point where MRSA patients do not exist in the population and hence no one is diagnosed with MRSA. To reach \({E}_{0}\), \({R}_{0}\) value(s) of the disease should be less than 1. \({E}_{0}\) of this model is unique and it is obtained as \({E}^{0}=\left({S}^{0},{C}_{C}^{0},{C}_{I}^{0},{H}_{C}^{0},{H}_{I}^{0},{G}^{0},{O}^{0},{T}^{0}\right)=\left(\frac{\Lambda }{\mu }, 0, 0, 0, 0,\text{0,0},0\right)\) and the endemic equilibrium point is given as \({E}^{*}=\left({S}^{*},{C}_{C}^{*},{C}_{I}^{*},{H}_{C}^{*},{H}_{I}^{*},{G}^{*},{O}^{*},{T}^{*}\right)\), It is clear that the endemic equilibrium point attracts the region so that, \({S}_{*}\) is the solution of,
where
Here
The other points are as follows:
Reproduction number of the system
In this model, since there are 4 different categories of disease, 4 different basic reproduction numbers, denoted by \({R}_{0}\), are obtained for each disease compartment. For the calculation of \({R}_{0}\) formulas, Next generation matrix (NGM) Method is used as follows12: The matrix that consists of new infections with MRSA is,
and the rest of the system is included in the below matrix:
According to the NGM method, \({R}_{0}\) formulas are computed by finding the dominant eigenvalues of the matrix \(F.{V}^{-1}\). Hence, \({R}_{0}\) formulas for the compartments \({C}_{C}, {C}_{I}, {H}_{C}\) and \({H}_{I}\) are obtained as below, respectively.
Sensitivity analysis
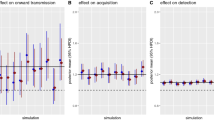
In order to facilitate analysis, 279 MRSA-positive samples were divided into groups based on age, year (before to/after the pandemic), and type (CA-MRSA/HA-MRSA). For this investigation, the information gleaned from the hospital system data regarding positive MRSA outpatients was referred to as CA-MRSA and that of inpatients as HA-MRSA. Appropriate statistical models and tests were run on the data groups to see whether there was any significance between these groups. It is verified by the authors that all procedures were followed in compliance with all applicable rules and regulations. Here we determined the sensitivity analysis of the model to check the impact of different parameters on reproductive number. The impact of parameters is shown in Fig. 2.
Impact of parameter through sensitivity index in reproductive number.
\(\frac{\partial {R}_{0, {H}_{l}}}{\partial {\beta }_{4}}=\frac{\Lambda }{{\mu (o}_{2}+{g}_{2}+{r}_{2}+d+\mu )} ,\) \(\frac{\partial {R}_{0, {H}_{l}}}{\partial {o}_{2}}= -\frac{\Lambda {\beta }_{4}{\mu }^{{{\prime}}}[d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}{{\mu [d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}^{2}},\) \(\frac{\partial {R}_{0, {H}_{l}}}{\partial {g}_{2}}= -\frac{\Lambda {\beta }_{4}{\mu }^{{{\prime}}}[d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}{{\mu [d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}^{2}},\)
\(\frac{\partial {R}_{0, {H}_{l}}}{\partial {r}_{2}}=-\frac{\Lambda {\beta }_{4}{\mu }^{{{\prime}}}[d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}{{\mu [d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}^{2}} ,\) \(\frac{\partial {R}_{0, {H}_{l}}}{\partial d}= -\frac{\Lambda {\beta }_{4}{\mu }^{{{\prime}}}[d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}{{\mu [d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}^{2}},\) \(\frac{\partial {R}_{0, {H}_{l}}}{\partial \mu }= -\frac{\Lambda {\beta }_{4}{\mu }^{{{\prime}}}[d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}{{\mu [d+\mu +{g}_{2}+{o}_{2}+{r}_{2}]}^{2}}.\)
Stability analysis of the model
Local stability
Theorem 3.1
The disease-free equilibrium (DFE) point of the system, \({E}_{0}\), is locally asymptotically stable if \({R}_{0, {C}_{C}}<1, {R}_{0, {C}_{I}}<1, {R}_{0, {H}_{C}}<1\) and \({R}_{0, {H}_{I}}<1\).
