Abstract
Ionized Mg (iMg) may offer a more reliable indicator of Mg status during acute illness than total Mg (tMg) concentrations. This study aimed to determine the prevalence of dysmagnesemia and their relationship using iMg and tMg. The clinical and biochemical characteristics as well as health outcomes and their association with iMg and tMg were also assessed. A prospective study including all eligible adult patients (≥18 years) who were hospitalized in the General Internal Medicine unit at Sultan Qaboos University Hospital (SQUH) for 3.5 months in 2023. The iMg and tMg concentrations were collected on all at the admission. In total 500 patients were included (females 49.2%) with a median age of 64.5 years (IQR: 48-77). The prevalence of hypomagnesemia and hypermagnesemia by iMg concentrations was 3.4% and 26.6%, respectively, while by tMg concentrations 13.2% and 11.0%, respectively. The agreement between both measurements was strong (r=0.665, p<0.01). An increased tMg concentration was independently associated with high dependency units’ admission (adjusted odds ratio (aOR): 4.34, 95%CI: 1.24-15.06, p=0.02) and cardiac arrest (aOR: 14.64, 95%CI: 3.04-70.57, p<0.01), and 6-month all-cause mortality (aOR: 11.44, 95%CI: 2.46-53.17, p<0.01). During follow-up hypermagnesemia using tMg had a higher mortality compared to other groups (hazard ratio (HR): 1.82, 95%CI: 1.11-3.01, p=0.02) while no significant findings were demonstrated using iMg concentrations. iMg and tMg concentrations had a strong correlation that might be supporting the potential use of point-of-care devices. Multivariant regression analysis showed that hypermagnesemia by tMg was associated with adverse outcomes. However, the generalizability of the study findings should be taken with caution and the difference in the associations with outcomes highlight the importance of further research to examine the complex associations and impacts of dysmagnesemia in various clinical settings.
Similar content being viewed by others
Introduction
Magnesium (Mg), the fourth most abundant cation in the body, serves as a cofactor for over 300 enzymatic reactions. It facilitates nucleic material and protein synthesis, enables neuromuscular conduction, supports cardiac contractility, aids in energy metabolism, and contributes to immune system function1, 2. Maintaining a proper Mg balance has been associated with reduced risk of inflammation, diabetes, colorectal cancer, stroke, and cardiovascular events3. Among hospitalized patients, previous research has indicated that hypomagnesemia and hypermagnesemia frequently occur, with prevalence rates ranging from 3.0 to 29.0% and 5–27%, respectively3,4,5,6,7,8.
Hypomagnesemia is associated with vascular dysfunction, inflammation, and increased risk of arrhythmias9. Also, hypomagnesemia in patients with heart failure, critically ill patients, and those undergoing dialysis is linked to higher mortality rates3. A meta-analysis indicated that a decrease of 0.49-mg/dL (0.2-mmol/L) in serum Mg concentration was associated with a 30% increase in cardiovascular diseases10.
Hypermagnesemia can also lead to serious complications such as muscle paralysis, respiratory depression, and cardiac arrest9. In a few studies, elevated mortality has been linked to hypermagnesemia, and this may be due to factors such as increased neurohormonal activation, suppressed parathyroid hormone, concurrent kidney dysfunction, and high phosphorus concentrations3, 8, 9, 11.
Most studies assessing the prevalence, and health outcomes of dysmagnesemia among hospitalized patients used total Mg (tMg)6, 11. However, approximately 99% of Mg in the body is found within the cells, while the remaining Mg in the extracellular space is either bound to albumin, complexed with anions or exists as the bioactive ionized form. Due to this distribution, measuring tMg may not be a reliable indicator of Mg status during acute illness12. Moreover, due to its lack of binding to albumin, ionized Mg (iMg) is believed to offer a more precise reflection of Mg concentrations within the body compared to tMg, particularly in critical care scenarios characterized by fluctuating hemodynamics6, 12. An older study suggested a correlation between tMg and iMg concentrations13. However, recent observational studies and clinical trials have not found a statistically significant positive correlation between tMg and iMg using Pearson’s correlation coefficients (with correlation values ranging from 0.50 to 0.77)12, 14.
This study aimed to determine the prevalence of dysmagnesemia using iMg and tMg concentrations among patients hospitalized in medical wards, as well as their correlation. The associated clinical and biochemical characteristics as well as health outcomes were also assessed.
Methods
Study design, setting, and population
This prospective cohort study was conducted at Sultan Qaboos University Hospital (SQUH) between May 10th and August 31st, 2023. The study included all eligible adult patients (≥ 18 years) who were hospitalized in the General Internal Medicine unit in general medical wards, i.e., not in high dependency unit or intensive care units. SQUH is a tertiary hospital with an academic setting, offering comprehensive medical services to inpatients from all regions of Oman15. Screening for inclusion was performed for all patients admitted under the care of the General Internal Medicine unit. Exclusion criteria were age younger than 18 years, non-consent, and readmission within 90 days. The iMg and tMg concentrations were collected for all participants once only during the first 24 h of admission.
