Abstract
The misuse and overtreatment of antibiotics in hospitalized patients with community-acquired pneumonia (CAP) can cause multi-drug resistance and worsen clinical outcomes. We aimed to analyze the trends and appropriateness of antibiotic changes in hospitalized patients with CAP and their impact on clinical outcomes. This retrospective study enrolled patients with CAP, aged > 18 years, admitted from January 2017 to December 2021 at Seoul National University Bundang Hospital, South Korea. We examined the pathogens identified, antibiotics prescribed, and the appropriateness of antibiotic changes as reviewed by infectious disease specialists. Antibiotic appropriateness was assessed based on adherence to the 2019 ATS/IDSA guidelines and the 2018 Korean national guidelines for CAP, targeting appropriate pathogens, proper route, dosage, and duration of therapy. Outcomes measured included time to clinical stability (TCS), length of hospital stay, duration of antibiotic treatment, and in-hospital mortality. The study included 436 patients with a mean age of 72.11 years, of whom 35.1% were male. The average duration of antibiotic treatment was 13.5 days. More than 55% of patients experienced at least one antibiotic change, and 21.7% had consecutive changes. Throughout their hospital stay, 273 patients (62.6%) received appropriate antibiotic treatment, while 163 patients (37.4%) received at least one inappropriate antibiotic prescription. Those who received at least one inappropriate prescription experienced longer antibiotic treatment durations and extended hospital stays, despite having similar TCS. In conclusion, inappropriate antibiotic prescribing in hospitalized patients with CAP is associated with prolonged antibiotic treatment and increased length of stay. Emphasizing the appropriate initial antibiotic selection may help mitigate these negative effects.
Similar content being viewed by others
Introduction
Community-acquired pneumonia (CAP) is a major cause of death, ranking third in South Korea in 20211. Clinicians often overuse antibiotics in pneumonia treatment because of the high morbidity and mortality rates and concerns of treatment failure or recurrence2,3. This overuse raises concerns about antibiotic side effects and the emergence of antibiotic-resistant bacteria.
Shorter-duration antibiotic therapy has been recommended as an effective approach for most patients, reducing antimicrobial resistance, medical costs, and adverse effects4,5,6. Although official guidelines on empirical antibiotic use have been proposed, clinicians may deviate from these recommendations owing to their prescribing habits or limited availability of specific antibiotic agents. Consequently, patients may receive excessive antibiotic therapy, even in cases of non-severe CAP. Moreover, individual preferences and practices, especially during hand-offs, often lead to notable changes in antibiotic prescriptions, which can negatively affect the patient’s prognosis. However, data on the effects of antibiotic changes are currently lacking.
In 2020, South Korea implemented the hospitalist system, where internists specialize in the care of hospitalized patients7. Given the shift-based scheduling of hospitalists, multiple hand-offs occur, potentially resulting in several changes in antibiotic prescriptions during patient care. Therefore, this study aimed to analyze the trends in antibiotic prescriptions and cases of antibiotic changes in patients with CAP. The appropriateness of these changes were evaluated and the impact of inadequate prescriptions and changes on patient outcomes were determined.
Methods
Study design and patients
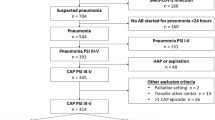
We retrospectively reviewed data of patients aged > 18 years hospitalized for CAP in the acute medical care unit of Seoul National University Bundang Hospital (SNUBH), South Korea between January 2017 and December 2021. Internists primarily managed the treatment of pneumonia. During the patient’s hospitalization, the internist in charge could change according to the schedule. Therefore, the antibiotics initiated by the previous internist were either continued, modified, or adjusted based on the evaluation of the new internist in charge. When necessary, treatment was administered in consultation with specialists in infectious disease and respiratory medicine at the discretion of the attending physician. Patients who were discharged or transferred to other departments or died within 48 h of admission, transferred from another department after 48 h of admission, and diagnosed with hospital-acquired pneumonia were excluded.
Definitions
A definite CAP diagnosis required the presence of a new pulmonary infiltrate on chest radiography and at least one additional lower respiratory infection symptom and physical examination finding: fever > 38 °C, cough, sputum, dyspnea, tachypnea, white blood count > 12,000 mm3 or < 6,000 mm3, abnormal lung sounds, and mental changes in patients aged > 70 years8. CAP severity was assessed using well-validated disease severity classification systems, including confusion, blood urea, respiratory rate, blood pressure, > 65 years old (CURB-65) scale; sequential organ failure assessment scale; and pneumonia severity index (PSI)9,10,11.
