Abstract
Sediment dredging and aeration are used as important technical measures to remediate internal loading of sediment in polluted rivers. However, previous studies have overlooked the impact of dredging and aeration on Greenhouse gases (GHGs) emission. We established three aeration rate(six different aeration intervals), one dredging treatment to investigate the effect of aeration and dredging on pollutant removals and CO2, CH4 and N2O emissions. The results indicated the pollutants and GHGs at 2.4, 3.4, 4.4 L min−1 aeration rates reached collaborative emission reduction after more than 3 h or within 1.5 h. Meanwhile, the GHGs fluxes after aeration decreased with the increasing aeration rate, with the mean CO2, CH4 and N2O fluxes of 69.74, 0.16, 7.53 mg m−2 h−1 and 33.64, 0.09, 4.17 mg m−2 h−1 before and after aeration, respectively. With respect to dredging, the pollutants and N2O reached synergic effects between reduction of pollution and carbon emissions after 1 h dredging. Specifically, the CO2 and CH4 emissions after dredging was lower than those of before dredging, but the N2O emissions was higher than those of before dredging. In addition, our analysis revealed that the dissolved oxygen (DO), oxidation-reduction potential (ORP), available potassium (AK) and ammoniacal nitrogen (NH4+-N) in the sediment influenced GHGs fluxes at the water-air interface in the aeration. Our study indicated moderate aeration and dredging can achieve the synergistic effect in reducing pollution and carbon emissions.
Similar content being viewed by others
Introduction
Although the global freshwater only covers 2.5% of the Earth’s surface1, freshwater contributes to high atmospheric CO2, CH4 and N2O emissions2,3,4. It has been estimated that global freshwater emits 3.9 Pg C to the atmosphere every year5, 159 (117–212) Tg CH4 and 0.12 Tg N (0.02–0.2) to the atmosphere every year2, accounting for approximately 42.9% and 40.0% of the total natural sources respectively2. However, global CO2 emissions from freshwater has not been accurately estimated, due mainly to inaccurate anthropogenic sources estimation and the high spatial variation of the effluxes2,6. Additionally, it has been underestimated the global CH4 and N2O emissions from freshwaters due to lack of anthropogenic sources and worldwide comprehensive data set of CH4 and N2O fluxes components2,3,7. Especially, Terrestrial carbon inputs to inland waters reached upward of 5.1 Pg of C annually6. Thus, the GHGs emissions of freshwater pollution from terrestrial inputs may play an important role in global freshwater GHGs budgets.
Many previous studies have demonstrated that freshwater pollution is as a strong source of atmospheric CO2, CH4 and N2O emissions6,7,8,9, Especially the urban black-odor rivers. Past studies showed that the GHGs emissions in polluted rivers were evidently higher than those in unpolluted rivers6,10,11. It has only been reported that the wastewater emits 0.4 Tg N (0.2–0.5) every year in IPCC AR62,3, which was twice as high as Intergovernmental Panel on Climate Change (IPCC) AR5. However, global GHGs emissions from freshwater pollution have not been accurately estimated due to lack of other polluted freshwater measurements, especially the impact of treatment technology on pollution.
Dredging and aeration are effective physical methods for controlling sediment pollution in black-odor rivers12,13,14,15. Sediment dredging has been used extensively to improve the water quality of lake eutrophication in China, but it can cause a second pollution due to disturbance sediment and need some storage location for dredged sediment15. On the other hand, aeration can offer an aerobic environment for microbial degradation of contaminants but not considering low-carbon goals16. In fact, dredging and aeration were used to control freshwater’ internal loading. Dredging can release pollutants into the water body associated with the change of environmental conditions (including physical, chemical and/or biological) in the sediment-water system. At the same time, aeration can remove the pollutants in the water system, but can remove the pollutants in the sediment when the aeration depth is deep enough. In particular, our previous studies have shown that the aeration and dredging promoted the removals of pollutants in the water17,18. It is noted that aeration remove nutrients from black and odorous water bodies and sediment. Previous studies have showed that moderate aeration facilitated the growths of related bacteria to N- and S-cycling and the Proteobacteria was dominant19. The impact of combining aeration and biofilm technology on nitrogen conversion in black and odorous rivers indicated that aeration can achieve a removal rate of 52.94% for total nitrogen in sediment20, and the removal rates of COD, NH4+-N and TP can reach 38%, 62% and 42% after aeration and setting elastic fillings in the river21. Past studies mainly focused on their impacts of the migration of nitrogen and phosphorus in sediment, the balance of nitrogen and phosphorus in water12. It has not yet been reported that dredging and aeration affects GHGs emissions in black-odor river ecosystems. A better understanding of the dredging and aeration affecting GHGs effluxes and their controlling factors is crucial to accurately estimating the reginal and global GHGs budgets from freshwater pollution.
