Abstract
Unionid mussels deposit growth rings (annuli) within the shell, which can be used to estimate age and growth. Thin-sectioning is a common technique for counting annuli, wherein a cross-section of a shell valve is taken and evaluated by multiple readers. Correctly identifying annuli can be challenging because ambiguous annuli can bias growth estimates. Staining with calcein, a fluorescent chemical, is a technique that has been used with marine and freshwater species to improve accuracy of growth estimates. This method chelates calcium, causing a permanent mark that fluoresces under ultraviolet light. Calcein has seen limited testing on unionid mussels so it remains unclear if this method has adverse effects on survival and growth. We evaluated calcein against 2 concentrations (125 mg L−1 and 250 mg L−1) at 2 exposure times (12 and 24 h) on Cyclonaias pustulosa, a common North American unionid. Survivorship remained above 80% 6 months post-immersion. Mark quality and retention for 250 mg L−1 were high for both 12- and 24-h immersions, although historical annuli were not highlighted. These findings corroborate studies indicating calcein immersion is generally safe and effective in juveniles and adults and suggest it may be useful in validating new growth.
Similar content being viewed by others
Introduction
Age, growth, and longevity are key indicators of individual performance within a given environment, which can provide insight on mechanisms shaping abundance, distribution, and persistence of aquatic organisms1,2,3. As a result, these data are useful for helping scientists and resource managers better anticipate how species will respond to environmental change and management actions4,5,6. Failing to consider this information for conservation, or using unvalidated estimates, can lead to poor understanding of life history traits and potential mismanagement5. For example, management actions intended for fast-growing or long-lived species but incorrectly applied to slow-growing or short-lived species could have detrimental outcomes. Thus, emerging methods on how to accurately measure age, growth, and longevity are critically important for conservation and natural resource management.
Given the importance of age, growth, and longevity, a number of methods have been developed to collect and validate this type of information, including recovery of known-age individuals, evaluation of length-frequency distributions, and interpretation of calcified structures3. Of these methods, counting annual deposits of calcium and protein in calcified structures such as otoliths and shells is the most widely used3. For freshwater unionid mussels, shell thin-sectioning is the primary method for estimating age, growth, and longevity, and entails taking a thin, radial cross-section of a shell valve using a low-speed saw and mounting it on an unfrosted microscope slide. Independent observers then count the historical growth rings (hereafter annuli) to determine age and measure the distance between annuli to estimate annual growth using a dissecting microscope1,7,8. Despite its wide use, obtaining accurate estimates can vary based on observer experience, whether or not a species produces unambiguous annuli, and how crowded the annuli are within the shell7,8.
Calcein, a fluorescent chemical (fluorochrome), is a chemical marking method that may help minimize some of the variation and sources of errors associated with shell thin-sectioning. This method entails immersing individuals in a bath containing dye, which chelates the calcium in calcified growing structures and forms a fluorescent mark that can be used to indicate growth when viewed under ultraviolet light9. Calcein has been used successfully to mark and evaluate growth in fish10, abalone11, marine mussels12,13, and oysters14,15,16,17 with minimal impacts to survivorship. For unionid mussels, fluorescent staining has been tested only on early juveniles of Lampsilis cardium, Plain pocketbook, and Actinonaias pectorosa, Pheasantshell, with little to no adverse effects on survival or growth18. However, because calcein has been tested on so few species it is unclear whether this is the case for other unionids or if the stain may manifest in the shell beyond just marking the annulus at the growing edge. Marking unionids with calcein entails submerging individuals in a chemical solution, which means other parts of the shell such as historical annuli could be illuminated if the dye is absorbed deeper into the shell matrix. To the best of our knowledge this has not been evaluated.