Healthcare-associated pneumonia (HCAP) characterizes pneumonia in individuals with significant healthcare exposure12. Individuals meeting HCAP criteria are identified as having a lower likelihood of being infected with multidrug-resistant pathogens. This recognition prompted the removal of the HCAP concept from the 2016 guidelines, with the intention that its principles would be assimilated into the CAP guidelines13,14. However, given prior studies highlighting the frequent use of broad-spectrum antibiotics among patients classified as HCAP15, this study analyzed patients previously categorized as HCAP both collectively and individually. This approach aimed to assess whether there have been changes in antibiotic prescribing practices over a 5-year period in real-world clinical settings.
Long-term acute care hospitals are defined as facilities that specialize in treating patients with serious medical conditions that require ongoing care but no longer require intensive care or extended diagnostic procedures, with an average length of stay (LOS) ≥ 25 days16.
The criteria for clinical stability were as follows: being afebrile (body temperature < 37.9 °C for > 48 h), a reduction in heart rate to < 100 beats/min and respiratory rate to < 24 breaths/min, elevation of systolic blood pressure to > 90 mmHg O2 saturation to > 90%17. Patients were considered stable if they remained afebrile for > 48 h and exhibited stability in three or all vital signs.
Side effects from antibiotic treatment were investigated through symptoms, laboratory abnormalities, or consultation reports from allergy specialists through chart reviews. Symptoms categorized as side effects included gastrointestinal problems (antibiotic-associated diarrhea, vomiting, and nausea), skin problems (itching, eruption, and allergic reactions), and drug fever. Laboratory abnormalities were defined as renal dysfunction, elevated liver function tests, and complete blood count abnormalities (neutropenia and thrombocytopenia), which are presumed to be associated with antibiotic use.
Antibiotics appropriateness
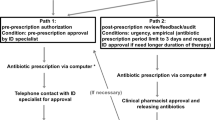
The Antimicrobial Stewardship Program (ASP) at SNUBH includes prior authorization for restricted antimicrobials, mandatory infectious disease physician consultation for patients with bacteremia, and daily monitoring of antibiotic prescriptions exceeding two weeks. Additionally, infectious disease physicians provide regular educational sessions for clinicians and distribute antibiotic dosing booklets based on renal function to support optimal antibiotic use18. The ASP program was also in place during the study period. The assessment of antibiotic prescriptions or modifications was conducted collaboratively by an infectious disease specialist and an antibiotic stewardship pharmacist through a thorough review of collected data. The appropriateness of antibiotic usage was evaluated at each point of antibiotic change, considering the patient’s clinical status and microbiology data available at that time. In instances where there was disagreement between the specialists, further consultation was sought from additional infectious disease or respiratory specialists to reach a consensus. This rigorous process ensured that decisions regarding antibiotic appropriateness were comprehensive and based on expert clinical judgment. Prescription appropriateness was evaluated based on several specific criteria: (1) initial empiric antibiotics adherence to established 2019 ATS/IDSA guidelines19 as well as the 2018 Korean national guidelines20 for the treatment of CAP; (2) modification to the antibiotic regimen were based on microbiological test results, such as culture and sensitivity data, to either de-escalate to a narrower spectrum antibiotics or escalate to a broader spectrum antibiotics if needed; (3) changes based on the clinical course of the patient were considered appropriate if they involved adjusting therapy in response to the patient’s clinical progression, such as worsening symptoms or lack of improvement, which could indicate the need of broader coverage or alternative agents; and (4) administration of antibiotics suitable for the identified or suspected pathogens, delivered through the appropriate route (oral or intravenous) and at an effective dosage, in accordance with the 2019 ATS/IDSA guidelines and the 2018 Korean national guidelines for CAP19,20,21; and (5) antibiotic therapy should be continued until the patient achieves stability and for ≥ 5 days. Prescriptions exceeding 14 days were evaluated for potential early discontinuation. In exceptional cases, evaluation was made based on an infectious disease specialist’s opinion. Antibiotic prescription or modification was deemed appropriate only when all these conditions were met. Conversely, instances where any of these criteria were not satisfied, or when unnecessary combination medication was employed, resulted in the evaluation of antibiotic prescription or modification as inappropriate22.