Proof
The Jacobian matrix16 evaluated at the DFE point \({E}_{0}\) is calculated as,
Here, \({S}_{0}=\frac{\Lambda }{\mu }\) and the eigenvalues of the matrix are \(-{\gamma }_{1}, -{\gamma }_{2}, -{\gamma }_{3}, -\mu , {\beta }_{1}{S}_{0}-a{\delta }_{1}-\left(1-a\right){\delta }_{2}-\mu , {\beta }_{2}{S}_{0}-{o}_{1}-{g}_{1}-{r}_{1}-\mu , {\beta }_{3}{S}_{0}-b{\delta }_{3}-\left(1-b\right){\delta }_{4}-\mu \) and \({\beta }_{4}{S}_{0}-{o}_{2}-{g}_{2}-{r}_{2}-d-\mu \). It is obvious that the eigenvalues \(-{\gamma }_{1}, -{\gamma }_{2}, -{\gamma }_{3}\) and \(-\mu \) are always negative since the parameters are always positive. The eigenvalues \({\beta }_{1}{S}_{0}-a{\delta }_{1}-\left(1-a\right){\delta }_{2}-\mu , {\beta }_{2}{S}_{0}-{o}_{1}-{g}_{1}-{r}_{1}-\mu , {\beta }_{3}{S}_{0}-b{\delta }_{3}-\left(1-b\right){\delta }_{4}-\mu \) and \({\beta }_{4}{S}_{0}-{o}_{2}-{g}_{2}-{r}_{2}-d-\mu \) are negative under the conditions \({R}_{0, {C}_{C}}<1, {R}_{0, {C}_{I}}<1, {R}_{0, {H}_{C}}<1\) and \({R}_{0, {H}_{I}}<1\). Thus, the DFE point is locally asymptotically stable if \({R}_{0, {C}_{C}}<1, {R}_{0, {C}_{I}}<1, {R}_{0, {H}_{C}}<1\) and \({R}_{0, {H}_{I}}<1\) and unstable if \({R}_{0, {C}_{C}}>1, {R}_{0, {C}_{I}}>1, {R}_{0, {H}_{C}}>1\) and \({R}_{0, {H}_{I}}>1\).
Global stability with Lyapunov function
Theorem 3.2
The disease-free equilibrium points \({E}^{0}\) are globally asymptotically stable if \({R}_{0}<1\).
Proof
Consider the Lyapunov function:
Taking derivative with respect to t on both sides, we get the following:
Now we’ll substitute the derivatives in the equation above.
Replacing S \(=S-{S}^{0},{ C}_{C}={C}_{C}-{{C}_{C}}^{0}, {C}_{I}={C}_{I}-{{C}_{I}}^{0},{ H}_{C}={H}_{C}-{{H}_{C}}^{0}, {H}_{l}={H}_{l}-{{H}_{l}}^{0}\), \(O=O-{O}^{0}, G=G-{G}^{0}, T=T-{T}^{0},\) we can have the following:
From the above equation, \(\frac{dV}{dt}\)<0 for \({R}_{0}<1,\text{ and }\frac{dV}{dt}\)=0 when \(S={S}^{0},{C}_{C}={{C}_{C}}^{0},{C}_{l}={{C}_{l}}^{0},{H}_{C}={{H}_{C}}^{0 },{H}_{l}={{H}_{l}}^{0 },O={O}^{0},G={G}^{0},\text{ and }T={T}^{0}\). Hence, we can conclude that the disease-free equilibrium points \({E}^{0}\) are globally asymptotically stable if \({R}_{0}<1\).
For the endemic Lyapunov function, we set all independent variables in the model, in our case, \(\left\{\text{S},{C}_{C},{C}_{l},{H}_{C},{H}_{l},\text{O},\text{G},\text{T}\right\},\dot{L}<0\) is the harmful equilibrium \({E}^{*}\).
Theorem 3.3
The endemic equilibrium point E* is globally asymptotically stable if the reproductive number \({R}_{0}>1\).
Proof
The following is how the Lyapunov function is used to prove the theorem:
As a result, when we apply the derivative with respect to t on both sides, we get the following:
And,
Now we’ll substitute the derivatives in the equation above.