Data collection
Relevant patient demographic information, including age and sex, was collected, along with medical history including medications with the potential to cause dysmagnesemia, such as loop diuretics, thiazides, and proton pump inhibitors (PPIs). Additionally, specific biochemical data were obtained, including electrolytes as well as measurements of albumin, creatinine, and estimated glomerular filtration rate (eGFR). The treatment of dysmagnesemia and any associated adverse events were carefully recorded. The primary diagnosis of patients was categorized according to the 10th revision of the International Classification of Diseases (ICD-10). Follow-up information was obtained through the review of medical files, and if necessary, by contacting either the patient or their next of kin.
Mg measurements
The concentration of tMg was determined by performing a colorimetric end-point reaction between Mg and xylidyl blue in an alkaline solution. This analysis was conducted using the Roche Cobas modular analyzer in the Biochemistry Department.
Samples of iMg were collected by dedicated research assistants. A 1–2 mL venous whole blood sample was collected from each patient after applying a tourniquet using heparinized syringes containing 60 IU heparin (self-filling sampler by Radiometer®, 2700 Bronshoj, Denmark). The ionized samples were subjected to analysis using the electrolyte analyzer, Stat Profile Prime Plus® (Nova Biomedicals, Waltman, MA, USA). This analyzer utilizes the direct integrated sensing electrode (ISE) method to determine the concentration of iMg. ISE has emerged as a proven potentiometric sensor for analyzing the analytical properties of biomarkers, as they exhibit high speed and precision. They require minimal sample volume and serve as a reliable foundation for electrolyte measurements in various medical applications, including hospital-based clinical laboratory and other research uses12, 16, 17. The method involves a neutral carrier-based membrane containing an ionophore that exhibits selectivity towards the size of the Mg ion. All samples were analyzed in the medical ward by trained research assistants, either immediately or within 1 h after collection, to ensure the stability of the samples and further details can be found elsewhere12.
Definitions
For iMg concentration measurement: hypomagnesemia was defined based on a local Omani population reference range as an initial iMg concentration of ≤ 0.46 mmol/L, while hypermagnesemia was determined by an iMg concentration of ≥ 0.69 mmol/L12.
For tMg concentration measurement: hypomagnesemia was defined as a tMg concentration of ≤ 0.69 mmol/L, and hypermagnesemia was defined as a tMg concentration of ≥ 1.01 mmol/L6.
Sample size
The sample size calculation was based on the main outcome, i.e., mortality. A previous study18 found 11.7% and 40% in-patient mortality in patients with hypomagnesemia and hypermagnesemia, respectively, compared to only 2.7% death in the normomagnesemia group. In a previous study from the SQUH the prevalence of dysmagnesemia was 22.0%6, and thus we hypothesized that the excess mortality due to dysmagenesemia would be approximately 8% compared to patients with normomagnesemia. Therefore, using a power of 90% and an alpha level of 5%, a sample size of 456 patients was needed. To account for any missing information and loss of follow-up, the sample size of this study was increased to 500 patients.
Ethical approval
Ethical approval was obtained from the Medical and Research Ethics Committee at the College of Medicine and Health Sciences at Sultan Qaboos University (SQU), (MREC #2719; SQU-EC/51/2022) and written informed consent was obtained from the patient or, in case of impaired capacity, next of kin. The study adhered strictly to the ethical guidelines and principles outlined in the Declaration of Helsinki.
Statistical analysis
Categorical variables were presented as frequencies and percentages. Continuous distributed variables were summarized using median and interquartile ranges (IQRs). Pearson’s correlation coefficient and Lin’s concordance correlation coefficients were used to determine the correlation between iMg and tMg concentrations. Bland-Altman plot was used to determine and illustrate the mean differences between iMg and tMg concentrations. The Kruskal-Wallis test determined the relationship between variables and different iMg concentration groups. The Chi-Square test was performed to examine the relationships between categorical variables and different Mg concentration groups, while Fisher’s exact test was applied if the cells had an anticipated frequency of less than five. The correlation between albumin concentrations and iMg and tMg concentrations was assessed using a Spearman’s rho test and illustrated using a scatter plot. All relevant characteristics were included in the backward stepwise regression analysis to identify potential independent factors associated with each group of dysmagnesemia using an adjusted odds ratio (aOR). Lastly, time-to-event survival analysis for 90-day re-admission and 6-month all-cause mortality was performed using the Kaplan–Meier method, and log-rank tests for comparisons among patients in the three different groups of iMg concentration to indignity hazard ratio (HR). The two-tailed level of significance was set at p < 0.05 level. Statistical analyses were conducted using STATA version 17.0 (STATA Corporation, College Station, TX, USA).
Results
In total 500 patients were included, 246 (49.2%) were females. The median age was 64.5 years (IQR: 48–77). Most of the patients had a history of hypertension (n = 330, 66.0%) and diabetes mellitus (DM) (n = 262, 52.4%). Other prevalent medical comorbidities included chronic kidney disease (CKD) (n = 122, 24.4%), heart failure (n = 103, 20.6%), and atrial fibrillation (AF) (n = 103, 20.6%). The commonly prescribed medications included PPIs (n = 389, 77.8%), followed by loop diuretics (n = 193, 38.6%), and thiazides (n = 51, 10.2%).
