Abstract
In this study, adsorption of volatile organic compounds (VOCs) (here just gasoline vapor) by activated carbon- modified UIO-66 was investigated. First, activated carbon prepared from mesquite grain (ACPMG) and then UIO/ACPMG nanohybrid was synthesized by the solvothermal method. In following, the effect of main key parameters which effect on the surface and adsorption capacity such as the ratio of ACPMG to UIO-66 was studied. Physiochemical changes of as- synthesized samples were investigated by TGA, HR-TEM, PSD, SEM, EDX/MAP, BET, FT-IR, XRD, and XPS. It was found the UIO/ACPMG20% nanohybrid had the highest adsorption capacity (391.304 mg/g) for VOCs compared with the other samples, while the adsorption capacity of UIO-66, UIO/ACPMG10% nanohybrid, and UIO/ACPMG30% nanohybrid was 298.871, 309.523, and 320 mg/g respectively. UIO/ACPMG20% nanohybrid desorbed 285.71 mg/g of the adsorbed gasoline, which is an excellent result in desorption. So, the sample of UIO/ACPMG20% nanohybrid was selected as the optimum nano-adsorbent. In other hand, all the nano-adsorbent showed a rapid kinetic behavior for gasoline vapor adsorption and the maximum time for reaching a high adsorption capacity approximately was obtained in 20 min. Density functional theory calculations also performed to understand the adsorption characteristics of gasoline vapor on activated carbon-modified UIO-66.
Similar content being viewed by others
Introduction
VOCs are considered as a series of organic materials including methane, carbon monoxide, benzene, xylene, carbon dioxide, carbonic acid, toluene and ammonium carbonate, which are existed in our environment surrounding while accounting as a serious problem to the organism healthy in the global1,2. In general, VOCs may be categorized to three main groups. First group is VOCs based on the boiling point which vaporized below 150–250 °C. The second group is the VOCs that have molecular structure for example ethane, propane, methanol, etc. the third group is the VOCs that have molecular polarity such as acetone, benzene, toluene, etc3,4. also, according to the boiling point the volatile organic compounds can be placed within very VOCs, VOCs, semi VOCs and particulate organic substances5. With the ascending of industrialization and urbanization in the world, the amount of VOCs emission has been increased significantly. In general, the amount of VOCs emission sources of industrial process, vehicles, daily life, and agriculture are almost about 43%, 28%,15% and 14%, respectively. Volatile organic compounds based on the aromatic and polycyclic hydrocarbon compound are stinky, venomous and carcinogenic to organism safety even at low concentration, causing a recessional aspiration and skin mucosa and injury of neural and blood systems6.
Until now, many techniques have been established to eliminate the environmental contaminations, such as adsorption, photocatalytic, condensation, adsorption, advanced oxidation, incineration, and electrochemical photocatalytic method7,8. However Cost-effective, low energy consumption, and reuse potential, sustainable, and higher removal efficient method for eliminating VOCs are still accounted a serious issue and need more attention in this regard. Among them, adsorption method due to ease to perform, low cost, stability, and simplicity in design, and environment friendly nature, is extensively utilized to eliminate the VOCs9,10. The selection of suitable adsorbent is the key to arising a high removal yield for VOCs. The important adsorbents can refer to metal-organic frameworks (MOFs)11,12,13,14,15,16,17,18,19, zeolites, activated carbon (AC), alumina, and clay20,21,22. AC is the most popular adsorbent for VOC/SVOC adsorption because of its rich porous structure, large specific surface area, and chemical stability10. Among them MOFs and AC are attracted much attention in this field due to promising properties. ACs are manufactured from a variety of carbonaceous materials. Metal–organic frameworks (MOFs) are a new porous crystalline hybrid compound, generated from two main parts (metal ions called node are clusters which are attached to the organic linkers called bridge by non-covalent bindings in 1-3D networks23,24,25. It can be considered as a potential adsorbent for removal of VOCs because of their controllable pore shape, tunable pore sizes high specific surface area, low density, bio compatibility, extremely large pore volumes, and various means available for post-synthesis organic-functionalization and established remarkable progress in the application of gas/VOC adsorption26,27. MOFs can be utilized in many fields such as drug delivery, gas adsorption, sensing, water splitting, separation, optic works, and catalysis. Today, MOFs are used for removing of VOCs due to above mentioned features and published many literatures in this respect. Vellingiri et al. demonstrated that the adsorption capacity amount of UIO-66, MOF-199, MIL-101(Fe)) were calculated about 166, 159, and 98.3 mg/g, respectively28. Zhao et al. revealed that HKUST-1 can uptake the benzene at 298 K and is up to 10.0 mmol/g29. Hao et al. prepared nitrogen-doped porous carbon/ ZIF-11 and understood that after KOH activation, surface area of the compound enhanced from 895 m2/g to 2188 m2/g30.
In this work a UIO-66/ACPMG nanohybrid was introduced as a promising nanoadsorbent for air filtration from VOCs with high adsorption efficiency, and low energy consumption in environmental conditions. Furthermore, the diffusion mechanism of VOCs in this nano-adsorbent was investigated by the kinetic diffusion model and intraparticle diffusion.
The novelty of adsorption of VOCs by UIO-66/ACPMG nanohybrid lies in its unique combination of two highly effective adsorbents. MOFs are a class of porous materials with high surface areas and tunable pore sizes, making them excellent candidates for gas adsorption. Activated carbon, on the other hand, has been widely used for the removal of VOCs due to its high adsorption capacity and low cost. The UIO-66/ACPMG nanohybrid combines the advantages of both materials to create a highly efficient adsorbent for VOCs. The MOF component provides a large surface area and specific binding sites for the target VOCs, while the activated carbon component enhances the overall adsorption capacity and stability of the hybrid material. Furthermore, the synthesis of UIO-66/ACPMG nanohybrid is a relatively new approach that has not been extensively studied for VOC removal. Therefore, this technology offers a promising alternative to traditional adsorbents and has the potential to be applied in various industrial applications where VOC emissions are a concern.
Experimental section
Materials
Mesquite grain collected from Mehran, Ilam, Iran. All chemicals and solvents used in this study were purchased with high purity from Merck (Darmstadt, Germany) as follows phosphoric acid (H3PO4, 99%), hydrochloric acid (HCl, 37%), dimethylformamide (DMF, 99.5%), zirconium tetrachloride (ZrCl4), terephthalic acid (BDC).
Fabrication of ACPMG
ACPMG was prepared in two following steps:
-
1.
Activation step: in this step, mesquite grain was washed by distilled water and dried at 80 °C for overnight. Subsequently, the precursor was grinded and meshed with mesh of 160 μm. Then, 5 g of raw powder of precursor was mixed with 3.5 mL of H3PO4 and in the oven and keep overnight at 115 °C.
-
2.