Replacing S \(=S-{S}^{*},{ C}_{C}={C}_{C}-{{C}_{C}}^{*}, {C}_{I}={C}_{I}-{{C}_{I}}^{*},{ H}_{C}={H}_{C}-{{H}_{C}}^{*}, {H}_{l}={H}_{l}-{{H}_{l}}^{*}\), \(O=O-{O}^{*}, G=G-{G}^{*}, T=T-{T}^{*},\) we can have the following:
We may simplify the above equality by rewriting it as follows:
where
\(\begin{aligned}\Sigma &=\Lambda +{\beta }_{1}{C}_{C}{S}^{*}+{\beta }_{1}{{C}_{C}}^{*}S+{\beta }_{2}{C}_{l}{S}^{*}+{\beta }_{2}{{C}_{l}}^{*}{S}+{\beta }_{3}{H}_{C}{S}^{*}+{\beta }_{3}{{H}_{C}}^{*}S+{\beta }_{4}{{H}_{l}}{S}^{*}+{\gamma }_{1}O+{\gamma }_{2}G+{\gamma }_{3}T\\ &\quad+a{\delta }_{1}C+b{\delta }_{3}{H}_{C}+\mu S+\Lambda {k}_{1}+{\beta }_{1}{{C}_{C}}S+{\beta }_{1}{{C}_{C}}^{*}{S}^{*}+a{\delta }_{1}{{C}_{C}}^{*}+{{C}_{C}}^{*}+a{\delta }_{2}{C}_{C}+\mu {{C}_{C}}^{*}+\Lambda {k}_{2}\\ &\quad+{\beta }_{2}{{C}_{l}}+{\beta }_{2}{{C}_{l}}^{*}{S}^{*}+a{\delta }_{2}{{C}_{C}}^{*}+{\delta }_{2}{C}_{C}+\mu {{C}_{l}}^{*}+\left({a}_{1}+{g}_{1}+{r}_{1}\right){{C}_{l}}^{*}+\Lambda {k}_{2}+{\beta }_{2}{{C}_{l}}S+{\beta }_{2}{{C}_{C}}^{*}{S}^{*}\\ &\quad+{C}_{C}+a{\delta }_{2}{{C}_{C}}^{*}+\left({a}_{1}+{g}_{1}+{r}_{1}\right){{C}_{l}}^{*}+\mu {{C}_{l}}^{*}+\Lambda {k}_{4}+{\beta }_{4}{{H}_{l}}S+{\beta }_{4}{{H}_{l}}^{*}{S}^{*}+b{\delta }_{4}{{H}_{C}}^{*}+{\delta }_{4}{H}_{C}\\ &\quad+\left(\mu +d\right){{H}_{l}}^{*}+\left({a}_{2}+{g}_{2}+{r}_{2}\right){{H}_{l}}^{*}+\Lambda +{o}_{1}{C}_{l}+{o}_{2}{{H}_{l}}^{*}+{\gamma }_{1}{O}^{*}+{g}_{1}{C}_{l}+{g}_{2}{H}_{l}+{\gamma }_{2}{G}^{*}+{\gamma }_{1}{{C}_{l}}\\ &\quad+{\gamma }_{2}{{H}_{l}+}{\gamma }_{3}{T}^{*}+\frac{{S}^{*}}{S }\left({k}_{1}+{k}_{2}+{k}_{3}+{k}_{4}+{\beta }_{1}{C}_{C}S+{\beta }_{1}{{C}_{C}}^{*}{S}^{*}+{\beta }_{2}{C}_{l}S+{\beta }_{2}{{C}_{l}}^{*}{S}^{*}+{\beta }_{3}{H}_{C}S\right. \\ & \quad \left. +{\beta }_{3}{{H}_{C}}^{*}{S}^{*}+{\beta }_{4}{H}_{l}S+\mu S\right)+\frac{{{C}_{C}}^{*}}{{C}_{C} }\left({\beta }_{1}{{C}_{C}}^{*}S+{\beta }_{1}{C}_{C}{S}^{*}+a{\delta }_{1}{C}_{C}+a{\delta }_{2}{C}_{C}+{\delta }_{2}{C}_{C}+a{\delta }_{2}{{C}_{C}}^{*}+\mu {C}_{C}\right)\\ &\quad+\frac{{{C}_{l}}^{*}}{{C}_{l} }\left({\beta }_{2}{{C}_{l}}^{*}S+{\beta }_{2}{C}_{l}{S}^{*}+{{C}_{C}}^{*}+a{\delta }_{2}{C}_{C}+\left({a}_{1}+{g}_{1}+{r}_{1}\right){C}_{l}+\mu {{C}_{l}}^{*}+{\delta }_{2}{{C}_{C}}^{*}+\mu {C}_{l}\right)\\ &\quad+\frac{{{H}_{C}}^{*}}{{H}_{C} }\left({\beta }_{2}{{C}_{l}}^{*}S+{\beta }_{2}{C}_{l}{S}^{*}+a{\delta }_{2}{C}_{C}+\left({a}_{1}+{g}_{1}+{r}_{1}\right){C}_{l}+{\delta }_{2}{{C}_{C}}^{*}+\mu {C}_{l}\right)+\frac{{{H}_{l}}^{*}}{{H}_{l} }\left({\beta }_{4}{{H}_{l}}^{*}S+{\beta }_{4}{H}_{l}{S}^{*}\right.\\ & \quad \left.+{\delta }_{4}{{H}_{C}}^{*}+b{\delta }_{4}{H}_{C}+\left({a}_{2}+{g}_{2}+{r}_{2}\right){H}_{l}+(\mu +d){H}_{l}\right)+\frac{{O}^{*}}{O }\left( {o}_{1}{C}_{l}+{o}_{2}{{H}_{l}}^{*}+{\gamma }_{1}O\right)+\frac{{G}^{*}}{G }\left({g}_{1}{{C}_{l}}^{*}\right. \\ & \quad \left.+{g}_{2}{{H}_{l}}^{*}+{g}_{2}{G}^{*}\right)+ \frac{{T}^{*}}{T } \left({\gamma }_{1}{{C}_{l}}^{*}+{\gamma }_{2}{{H}_{l}}^{*}+{\gamma }_{3}{T}^{*}\right),\end{aligned}\)and
If \(\Sigma <\Omega \), then \(\frac{dL}{dt}<0\). However,
If \(S={S}^{*},{C}_{C}={{C}_{C}}^{*},{C}_{l}={{C}_{l}}^{*},{H}_{C}={{H}_{C}}^{* },{H}_{l}={H}_{l},O={O}^{*},G={G}^{*},\text{ and }T={T}^{*}\).