Clinical assessment
Demographic data (age, sex, underlying disease, and Charlson Comorbidity Score (CCS) and laboratory data (hemoglobin, C-reactive protein, and procalcitonin levels) of patients were collected. Blood and sputum culture tests, sensitivity of the isolated microorganisms to the antibiotics tested, and respiratory virus cultures; urinary antigen testing for Streptococcus pneumoniae; serum antibody testing for Legionella spp.; and bronchoscopy cultures were investigated to identify infections. Viral cultures were not routinely performed unless clinically indicated. Bronchoscopic examinations were performed only on the recommendation of a respiratory specialist in cases of atypical symptoms or non-resolving pneumonia. When a microbiology test was performed again within 48 h of admission, the second test’s results were also reviewed. Information collected on antibiotic treatments encompassed the type of agent, dosage, treatment duration, any changes in agents, and the route of administration.
Clinical outcomes
We determined whether several outcomes varied based on changes in antibiotics and their appropriateness. The primary outcome assessed was time to clinical stability (TCS), defined as the duration required to normalize unstable vital signs observed upon admission. TCS was calculated as the number of days from the date of hospital admission to the date that the patient met clinical stability criteria.
For secondary outcomes, we analyzed the duration of antibiotic usage, including intravenous use and change to oral agents, LOS in the hospital, and in-hospital mortality. The duration of antibiotic usage was quantified by the number of days, distinguishing between intravenous and oral agents, starting from the initial antibiotics administered in the emergency room (ER) to the oral antibiotics prescribed post-discharge. LOS in the hospital was calculated by combining the number of days spent in the ER and subsequent days spent in the general ward. In-hospital mortality was defined as death from any cause occurring during the hospital stay.
This study was conducted in compliance with the Declaration of Helsinki and was approved by the Institutional Review Board (IRB) and Ethics Committee of Seoul National University Bundang Hospital. The informed consent was waived by IRB (IRB No. B-2202-736-103).
Statistical analysis
Statistical analysis was performed using R version 4.2.2. (R Core Team, Vienna, Austria), and graphs were created using the R package “ggplot2.” Descriptive data were expressed as mean (standard deviation) or count (%) unless otherwise specified. Student’s t-test was used to compare continuous variables, and the chi-square or Fisher’s exact test was used to compare categorical variables. Cox regression was conducted to evaluate the mortality according to antibiotics appropriateness with adjustments for demographic and laboratory data of patients.
Results
Patient characteristics
In total, 436 patients (average age, 72.11 [± 13.11] years; male, 35.1%) were reviewed (Table 1). The analysis revealed no significant differences in demographic characteristics and comorbid conditions when comparing the changes in the appropriateness of antibiotic prescriptions. Approximately half of the patients in this study (223 patients, 51.2%) met the criteria for HCAP and 21 patients (12.9%) patients were admitted from long-term acute care hospitals, with no significant difference in pneumonia severity.
Overall, 72.5% of patients had moderate-to-high PSI values, with meant PSI class III-V, and approximately 30% of patients had a CURB-65 score ≥ 3. The group with higher pneumonia severity showed a tendency towards having more inappropriate antibiotic prescriptions.
Identified microbiology and antibiotic treatment
The microbiology data of the patients are summarized in Supplementary Table S1. Sputum cultures predominantly identified Klebsiella spp., Pseudomonas spp., and S. aureus. MRSA was the most frequently detected multi-drug-resistant organism. Additionally, extended-spectrum beta-lactamase-positive gram-negative bacilli, carbapenem-resistant Pseudomonas aeruginosa, and carbapenem-resistant Acinetobacter baumannii were detected. These microbiological results were similar in patients with HCAP.
The antibiotic treatments and changes are listed in Table 2. Combination therapies of third-generation cephalosporin and azithromycin were most often chosen as the first-line antibiotic treatment (36.9%), and broad-spectrum antibiotics (piperacillin/tazobactam and carbapenem) were used in 24.6% of patients. More than half of the patients had their first antibiotic change after admission. Piperacillin/tazobactam (17.8%) was the most common initial change, followed by quinolones (15.8%). In subsequent changes, quinolones became the most frequently prescribed (25.3%).