Pyrolysis step: the materials obtained in the initial step were poured in the alumina ceramic boat crucible(10 cm×1 cm×1 cm), moved to a furan equipped by nitrogen flow, heated by rate 7 °C /min up to 600 °C, and kept 2 h in 600 °C. Then the sample was cooled until ambient temperature. For removing the residual H3PO4 and impurities, the sample was boiled in 70 mL HCl (10%) for 20 min to obtain a black powder of AC. In final the powder was filtered and washed by DI water to neutralize the pH. Then the produced AC was dried at 100 °C for 24 h.
Fabrication of UIO-66 nps
First, 0.124 g of ZrCl4(0.55 mmol), 10 mL of DMF, and 1 mL of HCl were added to a baker and sonicated for 25 min under ultrasonic bath. Simultaneously, in the other baker 0.123 g of BDC (0.75 mmol) were added in 5 mL of DMF and stirred until BDC was completely dissolved. Then two bakers were mixed together and sonicated for 20 min under ultrasonic bath. The obtained solution was stirred for 12 h at 85 °C to appear a milky solution. The milky solution was centrifuged with rate 6000 rpm, and then washed with DMF (three time) to gather the white precipitation. The white powder was dried at 90 °C for 24 h and then activated at 110 °C in a vacuum oven for overnight31.
Fabrication of UIO/ACPM nanohybrid
UIO/ACPMG nanohybrid was prapared based on pervious method. It was prepared by mixing 0.404 g of ZrCl4, 0.291 g of BDC, and 0.0157 g of ACPMG (the ratio of ACPMG/ Zr 0.1) in 25 mL of DMF. The synthesis step for UIO/ACPMG nanohybrid is similar to synthesis of UIO NPs. Synthesis of hybrid materials was carried out under different conditions to obtain the most efficient adsorbent with high adsorption capacity (Fig. 1).The ratio of ACPMG to Zr was 0.1, 0.2 and 0.3, the samples were named UIO/ACPMG10% nanohybrid, UIO/ACPMG20% nanohybrid and UIO/ACPMG30% nanohybrid, respectively32.
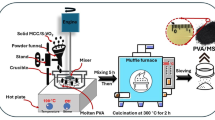
Schematic image of synthesis of ACPMG, UIO NPs, and UIO/ACPMG nanohybrid.
Experimental setup
The model of choice in this work is gasoline vapor, the main component of which is isobutane (ISO). According to the ASTM D-822,892 standard to calculate the dynamic adsorption treatment of VOCs as a function of time, an in-house built setup was designed and performed. the schematic of the setup is depicted in Fig. 2. This setup is capable to able to work under atmospheric pressure in two modes (adsorption and desorption). This setup is included of three important sections (a gas saturation tube, a U-shape adsorption column and a temperature controller system). In the adsorption mode, a mass flow controller (MFC-1) provided a constant flow rate of dried standard air (80–120 mL/min), as a carrier gas, for downstream facilities. In order to saturate carrier gas stream with a desired VOC sample, the outlet gas stream of MFC-1 was passed through a glassy gas saturation tube, containing the liquid VOC sample. Consequently, the VOC vapor was generated into the carrier air stream under atmospheric pressure. In such a situation, the con- centration of the VOC is directly proportional to its vapor pressure. Then, the outlet stream of the gas saturation tube entered to the U- shape adsorption column containing a weighted amount of adsorbent. The direction of the carrier gas in adsorption mode was downward, preventing adsorbent fluidization and providing sufficient contact time. By passing the gas mixture (i.e. air and VOC with equilibrium con- centration at 1 bar) through the U-shape adsorption column and its adsorption zone (see Fig. 2), only the VOC molecules were adsorbed and remained molecules left the zone and purged. In fact, it was as- sumed that the air adsorption on the adsorbent was negligible in the presence of VOC molecules, because of the significant difference in their molecular weights. Temperature of both the gas saturation tube and the U-shape adsorption column were controlled by using two glassy thermostatic jackets, connected to a main water circulator (model RK-20, Lauda- Brinkmann). The circulator provided a constant flow of DI water to jackets of the U-shape adsorption column and subsequently the saturation tube. Thus, the circulator was able to control the temperatures of both units at the same or any desired temperature in a range of 5–80 °C (with an accuracy of 1 °C). Heat transfer between the lines and the environment was negligible due to the meager approach temperature. Before each experiment, to remove adsorbed moisture and other undesired molecules, adsorbents were regenerated for 12 h (250 °C) under vacuum pressure. After a complete degasification, a weighted amount of selected adsorbent was placed in the U-shape adsorption column.
Points of the breakthrough curves were calculated by continuous recording of operating adsorbent weight at each time step. The experiment (e.g. adsorption or desorption) was ended up when a same weight (± 0.5%) was achieved at minimum three successive times.
Instead of weighing the adsorbents in each timed step, the U-shape package (containing U-shape adsorption column, adsorbents and adsorbed vapor of selected VOC) was weighed which substantially diminished the level of experimental uncertainty and errors. Since an isolated condition was needed for the U-shape package at each weighing step, two valves (V-7 and V-8) were located at top of the U-shape column, providing a complete disconnection between the U-shape package and other parts. Thus, the U-shape package and its contents were thoroughly isolated during each weighing process. Moreover, the weighting process was carried out with care to remove all water droplets at the exterior surface of the U-shape package. The amount of adsorbed VOC vapor at each time was calculated by following formula33:
The image schematic of VOC adsorption setup.
Computational details
To model the ACPMG substrate, a monolayer including carbon, oxygen and nitrogen atoms was utilized. The dangling bonds were avoided by adding hydrogen atoms to the edge atoms. The UIO NPs was simulated by a Zr6O4(OH)4cluster according to the previous study34. Then, the ACPMG-modified UIO NPs was obtained by placing the Zr6O4(OH)4cluster onto the ACPMG. Since the ISO is the main component of the gasoline vapor, the adsorption of a single ISO molecule on the ACPMG-modified UIO NPs was studied. All the geometry optimizations were performed with the PBE35functional and a double-numerical basis set with a polarization function (DNP). The weak van der waals interactions were included within the calculations by applying the Grimm’s D2 empirical dispersion correction36. The atomic charges were obtained by the Hirschfeld37 population analysis. All the calculations were performed using the DMol338.
To measure the strength of ISO adsorption on the ACPMG-modified UIO-66, the adsorption energy (Eads) was calculated as:
in the above equation, Etotal, EISO, and EmAC, respectively, represent the total energy of the complex (i.e., adsorbed ISO over ACPMG-modified UIO NPs), pristine activated carbon-modified UIO NPs and a free ISO molecule. A negative Eads value implies that the adsorption is exothermic, and therefore the obtained complex is thermodynamically desirable.