Freshwater unionid mussels are one of the most imperiled faunal groups globally due to human impacts19,20 and accurate age, growth, and longevity will be invaluable for better understanding “winners” and “losers” of these impacts and efforts to mitigate them. To facilitate a better understanding of these parameters we evaluated the efficacy of calcein in marking adult Cyclonaias pustulosa (pimpleback), a wide-ranging species in North America. This will be the first test of calcein in adult unionids, which means our findings should have broad applicability. The specific objectives of the study are (1) evaluate mussel survivorship and mark retention against 2 commonly used calcein dosages (125 mg L−1 and 250 mg L−1) and exposure periods (12- and 24-h immersion times); (2) test calcein’s usefulness in identifying annuli and validating age estimates of individual mussels; and (3) discuss the management and conservation implications of using fluorescent stains on unionid mussels.
Materials and methods
Study area description
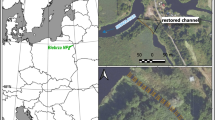
Individuals for this study were collected from the upper Sabine River in East Texas (Fig. 1). The Sabine River drains a total area of 25,267 km2 from its origin in Hunt County, Texas to the Gulf of Mexico21. The basin is heavily forested and is largely used for pasture, timber production, and livestock and poultry production22. The sample site was located within a riffle-run complex with gravel and sand substrate. Discharge at our sample site is characterized by high flows from December to February (averaging 37.71 m3s−1) and low flows in August and September (averaging 2.87 m3s−1), with a yearly average of 27.33 m3s-1 (USGS Gaging Station 08,019,200). Climate near the collection site is predominately humid and subtropical with an average annual rainfall of 1016 mm23 and an average annual temperature range of 1.1–34.4 °C22.
Map of study area showing location of collection site on the Sabine River in East Texas. Figure created using ArcMap 10.7.1 (https://www.esri.com/en-us/arcgis/products/arcgis-desktop/overview).
Study animals and collection
We evaluated calcein on C. pustulosa, which has a broad distribution in North America, ranging from the Nueces River basin in Texas to eastern reaches of the Great Lakes, Lake St. Clair, and Lake Erie, and is presently considered stable24,25. We chose this species because age (up to 48 years1) and growth (0.076–0.143 mm annually1) information is known. Additionally, its wide distribution means that our findings should have broad applicability. A total of 180 individuals were collected from our sample site on 7 December 2022 using a modified Surber sampler with a 0.25 m2 quadrat to excavate sediment to a depth of ~ 20 cm. Excavated sediment was passed through a 0.635 cm (0.25 inch) sieve to separate mussels. Collected individuals were wrapped in damp paper towels and transported in an insulated cooler to the Texas A&M AgriLife Research and Extension Center in Dallas, Texas. Upon arrival at the laboratory, mussels were held in quarantine for 3 weeks in a recirculating mesocosm system. Prior to the experiment, the mussels were weighed, measured for shell length, and tagged with a vinyl ID number (Hallprint) using super glue. All collected individuals were adults ranging between 40 and 70 mm in length. We chose this range in size based on specimen availability at our collection site and to keep sizes of individuals consistent between replicates.
Experimental design
We tested short- and long-term lethality of 2 different calcein dosages (125 mg L−1 and 250 mg L−1) and 2 different exposure periods (12- and 24-h immersion times)11,15,16,18. Immersion for all treatments began on 10 January 2023 and ended 11 January 2023. A 3-day acute and 10-day chronic observation period16 were used to measure immediate and short-term effects of calcein staining, which ended 14 January 2023 and 19 January 2023, respectively. After the 3-day observation period half of the individuals were culled for immediate analysis, and the other half was moved to recirculating aquaria for a 6-month period. This time, which ended on 10 July 2023, allowed us to evaluate long-term sublethal and lethal effects of staining and evaluate mark retention.
Collected individuals were randomly assigned to one of 4 experimental treatments and 2 controls, 10 individuals per replicate with 3 replicates per treatment and control (Fig. 2). All treatment individuals were immersed in calcein solutions of either 125 mg L−1 or 250 mg L−1 for 12 or 24 h. Controls were handled in the same manner as the treatments except for exposure to calcein. Calcein powder was acquired from Sigma-Aldrich, and the experimental solutions were made by weighing out either 1.25 g or 2.5 g of powder and adding it to reconstituted moderately hard water26 in an aerated container. These solutions were allowed to mix overnight before being used in the experiment. Concentrations and immersion times used in this study were based on published findings for freshwater and marine mollusks11,15,16,18.