Clinical outcomes
The average TCS was 3.29 (± 2.24) days. A variation in TCS was observed based on CAP severity; however, the difference was not significant. Patients received antibiotics for an average of 13.5 (± 4.13) days, comprising 7.76 (± 3.55) days of receiving intravenous agents and 6.26 (± 4.41) days of receiving oral agents.
The average LOS, encompassing ER stay, was 9.03 (± 4.08) days. The in-hospital all-cause mortality rate was 2.7% (12 cases), with 7 and 5 deaths in the severe CAP and non-severe CAP groups, respectively. Compared with patients without side effects, 29 (6.6%) patients with side effects showed a tendency for longer administration of intravenous antibiotics, although the difference was insignificant. However, patients experiencing side effects had a significantly longer LOS, expending 2–3 days more than those without side effects.
Appropriateness of antibiotic treatment
The information regarding antibiotics considered appropriate for initial treatment is summarized in Supplementary Table S2. Overall, antibiotic prescription was changed for 241 patients (55.3%) and 95 patients (21.8%) required a second change (Fig. 1). Initially, out of 436 patients, 302 (69.1%) received appropriate antibiotics prescriptions, while 134 (30.7%) received inappropriate ones, with piperacillin/tazobactam being the most frequently inappropriately prescribed antibiotics (40%) (Fig. 2). The first antibiotics change affected 241 patients (55.3%); among those initially appropriate, 140 (64.9%) were changed, with 111 (36.8%) remaining appropriate and 29 (9.6%) becoming inappropriate, while in the initially inappropriate group, 101 were changed, with 87 cases (64.9%) becoming appropriate and 14 cases (10.4%) remaining inappropriate. At this point, third-generation cephalosporin and fluoroquinolone (31%) were most frequently inappropriately prescribed in the initially appropriate group, while piperacillin/tazobactam and fluoroquinolone (57%) were most inappropriate in the initially inappropriate group. A second change was required for 95 patients (21.8%), resulting in 7 out of 14 cases (50%) from the initially inappropriate group becoming appropriate, in the group that appropriate, with only one case (3.4%) remaining inappropriate.
Appropriateness of initial and subsequent antibiotic changes.
Types and frequency of antibiotics based on initial and subsequent changes and appropriateness.
Most changes in antibiotics involved escalating the antibiotic class or adjusting for the identified microorganism, with 34 patients (31%) maintaining appropriateness throughout initial prescription and first change, although some changes remained inappropriate, as detailed in supplementary tables S3 and S4.
Effect of antibiotic alterations on clinical outcomes
Antibiotic appropriateness on clinical outcomes was investigated (Table 3). Data regarding the second change in antibiotics were not included in this outcome analysis due to the small numbers in each category. During their hospital stay, 273 patients (62.6%) received appropriate antibiotic treatment, whereas 163 patients (37.4%) received at least one inappropriate antibiotic prescription. There was no statistically significant difference in the TCS between cases that maintained appropriateness after the initial change (all-APP) and cases in which inappropriate antibiotics were administered more than once during the initial or subsequent changes (one-INAPP) (3.12 ± 1.88 vs. 3.34 ± 2.23 days, P = 0.261). Patients with one-INAPP antibiotics had significantly longer durations of intravenous antibiotic use and hospital stay than did those with all-APP (8.18 ± 3.57 vs. 7.27 ± 3.33 days, 9.59 ± 3.96 vs. 8.45 ± 3.94 days, P = 0.003). Overall, the use of one-INAPP antibiotics was associated with an extended duration of antibiotic use and LOS. When comparing the one-INAPP and all-APP groups, we found no significant differences in in-hospital mortality.
Discussion
This study highlights the significant impact of antibiotic appropriateness on clinical outcomes in hospitalized patients with CAP. Our findings indicate that over 55% of patients underwent at least one antibiotic change, with nearly 40% receiving at least one inappropriate antibiotic prescription during their hospital stay. This high rate of antibiotic changes and inappropriate prescriptions underscores the complexity of managing CAP and the need for stringent antibiotic stewardship23,24. The most common reason for the inappropriate initial antibiotic prescription was the use of broad-spectrum antibiotics that did not match the severity of the illness or past microbiological results. This inappropriate usage often led to extended intravenous antibiotic treatment and prolonged hospital stays, consistent with previous studies that have linked to inappropriate antibiotic use to adverse clinical outcomes, including the emergence of multi-drug resistant organisms and increased mortality.