In this study, we established three different aeration rates (at high, medium and low gradients) and one dredging treatment during six different time intervals. We measured the physical and chemical indicators in the water and sediment, and GHGs effluxes after aeration and dredging. We also analyzed the synergistic effects of GHGs emissions and the pollutants-removal rates to estimate the reduction of pollution and carbon emissions. Collectively, we aim to (1) examine the effect of aeration and dredging on the physical and chemical properties in the water and sediment; (2) explore the effect of aeration and dredging on GHGs effluxes; and (3) quantify the effect of aeration and dredging on the reduction of pollution and carbon emissions. We particularly focused on the relative contribution of different GHGs to global warming and the synergistic effects in order to provide the management decision-making for regulators.
Materials and methods
Site description
The study site is located in a greenhouse of the Chinese Research Academy of Environmental Sciences, in Beijing (40°3′ N and 116°25′ E). The climate is a warm temperate semi-humid continental monsoon with four distinct seasons. The highest temperature and lowest temperature are 40.6 °C and − 27.4 °C, respectively. The average annual active accumulated temperature greater than 0 °C and 10 °C are 4580 °C and 4168 °C respectively, with the average annual frost-free period of 181 days. The annual average precipitation and the annual average sunshine hours are 569.4 mm and 2764 h, respectively.
Experimental design
Plot setup
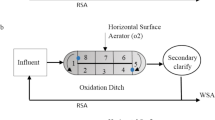
The experiment was set in a water tank with dimensions 142 cm × 97 cm × 76 cm in length × width × height with a volume of 900 L, the bottom was evenly overlaid with 0.1-m thick sediments from nearby river sediment (Black and odorous induced endogenous pollution). The water depth was 20 cm by continuously adding water to tank. After 15 days of setup, we started our experiment. We set up three plots with low (2.4 L min−1), medium (3.4 L min−1) and high aeration rate (4.4 L min−1) using a double tube oxygen pump (X6, Guangzhou Shuoxiang Electronic Technology Company, China) with setting six aeration intervals (0.5 h, 1 h, 1.5 h, 2 h, 3 h, 4 h) separately, with a total of 18 treatments. At the same time, the dredging treatment was set up, and the sediment at a depth of 10 cm was dredged to about 5 cm by a small shovel. The atmosphere, water and sediment samples were took before dredging and after 0.5 h, 1 h, 1.5 h, 2 h, 3 h, 4 h dredging (Fig. 1).
Scheme of setup and graphical presentation of the experiments.
Sampling
Samplings took place from January 10, 2022, to January 18, 2022, before and after aeration and dredging, which included greenhouse gases (GHGs) samples and sediment samples. We sampled GHGs at the water-air interface when the concentration of CO2 changed stable with a CO2 analyzer (LI-820, LI-COR Company, USA) after aeration and dredging. Then water quality parameters were measured through YSI EXO1 multi-parameter water quality meter (EXO1 Multiparameter System, YSI Company, USA). We collected sediment samples using a small scoop after obtaining gas samples and water measurements.
Measurements
GHGs flux measurements
GHGs fluxes at the water-air interface were measured using the floating static chambers22 The chamber system is composed of two parts: a movable cylindrical steel chamber body and a fixed base with a diameter of 16 cm, covering an area of 0.02 m2. The base was inserted into the water to a depth of 3 cm. The floating chamber was constructed using a PVC pipe 51.8 cm in length and 16 cm in diameter with styrofoam floats attached to the sides. The chamber was wrapped with aluminum foil to reduce the temperature change in the chamber during sampling. At the beginning of measurements, we positioned the chamber body on top of the base and used a silicon tube to seal the joint to form a closed system. A detailed description of the floating-chamber system and sampling can be found in Shao et al. (2017). The gas samples were each taken at a 10-minute interval to finish the measurements in about half an hour at each location. The air samples were soon transported to the laboratory for analyzing the GHGs concentrations with a gas chromatograph (GC 7890 A, Agilent, USA). The GHGs effluxes was calculated with a linear model by Liu et al. (2013). Cumulative emission of GHGs were calculated by the following Eq.