Conceptual diagram showing experimental design used to test efficacy and survivorship of mussels exposed to two different calcein dosages (125 mg L−1 and 250 mg L−1) and two different exposure periods (12- and 24-h immersion times). Each box represents an initial replicate (n = 10) used in calcein staining. Replicates for each treatment are labeled R1, R2, or R3.
We exposed individuals to calcein by placing each replicate in its own opaque tub with 10 L of moderate hard water mixed with calcein to the appropriate concentration and aerated with air stones. Water temperatures in the tubs were approximately 20 °C and mussels were fed 3 mL of a 50/50 mix Shellfish/Nano3600 diet (LPBtm Frozen Shellfish Diet® and Nanno3600tm, Reed Mariculture, Campbell, California) daily. Water quality parameters (ammonia, nitrite, and nitrate) were monitored daily and kept within ASTM27 ranges. At the 12-h mark all 12-h replicate individuals were removed from the immersion tubs and rinsed with reverse osmosis (RO) water. The immersion tubs were drained and refilled with 10 L of moderate hard water, and individuals were placed back in their respective tubs for a 3-day observation period. A complete water change was executed at the 48-h mark of the observation period. At the end of the 3 days half of each replicate was culled for immediate mark analysis and half was moved to aquariums for long-term maintenance and observation for sublethal and lethal effects of the calcein marking. Aquariums were outfitted with filters, PVC water recirculating frames, and gravel substrate. This same process was repeated for all 24-h replicates.
Evaluating mortality
During the long-term maintenance period aquaria were checked daily for mortality. A mussel was considered deceased if it was excessively gaping and unable to close its shell following physical stimulus. Dead mussels were removed from their respective tanks and placed in ethanol until they could be thin-sectioned. A 50% water change was performed on the tank in which the death occurred to maintain water quality.
Mortality statistics
We used a one-way ANOVA on survivorship data from the 6-month observation period to evaluate whether the 2 different calcein dosages (125 mg L−1 and 250 mg L−1) and 2 different exposure periods (12- and 24-h immersion times) had an effect on mussel survival. The proportion of individuals that survived were arcsin(x) transformed before analysis. Any significant treatment effects (P < 0.05) were further analyzed through a Bonferonni post hoc analysis to determine which treatments were different from each other.
Thin sectioning to evaluate fluorescent marking and growth
To evaluate mark retention and determine if calcein can be used for validating age estimates of mussels we thin-sectioned individuals following Haag and Commens-Carson7. Specifically, shells were cleaned and dried, then set in epoxy resin (Craft Resin Creative Liquid) on popsicle sticks. Once the resin was fully cured, radial thin sections were cut along the dorsal–ventral axis with a Buehler IsoMet 1000 Precision Cutter low speed saw equipped with a diamond wafering blade (12.7 mm)7. Thin sections were mounted on glass microscope slides with Crystalbond 509 clear mounting adhesive (SPI® Supplies, West Chester, Pennsylvania) and sanded with progressively finer grits of wet-sanding blocks until annuli were clearly visible under light microscopy. Annuli for each thin-section were identified using the following criteria described by Haag & Commens-Carson7. Specifically, a putative annulus must extend from the umbo to the periostracum (exterior layer) without interruption and terminate through the prismatic layer. A second counter was used to independently age each shell to validate annuli. Discrepancies between the readers were either reconciled or individual specimens with incongruence were removed from the data set.