Interestingly, inappropriate antibiotic changes often involved switching to broad-spectrum antibiotics unnecessarily, even when initial treatments were appropriate. This pattern was particularly evident in patients with HCAP, despite recent guideline updates excluding HCAP as a distinct category. This finding suggests a persistent trend toward overprescribing broad-spectrum antibiotics, highlighting the need for continuous education and reinforcement of guideline-based practices among healthcare providers.
Our study also found that patients who received at least one inappropriate antibiotic change had prolonged total courses of antibiotic therapy and longer hospital stays compared to those who received appropriate antibiotics throughout their treatment. This extended duration of therapy, often exceeding the recommended 3–5 days, reflects a cautious approach by physicians who may prioritize comprehensive care and prevention of readmissions over adherence to guidelines .
The implementation of an ASP is crucial in addressing these issues. ASPs aim to optimize antibiotic use, reduce inappropriate prescriptions, and ultimately improve patient outcomes25,26,27. Our study found that the rate of inappropriate antibiotic use decreased to 64.9% when patients were referred to internal medicine specialists, who appropriately changed the antibiotics in these cases. Among these appropriate changes, 23.7% involved switching to a narrow-spectrum antibiotic, and 34% were based on microbiological results. However, 24.6% of inappropriate antibiotics were not changed, and even when the initial antibiotic was appropriate, 9.6% of patients were switched to inappropriate antibiotics, 82.7% of which were broad-spectrum antibiotics. Despite the presence of an ASP, our study suggests that decisions regarding antibiotic type and duration still heavily rely on individual physician’s judgment, which may not always align with guideline recommendations. Enhancing the role of ASPs and ensuring adherence to evidence-based guideline can mitigate the risks associated with inappropriate antibiotic use.
To the best of our knowledge, this is the first study to evaluate the appropriateness of antibiotic changes and their impact on the clinical course of patients with CAP. We conducted a comprehensive review of the patients’ clinical data, including microbiological results and antibiotic side effects. However, this study has limitations, including its retrospective design, single-center setting, and relatively small cohort size. Additionally, we did not evaluate the readmission rates or long-term mortality which are important aspects of patient outcomes. Future multicenter studies with larger cohorts are needed to further investigate the appropriateness of antibiotic prescriptions and their impact on clinical outcomes in patient with CAP.
In conclusion, this study demonstrates that inappropriate antibiotic prescribing is prevalent among hospitalized patients with CAP and is associated with prolonged antibiotic treatment and extended hospital stays. Emphasizing the importance of appropriate initial antibiotic selection and adherence to established guidelines can improve clinical outcomes and reduce the burden of inappropriate antibiotic use.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to restrictions on data outflow from the hospital, but are available from the corresponding author on reasonable request.
References
Korean Statistical Information Service. Death by cause. Seoul: Statistic Korea. (2023). https://kosis.kr/eng/. Accessed 27 Nov 2023.
Fridkin, S. et al. Vital signs: Improving antibiotic use among hospitalized patients. MMWR Morb. Mortal. Wkly. Rep.63, 194–200 (2014).
Niederman, M. S. et al. Guidelines for the initial management of adults with community-acquired pneumonia: Diagnosis, assessment of severity, and initial antimicrobial therapy. American Thoracic Society. Medical section of the American Lung Association. Am. Rev. Respir. Dis.148, 1418–1426 (1993).
Uranga, A. et al. Duration of antibiotic treatment in community-acquired pneumonia: A multicenter randomized clinical trial. JAMA Intern. Med.176, 1257–1265 (2016).
Haas, M. K. et al. Effects of a syndrome-specific antibiotic stewardship intervention for inpatient community-acquired pneumonia. Open. Forum Infect. Dis.3, ofw186 (2016).
Avdic, E. et al. Impact of an antimicrobial stewardship intervention on shortening the duration of therapy for community-acquired pneumonia. Clin. Infect. Dis.54, 1581–1587 (2012).
Chae, W. et al. Development and evolution of hospital medicine in Korea. J. Hosp. Med.16, 247–250 (2021).
Prina, E., Ranzani, O. T. & Torres, A. Community-acquired pneumonia. Lancet. 386, 1097–1108 (2015).
Marti, C. et al. Prediction of severe community-acquired pneumonia: A systematic review and meta-analysis. Crit. Care16, R141 (2012).
