Abstract
This retrospective cohort study describes the real-world incidence and maintenance of clinically meaningful intraocular pressure (IOP) reduction (“response”) following stand-alone phacoemulsification for 667,987 eyes with suspected or confirmed glaucoma in the IRIS Registry (Intelligent Research in Sight) from 1/1/2013–9/30/2019. Intraocular pressure responders had ≥ 20% IOP reduction in daily mean IOP from baseline on two consecutive postoperative visits. We declared failure if a responder no longer maintained ≥ 20% IOP reduction. The estimated IOP responder rate was 41.3% by Kaplan–Meier analysis. Multivariate analysis demonstrated relationships between IOP response and baseline IOP (hazard ratio (HR) (95% confidence interval)) 1.48 (1.48–1.49), per 3 mmHg, P < 0.0001), age (HR 1.14 (1.13–1.14), per 10 years, P < 0.0001), male sex (HR 1.13 (1.12–1.15), P < 0.0001), prostaglandin analogue (HR 0.88 (0.87–0.90), P < 0.0001), and Rho-kinase inhibitor use (HR 1.50 (1.32–1.70), P = 0.01). Fifty percent of IOP responders failed at a median time of 14.3 months. Multivariate analysis demonstrated relationships between failure and baseline IOP (HR 0.75 (0.75–0.76), per 3 mmHg, P < 0.0001), nitric oxide donating prostaglandin (HR 1.78 (1.46–2.18), P < 0.0001) and Rho-kinase inhibitor use (HR 1.73 (1.43–2.09), P < 0.0001). Clinicians may counsel glaucoma patients with risk factors on whether to anticipate an IOP response and its expected duration after stand-alone phacoemulsification.
Similar content being viewed by others
Introduction
Glaucoma is the leading identifiable cause of irreversible vision impairment and blindness worldwide1. The goal of glaucoma treatment is to prevent or delay disease progression by lowering the intraocular pressure (IOP) to a level better tolerated by the optic nerve2. Several studies suggest that stand-alone phacoemulsification reduces IOP for eyes both with and without glaucoma3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21. The leading proposed mechanism involves changes from lens removal deepening the anatomic angle which may increase aqueous outflow by expanding the trabecular meshwork, lumen of Schlemm’s canal, and collapsed aqueous valves3,4. A better understanding of the factors associated with IOP reduction after cataract surgery and survival of this IOP response may not only help elucidate its etiology but also assist surgeons when considering stand-alone phacoemulsification versus a combined procedure.
The American Academy of Ophthalmology (Academy) IRIS Registry (Intelligent Research in Sight) is a large, de-identified clinical database that contains nearly 500 million patient visits from over 78 million unique patients22. Recent work with the IRIS Registry confirmed that there is a real-world post-operative IOP reduction in over a million eyes that underwent stand-alone phacoemulsification by comparing post-operative IOP to both baseline and unoperated fellow eye IOP5. The goal of the current analysis is to leverage the size of the IRIS Registry to assess for predictors of a clinically meaningful IOP response, defined as a 20% or greater confirmed IOP reduction from baseline, and survival of IOP response for eyes with glaucoma or pre-glaucoma.
Materials and methods
Subject eligibility
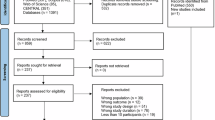
Because the IRIS Registry contains aggregated, de-identified data23, the University of Miami Institutional Review Board ruled no approval or informed consent were required for this study. The current analysis adhered to the Health Insurance Portability and Accountability Act and the tenets of the Declaration of Helsinki. Several aspects of the methodology have been previously described in a prior study investigating IOP changes after phacoemulsification in the IRIS Registry and are found elsewhere5. In summary, all patients with data included in the IRIS Registry who underwent stand-alone phacoemulsification, coded as either Current Procedural Terminology (CPT) code 66982 (complex surgery) or 66984 (standard surgery), from January 1, 2013, through September 30, 2019, were considered. Only the first eye per subject to have cataract surgery that met inclusion and exclusion criteria was included in this study. All participating eyes must have had a preoperative clinic visit within six months prior to the date of surgery with at least one IOP and one visual acuity (VA) measurement, at least two different dates with IOP measurements following surgery, and at least one IOP and VA measurement within 90 days after surgery. Our analysis included eyes with a diagnosis of “pre-glaucoma” (glaucoma suspect, ocular hypertension, primary angle-closure suspect (PACS), steroid responder, or multiple pre-glaucoma diagnoses) or glaucoma any time prior to and including the date of cataract surgery (International Classification of Disease (ICD) 9 code 365.XX, ICD-10 code H40.XXXX), any IOP-lowering medication any time prior to and including the date of surgery, any IOP-lowering procedure (CPT code 658XX, 661XX, 666XX, 667XX) any time prior to surgery, or a baseline IOP > 23 mmHg. Because the IRIS Registry does not specify laterality of topical medication, any IOP-lowering medication was conservatively assumed to be used in the surgical eye. We excluded eyes for the following reasons: age under 18 years; bilateral or unspecified laterality of surgery; any concurrent CPT code on date of surgery; no light perception (NLP) VA within six months prior or 90 days after surgery; any IOP ≤ 5 mmHg prior to surgery or within 90 days after surgery; diagnosis of pseudophakia (ICD codes V43.1 and Z96.1), either in the surgical eye or with an unspecified laterality, prior to surgery; diagnosis of corneal edema (370.2X, H18.2X), bullous keratopathy (371.2X, H18.1X), nanophthalmos (743.1, Q11.2), degenerative myopia (360.21, H44.2X), spherophakia (743.36, Q12.4), lens subluxation (379.32, H27.11X), or retained lens fragment (998.82, H59.02X); anterior vitrectomy following cataract surgery (CPT 67005 or 67,010); intravitreal injection (CPT 67028) or subconjunctival injection (CPT 68200), or injection into Tenon’s capsule (CPT 67515) within six months prior to surgery; unknown patient gender or surgeon practice region.