Once annuli were identified using light microscopy, a UV light system was used to evaluate calcein marking. Calcein fluoresces bright green at 490 nm, which we accomplished using a Nightsea Stereo Microscope Fluorescence adapter with an Olympus stereo microscope. Individual thin-sections were evaluated for the quality of the calcein mark and to determine whether the staining highlighted annuli visible under the microscope light by viewing the section under both the UV light and the normal microscope light and comparing the visibility of the historical annuli. Marked thin sections were also given a yes/no designation based on whether a clear “etch” mark created by the calcein could be identified at the growing edge of the sections12. Individuals in which growth could be observed relative to calcein etching were measured using a Moticam ProS5 Lite camera with Motic Images Plus 3.0 software, and growth measurements were analyzed using one-way ANOVA to identify any significant differences in growth between treatments.
Results
Mussel survivorship
No death or signs of stress such as gaping were observed in the 3-day observation period immediately post-immersion, and only 1 mussel failed to survive the 10-day chronic period. Cumulative mortality over the 6-month maintenance period across all treatments and exposure times was 16.7% of maintained individuals. Both the 12- and 24- h exposures at 125 mg L−1 had 20% mortality. Treatment mortality for 250 mg L−1 at 12 h was 13.3%, while 250 mg L−1 at 24 h was 26.7%. Control mortality was 13.3% for the 12-h control and 6.7% for the 24-h control (Table 1). There were no significant treatment effects on survivorship (F = 0.412; DF = 5; P = 0.831) when comparing dosage and exposure using an ANOVA (Fig. 3). Since there were no significant effects no further analysis was conducted. Several individuals that did not survive, regardless of treatment, appeared moribund weeks before they died, yet continued to respond to physical stimulus. This behavior was in stark contrast to individuals that died in controls, which did not show any evidence of prolonged stress prior to death.
Bar graph of 12- and 24-h treatments (125 mg L−1 and 250 mg L−1) and controls with 95% confidence intervals showing survivorship over 6 months as a proportion.
Marking quality, retention, and growth
All individuals showed evidence of calcein marking upon thin sectioning and analysis, both the cohort culled immediately post-immersion and those maintained for 6 months (Fig. 4). The staining was most prominent as an “etch” line at the growing edge of the shell, which was visible in 83.05% of individuals, but could also be seen in portions of the thin section where calcein had permeated down through portions of the periostracum (Fig. 5). There were no statistically significant differences in growth between treatments per the one-way ANOVA that was conducted, but the 250 mg L−1 for 12 h appeared to produce more vivid marks. Measurable growth occurred for 59 individuals with calcein etching, averaging 0.057 mm with a standard error of 0.007 mm (Table 1). There was no significant treatment effects on growth (F = 0.404; DF = 3; P = 0.754) when dosage and exposure were compared using an ANOVA (Fig. 6).
Pictures of etch marks resulting from calcein staining. Etching is indicated by red arrows. The left panel shows etching immediately post-immersion, and the right panel shows etching after six months of growth. Black marks on the shells indicate annuli. Images marked A represent the 125 mg L−1 concentration at a 12-h immersion, images marked B are 125 mg L−1 for 24 h, images marked C are 250 mg L−1 for 12 h, and images marked D are 250 mg L−1 for 24 h.
Two images of the same thin-section from the 250 mg L−1 × 12-h treatment. The top image is of the slide illuminated by UV light at 490 nm with red arrows indicating where calcein can be seen to have etched at the growing edge and seeped into portions of the shell. The bottom image is of the slide under ambient light with three of the annuli indicated with red arrows. Annuli are indicated in both images by pencil marks. Individual was determined to be 20 years old.
A bar graph of all staining treatments (12- and 24-h and 125 mg L−1 and 250 mg L−1; Total N = 59) with 95% confidence intervals showing average growth across treatments in mm.
Validation of annuli
All but 3 individuals were successfully aged, and ages ranged from 10 to 31 years with an average of 18.19 (standard error = 0.249). Calcein was ineffective for validating age estimates derived from thin-sectioning as applied in this study. We found that calcein staining did not highlight historical annuli in a manner that verified that the lines were annuli and was limited to etching at the growing edge and weak portions of the shell. For the latter, the stain appeared to penetrate from the outside of the shell, rather than be incorporated at the growing edge like the etch mark. This occurred in portions of the shell that appeared to have been eroded over time. The staining was typically most prominent near the umbo but could occasionally be seen towards the growing edge in especially damaged shells. (Fig. 5).