Result and discussion
Characterization
Figure 3A gives the FT-IR spectra of the UIO/ACPMG nanohybrid, UIO NPs, and ACPMG. As observed, the spectrum of UIO/ACPM nanohybrid has good accordance with the stimulation spectrum of UIO and ACPMG. The intensifying peaks in at ca. 1700 cm−1 and 1400 cm−1are attributed to the vibrations of symmetrical stretching of C = O bond and asymmetrical stretching in the -COO- group in the scaffold of UIO/ACPMG nanohybrid33,39,40,41. The small peak at 1504 cm−1 is correspond to the stretching vibration of the aromatic ring. The bands at the range of 658–808 cm−1are related to stretching and bending vibrations of benzene dicarboxylic acid ligand31. combined with C-H vibration, C = C stretch, OH bend and O-C-O bend in the ligand vibrations. In other hand, no adsorption peak was appeared in 1715 cm−1, corresponded to C = O vibration bands of the free benzene dicarboxylic acid ligand, confirming that the entire of carboxylate groups are attached to Zr ions42. Besides, the wide adsorption peak at the range of 3700 to 3200 cm−1 is ascribed to O-H vibrations, indicating that ACPMG is existed in the final product. Therefore, FT-IR spectra confirmed that UIO/ACPMG nanohybrid is well synthesized.
A Boehm method was used for the calculation of the number of acidic and basic groups on the particles’ surfaces. This method is based on acid–base titration with NaOH, Na2CO3, NaHCO3, and HCl. The following procedure was used: 0.5 g of the powder and 25 mL of 0.05 M NaOH, Na2CO3, NaHCO3, or HCl solution was agitated for 3 h. In the next step, 5 mL of each filtrate were pipetted and the excess of base or acid was titrated with 0.05 M HCl or NaOH, respectively. The results obtained through the Boehm titration showed that UIO/ACPMG nanohybrid approximately contain 0.71 mmol/g of basic group and 0.97 mmol/g of the acid group. The acid groups are due to lactonic (0.32 mmol/g), phenolic (0.78 mmol/g) and carboxylic group (0.05 mmol/g).
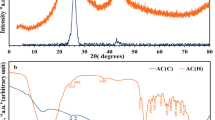
Figure 3B shows the XRD of UIO/ACPMG nanohybrid, UIO NPs, and ACPMG. The XRD obtained for the synthesized UIO showed that it has a crystalline structure. The 2θ values observed for UIO (about 7.2°, 111 and 8.9°, 222) were consistent with other reports43. Two peaks appeared in the region of 2θ = 25° (002) and 45° (100) confirms the quasi-crystalline structure of ACPMG44. XRD analysis confirmed that the as-synthesized compound was significantly crystalline structure and its pattern has good agreement with the simulation patterns. Compared with UIO NPs pattern, the peaks of the UIO/ACPMG nanohybrid pattern is sharper and no deformation than UIO NPs confirming that the UIO NPs structure had been preserved in the final structure of UIO/ACPMG nanohybrid and no impurities generated during the as-synthesized final compound were observed. On the other hand, since MOFs do not have pore stability and have low mechanical strength, their hybrid with activated carbon preserves its mechanical strength and stability, which is well confirmed by the results of XRD. On the other hand, the XRD of the cycled sample after the degradation process confirms the stability of the synthesized sample.
Figure 3C and D shows the BET adsorption- desorption and BJH plots of UIO NPs, ACPMG, and UIO/ACPMG nanohybrid in different concentrations of ACPMG. According to the IUPAC categorization, all the prepared compounds exhibited isotherm NO.1, indicating that compounds have microporous structure. As showed in Table 1, the specific surface area of prepared UIO/ACPMG nanohybrids is decreased in compared with free UIO NPs since ACPMG equipped the pores although the specific surface area is still high. The average pore diameter of UIO/ACPMG nanohybrids is nearly equal to UIO NPs, confirming that the introducing the ACPMG did not change the growth pattern of the UIO NPs framework.
In other words, ACPMG and UIO NPs both have structures with high internal surface area that act as a solid mass. In both types of adsorbents, the internal surface and the pore volume are important for adsorption. By increasing the internal surface and the pore volume, the effective adsorption area also increases. Therefore, the greater the internal surface and the pore volume, the greater the adsorption. But in the case of the UIO/ACPMG nanohybrid, with the increase in the average pore diameter compared to the pure UIO, the internal surface area decreases. This is because larger pores have less internal surface area and thus the adsorption area is also reduced. On the other hand, as the size of the cavities increases, the number of holes’ decreases, which also reduces the surface area. Therefore, it can be said that with the increase in the average pore diameter in the UIO/ACPMG nanohybrid, the internal surface area and the surface area decrease. But it should be noted that these changes are highly dependent on the structure and characteristics of each type of adsorbents. Based on the IUPAC classification, pore diameters are divided into three categories: microspore (the pore size < 2 nm), mesopore (the pore size 2–50 nm), and macrospore (the pore size > 50 nm)45,46,47. Since the diameter pores of the synthesized UIO/ACPMG nanohybrid is about 2 nm, it has a microporous structure. Additionally, the BJH plot (Fig. 3D) also clearly shows that the pore size distribution in the samples are in the range of 1–20 nm.
The specific surface area, pore size, and pore volume of UIO/ACPMG nanohybrids can vary depending on the ratio of UIO to ACPMG. The differences observed between UIO/ACPMG20%, UIO/ACPMG10%, and UIO/ACPMG30% can be attributed to several factors:
At UIO/ACPMG20%, there may be an optimal balance between the contributions of both UIO and ACPMG to the overall surface area. This ratio allows for sufficient pore formation from both materials without overcrowding the structure. The presence of ACPMG helps create additional pathways for gas or liquid molecules to access the pores of UIO, enhancing the effective surface area. Also, the interaction between UIO and ACPMG at this ratio may create a more favorable environment for maintaining the porosity of both materials, leading to a higher overall specific surface area. In the other hand, the microporous nature of UIO dominates the nanohybrids, especially at a moderate AC content. This results in smaller pore sizes as more space is occupied by the dense framework of UIO. At UIO/ACPMG20%, the presence of ACPMG may not significantly disrupt the formation of the smaller pores characteristic of UIO, leading to lower pore volumes. In the UIO/ACPMG10% sample, the nanohybrid may retain most of the high specific surface area of pure UIO, but the lack of sufficient ACPMG might limit additional surface area contribution from ACPMG. Therefore, fewer ACPMG pathways could mean less effective pore utilization, potentially leading to lower overall surface area compared to the UIO/ACPMG20% sample.
In the UIO/ACPMG30%, the high content of ACPMG can lead to crowding, which may hinder the formation of new pores and reduce overall porosity. The presence of too much ACPMG could also result in a denser packing that limits pore volume. Excessive ACPMG may disrupt the crystalline structure of UIO, leading to reduced surface area and pore volume due to potential collapse or densification.