Study design
We performed all data processing and statistical analyses with SAS version 9.4 software (SAS Institute, Cary, NC). Data processing included removal of any IOP with unspecified laterality and any VA documented as near, bilateral, or unspecified laterality. All VAs were converted to logMAR format. We defined the baseline visit date as the date which included the IOP most recently preceding cataract surgery within six months prior to surgery. We calculated the daily mean IOP as the mean of all available IOPs on one specific date. The baseline IOP was the mean of all daily mean IOPs within 30 days of the baseline visit date. We averaged all VAs of the same priority rank, based on prior published hierarchy5, from the same date to calculate a daily mean VA. The baseline VA was the daily mean VA closest to and including the baseline visit date. Preoperative medications were classified into groups such as prostaglandin analogue, beta blocker, etc. and because the Registry does not specify laterality, assumed to be in the surgical eye5. If any additional class of medication was added after the cataract surgery, that eye was censored as this could influence IOP. Thus, medications refer to preoperative medications.
We defined IOP responder as an eye with a daily mean IOP, on two consecutive postoperative visits, with 20% or greater IOP reduction from baseline. The first postoperative visit must be within the 90-day post-operative period with the second visit confirming the response. We defined an IOP non-responder as an eye without a 20% or greater reduction from baseline in daily mean IOP on two consecutive visits, at least one of which was within 90 days of surgery. The response date was considered the first of the two visits. The IOP at response is the daily mean IOP at the first of the two visits.
In addition to calculating an absolute incidence of IOP response, we estimated the IOP response rate over the 90-day post-operative period for all eyes with glaucoma or pre-glaucoma as well as by glaucoma diagnosis using Kaplan–Meier survival analyses. For the purpose of the survival curve, we considered an IOP reduction of 20% or greater from baseline IOP the tracked event. We censored eyes if there was any IOP-lowering procedure, any additional IOP-lowering medication compared to baseline, YAG (Yttrium–aluminum-garnet) laser capsulotomy (CPT code 66821), an intravitreal injection, subconjunctival injection, or injection into Tenon’s capsule within 90 days of cataract surgery for non-responders or before or at the time of response for responders, or reached 90 days after cataract surgery without a response having occurred (the censoring date is the date of the last IOP measurement before the end of the 90-day post-operative period). We also censored the eye if the censoring event occurred on or between the two visits with ≥ 20% IOP reduction from baseline.
All eyes classified as IOP responders were further analyzed for survival of IOP response with a Kaplan–Meier survival analysis. Day zero of the response survival analysis was the date of cataract surgery. Censoring and failure event criteria were only applied after the IOP response was confirmed as above. We defied failure as any one daily mean IOP which did not maintain a ≥ 20% IOP reduction from baseline following confirmation of IOP response. We defined an IOP response survivor as an eye with no failure prior to censorship. Eyes were censored in this survival analysis if there was any IOP-lowering procedure, any additional IOP-lowering medication compared to medication at baseline visit, YAG laser capsulotomy, an intravitreal injection, subconjunctival injection, or injection into Tenon’s capsule, any NLP VA, any IOP ≤ 5 mmHg or upon reaching the end of follow-up. If a failure and censoring event occurred on the same date, failure was declared.
We compared the incidence of IOP response versus non-response, and for IOP responders, the incidence of response failure versus survival, by Chi-square test for categorical parameters and independent samples t-test assuming unequal variance for continuous parameters. We used a multivariate Cox regression analysis to identify and calculate the hazard ratio (HR) with 95% confidence interval (CI) of characteristics significantly associated with both IOP response and, for IOP responders, the IOP response survival.