Discussion
The efficacy of 2 different calcein dosages (125 mg L−1 and 250 mg L−1) and 2 different exposure periods (12- and 24-h immersion times) in etching shells were successfully evaluated for adult C. pustulosa, which is a long-lived, widely distributed mussel species in North America. Thus, our findings should be applicable for congeners and other wide-ranging mussel species, especially those with similar life history characteristics. Individuals stained with calcein experienced little to no adverse effects on survival and produced fluorescent marking at the posterior margin of the shell, which we found to be most vivid for the 250 mg L−1 dosage and the 12-h immersion period. After 6 months, mark retention remained high as 83.05% of individuals retained their fluorescent marking on shells. Comparing calcein marks with annuli identified via shell thin-sectioning showed very little congruence, indicating this technique did not highlight historical annuli and was not useful for validating past annuli. Taken together, these findings indicate that calcein is generally non-lethal for adult mussels and produces a fluorescent mark on the inside of the shell that is long-lasting, which could be useful for evaluating mussel growth and survivorship.
Although calcein marking has not been widely used on unionid mussels, it has been used more frequently on marine mollusks (Supplementary Table 1). For marine burrowing bivalves optimal calcein concentrations range from 50 to 100 mg L−19, which is lower than concentrations used for unionid mussels, though immersion times in these studies were often greater. For unionid mussels, tested concentrations of calcein range from 125 mg L−1 to 250 mg L−1 for early juveniles. When tested on individuals older than 2 months, 250 mg L−1 at a 6- to 24-h immersion had less than 5% mortality and produced calcein marks that were readily identifiable 6 to 12 months post-immersion18. This is consistent with the findings from the present study, indicating 250 mg L−1 and a 12- or 24-h immersion period are suitable calcein concentrations for both juvenile and adult unionid mussels. Interestingly, freshwater hyriid mussels, which are related to our focal species, have shown mixed results when tested at 250 mg L−1 for 3 h28. Under this concentration and immersion individuals showed faint staining inside the shell but only a few had a definitive fluorescent mark, which is presumably due to inadequate immersion time.
Growth of mussels after being marked with calcein is important for its practical application in conservation and natural resource management. For unionid mussels, growth is often used to evaluate population performance (i.e., survival, growth, and reproduction), which is important for measuring health and resilience through time29. For marine mussels, fluorochrome marking appears to have generally negligible impacts on growth9, which mirrors findings for early age juvenile unionids. For example, 4- to 5-mo-old L. cardium immersed for 6 and 12 h in 250 mg L−1 calcein showed a 633% (3 to 22 mm) and 320% (5 to 21 mm) increase in maximum shell length (mm), respectively, 5 to 6 months post-immersion18. However, 7-mo-olds immersed for 24 h in 250 mg L−1 only showed a 31% (13 to 17 mm) increase in length18. Mortality across the three age-groups in that study was minimal (less than 4%18), which suggests the amount of expected growth post-immersion is dependent on size of the individuals, with smaller juveniles growing more than larger juveniles. Interestingly, life history may also play a role in growth post-immersion as species considered fast growing (i.e., L. cardium) had greater post-immersion increase, 31% (13 to 17 mm), in maximum shell length (mm) than those considered slow growing (i.e., A. pectorosa) at 7-mo-old18. In this study, we used adults of C. pustulosa, which is considered slow growing, so measuring changes in shell length (mm) over a six-month period was only feasible for marked individuals where growth past the calcein mark could be measured under a microscope. However, after the six-month monitoring period, all marked individuals, irrespective of treatment, showed growth that could be measured under a microscope after the calcein mark (averaging 0.057 mm with a standard error of 0.007 mm). This finding would indicate that growth in juveniles and adults is likely unaffected by the tested calcein and immersion concentrations, but does not evaluate whether there were any laboratory effects on the observed growth, either positive or negative.