The prevalence of hypomagnesemia measured by iMg concentration was 3.4% (95% CI 2.1% − 5.4%), while the prevalence of hypermagnesemia measured by iMg concentration was 26.6% (95% CI 22.9% − 30.7%). The prevalence of hypomagnesemia measured by tMg concentration was 13.2% (95% CI 10.5% − 16.5%), and the prevalence of hypermagnesemia measured by tMg concentration was 11.0% (95% CI 8.5% − 14.1%). The agreement between iMg and tMg concentrations was examined using a Bland-Altman Plot, demonstrating a Pearson’s correlation coefficient (r) of 0.665 (p < 0.01). This indicates a strong positive correlation. The negative correlation between difference and mean (-0.458), with a Bradley Blackwood F value of 959.964 (p < 0.01), further supports the relationship between iMg and tMg.
The prevalence of DM was higher in the hypomagnesemia group measured by iMg concentration compared to normomagnesemia and hypermagnesemia groups (82.4% vs. 54.9% vs. 42.1%; p < 0.01) but there were no differences between the groups by tMg (Table 1 and Table 2). Conversely, CKD was more common in the hypermagnesemia group measured by iMg concentration compared to normomagnesemia and hypomagnesemia groups (33.8% vs. 20.8% vs. 23.5%; p = 0.01), and a similar association was found among the groups measured by tMg concentrations. The hypomagnesemia group measured by iMg concentration had significantly higher sodium concentrations compared to the normomagnesemia and hypermagnesemia groups (138 vs. 136 vs. 137 mmol/L; p = 0.04). Conversely, the hypermagnesemia group measured by iMg concentration had significantly higher serum creatinine concentrations compared to the normomagnesemia and hypomagnesemia groups (92 vs. 78 vs. 72 mmol/L; p < 0.01) (Table 1).
In terms of medications, loop diuretics were more commonly used in patients with hypermagnesemia by tMg concentration (54.6% vs. 38.5% vs. 25.8%; p < 0.01) (Table 2).
There were no other significant differences in the characteristics, clinical features, and laboratory profiles of the patients when they were classified based on iMg and tMg concentrations (Table 1 and Table 2).
During hospitalization, 20.8% of patients (n = 104) received some form of Mg replacement. Among the hypomagnesemia group measured by iMg concentration, 58.8% (n = 10) of patients received Mg replacement, which is almost the same as patients treated for hypomagnesemia measured by tMg concentration (56.0%; n = 37) (Table 1 and Table 2).
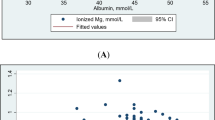
As shown in Fig. 1, the correlation between albumin concentrations and iMg and tMg concentrations was assessed using a Spearman’s rho test. The results revealed a non-significant weak positive correlation (Spearman’s rho = 0.075, p = 0.09) between albumin concentrations and iMg. In contrast, there was a significant weak positive correlation between albumin concentrations and tMg concentrations (Spearman’s rho = 0.088, p = 0.04).
Effect of albumin concentrations on ionized Mg and total Mg concentrations (n = 500). (A) Effect of albumin concentration on ionized Mg(B) Effect of albumin concentration on total Mgconcentration (Spearman’s rho = 0.075, p=0.09) (Spearman’s rho = 0.088, p=0.04).
Regarding clinical outcomes, there were no significant differences observed among the different groups of iMg concentrations (Table 3). However, the hypermagnesemia group measured by tMg concentration exhibited higher rates of transfers to high dependency units (HDUs) (38.2% vs. 21.4% vs. 21.2%; p = 0.02), cardiac arrest (20.0% vs. 7.7% vs. 9.1%; p = 0.01) and increased 6-month all-cause mortality (23.6 vs. 11.6% vs. 9.1%; p = 0.03) compared to the normomagnesemia and hypermagnesemia groups (Table 3).
Additionally, we calculated the fraction of iMg to tMg (iMg/tMg) *100 and studied its association with clinical outcomes (Table 3). The results demonstrated a significant association with adverse outcomes (including, HDU admission (p < 0.01), cardiac arrest (p < 0.01), inpatient mortality (p < 0.01), and 6-month mortality (p < 0.01)) when the fraction fell below 60%, in other words, a difference of 40% or more between iMg and tMg concentrations. Further, we calculated the positive predictive value (PPV) to examine the link between mortality and hypomagnesemia by tMg, and it was 9.1% while hypomagnesemia by iMg value was 17.9%. Moreover, the PPV between mortality and hypermagnesemia by tMg was 23.6% while hypermagnesemia by iMg value was 15.0%.
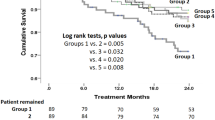
Multivariate regression analysis reinforced the association and identified tMg as an independent predictor associated with these adverse clinical outcomes, including; HDU admission (aOR: 4.34, 95% CI: 1.24–15.06, p = 0.02), cardiac arrest (aOR: 14.64, 95% CI: 3.04–70.57, p < 0.01), and 6-month all-cause mortality (aOR: 11.44, 95% CI: 2.46–53.17, p < 0.01) (Table 4). Survival analysis was conducted using both iMg and tMg. No significant difference was found between the groups regarding the first readmission within 90 days. However, in terms of all-cause mortality at 6 months, survival analysis using iMg did not reveal any significant difference (Fig. 2A), but the analysis using tMg indicated increased mortality in the hypermagnesemia group compared to the normomagnesemia and hypermagnesemia groups (HR: 1.87, 95% CI: 1.11–3.01, p = 0.02) (Fig. 2B).