Results
Demographics
We identified 667,897 eyes with glaucoma or pre-glaucoma that met inclusion and exclusion criteria (Fig. 1)5. Baseline and ocular characteristics of the patients are found online in Supplementary Tables 1 and 2, respectively. The mean age of patients was 72.2 + 8.9 (standard deviation (SD)) years with an average baseline IOP of 17.0 (4.3) mmHg and an average logMAR VA of 0.47 (SD, Snellen equivalent) (0.40, 20/59), prior to cataract surgery. Eyes were on a mean 0.46 (0.92) glaucoma medications before cataract surgery. Glaucoma severity was documented as 59.2% pre-glaucoma, 11.1% mild, 8.7% moderate, 3.5% severe, and 17.5% unspecified. The last available daily mean IOPs within the 90-day post-operative period as well as the follow-up period were at 45.4 (23.3) days and 22.3 (20.5) months after cataract surgery, respectively.
Flow chart of the IRIS Registry (Intelligent Research in Sight) subject selection. The number of patients excluded from the current study at each step is shown. CPT, Current Procedural Terminology; IOP, intraocular pressure; VA, visual acuity; CEIOL, cataract extraction and intraocular lens placement; NLP, no light perception.
Intraocular pressure response
In total, 139,991 (21.0%) of eyes had a confirmed IOP response and were classified as IOP responders. An additional 56,912 (8.5%) eyes had an initial response without a second confirmatory IOP response. Supplementary Tables 1 and 2 compare the incidence of various baseline parameters between IOP responders and non-responders. Systemic characteristics overrepresented in IOP responders include male sex, White race, diabetes, absence of hypertension, active smoker status, Medicare Fee For Service or Medicare Managed insurance, and surgeon practice in any geographic region other than the Northeast (P < 0.0001 for all parameters, excluding hypertension P = 0.005). IOP responders were older than non-responders with a mean age of 72.7 (8.8) versus 71.1 (8.9) years (P < 0.0001), respectively. Ocular characteristics overrepresented in IOP responders include right eyes, senile or age-related cataracts, complex cataract surgery (CPT 66982), eyes documented as pseudophakic after surgery rather than aphakic or unspecified lens status, and severe or unspecified glaucoma stage (P < 0.0001). Intraocular pressure responders had a greater baseline IOP than non-responders with a mean baseline IOP of 19.8 (4.8) versus 17.0 (4.3) mmHg (P < 0.0001), respectively.
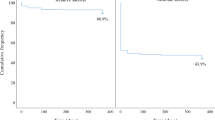
The response outcome was declared at a mean of 20.6 (21.2) days after surgery for responders and 40.9 (25.5) days for non-responders (P < 0.0001). The last available daily mean IOP within the 90-day postoperative period was at 50.7 (21.9) days for responders and 44.0 (23.4) days for non-responders (P < 0.0001). The last available daily mean IOP was at 22.7 (20.0) months for responders and 22.3 (20.6) months for non-responders (P < 0.0001). A Kaplan–Meier survival analysis estimated an IOP response rate for all eyes with glaucoma of 41.3% through the 90-day post-operative period (Fig. 2 and online Supplementary Table 3). Censoring events include 55,373 eyes with addition of a new IOP lowering medication (8.5%), 5523 Yttrium–Aluminum-Garnet capsulotomy (0.8%), 3948 injections (0.6%), and 2777 IOP lowering procedures (0.4%). The IOP response rate as estimated by Kaplan–Meier survival analysis by glaucoma diagnoses followed a similar pattern as the absolute incidence of IOP response and is displayed in Fig. 3 with additional data online in Supplementary Table 4.
Kaplan–Meier survival analysis estimate of intraocular pressure response through the 90-day post-operative period. We defined IOP responder as an eye with a daily mean IOP, on two consecutive post-operative visits, with 20% or greater IOP reduction from baseline. The first post-operative visit must be within the 90-day post-operative period with the second visit confirming the response. The date of response was considered the first of the two visits. Note no 95% confidence bands are visible given the range is narrower than the survival curve. IOP, intraocular pressure.
Kaplan–Meier survival analysis estimates of intraocular pressure response by diagnosis. Bars denote 95% confidence interval. Additional data available online in Supplementary Table 5. IOP, intraocular pressure; PACS, primary angle-closure suspect; POAG, primary open-angle glaucoma; PACG, primary angle-closure glaucoma.