The findings of this study combined with those of Eads and Layzer18 indicate that calcein immersion is effective and non-lethal for marking unionid adults and juveniles. To date, the species tested with calcein are not species of conservation concern so additional studies are needed to test multiple life stages of threatened species or closely related congeners. Also, all of the studies evaluating the effectiveness of calcein have focused on survivorship and growth so impacts to mussel reproduction remain unknown. Unionid mussels possess a unique reproductive life history involving larval (hereafter glochidia) parasitism, typically of fish29. Future studies could begin addressing potential impacts of calcein to reproduction by immersing gravid females with known glochidia viability for 12- or 24-h at 125 mg L−1 or 250 mg L−1 calcein and then comparing viability afterwards. Other studies could look at the impact of calcein on transformation success by immersing glochidia and host fish simultaneously to different concentrations and exposure periods. Metamorphosis rate, measured as the number of live juveniles divided by the sum of live juveniles and sloughed glochidia30, could be compared afterwards. Finally, in situ mark-recapture studies using individuals exposed to calcein are needed to determine how useful calcein can be used in validating age and longevity estimates.
Conclusion
The ability to identify fluorescence marks on the outside of the shell broadens the application of this method to evaluating stocking of mussels, a point made by Eads and Layzer18. Ideally, juveniles or adults raised in a hatchery or translocated would be stained and then released for reintroduction or to supplement an existing population. Subsequent monitoring efforts could easily track the performance of those individuals using UV light and growth could be measured for vouchered individuals using thin-sectioning. For example, calcein staining to monitor stocking efforts for Chinook salmon (Oncorhynchus tshawytscha) and rainbow trout (O. mykiss) effectively used a handheld light to identify marked individuals for nearly 3 years afterwards31. Pediveligers (i.e., larvae) of the eastern oyster, Crassostrea virginica, exposed to calcein maintained their marks through transformation and settlement and were identifiable afterwards using blue light (490 nm) without sacrificing the individuals32. If calcein is found to have minimal to no impacts on mussel reproduction then a similar approach could be taken, which would save time and funding because conventional marking relies on gluing vinyl or PIT tags onto the outside of the shell of a live individual. Depending on the number of individuals, which often can be in the thousands, this process can encumber a significant amount of resources. Additionally, emerging techniques focused on minimally destructive sampling33 for age and growth analysis could potentially be used in conjunction with calcein staining to increase the accuracy of the generated estimates.
Data availability
Datasets generated during the current study may be made available by the corresponding author upon reasonable request.
References
Haag, W. R. & Rypel, A. L. Growth and longevity in freshwater mussels: evolutionary and conservation implications. Biol. Rev. 86, 225–247 (2011).
Piovesan, G. & Biondi, F. On tree longevity. New Phytol. 231, 1318–1337 (2021).
Isely, J. & Grabowski, T. Analysis and Interpretation of Freshwater Fisheries Data: Age and Growth (American Fisheries Society, 2007).
Olden, J. D., Poff, N. L. & Bestgen, K. R. Life-history strategies predict fish invasions and extirpations in the Colorado River basin. Ecol. Monogr. 76, 25–40 (2006).
Winemiller, K. O. Life history strategies, population regulation, and implications for fisheries management. Can. J. Fish. Aquat. Sci. 62, 872–885 (2005).
Winemiller, K. O., Fitzgerald, D. B., Bower, L. M. & Pianka, E. R. Functional traits, convergent evolution, and periodic tables of niches. Ecol. Lett. 18, 737–751 (2015).
Haag, W. R. & Commens-Carson, A. M. Testing the assumption of annual shell ring deposition in freshwater mussels. Can. J. Fish. Aquat. Sci. 65, 493–508 (2008).