(A) FT-IR of (a) ACPMG, (b) UIO NPs, (c) UIO/ACPMG nanohybrid; (B) XRD pattern of (a) UIO NPs, (b) ACPMG, (c) UIO/ACPMG nanohybrid; (C) Nitrogen adsorption-desorption isotherm of UIO NPs, ACPMG, UIO/ACPMG10% nanohybrid, UIO/ACPMG20% nanohybrid, UIO/ACPMG30% nanohybrid; (D) BJH plots of UIO NPs, ACPMG, UIO/ACPMG10% nanohybrid, UIO/ACPMG20% nanohybrid, UIO/ACPMG30% nanohybrid.
Based to the X-ray photoelectron spectroscopy (XPS) survey spectra of ACPMG (Fig. 4A), there were three main peaks at 281.9, 401, and 527 eV corresponding to C1s, N1s, and O1s, respectively. Table 2 lists the C, N, and O content of the ACPMG. Also, the presence of peaks related to carbon in sample UIO/ACPMG confirms the correct synthesis of the sample (see Table 2; Fig. 4B).
(A) XPS spectra of (a) full survey spectrum (ACPMG and UIO/ACPMG), (b) O1s of ACPMG, (c) C1s of ACPMG, (d) N1s of ACPMG; B) XPS spectra of a) O1s of UIO/ACPMG, (b) C1s of UIO/ACPMG, (c) Zr 3d of UIO/ACPMG, (d) N1s of UIO/ACPMG.
The morphology of UIO/ACPMG nanohybrid, and ACPMG was investigated by SEM analysis. ACPMG have porous structure, respectively (Fig. 5A). The SEM image in Fig. 5B show the nanohybrid at 2 μm magnification, and reveal a rough and uneven surface of ACPMG and UIO NPs. It can be observed that clusters of semi-spherical UIO NPs particles were present although unevenly distributed throughout the surface of ACPMG. A closer look, at 5 μm magnification (Fig. 5C), shows a more defined image of the UIO NPs structures formed on the surface of the ACPMG. The size distribution of UIO/ACPMG nanohybrid is in the range 58–130 nm which is due to the presence of macromolecules of ACPMG. SEM images reveal that that in situ addition of the ACPMG sample during the synthesizing the UIO NPs does not effect on the fabrication and monotonous growth of the UIO/ACPMG nanohybrid.
Furthermore, Fig. 5D, E, is displayed the HR-TEM result which is a clear reason for creating the UIO/ACPMG nanohybrid structure. The energy dispersive X-ray (EDX) results of the UIO/ACPMG nanohybrid displays in Fig. 6A. Since most of the nanohybrid is composed of UIO NPs, the peaks related to Zr are more intense than those of C, N, and O. The presence of well-distributed C (30.09 Wt.%), N (12.15 Wt.%), O (37.34 Wt.%), and Zr (20.42 Wt.%) elements on UIO/ACPMG nanohybrid was showed by mapping (Fig. 6B) which confirm uniform distribution of ACPMG on UIO NPs.
SEM images of (A) ACPMG, (B, C) UIO/ACPMG nanohybrid; (D, E) HR-TEM Images UIO/ACPMG nanohybrid.
(A) EDX analysis of UIO/ACPMG nanohybrid; (B) Elemental mapping images of UIO/ACPMG nanohybrid.
Figure 7A shows the particle size distribution (PSD) of samples UIO/ACPMG nanohybrid obtained from images SEM. For this purpose, about 50 particles of the particles in the image have been measured. As can be seen, particles with a size of 20–30 nm have the highest frequency for UIO NPs. While after the hybrid with carbon, the most frequent particle size is 85–130 nm, which is due to the presence of macromolecules of ACPMG.
The heating stability of the UIO/ACPMG nanohybrid, UIO NPs, and ACPMG was determined by TGA analysis (Fig. 7B). As can be seen, ACPMG indicated the primary weight loss step accrue in the around 250–400 °C. Biomass is composed of extractives, lignin, cellulose, and hemicelluloses. Therefore, initial decomposition TGA curves of ACPMG are associated with moisture elimination (see around 58.33 °C in Fig. 7B-a)33,48. The second decomposition is due to the degradation of cellulose and hemicelluloses, which occurs at a temperature of about 330 ºC (The place of the evolution of light volatile compounds). Since the lignin is slowly degraded, so it degradation continues to higher temperatures. In the > 600 °C, no decomposition because of the activation process of RM performance in this temperature, therefore means a stable state and no remains could be found. The first weight loss in Fig. 7B-b in the < 100 °C indicates the loss of water, the evaporation of remain solvents in fabricated samples, and or trapped solvent molecules inside the framework49. The second stage in Fig. 7B-b, between 100 and 380 °C- 500 °C, is that the UIO framework undergoes dihydroxylation. This stage represents the dehydroxylation of the zirconium oxo-clusters and the removal of DMF49. The third weight drop take place at around of 500 °C is refers to the decomposition of UIO due to the burning of organic linker molecules in the framework49and the breakdown of the polymeric molecule chains49. TGA indicated that UIO/ACPMG nanohybrid (Fig. 7B-c)is more stable than the free UIO NPs. In other hand, TGA curves shows that UIO/ACPMG nanohybrid is decomposed in three steps. In the first step, the weight loss takes place at 95 °C which can be because of the evaporation of solvents adsorbed in the pores or surface of the sample31,33. The second loss weight loss takes place at 380 °C which is related to the loss of trapped solvent and unreacted linkers of UIO/ACPMG nanohybrid due to higher temperature than their boiling point. The last weight loss occurs at 550 °C is attributed to decomposition and degradation of skeleton of UIO/ACPMG nanohybrid, turned to CO2, CO, and ZrO2. Therefore, it can be said that the increase in the stability of free UIO NPs is due to the presence of ACPMG in the UIO/ACPMG nanohybrid structure. In TGA curves, the ash content represents the residue left behind after a material has been thermally decomposed or incinerated. In the case of a UIO/ACPMG nanohybrid, the higher ash content compared to pure MOF can be attributed to follow factors: ACPMG is a highly porous material with a high carbon content. When activated carbon is incorporated into a nanohybrid with a MOF, it can contribute significantly to the ash content of the material. During thermal decomposition in TGA, the ACPMG may not completely combust, leaving behind a higher ash content in the nanohybrid compared to pure MOF.
The presence of inorganic components in the activated carbon or other additives used in the synthesis of the nanohybrid can also contribute to the higher ash content. These inorganic components may not decompose completely during TGA analysis, leading to a higher residual ash content in the nanohybrid.
Also, the synthesis process of the UIO/ACPMG nanohybrid may involve high-temperature treatments or other processes that could lead to carbonization or char formation. These processes can result in the incorporation of additional carbonaceous residues in the nanohybrid, contributing to a higher ash content compared to pure MOF. General, the presence of activated carbon, inorganic components, and the synthesis process are likely factors that contribute to the higher ash content of a UIO/ACPMG nanohybrid compared to pure UIO in TGA curves.
PSD curve of (A) UIO/ACPMG nanohybrid; (B) TGA curves of (a) ACPMG, (b) UIO NPs, (c) UIO/ACPMG nanohybrid.