Hazard ratios for IOP response in the multivariate Cox regression analysis are found in Table 1. For every 3 mmHg increase in baseline IOP, there was a 1.48 HR (95% CI 1.48–1.49) favoring IOP response (P < 0.0001). For every 10-year increase in baseline age, there was a 1.14 HR (1.13–1.14) favoring IOP response (P < 0.0001). Males were more likely to have IOP response (HR 1.13 (1.12–1.15), P < 0.0001). Compared to glaucoma suspects, eyes with diagnoses of PACS (HR 1.26 (1.23–1.28), P < 0.0001), multiple pre-glaucoma diagnoses (HR 1.18 (1.14–1.22), P < 0.001), pseudoexfoliative glaucoma (1.14 (1.10–1.19), P < 0.0001), and PACG (HR 1.13 (1.09–1.17), P < 0.0001) were more likely to be IOP responders while eyes with unclassified glaucoma (HR 0.86 (0.82–0.89), P < 0.0001), multiple glaucoma diagnoses (HR 0.88 (0.82–0.93), P < 0.001) and POAG (HR 0.94 (0.93–0.96), P < 0.0001) were less likely to be IOP responders. In addition, eyes with baseline use of prostaglandin analogues were less likely to have IOP response (HR 0.88 (0.87–0.90), P < 0.0001) while eyes on a Rho-kinase inhibitor were more likely to have IOP response (HR 1.50 (1.32–1.70), P = 0.01).
Intraocular pressure response survival
A total of 69,956 (50.0%) IOP responders had their IOP reduction survive until censorship while 69,955 (50.0%) had an observed failure. Online Supplementary Tables 5 and 6 compare the incidence of various systemic and ocular parameters between IOP responders that their IOP reduction survived versus failed. Systemic characteristics overrepresented in IOP response survivors include male sex, unknown race, no diabetes or hypertension, active or unknown smoker status, all insurance other than Medicare Fee For Service or government, or surgeon practice in the Midwest or West region (P < 0.0001). IOP response survivors were older than failures with a mean age of 72.9 (8.9) versus 72.6 (8.8) years (P < 0.0001), respectively. Ocular characteristics overrepresented in IOP response survivors include right eyes, senile or age-related, hypermature, and secondary cataracts, eyes documented as aphakic or unspecified lens status after surgery, eyes with pre-glaucoma or unspecified glaucoma severity, and eyes noted to be IOP responders by postoperative day one, week one, and month one (P < 0.0001 other than P = 0.016 for eye laterality). Intraocular pressure response survivors had a greater baseline IOP than failures with a mean baseline IOP of 21.0 (5.1) versus 18.5 (3.9) mmHg (P < 0.0001), respectively, as well as fewer baseline glaucoma medications (0.40 (1.0) versus 0.6 (1.0), P < 0.0001).
The survival of response outcome was declared at a mean of 12.0 (13.7) months after surgery for survivors and 9.7 (11.1) months for failures (P < 0.0001). The last available daily mean IOP was at 16.3 (17.8) months for survivors and 29.1 (20.1) months for failures (P < 0.0001). A Kaplan–Meier survival analysis estimated a mean survival of 23.7 months with a cumulative survival rate of 3.56% through 83.2 months after cataract surgery (Fig. 4 and online Supplementary Table 7). Quartile estimates found a 25% failure rate by 4.5 months, 50% failure rate by 14.3 months, and 75% failure rate by 35.8 months. Censoring events include 8098 Yttrium–Aluminum-Garnet capsulotomy (5.8%), 7082 additions of a new IOP lowering medication (5.1%), 982 injections (0.7%), and 11 IOP lowering procedures (0.0%). The IOP response survival rate as estimated by Kaplan–Meier survival analysis by glaucoma diagnoses followed a similar pattern as the absolute incidence of IOP response and is displayed in Fig. 5 with additional data online in Supplementary Table 8.
Kaplan–Meier survival curve with 95% confidence bands of intraocular pressure response survival for eyes that met intraocular pressure responder criteria. Day zero of the survival of response analysis was the date of cataract surgery. The failure event was any single date without a maintained 20% or greater reduction in daily mean intraocular pressure from baseline. IOP, intraocular pressure.
Kaplan–Meier survival analysis estimates of median time to failure of intraocular pressure response in months by diagnosis. Bars denote 95% confidence interval. Additional data available online in Supplementary Table 10. IOP, intraocular pressure; PACS, primary angle closure suspect; POAG, primary open-angle glaucoma; PACG, primary angle closure glaucoma.
Hazard ratios for failure in the multivariate Cox regression analysis are found in Table 2. For every 3 mmHg increase in baseline IOP, there was a 0.75 HR (95% CI 0.75–0.76) against IOP response failure (P < 0.0001). The relationship between race and IOP response survival was complex. Compared to White eyes, Black (HR 1.23 (1.20–1.26), P < 0.0001) and Hispanic eyes (1.05 (1.02–1.08), P = 0.0005) had greater odds while Asian eyes had lower odds of failure (HR 0.94 (0.90–0.98), P = 0.0007). Black (HR 1.31 (1.25–1.38), P < 0.0001) and Hispanic eyes (HR 1.12 (1.06–1.18), P < 0.0001) also had greater odds of failure compared to Asian eyes. Black eyes had greater odds of failure compared to Hispanic eyes (HR 1.17 (1.13–1.21), P < 0.0001) as well.