Neves, R. & Moyer, S. Evaluation of techniques for age determination of freshwater mussels (unionidae). Am. Malacol. Bull. 6, 179–188 (1988).
Wu, L. et al. Evaluation of three fluorochromes as in situ growth markers in Manila clam Ruditapes philippinarum. Aquac. Rep. 28, 101463 (2023).
Wilson, C., Beckman, D. & Dean, J. M. Calcein as a fluorescent marker of otoliths of larval and juvenile fish. Trans. Am. Fish. Soc. 116, 668–670 (1987).
Day, R., Williams, M. & Hawkes, G. A comparison of fluorochromes for marking abalone shells. Mar. Freshw. Res. 46, 599 (1995).
Van Der Geest, M., Van Gils, J. A., Van Der Meer, J., Olff, H. & Piersma, T. Suitability of calcein as an in situ growth marker in burrowing bivalves. J. Exp. Mar. Biol. Ecol. 399, 1–7 (2011).
Kaehler, S. & McQuaid, C. D. Use of the fluorochrome calcein as an in situ growth marker in the brown mussel Perna perna. Mar. Biol. 133, 455–460 (1999).
Cáceres-Puig, J. I., Huato-Soberanis, L., Melo-Barrera, F. N. & Saucedo, P. E. Use of calcein to estimate and validate age in juveniles of the winged pearl oyster Pteria sterna. Aquat. Living Resour. 24, 329–335 (2011).
Linard, C. et al. Calcein staining of calcified structures in pearl oyster Pinctada margaritifera and the effect of food resource level on shell growth. Aquaculture 313, 149–155 (2011).
Spires, J. E. & North, E. W. Marking the shells of juvenile and adult eastern oysters, Crassostrea virginica, with the fluorochrome dye calcein and measuring growth and mortality after marking. J. Molluscan Stud. 88, eyac004 (2022).
Merk, V. et al. Potential of calcein staining as growth monitoring marker in Ostrea edulis. Aquat. Living Resour. 36, 17 (2023).
Eads, C. B. & Layzer, J. B. How to pick your mussels out of a crowd: Using fluorescence to mark juvenile freshwater mussels. J. N. Am. Benthol. Soc. 21, 476–486 (2002).
Haag, W. R. & Williams, J. D. Biodiversity on the brink: An assessment of conservation strategies for North American freshwater mussels. Hydrobiologia 735, 45–60 (2014).
Galbraith, H. S., Spooner, D. E. & Vaughn, C. C. Synergistic effects of regional climate patterns and local water management on freshwater mussel communities. Biol. Conserv. 143, 1175–1183 (2010).
Texas Commision on Environmental Quality (TCEQ). Basin Narrative Summary. https://wayback.archive-it.org/414/20190908091155/https://www.tceq.texas.gov/assets/public/waterquality/swqm/assess/02twqi/basin5.pdf (2002).
Griffith, G., Bryce, S., Omernik, J. & Rogers, A. Ecoregions of Texas. (2007).
Sabine River Authority. Sabine River Basin Highlights 2021. https://www.sratx.org/wp-content/uploads/2021-Sabine-Basin-Highlights.pdf (2021).
Cordeiro, J. & Ormes, M. NatureServe Network Biodiversity Location Data: Pustulosa pustulosa. https://explorer.natureserve.org/Taxon/ELEMENT_GLOBAL.2.1156878/Pustulosa_pustulosa (2020).
Randklev, C. R. et al. Mussels of Texas Project Database. (2023).
Smith, M. E., Lazorchak, J. M., Herrin, L. E., Brewer-Swartz, S. & Thoeny, W. T. A reformulated, reconstituted water for testing the freshwater amphipod, Hyalella azteca.
ASTM International. Standard guide for conducting laboratory toxicity tests with freshwater mussels. In: In Annual Book of ASTM Standards vol. 11.06 1393–1444 (American Society of Testing Materials International, 2006).