Kaplan Meier analysis for the first 6-month all-cause mortality stratified according to ionized Mg and total Mg concentrations for all survived patients (n = 500). (A) 6-month all-cause mortality. (B) 6-month all-cause mortality survival analysis for ionized Mg concentration survival analysis for total Mg concentration HR; 1.18, p = 0.56 [95% CI: 0.72 - 1.92] HR: 1.82, p = 0.02 [95% CI: 1.11 - 3.01].
Subgroup analysis
After excluding patients with eGFR < 60 ml/min/1.73 m² (n = 318), hypermagnesemia by tMg concentrations were significantly associated with a higher incidence of cardiac arrest (27.3%, p = 0.04). The iMg/tMg ratio showed a significant association with higher rates of inpatient mortality and within 6-month mortality (6.6%, p < 0.01) (Supplementary Table 1).
Multivariate logistic regression further identified hypermagnesemia by tMg concentrations (aOR 12.75, 95% CI: 1.47–110.59, p = 0.02) as a significant predictor of cardiac arrest (Supplementary Table 2).
Further exclusion of both CKD patients and those with ≥ 3 comorbidities (n = 290) reinforced these findings. Hypermagnesemia by tMg concentrations remained significantly associated with increased cardiac arrest (27.3%, p = 0.002) and 6-month mortality (27.3%, p = 0.028).
Discussion
The study is the first study that provides data using both iMg and tMg to assess the prevalence of dysmagnesemia and associated health outcomes among hospitalized patients in general medical wards. A strong agreement between iMg and tMg concentrations was found. Hypomagnesemia measured by iMg concentration was associated with a higher prevalence of diabetes, while hypermagnesemia by both iMg and tMg correlated with CKD. Also, the increase in tMg concentrations showed an independent association with adverse health outcomes.
The biologically active fraction of circulating Mg is iMg. While iMg could potentially serve as a more physiologically relevant marker compared to tMg, previous studies have suggested that both concentrations might not correlate very well during hospitalization19, 20.
There is a lack of agreement regarding the precise range of serum tMg concentrations deemed sufficient, resulting in varying suggested intervals for both research and clinical purposes. Regarding iMg, intervals of 0.53–0.67 mmol/L and 0.44–0.59 mmol/L have predominantly been utilized, while for tMg, ranges of 0.75–0.95 mmol/L, 0.70–1.00 mmol/L, and 0.70–1.10 mmol/L have commonly been used21.
A strong positive correlation (Pearson’s correlation) between iMg and tMg concentrations was found in the current study. The iMg samples were analyzed using fresh blood within 1 h by trained research assistants in the medical ward, which may account for the observed positive correlation. These findings support to the utilization of point-of-care analyzers for assessing iMg status in hospitalized patients, particularly when timely clinical decisions are necessary to guide patient management, including cases involving cardiac arrhythmia22.
The prevalence of hypomagnesemia measured by tMg concentrations was 13.2%, which falls within the previously reported range of hypomagnesemia in patients admitted to medical wards, ranging between 3.0% and 29%3, 5,6,7. This prevalence is also similar to hypomagnesemia rate previously reported from our site6. The prevalence of hypermagnesemia measured by tMg concentrations was 11.0%, which falls within the range of previously reported prevalence of hypermagnesemia among hospitalized patients, ranging from 5 to 27%3, 6, 8. The prevalence of dysmagnesemia measured by tMg concentrations can vary due to different factors. These factors include the use of various cut-off values to define normal Mg concentrations21, differences in clinical care settings, variations in patient characteristics, and the wide range of medications that can potentially lead to hypomagnesemia6, 23.
In our study, the prevalence of hypomagnesemia measured by iMg concentrations was found to be 3.4%. In comparison, previously reported rates of hypomagnesemia among patients admitted to the intensive care unit (ICU) ranged from 9.7 to 22.0%19, 24, 25. However, there have been no previous studies reporting on hypomagnesemia measured by iMg concentrations in medically admitted patients. While prevalence of hypermagnesemia measured by iMg concentrations was found to be 26.6%, which is comparable to the previously reported prevalence from ICU (23.6%)25.
In the present study, significant discrepancies were observed in the prevalence of dysmagnesemia when comparing the use of iMg and tMg concentrations. Which is in concordance with previous studies19, 24, 25. These differences could be attributed to variations in defining the normal Mg concentration for iMg and tMg. For example, in our setting, the reference range for iMg was established using fresh samples obtained from healthy volunteers specifically from the Omani population12. Furthermore, tMg concentrations could be underestimated in acutely ill medical patients due to low albumin concentrations, which is also the case in this study26.
Despite iMg being the active form of Mg, there have been very few studies assessing iMg concentrations among hospitalized patients13. One study found that the prevalence of hypomagnesemia defined as iMg concentrations less than 0.48 mmol/L, was 12.7% in patients presenting to the emergency department with de novo AF22. Another study including 31 patients following abdominal surgery, found that both tMg and iMg concentrations decreased during surgery, with a strong correlation between both13.