Compared to eyes with glaucoma suspects, eyes with diagnoses of steroid responder (HR 1.51 (1.34–1.73), P < 0.0001) and ocular hypertension (HR 1.18 (1.15–1.22), P < 0.0001) had greater odds of failure while eyes with PACS were less likely to fail (HR 0.92 (0.90–0.95), P < 0.0001). All glaucoma diagnoses had greater HR for failure compared to glaucoma suspects. There were also significant associations between IOP response failure and baseline nitric oxide donating prostaglandin analogue (HR 1.78 (1.46–2.18), P < 0.0001) and Rho-kinase inhibitor use (HR 1.73 (1.43–2.09), P < 0.0001).
Discussion
The IRIS Registry is a large, de-identified database that is a compelling example of big data as a powerful research tool. By combining both clinical and billing data from tens of millions of unique patients, the IRIS Registry is well powered to explore clinical questions that may inform and advance patient care. While several studies have either directly or tangentially explored IOP reduction following cataract surgery, most have been limited in size or were retrospective in nature. Recent publications described IOP changes following stand-alone cataract surgery in the IRIS Registry by comparing post-operative IOP to phakic fellow eyes as a control5,24. Eyes had a mean IOP reduction of about 1.5 mmHg or 8% of baseline for all eyes, about 2 mm Hg or 9% of baseline for eyes with glaucoma, and about 1 mmHg or 7% of baseline for eyes without glaucoma5. The purpose of the current analysis is to explore predictors of a clinically significant IOP response, defined as a confirmed 20% or greater reduction in IOP from baseline, as well as survival of the IOP response for eyes with glaucoma and pre-glaucoma.
The current study estimated approximately 41% of glaucomatous eyes would have an IOP response. The observed absolute IOP response rate is lower but a Kaplan–Meier analysis incorporates the constantly changing pool of eligible eyes with variable follow-up at different time points and yielded estimated IOP response rates consistent with the literature. Some of the most robust data available regarding IOP reduction following cataract surgery is derived from the control arms of clinical trials. Mansberger and colleagues6 assessed IOP changes after cataract surgery for eyes with data prospectively collected as part of the Ocular Hypertension Treatment Study. Similar to the current study, they found approximately 40% of eyes with ocular hypertension that were not randomized to medical therapy had a ≥ 20% IOP reduction. The IRIS Registry results are also congruent with the control arm of the first generation iStent (Glaukos Corporation, San Clemente, CA) pivotal trial that reported a ≥ 20% IOP reduction in 48% of stand-alone cataract surgery eyes at one year7. However, there was a greater IOP response for the control arms of the second generation iStent (62% at two years) as well as the two-year results of the HORIZON trial for the Hydrus trabecular micro-stent (58%, Alcon, Fort Worth, TX)8,9. While randomized controlled clinical trials provide a robust environment for comparing the efficacy of interventions, the artificial constraints of these studies such as strict inclusion and exclusion criteria and medication washouts can produce results that may not be as applicable to the actual experiences of many patients.
The current study augments our understanding of factors associated with an IOP response which may help guide surgical decision-making. The present multivariate analysis found baseline IOP was the greatest predictor of IOP response with a hazard ratio of 1.48 for every 3 mm Hg. The importance of baseline IOP is consistent with the literature, as emphasized in a 2015 AAO Ophthalmic Technology Assessment on IOP after cataract surgery10. Wang et al.11 created an algorithm to extract data from the electronic health record of 4883 patients, 1042 patients with glaucoma, and found a 6% increase in the hazard of achieving IOP reduction after cataract surgery with every 1 mmHg increase in baseline IOP. The Swedish National Cataract Register prospectively collected data on over 20,000 cataract surgeries and also found relationships between lower post-operative IOP and greater baseline IOP and age12. Slabaugh et al.13 similarly identified age as a risk factor for lower post-operative IOP in patients with controlled POAG. The present study suggests the causative mechanism attributed to age likely involves more than lens density because cataract diagnosis, such as total or mature cataract, was not an independent predictor of IOP response. While sex hormones may influence outflow facility and IOP25, there is limited data available on this relationship after cataract surgery. Unlike the present study that found males were more likely to have an IOP response, Moghimi and colleagues14 found a greater IOP reduction after phacoemulsification for females with PACG but their cohort included some patients with concurrent viscogoniosynechialysis. In addition, eyes that were aphakic after cataract surgery were less likely to have an IOP response. These surgeries likely had complications that often require an intraoperative injection of corticosteroid for staining of vitreous as well as a prolonged postoperative corticosteroid course. No independent differences in the odds of IOP response were identified by race in the current multivariate analysis while Wang et al.11 found a greater chance of IOP reduction for Black and Hispanic patients compared to White patients, but their cohort also included eyes without glaucoma.