The proposed model, we conclude, has the largest compact invariant set:
Lasalle's invariance concept suggests that endemic equilibrium is globally asymptotically stable in \(\Gamma \) if \(\Sigma <\Omega .\)
Numerical results with non-standard finite difference method
This section designs an NSFD scheme that resembles the dynamics of the continuous model of the system of Eqs. (1). Let \({Y}_{k} = {\left({S}_{k}, {E}_{k}, {I}_{k}, {Q}_{k}, {R}_{k}\right)}^{T}\) denotes an approximation of \(X({t}_{k})\) where \({t}_{k} = k\Delta t\) with \(k \in N\), \(h = \Delta t > 0\) be a step size, then,
which is the proposed NSFD Scheme for the given model, where,
Since the discrete approach is built by following the Mickens criteria, it is, in fact, an NSFD scheme.
Rule 1: Eq. (9) substitutes the complex denominator function for the conventional denominator \(h = \Delta t\) of the discrete derivatives, satisfying the asymptotic connection.\(\vartheta (h) = h + O({h}_{2}).\)
Rule 2: The non-local approximation of the nonlinear variables on the right-hand side of the system of Eqs. (2). For instance, \(E({t}_{k}) I({t}_{k}) \sim = {E}_{k+1} {I}_{k}\) we have instead of \(E({t}_{k}) I({t}_{k}) \sim = {E}_{k} {I}_{k}.\)
Analysis of the scheme
Theorem 4.1
The continuous model system of Eq. (2) has a dynamical system called the NSFD scheme (40) on the biological feasible domain κ.
Proof:
Firstly, we establish that the scheme (40) is positive. It is simple to demonstrate that the NSFD scheme (40) adopts an explicit form.
Thus \({S}^{k+1}\ge 0,\) \({C}_{c}^{k+1}\ge 0\),\({C}_{l}^{k+1}\ge 0\),\({H}_{c}^{k+1}\ge 0\),\({H}_{l}^{k+1}\ge 0\), \({O}^{k+1}\ge 0,\) \({G}^{k+1}\ge 0\),\({T}^{k+1}\ge 0\). whenever in the previous iteration the discrete variables are not negative. The positive invariance of κ has to be demonstrated. we get,
Therefore \({M}_{K+1}\le \frac{\vartheta\Lambda \left({K}_{1}+{k}_{2}\right)}{2\mu +{o}_{1}+{g}_{1}+{r}_{1}a{\delta }_{1}}\) whenever \({M}_{K}\le \frac{\vartheta\Lambda \left({K}_{1}+{k}_{2}\right)}{2\mu +{o}_{1}+{g}_{1}+{r}_{1}a{\delta }_{1}}\).
The fact that \({C}_{c}^{k+1}\) and \({S}^{k+1}\) are less than or equal to \({M}^{k+1}\) makes it easy to determine the priori limits for \({C}_{l}^{k+1}\) and \({H}_{c}^{k+1}\). This completes the proof.
Simulations of the proposed scheme
The data used in this paper is gathered from the Near East University Hospital Centre Laboratory with the approval of the Near East University Ethics Review Board (project no: YDU/2022/108-1653). For presenting the study, the Near East University Ethics Review Board waived informed consent to the authors of the paper. This study includes retrospective data from 48,835 patients who were administered to the Near East University Hospital Microbiology Laboratory on the dates between January 2016 and December 2022. Microorganisms were detected in 13,350 of the studied patient samples. S. aureus was detected in 612 of the 13,350 samples. Among the 612 S. aureus samples, 279 were determined to be MRSA-positive. In this study, MRSA infections were analyzed in both community and hospitals in the north side of Cyprus. In this regard, a compartmental mathematical model was constructed with and without control to visualize the efficacy of the applied control and increase the awareness of MRSA cases given in Tables 1 and 2. According to the results of the compartmental mathematical model without control, a DFE point exists and is locally asymptotically stable in the population. Moreover, numerical simulations of the model without the optimal control revealed that in the upcoming years, there will be a rise in the four compartments studied, including community-acquired S. aureus, community-acquired MRSA, hospital-acquired S. aureus, and hospital-acquired MRSA. On the other hand, these increases are more dangerous for CA-MRSA patients that are MR positive since the trend of these patients is not stable after some point like the other compartments. We construct the numerical results through simulation in Figs. 3 and 4 for all compartments whose parameter values are given in Table 2 by using the NSFD scheme. Here, we can easily observe the solution is bounded to a steady state point and lies in a feasible region. Applied method via Matlab gives better efficiency and good agreement to understand the phenomenon and harmful effects of bacteria in society which cause several diseases.