Asai, N. et al. Efficacy and accuracy of qSOFA and SOFA scores as prognostic tools for community-acquired and healthcare-associated pneumonia. Int. J. Infect. Dis.84, 89–96 (2019).
Fine, M. J. et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N. Engl. J. Med.336(4), 243–250 (1997).
American Thoracic Society, Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am. J. Respir. Crit. Care Med.171, 388–416 (2005).
Yap, V., Datta, D. & Metersky, M. L. Is the present definition of health care–associated pneumonia the best way to define risk of infection with antibiotic-resistant pathogens?. Infect. Dis. Clin. N. Am.27, 1–18 (2013).
Kalil, A. C., Metersky, M. L. & Klompas, M. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin. Infect. Dis.63, e61–111 (2016).
Vallés, J. et al. Epidemiology, antibiotic therapy and clinical outcomes of healthcare-associated pneumonia in critically ill patients: A Spanish cohort study. Intensive Care Med.40, 572–581 (2014).
Kahn, J. M., Benson, N. M., Appleby, D., Carson, S. S. & Iwashyna, T. J. Long-term acute care hospital utilization after critical illness. JAMA. 303, 2253–2259 (2010).
Halm, E. A. et al. Time to clinical stability in patients hospitalized with community-acquired pneumonia: implications for practice guidelines. JAMA. 279, 1452–1457 (1998).
Shin, D. H. et al. Stepwise expansion of antimicrobial stewardship programs and its impact on antibiotic use and resistance rates at a tertiary care hospital in Korea. Microbiol. Spectr.10(3), e0033522 (2022).
Metlay, J. P. et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am. J. Respir Crit. Care Med.200(7), e45–e67 (2019).
Lee, M. S. et al. Guideline for antibiotic use in adults with community-acquired pneumonia. Infect. Chemother.50, 160 (2018).
Gyssens, I. C. Quality measures of antimicrobial drug use. Int. J. Antimicrob. Agents. 17, 9–19 (2001).
Kariyawasam, R. M. et al. Antimicrobial resistance (AMR) in COVID-19 patients: A systematic review and meta-analysis (November 2019-June 2021). Antimicrob. Resist. Infect. Control11, 45 (2022).
Cdc, A. Antibiotic resistance threats in the United States. US Department of Health and Human Services. Published online 2019. (1996). http://www.hcfa.gov/stats/stathili.com. Accessed December 2.
Mahmood, R. K., Gillani, S. W., Alzaabi, M. J. & Gulam, S. M. Evaluation of inappropriate antibiotic prescribing and management through pharmacist-led antimicrobial stewardship programmes: A meta-analysis of evidence. Eur. J. Hosp. Pharm. Sci. Pract.29, 2–7 (2022).
Wagner, B. et al. Antimicrobial stewardship programs in inpatient hospital settings: A systematic review. Infect. Control Hosp. Epidemiol.235, 1209–1228 (2014).
Dik, J. W. H. et al. Financial evaluations of antibiotic stewardship programs—A systematic review. Front. Microbiol.6, 317 (2015).
Schuts, E. C. et al. Current evidence on hospital antimicrobial stewardship objectives: A systematic review and meta-analysis. Lancet Infect. Dis.16, 847–856 (2016).
Funding
This study was supported by a grant from the Seoul National University Bundang Hospital Research Fund (no. 02-2021-0039). The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Author information
Authors and Affiliations
Contributions
Conception and design: E.S.K. and R.R.; Analysis and interpretation of the data: E.S.K., E.S.K., J.R., S.Y., N.-H.K., J.H.O., Y.L., J.L., H.W.K., S.-W.K., H.-S.P. and E.H; Drafting of the article: J.R., E.S.K., and S.Y.; Critical revision of the article for important intellectual contents: E.S.K., J.R., S.Y., E.S.K., and N.-H.K.; Final approval of the article: J.R., N.-H.K., J.H.O., Y.L., J.L., H.W.K., S.-W.K., H.-S.K., E.S.K., S.Y., and E.S.K..
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ryu, J., Kim, NH., Ohn, J.H. et al. Impact of antibiotic changes on hospital stay and treatment duration in community-acquired pneumonia. Sci Rep 14, 22669 (2024). https://doi.org/10.1038/s41598-024-73304-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-73304-z