The study of adsorption and desorption of VOC by nanoadsorbents
Gasoline breakthrough curves of adsorbents
Gasoline vapor breakthrough curves of UIO NPs and UIO NPs hybridized by ACPMG are given in Fig. 8A-F. In this work, gasoline vapor is chosen as the model, since the dominant component of gasoline vapor is ISO molecules, so we focus on ISO adsorption. As the XPS results show, the structure of the sample mainly consists of C, N, and O atoms. The H atoms of ISO interact with the O atoms (negative potential) of the sample through the formation of hydrogen bonds and lead to the adsorption of ISO. As it is clear from the figure, the UIO/ACPMG20% nanohybrid has better gasoline adsorption capacity (GAC) (391.304 mg/g) among the samples due to its extremely high pore volume, and includes both mesoporous and microspores, as discussed earlier. Micro mesoporous structures improve the adsorption of gasoline and organic components such as isoparaffinic, natural hydrocarbons, aromatic compounds and cyclic hydrocarbons with diverse polarity, shape and molecular size. In general, all the prepared adsorbents showed fast kinetic behaviors for gasoline vapor adsorption and their maximum adsorption capacity was reached in the first 15 min. In other words, complete adsorption occurred in the first 15 min, and then gasoline vapor was continuously adsorbed at a decreasing rate until equilibrium conditions were reached. Also, the desorption of gasoline on the synthesized nanohybrids was investigated (Fig. 8C-F). UIO/ACPMG20% nanohybrid desorbed 285.71 mg/g of the adsorbed gasoline, which is an excellent result in desorption. Therefore, this sample was selected as the best sample. The two main parameters for high adsorption and desorption are the small pore diameter and the large volume of pores. The higher adsorption of the UIO/ACPMG20% nanohybrid compared to other samples is due to the average pore diameter is smaller and the total volume is larger.
(A) Adsorption capacity and (B) Desorption capacity of UIO NPs, UIO/ACPMG10%, UIO/ACPMG20%, and UIO/ACPMG30% at 298 K and 1 atm; The breakthrough curves of VOC on (C) UIO (D) UIO/ACPMG10%, (E) UIO/ACPMG20%, (F) UIO/ACPMG30% at 298 K and 1 atm.
Adsorption kinetics
One of the most cited methods to investigate the effect of time on the adsorption process is adsorption kinetics. In this research work, three well-known kinetic models, pseudo-first-order (PFO)50,51, pseudo-second-order (PSO)52,53, and intraparticle diffusion50, were used to study the kinetic data. The linear form of these kinetic models and the parameters obtained from the synthetic analysis are presented in Tables 3 and 4 respectively.
The adsorption kinetics of the adsorbent are determined by the intraparticle diffusion model. Since the R2 values of all samples for the PFO (Fig. 9A-D) and intraparticle diffusion models are very different from PSO (Fig. 10A-D), it can be said that these two models are not able to describe the VOC adsorption process by the samples (Table 4). Possible mechanisms of VOC sorption are as follows: (1) bulk diffusion, (2) film diffusion, and (3) pore diffusion or intraparticle diffusion (Fig. 11A-D). According to previous reports, these diagrams show two or more stages, which confirm the multi-linear adsorption process. Kinetic studies showed that the PSO model fits well with the experimental data of VOC adsorption by four adsorbents.
PFO curves of (A) UIO NPs, (B) UIO/ACPMG10%, (C) UIO/ACPMG20%, and (D) UIO/ACPMG30%.
PSO curves of (A) UIO NPs, (B) UIO/ACPMG10% nanohybrid, (C) UIO/ACPMG20% nanohybrid, (D) UIO/ACPMG30% nanohybrid.
Steps of the intraparticle diffusion model for gasoline adsorption onto (A) UIO NPs, (B) UIO/ACPMG10% nanohybrid, (C) UIO/ACPMG20% nanohybrid, (D) UIO/ACPMG30% nanohybrid.
Study of reusability
One of the essential parameters to the widespread applications of sorbents is their stability and reusability. Therefore, the durability of the UIO/ACPMG20% nanohybrid was investigated by the adsorption-desorption recycling test (Fig. 12). After any cycle of using the adsorbent, the desorption of the VOS from UIO/ACPMG20% nanohybrid was completed by degassed in vacuum oven at 100 °C and then reused for the next run of adsorption. As Fig. 12 show, there is no significant deficiency of adsorption site after four cycles, which confirm the excellent stability and constancy of the adsorbent. The excellent reproducibility of UIO/ACPMG of 20% indicates its potential application in non-contaminating removal processes.
Reusability of UIO/ACPMG20% nanohybrid by adsorption–desorption process.
Comparison of the adsorption capacities of AC reported in this study with other adsorbents
The UIO/ACPMG nanohybrid described in this research is an excellent adsorbent for VOC adsorption. VOC is improved by using AC prepared from waste materials in a simple way. Comparison of UIO/ACPMG nanohybrid adsorption capacity with adsorbents available in the literature for VOCs adsorption (Table 5) shows that this adsorbent has excellent adsorption capacity.
DFT results
DFT calculations may provide useful information about the nature of interaction between the gasoline vapor and the modified carbon substrate. Figure 13A and B depicts the optimized geometry of the ACPMG model used in this study. According to our previous study33, this simple model is reliable enough to properly describe the adsorption properties of gasoline vapor. Besides, due to the presence of various defects sites, it is likely that this nonporous carbon cluster also exhibits a large surface reactivity towards the UIO NPs. Figure 13C shows also the optimized geometry of Zr6O4(OH)4 cluster, which is likely a proper model to describe the UIO NPs. The Hirshfeld analysis reveals that the average charge of Zr atoms in this cluster is about 0.35 |e|, while those of oxygen atoms are − 0.38 |e|. This indicates the Zr-O bonds of Zr6O4(OH)4 are significantly polarized, which is essential to adsorb the ISO molecule. Let us now consider the adsorption of an ISO molecule over the Zr6O4(OH)4 cluster. To this aim, a single ISO molecule was placed around the Zr6O4(OH)4 and all possible geometry configurations were considered carefully. As shown in Fig. 13D, the ISO molecule is adsorbed on the Zr atom of Zr6O4(OH)4 with a minimum binding distance of about 2.8 Å. Moreover, the corresponding negative adsorption energy (-8.25 kcal/mol) confirms that the ISO molecule is thermodynamically favorable at normal condition. On the other hand, the adsorption of ISO is accompanied by a transfer of 0.22 electrons from the ISO into the Zr6O4(OH)4 cluster. This means that the Zr6O4(OH)4 acts as the electron accepter when interacts with the ISO.
(A) and (B) optimized structure of ACPMG from top and side views, (C) Zr6O4(OH)4 cluster, (D) ISO adsorbed onto Zr6O4(OH)4, (E) Zr6O4(OH)4/AC and (F) ISO adsorbed onto UIO/ACPMG.