The multivariate analysis found, with glaucoma suspect as a reference, diagnoses of PACS, pseudoexfoliative glaucoma, and PACG were more likely to have an IOP response while NTG and POAG were less likely to have an IOP response. The Academy Ophthalmic Technology Assessment similarly found a more robust IOP response for eyes with pseudoexfoliative glaucoma and PACG10. Compared to eyes without glaucoma, Wang et al.11 found both POAG and narrow angle diagnoses as independent variables associated with IOP reduction. The IOP-lowering effect of cataract removal for eyes with either PACS or PACG is likely due to anatomic changes from lens removal unblocking the occluded trabecular meshwork. Huang and colleagues3 used anterior segment optical coherence tomography to quantify the inverse relationship between preoperative anterior chamber depth and angle opening distance and changes to these parameters after cataract removal. They found that the percent change in IOP after surgery was nearly twice as large for eyes classified as having a narrow angle versus open angle. Yang and colleagues15 similarly described that, in addition to preoperative IOP, both change in anterior chamber area and angle opening distance were related to IOP change for 999 healthy eyes without glaucoma. Phacoemulsification may have a greater effect on IOP for eyes with pseudoexfoliative glaucoma because pseudoexfoliative material is removed with the anterior lens capsule and the intraoperative fluidics may clear this material from the trabecular meshwork and downstream collector channels16. Patients on Rho-kinase inhibitors were also much more likely to be IOP responders. This may be related to the mechanism of the drug that acts directly on the trabecular meshwork26 or Rho-kinase inhibitor use may be a surrogate for another unidentified parameter, such as socioeconomic status. Conversely, patients on prostaglandin analogues were less likely to be IOP responders, which may be due to the drug’s pro-inflammatory properties or perhaps these patients already have increased uveoscleral outflow and benefit less from the anatomic changes of lens removal.
There is limited research available on survival of IOP reduction following cataract surgery. The initial work of Mansberger and colleagues with Ocular Hypertension Treatment Study control arm data suggested that the IOP response persisted but diminished at a mean rate of 0.05 mmHg/month over the 3 years following cataract surgery6. Their more recent work with eyes with ocular hypertension that were assigned to medication had an initial IOP reduction compared to controls but this difference was gone by a year17. The control cohort of the HORIZON trial for the Hydrus trabecular micro-stent had sustained IOP reduction in eyes with stand-alone cataract surgery through five years but, because this question was not the primary aim of the study, this was not explored further18. A retrospective chart review of 240 eyes with pseudoexfoliative glaucoma found a significant IOP reduction after cataract surgery lasted only one year19.
The present study utilized Kaplan–Meier survival analysis to estimate the median time to failure of IOP responders but suggests that the durability of a clinically significant IOP reduction after cataract surgery is modest as half of all IOP responders did not maintain at least a 20% IOP reduction through 14.1 months. However, the multivariate analysis suggests several characteristics are associated with a longer sustained IOP response. Similar to predictors of IOP response, baseline IOP was the characteristic with the strongest relationship to IOP response failure as eyes with higher baseline IOP were more likely to maintain this reduction after surgery. Nearly all glaucoma diagnoses had greater odds of IOP response failure compared to glaucoma suspect eyes. This is not unexpected as these eyes have issues with IOP regulation inherent to their glaucoma. This finding is similar to the pattern described by Shingleton et al.20 who retrospectively compared the percentage of patients with a significantly lower IOP after cataract surgery by Kaplan–Meier analysis and found a lower incidence of IOP reduction for eyes with than without glaucoma at both years three and five. Interestingly, the current study found that only a diagnosis of PACS was protective against IOP response failure. For these eyes, removal of the lens and opening of the angle structures before peripheral anterior synechiae formation or chronic degradation of the trabecular meshwork affects IOP regulation appears to be protective towards improved long-term IOP control.