DM was highly prevalent among the hypomagnesemia group measured by iMg concentrations, which could be attributed to the increased excretion of Mg in urine due to glucosuria, nutritional deficiencies, and the development of hypomagnesemia induced by hyperinsulinemia. Hypomagnesemia is linked to an accelerated and enduring decline in renal function among patients with DM27. Furthermore, epidemiological investigations consistently reveal a negative correlation between the intake of Mg through the diet and the risk of developing T2DM27, 28. An increasing number of reported evidence has linked to worsening T2DM control and increased microvascular and macrovascular complications29,30,31. Glycated hemoglobin A1c (HbA1c), fasting blood glucose (FBG), and 2-hour prandial glucose (2hPG) were found to be significantly higher in patients with hypomagnesemia indicating poor control32, 33. Mg is mainly excreted via the kidneys and its homeostasis is affected by intestinal absorption. Thus, kidney disorders can potentially lead to both its depletion and as a result increase the risk of CVD in T2DM34. On the other hand, patients with hypermagnesemia by both iMg and tMg concentrations had a higher prevalence of CKD, elevated serum creatinine concentrations, and eGFR35.
The rate of Mg replacement was higher in both hypomagnesemia groups measured by iMg and tMg compared to other groups, which aligns with findings from a prior RCT which showed that the administered amount of Mg oxide replacement, guided by both iMg and tMg measurements, was similar and had no discernible impact on the development of arrhythmia36. However, there remains insufficient evidence supporting the use of iMg as a guiding factor in treatment decisions, particularly regarding its impact on the increment concentration and subsequent clinical outcomes. We believe that an RCT is needed to look into this hypothesis20. Nevertheless, it is crucial to note that in our setting, the replacement was not guided by iMg concentrations but rather by tMg concentrations and we have not looked into other subsequent concentrations post-replacement.
Evaluating Mg status is intricately complex since only less than 1% of the total body Mg is present in both serum and red blood cells, with the majority residing intracellularly. The serum Mg, constituting approximately 0.3% of the total body Mg, exists in three states: 62% as iMg, 33% bound to proteins, primarily albumin, and the remaining 5% forming complexes with anions37, 38. Given the complex nature of Mg metabolism, multiple associations were examined to explore the connections between various forms of Mg and other biochemical parameters. Our study revealed a weakly positive association between albumin and both tMg and iMg concentrations. However, the statistical significance was observed only with tMg concentrations. These findings align with a systematic review and meta-analysis investigating Mg metabolism in individuals with chronic alcohol use disorder. A substantial reduction in both tMg and iMg among these patients was reported, but it did not identify a direct association with albumin39.
Dysmagnesemia is associated with poor health outcomes as demonstrated in previous studies among hospitalized patients3, 11. The current study showed that hypermagnesemia measured by tMg concentration was an independent factor associated with an increased need for HDU admission, an elevated rate of cardiopulmonary arrest and poor survival. In a large study involving 65,974 patients, hypermagnesemia by tMg was associated with prolonged length of hospital stay and increased hospital mortality3. Another study concluded that total hypermagnesemia was associated with increased mortality among patients who were admitted with acute decompensation of heart failure40.
In a study involving 20,438 participants, it was found that hypomagnesemia and hypermagnesemia measured by tMg were associated with increased in-hospital mortality. Particularly, the risk was higher in patients with hypermagnesemia18, 41. Moreover, among patients admitted with community-acquired pneumonia, both hypomagnesemia and hypermagnesemia measured by tMg at admission were associated with an increased rate of 30-day mortality42.
Limited research has investigated the relationship between dysmagnesmia by iMg and health outcomes among hospitalized patients. Furthermore, most of the existing studies have focused on critically ill patients admitted to ICU25. A study that included 144 ICU patients showed that hypermagnesemia by iMg was associated with mortality25. Another study, consisting of 446 patients, found that the development of hypomagnesemia by iMg during an ICU stay was associated with a poor prognosis which is often associated with the use of diuretics and sepsis43.
We further investigated this complex relation between different measurements of iMg and tMg concentrations and the associated outcomes by calculating the fraction of iMg to tMg. We identified an association with adverse clinical outcomes when the difference between both measurements exceeded 40%. A study found an association between the iMg/tMg fraction when exceeding 72% and an elevated APACHE II score surpassing 20 in the ICU setting. They also found that this increased score was associated with an increased in-hospital mortality risk of 40%, with a PPV exceeding 53%24. Furthermore, the elevated iMg/tMg fraction exhibited a negative correlation with albumin concentrations and we know that low albumin concentrations are associated with increased mortality risk24, 25.
Additionally, in this study, the iMg/tMg fraction was predominantly associated with a significant decrease in iMg and concurrently inappropriately normal tMg concentrations when the difference between the two exceeded 40%. This observation may contribute to a more comprehensive understanding of the stronger association with adverse clinical outcomes when compared to tMg or iMg in isolation. Notably, our findings revealed a higher PPV for hypomagnesemia by iMg with 6-month mortality (17.6%) compared to hypomagnesemia measured by tMg (9.1%). This is in concordance with the literature and supports the fact that hypomagnesemia measured by iMg is associated with unfavorable clinical outcomes, including mortality21, 43. Besides that, the inverse correlation between serum albumin concentrations and iMg/tMg fraction can be explained by positing that individuals with lower albumin concentrations tend to have a higher proportion of their serum Mg in the ionized form44. This was described as “pseudo-hypomagnesemia” of tMg concentration as a result of hypoalbuminemia with normal serum iMg concentrations45.