To construuct the UIO NPs-modified with the ACPMG (UIO/ACPMG), the Zr6O4(OH)4 cluster was placed at different sites of the ACPMG. After geometry optimizations, it was found the Zr6O4(OH)4 cluster tends to adsorb on the defect site located in the center of the carbon cluster. As evident from Fig. 13E, the Zr atom of Zr6O4(OH)4 interacts chemically with the C atoms of the carbon cluster. This is also verified by the large negative adsorption energy of the Zr6O4(OH)4 cluster (-26.58 kcal/mol), suggesting that the Zr6O4(OH)4 is stabilized efficiently on the defect site of the carbon substrate. Besides, the Hirshfeld analysis reveals that during the adsorption process, about 0.42 electrons is transferred from the Zr6O4(OH)4 to the carbon cluster. Accordingly, the atomic charges on the Zr atoms of Zr6O4(OH)4 becomes more positive following its adsorption.
The adsorption of ISO molecule onto the UIO/ACPMG was also investigated to see how the addition of Zr6O4(OH)4 onto the ACPMG modifies the adsorption strength of ISO. Figure 13F shows the most stable configuration of the ISO molecule adsorbed on the UIO/ACPMG. It is found that the ISO is adsorbed on the Zr atom of Zr6O4(OH)4 moiety with a minimum binding distance of 2.65 Å. The adsorption of ISO onto UIO/ACPMG is calculated to be -11.84 kcal/mol, which is more negative than that of pristine Zr6O4(OH)4(ca. -8.25 kcal/mol) or ACPMG (-8.94 kcal/mol)33. This implies that the addition of Zr6O4(OH)4 onto the ACPMG improves the adsorption properties of the ISO. To illustrate this, the charge-transfer from the ISO into the Zr6O4(OH)4 moiety is also calculated. It is found that the adsorption of ISO onto the UIO/ACPMG is followed by a transfer of 0.29 electrons from the ISO into the Zr6O4(OH)4 subunit. This improvement of adsorption energy and the charge-transfer values can be understanding by the fact that when Zr6O4(OH)4 is added onto the ACPMG to form UIO/ACPMG, it acts as the electron donor and this makes Zr atoms to be more positive. As a result, Zr6O4(OH)4 in UIO/ACPMG becomes more favorable electron acceptor towards the ISO compared to the pristine Zr6O4(OH)4. Therefore, the ISO molecule tends more strongly adsorb on the Zr6O4(OH)4.
Also, to understand the activation energy during the adsorption of ISO, we performed slow-growth free energy calculations60,61 at varying percentages of UIO/ACPMG: 25%, 50%, and 75%. This analysis helps us assess how different compositions affect adsorption efficiency. The distance between ISO and UIO/ACPMG was used as the constraint variable (R). We measured the average constraint force over a 2400 fs trajectory with a growth rate of 0.00025 Å fs⁻¹, allowing R to vary from − 0.1 Å to 0.5 Å. From these measurements, we obtained the free energy profile using thermodynamic integration over R (Fig. 14). The results showed that increasing the percentage of UIO/ACPMG decreased the activation energy, suggesting that higher levels of Zr6O4(OH)4 enhance the adsorption process. This is likely due to increased interaction sites, which facilitate better adsorption of ISO molecules.
Activation energy during ISO adsorption with UIO/ACPMG at 25%, 50%, and 75% using slow-growth free energy calculations.
The London dispersive surface energy was calculated for ISO adsorbed on UIO/ACPMG with avogadro softwar62. This analysis is important because it helps us understand the interactions between ISO and the UIO/ACPMG material, which is crucial for applications in adsorption, catalysis, and material design. We determined this parameter by calculating the interaction energy with and without dispersion. Using the following equation,
\(\:{{\upgamma\:}}_{disp}\) is dispersive surface energy, \(\:{E}_{dispersion}\:\) is energy from dispersion forces, and \(\:A\) is surface area. The value of London dispersive surface energy obtained equal to -0.014 eV/ Å2, represents the strength of the dispersive forces between the surface and the adsorbed ISO molecules. A negative value indicates an attractive interaction, suggesting that ISO is likely to cling to the surface, enhancing the material’s effectiveness in applications like gas storage or separation.
Van der Waals electrostatic potential maps are essential in chemistry as they visually represent electron density distribution in a molecule. They help predict reactivity by highlighting electron-rich and electron-deficient areas. Electron-deficient regions (often blue) can function as Lewis acids, while electron-rich regions (often red) act as Lewis bases, providing insights into potential interactions like hydrogen bonding and van der Waals forces. In this study, ISO and Zr6O4(OH)4 show blue areas, indicating their ability to accept electron pairs and act as Lewis acids. Conversely, red areas in ACPMG suggest electron abundance, making it a potential Lewis base capable of donating electron pairs, Fig. 15. This understanding is crucial for designing new materials and predicting molecular behavior in different chemical environments.
Electrostatic potential maps showing electron density. Blue areas in ISO and Zr6O4(OH)4 indicate Lewis acid potential, while red areas in ACPMG suggest Lewis base behavior, this visualization was created using Avogadro 1.2.0 (http://avovadro.cc/)62.
Conclusions
The aim of this work is to conduct a comparative analysis on the effectiveness of different adsorbents, for example UIO NPs, UIO/ACPMG10%, UIO/ACPMG20%, and UIO/ACPMG30% nanoadsorbents in the removal of VOC from the air. Therefore, a set of UIO NPs improved with ACPMG as nanoadsorbents were successfully synthesized. The weight ratio of ACPMG to UIO NPs was optimized and to study the performance of the prepared nanoadsorbents. Then structural characteristics and VOC adsorption properties of the synthesized UIO/ACPMG nanohybrid were then investigated. XRD results confirmed the successful synthesis of the UIO/ACPMG nanohybrid. SEM images indicated that the addition of ACPMG did not result in any observable defects on the surface of UIO NPs. UIO/ACPMG10%, UIO/ACPMG20%, and UIO/ACPMG30% nanoadsorbents illustrated a high adsorption capacity for adsorption of VOC, nearly 4.73%, 7.85%, and 24.64% more than pure UIO NPs. UIO/ACPMG20% nano-adsorbent showed higher surface (391.304 mg/g), due to higher pore volume than other UIO/ACPMG nano-adsorbents. DFT calculations revealed that cooperative charge-transfer between the ACPMG, Zr6O4(OH)4 and ISO is the main reason for the improvement of adsorption energy of ISO molecule onto the UIO/ACPMG. The findings presented in these work are expected to serve as a guide for optimizing the operational conditions for the effective utilization of the developed adsorbents.
Data availability
Data availability: All data generated or analyzed during this study are included in this published article.
References
Zhao, Q. et al. Effects of regulator ratio and guest molecule diffusion on VOCs adsorption by defective UiO-67: experimental and theoretical insights. Chem. Eng. J. 433, 134510 (2022).
Zhan, J. et al. Ozone and SOA formation potential based on photochemical loss of VOCs during the Beijing summer. Environ. Pollut. 285, 117444 (2021).