There are several limitations to the study, many of which are inherent to the retrospective nature of the IRIS Registry. Analyses rely on accurate data entry and maintenance of the electronic health record, including IOP measurements, diagnostic and procedural coding, and medication regimens. A potential history of prior filtering surgery such as trabeculectomy was not considered in the exclusion criteria as this data would only be captured if performed by a surgeon who participated in the Registry during the limited time period of study. Data from clinical visits with providers who do not participate in the IRIS Registry are not available for analysis. Intraocular pressure and VA were obtained by variable methods; a VA hierarchy ranking system was used to prioritize selection of higher quality VA while the method of IOP measurement is not specified in the IRIS Registry5. The baseline IOP was retrospectively calculated using a 30-day window which might not capture the most accurate baseline level, especially if there are fluctuations in IOP. The definition of IOP responder conservatively required a confirmatory IOP reduction while failure of IOP response conservatively required only one daily mean IOP with less than 20% reduction from baseline; both IOP response and survival would be greater if these definitions were more lax. There were differences as noted in length of follow-up between IOP responders and non-responders as well as eyes with IOP response survival versus failure; thus, Kaplan–Meier survival analyses were utilized to address variable follow-up length. Of note, the sample size drops dramatically after four months. Topical medications in the IRIS Registry do not have a specified laterality and were conservatively assumed to be bilateral. A large proportion of eyes were not treated with topical IOP lowering medication. Eyes with concurrent procedures, such as microinvasive glaucoma surgery, were excluded which may introduce bias as providers may have decided to not pursue concurrent intervention for reasons that affect IOP such as peripheral anterior synechiae or neovascularization. Given the size of the Registry and breadth of data available, some parameters, such as geographic region, have statistically significant relationships without a clear underlying pathophysiologic explanation. These variables may be surrogates of sociologic influences or an artifact of the study.
Despite these limitations, the current analysis increases our understanding of predictors of clinically meaningful IOP reduction and survival of this IOP response following cataract surgery in eyes with glaucoma. While approximately 40% of eyes with glaucoma were estimated to have a 20% or greater IOP reduction following cataract surgery, the survival of this response appears modest. Factors associated with IOP response include higher baseline IOP, older age, male sex, and baseline Rho-kinase inhibitor use while eyes that were aphakic after surgery or on baseline prostaglandin medication were less likely to be IOP responders. Additionally, compared to glaucoma suspects, IOP response was more likely in PACS, pseudoexfoliative glaucoma, and PACG but less likely in POAG. Half of all eyes had an observed failure before censorship with a median time to failure of 14.3 months. Factors associated with failure of IOP response survival include lower baseline IOP, Black and Hispanic race, and baseline nitric oxide donating prostaglandin or Rho-kinase inhibitor use. Most glaucoma diagnoses had a greater risk of failure when compared to glaucoma suspects, with the notable exception of PACS. These relationships may help guide surgical planning to better predict which eyes with glaucoma or pre-glaucoma may or may not have a clinically meaningful and sustained IOP reduction following cataract surgery. Given the limited long-term survival of IOP reduction from stand-alone phacoemulsification suggested in this study, surgeons may consider a concurrent procedure such as a microinvasive glaucoma surgery if there is a concern about IOP control with a patient with unfavorable odds of IOP response or survival. The precision and volume of large data repositories such as the IRIS Registry could be used to train predictive models that could enhance personalized medicine and aid in this decision-making process.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to the database being funded and supported from nonpublic sources, but are available from the corresponding author on reasonable request.
References
Pascolini, D. & Mariotti, S. P. Global estimates of visual impairment: 2010. Br. J. Ophthalmol. 96, 614–618 (2012).
Gedde, S. J. et al. Primary open-angle glaucoma preferred practice pattern. Ophthalmology 128, 71–150 (2021).
Huang, G. et al. Anterior chamber depth, iridocorneal angle width, and intraocular pressure changes after phacoemulsification: narrow vs open iridocorneal angles. Arch. Ophthalmol. 129, 1283–1290 (2011).
Poley, B. J., Lindstrom, R. L., Samuelson, T. W. & Schulze, R. Jr. Intraocular pressure reduction after phacoemulsification with intraocular lens implantation in glaucomatous and nonglaucomatous eyes: Evaluation of a causal relationship between the natural lens and open-angle glaucoma. J. Cataract. Refract. Surg. 35, 1946–1955 (2009).
Rothman, A. L., Chang, T. C., Lum, F. & Vanner, E. A. Intraocular pressure changes following stand-alone phacoemulsification: an IRIS Registry analysis. Am. J. Ophthalmol. 245, 25–36 (2023).
Mansberger, S. L. et al. Reduction in intraocular pressure after cataract extraction: The ocular hypertension treatment study. Ophthalmology 119, 1826–1831 (2012).
Samuelson, T. W. et al. Randomized evaluation of the trabecular micro-bypass stent with phacoemulsification in patients with glaucoma and cataract. Ophthalmology 118, 459–467 (2011).
Samuelson, T. W. et al. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology 126, 811–821 (2019).
Samuelson, T. W. et al. A Schlemm canal microstent for intraocular pressure reduction in primary open-angle glaucoma and cataract: The HORIZON study. Ophthalmology 126, 29–37 (2019).
Chen, P. P. et al. The effect of phacoemulsification on intraocular pressure in glaucoma patients: A report by the American Academy of Ophthalmology. Ophthalmology 122, 1294–1307 (2015).