In general, there is a dearth of comprehensive research on the significance of dysmagnesemia by iMg, especially among patients hospitalized in general medical wards. The absence of prior studies primarily focused on this specific population emphasizes the necessity for further investigation in this field. In our study, we further examined the association between dysmagnesemia and adverse clinical outcomes after excluding patients with eGFR < 60 ml/min/1.73 m² and/or multimorbidity. We found a significant association between elevated tMg levels and increased risks of cardiac arrest and 6-month mortality. This association was particularly pronounced when there was a substantial dissociation in the iMg/tMg ratio. These findings highlight the importance of dysmagnesemia as a predictor of adverse clinical outcomes. In fact, acid-base disturbances and CKD play critical roles in magnesium homeostasis, influencing its absorption, excretion, and overall balance in the body. In CKD, the kidneys’ ability to maintain magnesium balance is progressively compromised as glomerular filtration rate declines. This is because the kidneys lose their ability to effectively excrete magnesium, leading to its accumulation in the blood. Additionally, acid-base imbalances, commonly seen in CKD and critically ill patients, further complicate magnesium homeostasis46,47,48,49. Thus, these results should be interpreted with caution, as they are based on a single measurement of magnesium concentrations and do not account for the potential influence of acid-base status, which can dynamically affect magnesium regulation.
This study has several strengths, including the prospective design and the inclusion of both tMg and iMg measurements. The study findings raise questions about the potential confounding effect behind the previously reported association between total dysmagnesemia and poor health outcomes. Limitations of the study include that it was a single-center study, the measurements of Mg concentration were based on a single reading and did not account for the potential influence of acid-base status, which can dynamically affect magnesium regulation and leads to misclassification. Hence, this might affect the ability to assess causality and limits generalizability, especially that some patients received Mg correction during the hospital stay. Finaly, our reference range used for both iMg and tMg may not be adequate.
Conclusion
There was a strong positive correlation between concentrations of iMg and tMg, that might be supporting the potential use of point-of-care devices for rapid assessment of iMg in critical situations. Notably, hypomagnesemia by iMg was associated with a higher prevalence of diabetes, while hypermagnesemia by both iMg and tMg showed associations with CKD. Multivariant regression analysis showed that hypermagnesemia by tMg was assocciated with adverse health outcomes. The generalizability of the study findings should be taken with caution, highlighting the importance of further research to examine the complex associations and impacts of dysmagnesemia in various clinical settings.
Data availability
All necessary data are included, and further data are available on reasonable request to the corresponding author.
References
Al Alawi, A. M., Al Badi, A., Al Huraizi, A. & Falhammar, H. Magnesium: the recent research and developments. Adv. Food Nutr. Res. 96, 193–218 (2021).
Al Alawi, A. M., Majoni, S. W. & Falhammar, H. Magnesium and human health: perspectives and research directions. Int. J. Endocrinol. 2018, 9041694 (2018).
Cheungpasitporn, W., Thongprayoon, C. & Qian, Q. Dysmagnesemia in hospitalized patients: prevalence and prognostic importance. Mayo Clin Proc. 90(8), 1001-10. (2015).
Thongprayoon, C., Sy-Go, JPT., Nissaisorakarn, V., Dumancas, CY., Keddis, MT., Kattah, A. G., Pattharanitima, P., Vallabhajosyula, S., Mao, M. A., Qureshi, F., Garovic, V. D., Dillon. J. J., Erickson, S. B. & Cheungpasitporn, W. Machine learning consensus clustering approach for hospitalized patients with dysmagnesemia. Diagnostics (Basel). 11(11), 2119. https://doi.org/10.3390/diagnostics11112119. (2021).
Shin, H. J., Han, J., Yim, S. & Do, S. H. Incidence of ionized hypomagnesemia in adult patients undergoing noncardiac major surgery: a prospective observational trial. Magnes Res. 35 (1), 11–17 (2022).
Al Shukri, Z. et al. Incidence of dysmagnesemia among medically hospitalized patients and associated clinical characteristics: a prospective cohort study. Int. J. Endocrinol. 2023, 6650620 (2023).
Gautam, S. & Khapunj, A. Prevalence of hypomagnesemia among elderly patients attending a tertiary care center: a descriptive cross-sectional study. JNMA J. Nepal. Med. Assoc. 59 (233), 35–38 (2021).
Aal-Hamad, A. H., Al-Alawi, A. M., Kashoub, M. S. & Falhammar, H. Hypermagnesemia in clinical practice. Med. (Kaunas) 59(7), 1190. https://doi.org/10.3390/medicina59071190. (2023).
Cheungpasitporn, W. et al. Impact of admission serum magnesium levels on long-term mortality in hospitalized patients. Hosp. Pract. (1995). 48(2), 80–85 (2020).
Del Gobbo, L. C. et al. Circulating and dietary magnesium and risk of cardiovascular disease: a systematic review and meta-analysis of prospective studies. Am. J. Clin. Nutr. 98(1), 160–173 (2013).
Al Alawi, A. M., Berhane, T., Majoni, S. W. & Falhammar, H. Characteristics and health outcomes of patients hospitalised with hypomagnesaemia: a retrospective study from a single centre in the Northern Territory of Australia. Intern. Med. J. 52 (9), 1544–1553 (2022).
Al-Maqbali, J. S., Al Alawi, A. M., Al-Falahi, Z., Falhammar, H. & Al-Zakwani, I. & Al Za’abi M. The stability of analytes of ionized magnesium concentration and its reference range in healthy volunteers. Biomedicines 11(9), 2539. https://doi.org/10.3390/biomedicines11092539. (2023).