Klunzinger, M. W., Beatty, S. J., Morgan, D. L., Lymbery, A. J. & Haag, W. R. Age and growth in the Australian freshwater mussel, Westralunio carteri, with an evaluation of the fluorochrome calcein for validating the assumption of annulus formation. Freshw. Sci. 33, 1127–1135 (2014).
Haag, W. R. North American Freshwater Mussels: Natural History, Ecology, and Conservation (Cambridge University Press, 2012).
Dudding, J. et al. Host Fish Associations for Two Highly Imperiled Mussel Species from the Southwestern United States: Cyclonaias necki (Guadalupe Orb) and Fusconaia mitchelli (False Spike). Freshw. Mollusk Biol. Conserv. 22, 12–19 (2019).
Negus, M. T. & Tureson, F. T. Retention and nonlethal external detection of calcein marks in rainbow trout and chinook salmon. N. Am. J. Fish. Manag. 24, 741–747 (2004).
Spires, J. E., Dungan, C. F. & North, E. W. Marking the shells of Pediveliger eastern oysters crassostrea virginica, with a calcein fluorochrome dye. J. Shellfish Res. https://doi.org/10.2983/035.040.0304 (2022).
Nykänen, S., Taskinen, J., Hajisafarali, M. & Kuparinen, A. Growth and longevity of the endangered freshwater pearl mussel (Margaritifera margaritifera): Implications for conservation and management. Aquat. Conserv. Mar. Freshw. Ecosyst. 34, e4205 (2024).
Moran, A. & Marko, P. A simple technique for physical marking of larvae of marine bivalves. J. Shellfish Res. 24, 567–571 (2005).
Loría, P. L. M. & Huato-Soberanis, L. Efficacy of calcein and Coomassie Blue dyeing of shell growing-edges and micro growth-bands: Ageing juvenile of Pinctada mazatlanica (Pterioida: Pteriidae). Rev. Biol. Trop. 62, 969–76 (2014).
Fitzpatrick, M. P., Jeffs, A. G. & Dunphy, B. J. Efficacy of calcein as a chemical marker of green-lipped mussel (Perna canaliculus ) larvae and its potential use for tracking larval dispersal. Aquac. Res. 44, 345–353 (2013).
Zhou, S., Zhang, X., Li, W., Li, L. & Cai, X. Experimental evaluation of fluorescent (alizarin red S and calcein) and clip-tag markers for stock assessment of ark shell, Anadara broughtonii. Chin. J. Ocean. Limnol. 35, 265–274 (2017).
Gancel, H. N., Carmichael, R. H., Park, K., Krause, J. W. & Rikard, S. Field mark-recapture of calcein-stained larval oysters (Crassostrea virginica) in a freshwater-dominated estuary. Estuaries Coasts 42, 1558–1569 (2019).
Acknowledgements
We also thank Dorothea Mildenberger, Alexander Kiser, and Ajoi Harris from the Texas A&M Natural Resources Institute for their assistance with laboratory experiments. Three anonymous reviewers provided insightful comments and greatly improved the quality and clarity of the final manuscript.
Author information
Authors and Affiliations
Contributions
ALE contributed to experimental design and manuscript writing, generated, compiled, and analyzed data, and prepared all figures and tables. MD contributed to experimental design and manuscript writing. RRL, CRRandklev and CRRobertson contributed to funding acquisition. RRL and CRRobertson contributed to manuscript writing. CRRandklev contributed to experimental design, data analysis, and manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
Collection of adult mussels was covered under Texas Parks and Wildlife Scientific Collection Permit No. SPR-0511-142. All experimental protocols were approved by the Texas A&M University Institutional Animal Care and Use Committee and performed in accordance with relevant guidelines and regulations. No testing was conducted on vertebrates. All methods are reported in accordance with ARRIVE guidelines.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Eismont, A.L., deMoulpied, M., Robertson, C.R. et al. Evaluation of survivorship and annulus validation in calcein-stained freshwater unionid mussels. Sci Rep 14, 20659 (2024). https://doi.org/10.1038/s41598-024-71597-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71597-8