Where CE is the cumulative emission of GHGs (mg m−2), F is the GHGs flux (mg m−2 d−1), Dj+1-Dj is the interval between two adjacent sampling, J is the jth sampling, m is the total number of samplings.
Global warming potential (GWP) can be used to quantitatively calculate the relative contribution of different GHGs to global warming. A detailed description of the GWP can be found in previous studies2,24. In addition, it should be noted that the GWP before dredging was calculated using the GHGs fluxes (as a constant) multiplying 4 h. The GWP was calculated by the following Eq.
Where GWP is the CO2 equivalent of three GHGs emissions (mg m−2), CE is the cumulative GHGs emissions (mg m−2).
The synergistic effect coefficient is a dimensionless number. Past studies referred to the calculation formula of synergistic effect coefficient. and proposed the calculation system as shown in Eq. (5)25. The coefficient and based on the characteristics of this study, proposes the calculation system as shown in Eq. (5). This coefficient can reflect the “quantity” and “rate” changes of carbon (water Chl.a), nitrogen (sediment NH4+-N element removal in water and greenhouse gas (CO2, CH4 and N2O) emissions and quantitatively evaluate the synergistic effect of decarbonization and nitrogen removal on greenhouse gas (CO2, CH4 and N2O) emissions.
Where E(G, W) is the synergistic effect coefficient (dimensionless); ∆FCO2, ∆FCH4 and ∆FN2O are the changes of CO2, CH4 and N2O fluxes before and after the aeration and dredging (mg m−2 h−1), respectively; FCO2, FCH4 and FN2O are the CO2, CH4 and N2O fluxes before the aeration and dredging (mg m−2 h−1), respectively; ∆Cc is the change of pollutant (water Chl.a) before and after the aeration and dredging (mg L−1), respectively; Cc is the concentration of Chl.a in the water on the before the aeration and dredging (mg L−1).\(\mathop {\Delta C}\nolimits_{{\mathop {{\text{NH}}_{4}^{+} - {\text{N}}}\limits_{{}} }}\) is the change of pollutants (sediment NH4+-N before and after the aeration and dredging (mg L−1); \(\mathop C\nolimits_{{\mathop {{\text{NH}}_{4}^{+} - {\text{N}}}\limits_{{}} }}\) is the concentration of NH4+-N in the sediment before the aeration and dredging (mg L−1).
When the coefficient of synergistic effect E (G, W) > 0, it indicates that the freshwater ecosystem has a positive synergistic effect on the removal of water pollutants and the reduction of GHGs emissions; when E (G, W) < 0, it indicates that the freshwater ecosystem has a negative effect on the removal of water pollutants and the reduction of GHGs emissions. In other words, freshwater ecosystems only have the reduction effect on either water pollutants or GHGs emissions. Referring to the collaborative state classification standard in Sun et al. (2023), he collaborative state is divided into 8 categories as follows (Table 1).
Water and sediment measurements
Dissolved oxygen (DO), pH, oxidation-reduction potential (ORP) and chlorophyll a (Chl.a) in the water were measured using the YSI EXO1 Multiparameter System (EXO1 Multiparameter System, YSI Company, USA). In the laboratory, the ammoniacal nitrogen (NH4+-N), the available phosphorus (AP) and the available potassium (AK) in the sediment were measured using the high intelligence Soil Nutrient Detector (TR-G01S, Beijing Saiyasi Technology, China).
Data analysis
We also conducted normality tests for the GHGs fluxes and found that GHGs fluxes followed normal frequency distribution. We also used correlation analyses to determine the relationships between environmental variables and GHGs fluxes. All statistical analyses were performed using the SPSS 23.0 statistical software (SPSS Inc., Chicago, IL, USA), and graphs were created using the Sigma Plot 11.0 program (Systat Software Inc., San Jose, CA, USA).