Lanzinger, M. J. et al. The relationship between ionized and total serum magnesium concentrations during abdominal surgery. J. Clin. Anesth. 15 (4), 245–249 (2003).
Rooney, M. R., Rudser, K. D., Alonso, A., Harnack, L., Saenger, A. K. & Lutsey, P. L. Circulating ionized magnesium: comparisons with circulating total magnesium and the response to magnesium supplementation in a randomized controlled trial. Nutrients 12(1), 263. https://doi.org/10.3390/nu12010263. (2020).
Al-Yarabi, A. et al. Inappropriate hospital stay of patients admitted under care of General Medicine Units: a retrospective study. Sultan Qaboos Univ. Med. J. 23 (2), 174–181 (2023).
Sun, Q., Welsh, K. J., Bruns, D. E., Sacks, D. B. & Zhao, Z. Inadequate reporting of analytical characteristics of biomarkers used in clinical research: a threat to interpretation and replication of study findings. Clin. Chem. 65 (12), 1554–1562 (2019).
Datta, S. K. & Chopra, P. Interference in Ion-Selective electrodes due to proteins and lipids. J. Appl. Lab. Med. 7 (2), 589–595 (2022).
Malinowska, J., Małecka, M. & Ciepiela, O. Variations in magnesium concentration are associated with increased mortality: study in an unselected population of hospitalized patients. Nutrients 12(6), 1836. https://doi.org/10.3390/nu12061836. (2020).
Yeh, D. D. et al. Total and ionized magnesium testing in the surgical intensive care unit - opportunities for improved laboratory and pharmacy utilization. J. Crit. Care. 42, 147–151 (2017).
Scarpati, G., Baldassarre, D., Oliva, F., Pascale, G. & Piazza, O. Ionized or total magnesium levels, what should we measure in critical ill patients? Transl Med. UniSa. 23, 68–76 (2020).
Gagliano, V., Schäffeler, F., Del Giorno, R., Bianchetti, M., Carvajal Canarte, C. F., Caballero Regueira, J. J. & Gabutti, L. Does ionized magnesium offer a different perspective exploring the association between magnesemia and targeted cardiovascular risk factors? J. Clin. Med. 11(14), 4015. https://doi.org/10.3390/jcm11144015. (2022).
Bouillon-Minois, J. B., Khaled, L., Vitte, F., Miraillet, L., Eschalier, R., Jabaudon, M., Sapin, V., Derault, L., Kahouadji, S., Brailova, M., Durif, J., Schmidt, J., Moustafa, F., Pereira, B., Futier, E. & Bouvier, D. Ionized magnesium: interpretation and interest in atrial fibrillation. Nutrients 15(1), 236. https://doi.org/10.3390/nu15010236. (2023).
Rosanoff, A. et al. Recommendation on an updated standardization of serum magnesium reference ranges. Eur. J. Nutr. 61 (7), 3697–3706 (2022).
Huijgen, H. J. et al. Magnesium levels in critically ill patients. What should we measure? Am. J. Clin. Pathol. 114 (5), 688–695 (2000).
Escuela, M. P. et al. Total and ionized serum magnesium in critically ill patients. Intensive Care Med. 31 (1), 151–156 (2005).
Hansen, B. A. & Bruserud, Ø. Hypomagnesemia in critically ill patients. J. Intensive Care. 6, 21 (2018).
Gommers, L. M., Hoenderop, J. G., Bindels, R. J. & de Baaij, J. H. Hypomagnesemia in type 2 diabetes. Vicious Circle? Diabetes. 65 (1), 3–13 (2016).
Pham, P. C. et al. Lower serum magnesium levels are associated with more rapid decline of renal function in patients with diabetes mellitus type 2. Clin. Nephrol. 63 (6), 429–436 (2005).
Batar, P. K. Study of serum magnesium level in diabetes mellitus and it’s correlation with micro and macro complications. J. Assoc. Physicians India. 70 (4), 11–12 (2022).
Bherwani, S. et al. Hypomagnesaemia: a modifiable risk factor of diabetic nephropathy. Horm. Mol. Biol. Clin. Investig. 29 (3), 79–84 (2017).
Chu, C. et al. Low serum magnesium levels are associated with impaired peripheral nerve function in type 2 diabetic patients. Sci. Rep. 6, 32623 (2016).
Agrawal, P., Arora, S., Singh, B., Manamalli, A. & Dolia, P. B. Association of macrovascular complications of type 2 diabetes mellitus with serum magnesium levels. Diabetes Metab. Syndr. 5 (1), 41–44 (2011).
Wang, S. et al. Serum electrolyte levels in relation to macrovascular complications in Chinese patients with diabetes mellitus. Cardiovasc. Diabetol. 12, 146 (2013).
Pethő, Á. G., Tapolyai, M., Browne, M. & Fülöp, T. Hypomagnesemia as a risk factor and accelerator for vascular aging in diabetes Mellitus and chronic kidney disease. Metabolites 13(2) 306. https://doi.org/10.3390/metabo13020306. (2023).
Galán Carrillo, I. et al. Impact of serum magnesium levels on kidney and cardiovascular prognosis and mortality in CKD patients. J. Ren. Nutr. 31 (5), 494–502 (2021).