Simulation of data by using NSFD scheme for all compartments.
Simulation of data by using NSFD scheme for all compartments at different populations.
Mathematical model with optimal control
In this section, optimal control theory was introduced to the proposed model to evaluate its efficacy on the disease. The control \(u\) denotes the efficacy of precautions taken by people who are aware of MRSA infection, where \(0\le u\le 1\). The control is added to the model as \(1-u\) since it is assumed that awareness exists in some of the population. The model is revised as follows:
The objective functional to be minimized is
The number of individuals diagnosed with MRSA in all compartments and costs of control are expected to be minimized. \(K\) is a weight factor representing benefit/cost and the level of the patient’s increase of awareness. A quadratic control \(\frac{1}{2}K{u}^{2}\) is used for convenience in finding an analytic representation of the control \(u\in\Omega \).
The goal is to find \({u}^{*}\) that will satisfy \(J\left({u}^{*}\right)=\underset{u\in\Omega }{\text{min}}J\left(u\right),\) where \(\Omega =\left\{u\left(t\right): 0\le u\le {u}_{max}=l, u \text{piecewise continuous function}, l \text{is a fixed constant}, t\in \left[0, T\right]\right\}.\) For optimal control, Pontryagin’s Maximum Principle’s conditions should be satisfied29. In this regard, the Hamiltonian \(H\) is obtained as
where \({f}_{i}^{\prime}s\) represent the right-hand side of the proposed system and \({\lambda }_{i}^{\prime}s\) represent the adjoint variables for \(i=1, 2, \dots , 8\). That is,
Theorem 5.1
Given an optimal control \({u}^{*}\) and solutions \({S}^{*}, {C}_{C}^{*}, {C}_{I}^{*}, {H}_{C}^{*}, {H}_{I}^{*}, {O}^{*}, {G}^{*}, {T}^{*}\) pf the corresponding system which minimizes \(J\left(u\right)\) over \(\Omega \). Then there exist adjoint variables \({\lambda }_{1}, {\lambda }_{2},{\lambda }_{3}, {\lambda }_{4}, {\lambda }_{5}, {\lambda }_{6}, {\lambda }_{7}, {\lambda }_{8}\) satisfying,
with transversely conditions.
and the optimal control satisfying the optimality condition,
Proof:
The adjoint system is computed by taking partial derivatives of Hamiltonian function \(H\) with respect to the state variables, separately. That is,
with transversality conditions.
On the interior of the given control set, for \(0\le u\le 1\), we get,
Thus,
Therefore,
Numerical simulations and discussion for the constructed mathematical model
Numerical simulations were calculated by the proposed compartmental mathematical model. In Figs. 5a, 6a, 7a and 8a, the expected trend of \({C}_{c}, {C}_{I}, {H}_{C}\) and \({H}_{I}\) cases are given, respectively. These trends were calculated via MatLab according to the proposed model. Numerical simulations of the given compartmental mathematical model with optimal control were calculated. Optimal control was applied to the community and the expected trend of \({C}_{c}, {C}_{I}, {H}_{C}\) and \({H}_{I}\) cases were estimated in Figs. 5b, 6b, 7b and 8b, respectively. These trends were calculated via MatLab according to the proposed model.
(a) The trend of community-acquired Staphylococcus aureus-infected individuals \(\left({C}_{c}\right)\) cases in the community. The graph demonstrates that according to the constructed model, community-acquired Staphylococcus aureus cases indicate an increasing trend for 10 years with a start date of early 2023 followed by a plateau of up to 700 cases. (b) The trend of community-acquired Staphylococcus aureus-infected individuals \(\left({C}_{C}\right)\) cases in the community with control. The graph demonstrates that according to the constructed model with optimal control, community-acquired Staphylococcus aureus cases indicate an increasing trend starting from the earliest of 2023 followed by a plateau with up to 550 cases.