Lin, Q., Gao, Z., Zhu, W., Chen, J. & An, T. Underestimated contribution of fugitive emission to VOCs in pharmaceutical industry based on pollution characteristics, odorous activity and health risk assessment. J. Environ. Sci. 126, 722–733 (2023).
Zhang, L. et al. Improved speciation profiles and estimation methodology for VOCs emissions: a case study in two chemical plants in eastern China. Environ. Pollut. 291, 118192 (2021).
Lu, F. et al. Exposure characteristics and risk assessment of VOCs from Chinese residential cooking. J. Environ. Manage. 289, 112535 (2021).
Tsai, W. T. An overview of health hazards of volatile organic compounds regulated as indoor air pollutants. Rev. Environ. Health. 34, 81–89 (2019).
Chung, W. C., Mei, D. H., Tu, X. & Chang, M. B. Removal of VOCs from gas streams via plasma and catalysis. Catal. Reviews. 61, 270–331 (2019).
Shi, Y., Wang, J. & Zhou, R. Pt-support interaction and nanoparticle size effect in Pt/CeO2–TiO2 catalysts for low temperature VOCs removal. Chemosphere. 265, 129127 (2021).
Li, X. et al. Adsorption materials for volatile organic compounds (VOCs) and the key factors for VOCs adsorption process: a review. Sep. Purif. Technol. 235, 116213 (2020).
Yang, C. et al. Abatement of various types of VOCs by adsorption/catalytic oxidation: a review. Chem. Eng. J. 370, 1128–1153 (2019).
Yuan, G. et al. Facile construction of a core-shell structured metal-organic frameworks nanofiber membrane for removing Co (II) from simulated radioactive wastewater. Sep. Purif. Technol. 336, 126295 (2024).
Peng, Y. et al. In situ synthesis of NH2-MIL-53-Al/PAN Nanofibers for Removal Co (II) through an Electrospinning process. Langmuir. 40, 2567–2576 (2024).
Li, J. et al. Novel MOFs-based ion-imprinted polymer for selective separation of cobalt ions from waste battery leaching solution. Inorg. Chim. Acta. 536, 120922 (2022).
Anum, A. et al. A. ur Rehman, Construction of hybrid sulfur-doped MOF-235@ g-C3N4 photocatalyst for the efficient removal of nicotine. Inorg. Chem. Commun. 157, 111268 (2023).
Eleryan, A. et al. Isothermal and kinetic screening of methyl red and methyl orange dyes adsorption from water by Delonix regia biochar-sulfur oxide (DRB-SO). Sci. Rep. 14, 13585 (2024).
Ahmad, U. et al. Ahmad Shah, ZIF-8 composites for the removal of wastewater pollutants. ChemistrySelect. 9, e202401719 (2024).
Khalil, A., Nazir, M. A., Salem, M. A., Ragab, S. & Nemr, A. E. Magnetic pomegranate peels activated carbon (MG-PPAC) composite for Acid Orange 7 dye removal from wastewater. Appl. Water Sci. 14, 178 (2024).
Nazir, M. A. et al. A. ur Rehman, synthesis of bimetallic Mn@ ZIF–8 nanostructure for the adsorption removal of methyl orange dye from water. Inorg. Chem. Commun. 165, 112294 (2024).
Ullah, S. et al. Activated carbon derived from biomass for wastewater treatment: Synthesis, application and future challenges, Journal of Analytical and Applied Pyrolysis, 106480. (2024).
Veerapandian, S. K. P., De Geyter, N., Giraudon, J. M., Lamonier, J. F. & Morent, R. The use of zeolites for VOCs abatement by combining non-thermal plasma, adsorption, and/or catalysis: a review. Catalysts. 9, 98 (2019).
Lee, J. E. et al. Recent advances in volatile organic compounds abatement by catalysis and catalytic hybrid processes: a critical review. Sci. Total Environ. 719, 137405 (2020).
Zhang, Y. et al. Anchoring nanosized MOFs at the interface of porous millimeter beads and their enhanced adsorption mechanism for VOCs. J. Clean. Prod. 353, 131631 (2022).
Liu, C., Wang, J., Wan, J. & Yu, C. MOF-on-MOF hybrids: synthesis and applications. Coord. Chem. Rev. 432, 213743 (2021).
Masoomi, M. Y., Morsali, A., Dhakshinamoorthy, A. & Garcia, H. Mixed-metal MOFs: unique opportunities in metal–organic framework (MOF) functionality and design. Angew. Chem. 131, 15330–15347 (2019).
Ebrahimi, A. K., Barani, M. & Sheikhshoaie, I. Fabrication of a new superparamagnetic metal-organic framework with core-shell nanocomposite structures: characterization, biocompatibility, and drug release study. Mater. Sci. Engineering: C. 92, 349–355 (2018).
Ebrahimi, A. K., Sheikhshoaie, I., Salimi, S. & Arkaban, H. In-situ facile synthesis of superparamagnetic porous core-shell structure for dye adsorption. J. Mol. Struct. 1228, 129797 (2021).
Vikrant, K., Kim, K. H., Kumar, V., Giannakoudakis, D. A. & Boukhvalov, D. W. Adsorptive removal of an eight-component volatile organic compound mixture by Cu-, Co-, and Zr-metal-organic frameworks: experimental and theoretical studies. Chem. Eng. J. 397, 125391 (2020).
Vellingiri, K., Kumar, P., Deep, A. & Kim, K. H. Metal-organic frameworks for the adsorption of gaseous toluene under ambient temperature and pressure. Chem. Eng. J. 307, 1116–1126 (2017).
Zhao, Z. et al. Competitive adsorption and selectivity of benzene and water vapor on the microporous metal organic frameworks (HKUST-1). Chem. Eng. J. 259, 79–89 (2015).
Hao, F., Li, L., Zhang, X. & Chen, J. Synthesis and electrochemical capacitive properties of nitrogen-doped porous carbon micropolyhedra by direct carbonization of zeolitic imidazolate framework-11. Mater. Res. Bull. 66, 88–95 (2015).
Sharafinia, S., Farrokhnia, A., Lemraski, E. G. & Rashidi, A. Decoration of ZnFe2O4 and UiO-66 over g-C3N4 as magnetically novel reusable visible light photocatalyst for degradation of Rh–B. Opt. Mater. 132, 112838 (2022).
Dizoğlu, G. & Sert, E. Fuel additive synthesis by acetylation of glycerol using activated carbon/UiO-66 composite materials. Fuel. 281, 118584 (2020).
Sharafinia, S., Rashidi, A. & Esrafili, M. D. Optimized adsorption of volatile organic compounds on the activated carbon prepared from mesquite grain: a combined experimental and computational study. J. Environ. Chem. Eng. 10, 108528 (2022).