Steinberger, H. A. & Hanson, C. W. 3 Outcome-based justification for implementing new point-of-care tests: there is no difference between magnesium replacement based on ionized magnesium and total magnesium as a predictor of development of arrhythmias in the postoperative cardiac surgical patient. Clin. Lab. Manage. Rev. 12 (2), 87–90 (1998).
Fawcett, W. J., Haxby, E. J. & Male, D. A. Magnesium: physiology and pharmacology. Br. J. Anaesth. 83 (2), 302–320 (1999).
RayBS Louise Eisenhardt. 1891–1967. Surg. Neurol. 8 (4), 227–228 (1977).
Vanoni, F. O., Milani, G. P., Agostoni, C., Treglia, G., Faré, P. B., Camozzi, P., Lava, S. A. G., Bianchetti, M. G. & Janett, S. Magnesium metabolism in chronic alcohol-use disorder: meta-analysis and systematic review. Nutrients 13(6), 1959. https://doi.org/10.3390/nu13061959. (2021).
Cidade-Rodrigues, C. et al. The prognostic impact of magnesium in acute heart failure is different according to the presence of diabetes mellitus. Porto Biomed. J. 7 (6), e197 (2022).
Angkananard, T. et al. The association of serum magnesium and mortality outcomes in heart failure patients: a systematic review and meta-analysis. Med. (Baltim). 95 (50), e5406 (2016).
Nasser, R., Naffaa, M. E., Mashiach, T., Azzam, Z. S. & Braun, E. The association between serum magnesium levels and community-acquired pneumonia 30-day mortality. BMC Infect. Dis. 18 (1), 698 (2018).
Soliman, H. M., Mercan, D., Lobo, S. S., Melot, C. & Vincent, J. L. Development of ionized hypomagnesemia is associated with higher mortality rates. Crit. Care Med. 31 (4), 1082–1087 (2003).
Saha, H., Harmoinen, A., Karvonen, A. L., Mustonen, J. & Pasternack, A. Serum ionized versus total magnesium in patients with intestinal or liver disease. Clin. Chem. Lab. Med. 36 (9), 715–718 (1998).
Kulpmann, W. R., Rossler, J., Brunkhorst, R. & Schuler, A. Ionised and total magnesium serum concentrations in renal and hepatic diseases. Eur. J. Clin. Chem. Clin. Biochem. 34 (3), 257–264 (1996).
Rylander, R., Tallheden, T. & Vormann, J. Acid-base conditions regulate calcium and magnesium homeostasis. Magnes Res. 22 (4), 262–265 (2009).
Cunningham, J., Rodriguez, M. & Messa, P. Magnesium in chronic kidney disease stages 3 and 4 and in dialysis patients. Clin. Kidney J. 5 (Suppl 1), i39–i51 (2012).
Felsenfeld, A. J., Levine, B. S. & Rodriguez, M. Pathophysiology of calcium, phosphorus, and magnesium dysregulation in chronic kidney disease. Semin Dial. 28 (6), 564–577 (2015).
Rylander, R., Remer, T., Berkemeyer, S. & Vormann, J. Acid-base status affects renal magnesium losses in healthy, elderly persons. J. Nutr. 136 (9), 2374–2377 (2006).
Cockwell, P. & Fisher, L. A. The global burden of chronic kidney disease. Lancet. 395 (10225), 662–664 (2020).
McCluskey, A., Thomas, A. N., Bowles, B. J. & Kishen, R. The prognostic value of serial measurements of serum albumin concentration in patients admitted to an intensive care unit. Anaesthesia. 51 (8), 724–727 (1996).
Funding
The research was supported by a grant from the Sultan Qaboos University IG/MED/MEDE/23/01. Nova Biomedical (Waltman, Massachusetts, United States) supplied the electrolytes analyzer for ionized magnesium detection with its consumables (Stat Profile Prime Plus®). Technical support and maintenance services were provided by Nova Biomedical and Global Source Trading. The funding organizations had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit.
Author information
Authors and Affiliations
Contributions
Conceptualization: AA and HF: Data curation: ZS, DA, QM, MT, AS and RB: Formal analysis: JM: Funding acquisition: AA, and SB: Investigation: AA, JM, ZS and SB: Methodology: AA, JM and ZS: Project administration: AA and JM: Resources: AA, JM, ZS and SB: Machine handling: QM, MT and DA. Supervision: AA and JM. Validation: AA and JM: Drafting the manuscript: AA, SB, HF and JM. Critical review of the manuscript for important intellectual content: All authors.
Corresponding author
Ethics declarations
Institutional review board statement
The study was approved by the Medical and Research Ethics Committee at the College of Medicine and Health Sciences, SQU, Muscat, Oman (MREC #2719; SQU-EC/51/2022; dated: April 1st, 2022).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Al Alawi, A.M., Al Shukri, Z., Al-Busaidi, S. et al. Prevalence, clinical characteristics, and health outcomes of dysmagnesemia measured by ionized and total body concentrations among medically hospitalized patients. Sci Rep 14, 23668 (2024). https://doi.org/10.1038/s41598-024-74920-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-74920-5
Keywords
This article is cited by
-
Elevation of ionized magnesium level in the blood is a risk factor for mortality in patients undergoing maintenance hemodialysis
Renal Replacement Therapy (2025)
-
Ionized and total magnesium levels and health outcomes in patients with type 2 diabetes mellitus
Scientific Reports (2025)