(a) The trend of community-acquired methicillin-resistant Staphylococcus aureus-infected individuals \(\left({C}_{I}\right)\) cases in the community. The graph demonstrates that according to the constructed model, community-acquired methicillin-resistant Staphylococcus aureus cases indicate an increasing trend starting from the earliest of 2023 and continuing to show an increasing trend for the following years with up to 7000 cases. (b) The trend of community-acquired methicillin-resistant Staphylococcus aureus-infected individuals \(\left({C}_{I}\right)\) cases in the community with control. The graph demonstrates that according to the constructed model with optimal control, community-acquired methicillin-resistant Staphylococcus aureus cases indicate an increasing trend starting from the earliest of 2023 and continuing to show an increasing trend for the following years with up to 2000 cases.
(a) The trend of hospital-acquired Staphylococcus aureus-infected individuals \(\left({H}_{C}\right)\) cases. The graph demonstrates that according to the constructed model, hospital-acquired Staphylococcus aureus cases indicate an increasing trend for 20 years with a start date of early 2023 followed by a plateau of up to 900 cases. (b) The trend of hospital-acquired Staphylococcus aureus-infected individuals \(\left({H}_{C}\right)\) cases with control. The graph demonstrates that according to the constructed model with optimal control, hospital-acquired Staphylococcus aureus cases indicate an increasing trend starting from the earliest of 2023 followed by a plateau with up to 650 cases.
(a) The trend of hospital-acquired methicillin-resistant Staphylococcus aureus-infected individuals \(\left({H}_{I}\right)\) cases. The graph demonstrates that according to the constructed model, hospital-acquired methicillin-resistant Staphylococcus aureus cases indicate an increasing trend for 30 years with a start date of early 2023 followed by a plateau of up to 800 cases. (b) The trend of hospital-acquired methicillin-resistant Staphylococcus aureus-infected individuals \(\left({H}_{I}\right)\) cases with control. The graph demonstrates that according to the constructed model with optimal control, hospital-acquired methicillin-resistant Staphylococcus aureus cases indicate an increasing trend starting from the earliest of 2023 followed by a plateau with up to 650 cases.
When the optimal control theory was applied to the constructed model, it was observed that all compartments indicated an increasing trend. These increases in the number of cases were almost half of the results of the mathematical model without the control. For example, community-acquired S. aureus cases indicated an increasing trend for 10 years with a start date of early 2023 followed by a plateau of up to 700 cases with the constructed compartmental mathematical model. The number of cases for the same group indicated an increasing trend starting from the earliest of 2023 followed by a plateau with up to 550 cases by the constructed compartmental mathematical model with an optimal control. There is a significant drop in the number of community-acquired S. aureus cases when the control is applied. The same decreasing trends of the number of cases occurred in different compartments when the optimal control was applied to the model. In CA-MRSA cases, a rise up to 7000 is expected with the compartmental mathematical model. When optimal control was applied, the number of cases dropped to 550. The same pattern of decrease in trend occurred in the hospital-acquired Staphylococcus aureus cases from 900 to 650, and HA-MRSA cases from 800 to 650. These results firmly suggested that there is an impact of the control measures on the number of cases when applied to the mathematical model. Others have also implemented deterministic mathematical models to study CA- and HA-MRSA dynamics. D'Agata et al.’s model strongly suggested that CA-MRSA will be replaced with the dominant traditional HA-MRSA strain in hospitals and healthcare facilities. This was due to the well-documented expanding CA-MRSA reservoir and increasing influx of CA-MRSA harboring individuals into the hospitals. CA-MRSA infections result in longer hospitalizations. Hence, D'Agata et al. evoked effective strategies for hand hygiene, screening, and decolonization for CA-MRSA carriers to prevent this transmission35.
Another group McBryde et al., also applied stochastic and deterministic mathematical models to determine the transmission dynamics in an intensive care unit. In addition, the group aimed to predict the impact of interventions. The result of this study revealed that increasing the length of stay of all patients, especially the stay of colonized patients increased the transmission. The model predicted that the most effective intervention was hand hygiene to prevent transmission of HA-MRSA36.
Conclusion
The analysis and the methods used for the constructed model allowed us to understand the phenomenon and harmful effects of bacteria in society which cause several diseases. It is determined that the disease-free equilibrium point is globally asymptotically stable if \({R}_{0}<1\) while the endemic equilibrium point is globally asymptotically stable if \({R}_{0}>1\).
Overall, MRSA cases are expected to increase over time, which is a threat to public health. Thus, this study aimed to suggest which control measures should be improved in preventing the transmission of MRSA by applying optimal control theory. As a control, one of the parameters was to increase awareness and consciousness in both society and the hospital. Here it was assumed that; some part of the society was conscious, meaning that they were aware of the MRSA transmission. In the applied optimal control theory, parameters were increased to evaluate its effectiveness on transmission (the u function in the second model is this control). As a result, the model firmly suggested that the spread of this disease can be reduced if people use masks, disinfectants, and gloves (patient relatives, companions) when going to the hospital for a visit or examination. In addition, caregivers and nurses should regularly change gloves, wear masks, pay attention to hygiene, and use disinfectants when examining patients. To conclude, the model showed that increasing the awareness of control measures in the model can significantly reduce the number of MRSA cases in both the community and hospitals.