Wang, S. Y. et al. Intraocular pressure changes after cataract surgery in patients with and without glaucoma: An informatics-based approach. Ophthalmol. Glaucoma. 3, 343–349 (2020).
Zetterström, C. et al. Changes in intraocular pressure after cataract surgery: Analysis of the Swedish national cataract register data. J. Cataract Refract. Surg. 41, 1725–1729 (2015).
Slabaugh, M. A., Bojikian, K. D., Moore, D. B. & Chen, P. P. The effect of phacoemulsification on intraocular pressure in medically controlled open-angle glaucoma patients. Am. J. Ophthalmol. 157, 26–31 (2014).
Moghimi, S. et al. Phacoemulsification versus combined phacoemulsification and viscogonioplasty in primary angle-closure glaucoma: a randomized clinical trial. J. Glaucoma. 24, 575–582 (2015).
Yang, H. S., Lee, J. & Choi, S. Ocular biometric parameters associated with intraocular pressure reduction after cataract surgery in normal eyes. Am. J. Ophthalmol. 156, 89–94 (2013).
Merkur, A., Damji, K. F., Mintsioulis, G. & Hodge, W. G. Intraocular pressure decrease after phacoemulsification in patients with pseudoexfoliation syndrome. J. Cataract. Refract. Surg. 27, 528–532 (2001).
Mansberger, S. L. et al. Cataract surgery lowers intraocular pressure and medication use in the medication group of the Ocular Hypertension Treatment Study. Am. J. Ophhthalmol. 236, 53–62 (2022).
Ahmed, I. I. K. et al. Long-term outcomes from the HORIZON randomized trial for a Schlemm’s canal microstent in combination cataract and glaucoma surgery. Ophthalmology 129, 742–751 (2022).
Shingleton, B. J. et al. Effect of phacoemulsification on intraocular pressure in eyes with pseudoexfoliation: single-surgeon series. J. Cataract. Refract. Surg. 34, 1834–1841 (2008).
Shingleton, B. J., Pasternack, J. J., Hung, J. W. & O’Donoghue, M. W. Three and five year changes in intraocular pressures after clear corneal phacoemulsification in open angle glaucoma patients, glaucoma suspects, and normal patients. J. Glaucoma. 15, 494–498 (2006).
Rothman, A. L. & Lee, R. H. Cataract extraction in glaucoma. Int. Ophthalmol. Clin. 63, 137–152 (2023).
American Academy of Ophthalmology. IRIS Registry Data Analysis. Accessed May 9, 2023. https://www.aao.org/iris-registry/data-analysis/requirements.
Chiang, M. F. et al. The 2016 American Academy of Ophthalmology IRIS Registry (Intelligent Research in Sight) database: Characteristics and methods. Ophthalmology 125, 1143–1148 (2018).
Lidder, A. K., Vanner, E. A., Chang, T. C., Lum, F. & Rothman, A. L. Intraocular pressure spike following stand-alone phacoemulsification in the IRIS Registry (Intelligent Research in Sight). Ophthalmology. https://doi.org/10.1016/j.ophtha.2024.01.022 (2024).
Toker, E., Yenice, Ö. & Temel, A. Influence of serum levels of sex hormones on intraocular pressure in menopausal women. J. Glaucoma. 12, 436–440 (2003).
Saha, B. C. et al. Status of Rho kinase inhibitors in glaucoma therapeutics—an overview. Int. Ophthalmology. 42, 281–294 (2022).
Author information
Authors and Affiliations
Contributions
Authors ALR, TCC, FL, and EAV contributed to the conception and design of the work and interpretation of data. Author EAV contributed to the analysis of data and creation of software used in the work. All authors reviewed the manuscript and approved of the submitted version and agreed to be accountable for the work.
Corresponding author
Ethics declarations
Competing interests
This research was funded in part by Research to Prevent Blindness (New York, NY, USA)/American Academy of Ophthalmology (San Francisco, CA, USA) 2020 Award for IRIS Registry Research; NIH Center Core Grant (P30EY013810, Bethesda, MD, USA); Research to Prevent Blindness Unrestricted Grant (New York, NY, USA). The sponsor or funding organizations participated in the design of the study, conducting the research, data collection, data management, and review and approval of the manuscript. Dr. Lum is an employee of the American Academy of Ophthalmology. Dr Vanner is a stockholder for Greenlight Biosciences (Medford, MA, USA). Drs. Rothman, Chang, and Lum have no financial disclosures.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Tables
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rothman, A.L., Chang, T.C., Lum, F. et al. Predictors of intraocular pressure response and survival after phacoemulsification for glaucomatous eyes in the IRIS registry (Intelligent Research in Sight). Sci Rep 14, 19050 (2024). https://doi.org/10.1038/s41598-024-70148-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70148-5