Embaby, M. S., Elwany, S. D., Setyaningsih, W. & Saber, M. R. The adsorptive properties of UiO-66 towards organic dyes: a record adsorption capacity for the anionic dye alizarin Red S. Chin. J. Chem. Eng. 26, 731–739 (2018).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Grimme, S. Semiempirical GGA-type density functional constructed with a long‐range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Hirshfeld, F. L. Bonded-atom fragments for describing molecular charge densities. Theoret. Chim. Acta. 44, 129–138 (1977).
Delley, B. From molecules to solids with the DMol 3 approach. J. Chem. Phys. 113, 7756–7764 (2000).
Lemraski, E. G. & Sharafinia, S. Kinetics, equilibrium and thermodynamics studies of Pb2 + adsorption onto new activated carbon prepared from persian mesquite grain. J. Mol. Liq. 219, 482–492 (2016).
Sharafinia, S., Farrokhnia, A. & Ghasemian, E. Comparative study of adsorption of safranin o by TiO2/activated carbon and chitosan/TiO2/activated carbon adsorbents. Phys. Chem. Res. 9, 605–621 (2021).
Ghasemian Lemraski, E., Sharafinia, S. & Alimohammadi, M. New activated carbon from persian mesquite grain as an excellent adsorbent. Phys. Chem. Res. 5, 81–98 (2017).
Sharafinia, S. et al. Effective adsorption of Amoxicillin by using UIO-66@ Cr-MIL-101 nanohybrid: isotherm, kinetic, thermodynamic, and optimization by central composite design. Sci. Rep. 13, 22689 (2023).
Erfaninia, N., Tayebee, R., Dusek, M. & Amini, M. Ethylene diamine grafted nanoporous UiO-66 as an efficient basic catalyst in the multi‐component synthesis of 2‐aminithiophenes. Appl. Organomet. Chem. 32, e4307 (2018).
Sharafinia, S., Rashidi, A., Babaei, B. & Orooji, Y. Nanoporous carbons based on coordinate organic polymers as an efficient and eco-friendly nano-sorbent for adsorption of phenol from wastewater. Sci. Rep. 13, 13127 (2023).
Sharafinia, S. & Rashidi, A. MIL-101 (cr) hybrid nanoporous carbon derived MOF as a nano-adsorbent for dye removal using RSM-CCD. Arab. J. Chem. 16, 105288 (2023).
Sharafinia, S. et al. Adsorption of phenol from both acidic and basic industrial waste via newly synthesized metal organic framework hybrid smart adsorbents. ACS Appl. Eng. Mater. 1, 1554–1565 (2023).
Lemraski, E. G. et al. Polyvinyl alcohol/chitosan/silver nanofibers as antibacterial agents and as efficient adsorbents to remove methyl orange from aqueous solutions. J. Iran. Chem. Soc. 19, 1287–1299 (2022).
Sharafinia, S., Farrokhnia, A. & Lemraski, E. G. The adsorption of cationic dye onto ACPMG@ ZIF-8 core-shell, optimization using central composite response surface methodology (CCRSM). Colloids Surf., a. 634, 128039 (2022).
Sharafinia, S., Farrokhnia, A. & Lemraski, E. G. Optimized safranin adsorption onto poly (vinylidene fluoride)-based nanofiber via response surface methodology. Mater. Chem. Phys. 276, 125407 (2022).
Gholamiyan, S., Hamzehloo, M. & Farrokhnia, A. RSM optimized adsorptive removal of erythromycin using magnetic activated carbon: Adsorption Isotherm, kinetic modeling and thermodynamic studies. Sustainable Chem. Pharm. 17, 100309 (2020).
Wang, J. & Guo, X. Adsorption kinetic models: physical meanings, applications, and solving methods. J. Hazard. Mater. 390, 122156 (2020).
LagergrenS. Zur Theorie Der Sogenannten adsorption geloster stoffe. Kungliga Svenska Vetenskapsakademiens Handlingar. 24, 1–39 (1898).
Weber, W. J. Jr & Morris, J. C. Kinetics of adsorption on carbon from solution. J. Sanit. Eng. Div. 89, 31–59 (1963).
Zhang, X. et al. Adsorption of Acetone and Cyclohexane onto CO2 Activated Hydrochars245125664 (Chemosphere, 2020).
Zhu, D., Kwon, S. & Pignatello, J. J. Adsorption of single-ring Organic Compounds to wood Charcoals Prepared under Different Thermochemical Conditions393990–3998 (Environmental science & technology, 2005).
Sun, Y. & Webley, P. A. Preparation of activated carbons from corncob with large specific surface area by a variety of chemical activators and their application in gas storage. Chem. Eng. J. 162, 883–892 (2010).
Tham, Y., Latif, P. A., Abdullah, A., Shamala-Devi, A. & Taufiq-Yap, Y. Performances of toluene removal by activated carbon derived from durian shell. Bioresour. Technol. 102, 724–728 (2011).
Wibowo, N., Setyadhi, L., Wibowo, D., Setiawan, J. & Ismadji, S. Adsorption of benzene and toluene from aqueous solutions onto activated carbon and its acid and heat treated forms: influence of surface chemistry on adsorption. J. Hazard. Mater. 146, 237–242 (2007).
Popescu, M., Joly, J., Carre, J. & Danatoiu, C. Dynamical adsorption and temperature-programmed desorption of VOCs (toluene, butyl acetate and butanol) on activated carbons. Carbon. 41, 739–748 (2003).
Hadizadeh, M. H. & Xu, F. First results from the interaction between hydroxyl radicals and condensed water droplets: a spin-polarized DFT-MD study. J. Mol. Liq. 393, 123557 (2024).
Hadizadeh, M. H., Yang, L., Fang, G., Qiu, Z. & Li, Z. The mobility and solvation structure of a hydroxyl radical in a water nanodroplet: a born–Oppenheimer molecular dynamics study. Phys. Chem. Chem. Phys. 23, 14628–14635 (2021).
Hanwell, M. D. et al. Avogadro: an advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 4, 1–17 (2012).
Acknowledgements
The authors are grateful for the financial support received from the Petroleum Industry Research Institute Iran National Science Foundation (INSF, Code Number: 99027224).
Author information
Authors and Affiliations
Contributions
Soheila Sharafinia: Doing the lab work and collecting data, writing of the original draft, literature review, and creating graphs and tables.Alimorad Rashidi: Conceptualization, supervision, and correspondence. Ahmad Ebrahimi: Writing and characterization.Behnam Babaei: Creating figures and editing.Mohammad Hassan Hadizadeh: Conceived and designed the analysis, Contributed data or analysis tools, Validation.Mehdi D. Esrafili: Visualization and editing.Mahnaz Pourkhalil: Sample characterization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sharafinia, S., Rashidi, A., Ebrahimi, A. et al. Enhanced VOCs adsorption with UIO-66–porous carbon nanohybrid from mesquite grain: A combined experimental and computational study. Sci Rep 14, 25177 (2024). https://doi.org/10.1038/s41598-024-74853-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-74853-z