Data availability
The data for this study are available from the corresponding author upon a reasonable request.
References
Sevgican, E., Sinirtas, M., Ozakin, C. & Gedikoglu, S. Detection of methicillin resistance in Staphylococcus species with different methods. Turk. J. Infect. 23, 63–68 (2009).
Ip, M., Lyon, D. J. & Cheng, A. F. A longitudinal analysis of methicillin-resistant Staphylococcus aureus in a Hong Kong teaching hospital. Infect. Control Hosp. Epidemiol. 25, 126–129 (2004).
Wenzel, R. P., Reagen, D. R., Bertino, J. S., Baron, E. J. & Arias, K. Methicillin-resistant Staphylococcus aureus outbreak: A consensus panel’s definition and management guidelines. Am. J. Infect. Control 26, 102–110 (1998).
Lowy, F. D. Staphylococcus aureus infections. N. Engl. J. Med. 339, 520–532 (1998).
Guler, I., Kilic, H., Atalay, M. A., Percin, D. & Ercal, B. D. In-vitro susceptibility of methicillin-resistant Staphylococcus aureus strains to antibiotics. Dicle Med. J. 38, 466–470 (2011).
Hiramatsu, K., Katayama, Y., Yuzawa, H. & Ito, T. Molecular genetics of methicillin-resistant Staphylococcus aureus. Int. J. Med. Microbiol. 292, 67–74 (2002).
Cetinkaya, Y. & Unal, S. The importance and treatment of staphylococcal nasal carriage. Hosp. Infect. J. 3, 22–32 (1999).
Diekema, D. J. et al. Survey of infections due to Staphylococcus species: Frequency of occurrence and antimicrobial susceptibility of isolates collected in the United States, Canada, Latin America, Europe, and the Western Pacific region for the Sentry Antimicrobial Surveillance Program, 1997–1999. Clin. Infect. Dis. 32, 114–132 (2001).
Stefani, S. et al. Methicillin-resistant Staphylococcus aureus (MRSA): Global epidemiology and harmonisation of typing methods. Int. J. Antimicrob. Agents 39, 273–282 (2012).
Cassidy, R. et al. Mathematical modelling for health systems research: A systematic review of system dynamics and agent-based models. BMC Health Serv. Res. 19, 1–24 (2019).
Jodar, L. & Company, R. Preface to “Mathematical methods, modelling and applications”. Mathematics 10, 1607 (2022).
Gokbulut, N., Hincal, E., Besim, H. & Kaymakamzade, B. Reducing the range of cancer risk on BI-RADS 4 subcategories via mathematical modelling. CMES 133, 93–109 (2022).
Hurdoganoglu, U. et al. Evaluation of ESBL resistance dynamics in Escherichia coli isolates by mathematical modeling. Open Phys. 20, 548–559 (2022).
Yegorov, I., Mairet, F., de Jong, H. & Gouze, J. L. Optimal control of bacterial growth for the maximization of metabolite production. J. Math. Biol. 78, 985–1032 (2019).
Mondragon, E. I., Leiton, J. P. R., Esteva, L. & Rosero, E. M. B. Mathematical modelling of bacterial resistance to antibiotics by mutations and plasmids. J. Biol. Syst. 24, 129–146 (2016).
Teklu, S. W. & Rao, K. P. HIV/AIDS-pneumonia codynamics model analysis with vaccination and treatment. Comput. Math. Methods Med. 2022, 3105734 (2022).
Teklu, S. W. & Mekonnen, T. T. HIV/AIDS-pneumonia coinfection model with treatment at each infection stage: Mathematical analysis and numerical simulation. J. Appl. Math. 2021, 5444605 (2021).
Teklu, S. W. Investigating the effects of intervention strategies on pneumonia and HIV/AIDS coinfection model. Biomed Res. Int. 2023, 5778209 (2023).
Teklu, S. W., Terefe, B. B., Mamo, D. K. & Abebaw, Y. F. Optimal control strategies on HIV/AIDS and pneumonia co-infection with mathematical modelling approach. J. Biol. Dyn. 18, 2288873 (2024).
Teklu, S. W. Impacts of optimal control strategies on the HBV and COVID-19 co-epidemic spreading dynamics. Sci. Rep. 14, 5328 (2024).
Savasan, A., Kaymakamzade, B., Gokbulut, N., Hincal, E. & Yoldascan, E. Sensitivity analysis of COVID-19 in mediterranean island. CMES 130, 133–148 (2021).
Agusto, F. B., Marcus, N. & Okosun, K. O. Application of optimal control to the epidemiology of malaria. Elect. J. Diff. Equ. 2012, 1–22 (2012).
Hethcote, H. W. The mathematics of infectious diseases. SIAM Rev. 42, 599–653 (2000).
Grundmann, H. & Hellriegel, B. Mathematical modelling: A tool for hospital infection control. Lancet Infect. Dis. 6, 39–45 (2006).
Van Kleef, E., Robotham, J. V., Jit, M., Deeny, S. R. & Edmunds, W. J. Modelling the transmission of healthcare associated infections: A systematic review. BMC Infect. Dis. 13, 294 (2013).
Kwok, K. O. et al. A systematic review of transmission dynamic studies of methicillin-resistant Staphylococcus aureus in non-hospital residential facilities. BMC Infect. Dis. 18, 188 (2018).
Danfeng, P., Yanni, X. & Zhao, X. Q. A cross-infection model with diffusive environmental bacteria. J. Math. Anal. Appl. 505, 125637 (2022).
Alsolami, A. et al. Community-acquired methicillin resistant Staphylococcus aureus in hospitals: Age-specificity and potential zoonotic-zooanthroponotic transmission dynamics. Diagnostics 13, 2089 (2023).
Durazzi, F. et al. Modelling antimicrobial resistance transmission to guide personalized antimicrobial stewardship interventions and infection control policies in healthcare setting: A pilot study. Sci. Rep. 13, 15803 (2023).
Gowler, C. D., Slayton, R. B., Reddy, S. C. & O’Hagan, J. J. Improving mathematical modeling of interventions to prevent healthcare-associated infections by interrupting transmission or pathogens: How common modeling assumptions about colonized individuals impact intervention effectiveness estimates. PLoS One 17, e0264344 (2022).
Farman, M., Alfiniyah, C. & Shehzad, A. Modelling and analysis tuberculosis (TB) model with hybrid fractional operator. Alex. Eng. J. 72, 463–478 (2023).
Ahmad, A., Farman, M., Akgul, A., Bukhari, N. & Imtiaz, S. Mathematical analysis and numerical simulation of co-infection of TB-HIV. Arab. J. Basic Appl. Sci. 27, 431–441 (2020).
Owolabi, K. M. & Pindza, E. A nonlinear epidemic model for tuberculosis with Caputo operator and fixed point theory. Healthc. Anal. 2, 100111 (2022).
Yusuf, T. T. & Abidemi, A. Effective strategies towards eradicating the tuberculosis epidemic: An optimal control theory alternative. Healthc. Anal. 3, 100131 (2023).
D’Agata, E. M. C., Webb, G. F., Horn, M. A., Moellering, R. C. & Ruan, S. Modeling the invasion of community-acquired methicillin-resistant Staphylococcus aureus into hospitals. Clin. Infect. Dis. 48, 274–284 (2009).
McBryde, E. S., Pettitt, A. N. & McElwain, D. L. S. A stochastic mathematical model of methicillin resistant Staphylococcus aureus transmission in an intensive care unit: Predicting the impact of interventions. J. Theor. Biol. 245, 470–481 (2007).
Acknowledgements
We would like to thank members of the Near East University Hospital Microbiology Laboratory and Mathematics Research Center.
Author information
Authors and Affiliations
Contributions
The contributions of each author to this research are as follows: N.G., M.F.: Conceptualization, Methodology, Formal Analysis, Writing—Original Draft, Writing—Review and Editing, Visualization and Numerical Analysis. U.H.: Conceptualization, Methodology, Investigation, Writing—Original Draft, Writing—Review and Editing, Visualization. N.S: Conceptualization, Investigation, Writing—Original Draft, Writing—Review and Editing, Visualization. E.G: Conceptualization, Investigation, Writing—Review and Editing, Visualization. E.H.: Methodology, Formal Analysis, Investigation, Writing—Review and Editing, Visualization. K.S.: Methodology, Investigation, Writing—Review and Editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gokbulut, N., Farman, M., Hurdoganoglu, U. et al. Dynamical analysis of methicillin-resistant Staphylococcus aureus infection in North Cyprus with optimal control: prevalence and awareness. Sci Rep 14, 18531 (2024). https://doi.org/10.1038/s41598-024-68893-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-68893-8
Keywords
This article is cited by
-
Analyzing fractional glucose-insulin dynamics using Laplace residual power series methods via the Caputo operator: stability and chaotic behavior
Beni-Suef University Journal of Basic and Applied Sciences (2025)
-
Mathematical modeling of tumor-immune dynamics: stability, control, and synchronization via fractional calculus and numerical optimization
Scientific Reports (2025)