Results
The synergistic effect of aeration and dredging on the reduction of pollution and carbon emissions in microcosms
Our results revealed synergic effects between reduction of pollution and carbon emissions in aeration and dredging. When the aeration time reached more than 3 h or within 1.5 h, the pollutants and GHGs at 2.4 L min−1, 3.4 L min−1, 4.4 L min−1 aeration rates reached collaborative emission reduction, that is, the pollutants-removal rates and GHGs reduction rates (especially N2O) were less than zero respectively, but the coefficient of synergistic effect for pollution reduction and carbon reduction was greater than zero (Tables 1 and 2). It should be noted that after 2 h aeration time the pollutants and GHGs (especially N2O) at 2.4 L min−1 and 4.4 L min−1 aeration rates reached synergic effects between reduction of pollution and carbon emissions. After 1 h dredging, the pollutants and N2O reached synergic effects between reduction of pollution and carbon emissions (Table 3).
The effect of aeration and dredging on the physical and chemical properties in Microcosms
The effect of aeration and dredging on the physical and chemical properties of water
Although the pattern of DO concentration and ORP in the water before and after aeration was similar, but the trends of DO concentration and ORP were different at three different aeration rates. It should be noted that after aeration only at 2.4 L min−1 aeration rate the DO concentration was larger than that of before aeration. After aeration, the maximum DO concentration in the water under three aeration rates were 10.11 mg L−1, 9.55 mg L−1 and 9.71 mg L−1 after 4 h, 4 h and 3.5 h aeration respectively, while ORP was 356.9, 362.1 and 230.4 after 0.5 h, 4 h and4 h at 2.4 L min−1, 3.4 L min−1, 4.4 L min−1 aeration rates, respectively. In addition, the pattern and trend of pH were both different before and after aeration at three different aeration rates (Fig. 2).
With respect to dredging, the DO concentration and pH after dredging was generally lower than those of before dredging, and the pattern of ORP demonstrated a “W” shape. The pH decreased sharply after dredging then increased slowly after 0.5 h dredging, but DO concentration increased slowly after 2 h dredging (Fig. 3).
Effects of aeration on DO, ORP and pH of water. (a) aeration rate of 2.4 L min−1; (b) aeration rate of 3.4 L min−1 and (c) aeration rate of 4.4 L min−1.
Effects of dredging on DO, ORP and pH of water.
The effect of aeration and dredging on the physicochemical properties of sediment
Among three aeration rates, the medium aeration rate has the best removal effect on nitrogen, phosphorus and kalium concentration (NH4+-N, AP and AK) in the sediment, and the removal rate increased with the aeration time. With increasing aeration time, the change trend of NH4+-N content in the sediment before and after aeration was consistent. At low aeration rates, the NH4+-N content in the sediment after aeration was higher than before aeration, while at medium and high aeration rates, the NH4+-N content in the sediment after aeration was lower than before aeration. At low aeration rates, the changes in sediment AP content and sediment AK content were opposite with increasing aeration time before and after aeration, while at medium and high aeration rates, the changes in sediment AP content and sediment AK content before and after aeration were consistent. After aeration, the AP content of the sediment was generally higher than before aeration. The AK content of the sediment is increased by aeration with low aeration rate, while aeration with medium aeration rate decrease it. At high aeration rate, the AK content of the sediment hardly changed before and after aeration (Fig. 4).
In terms of dredging, the patterns of NH4+-N, AP and AK generally demonstrated “W” shape at different time stages after dredging, and the concentrations of NH4+-N, AP and AK increased after 2 h dredging. It should be noted that the he concentrations of NH4+-N, AP and AK at 4 h stage after dredging was notably higher than those of before dredging, but at 1 h and 2 h was lower than those of before dredging (Fig. 5).
Effect of aeration on NH4+-N, AP and AK of sediment. (a) aeration rate of 2.4 L min−1; (b) aeration rate of 3.4 L min−1 and (c) aeration rate of 4.4 L min−1.
Effect of dredging on NH4+-N, AP and AK of sediment.
The effect of aeration and dredging on GHGs
The effect of aeration and dredging on GHGs emissions in Microcosms
At three different aeration rates, the GHGs emissions after aeration decreased with the increasing aeration rate. At low and high aeration rates, the GHGs emissions before aeration were higher than those of after aeration during a 4-h period, while at medium aeration rates, CO2 and N2O emissions before aeration were higher than those of after aeration. At three different aeration rates, the maximum reduction of CO2, CH4 and N2O emissions occurred at high aeration rate, low aeration rate and low aeration rate compared to no aeration, respectively, while the maximum reduction rate (81.15%) of GHGs emissions both occurred at high aeration rate.
Specifically, the contribution of GHGs to the GWP followed by the N2O, CO2 and CH4 in an increasing order both before and after aeration. We found that the N2O contributed the most to the GWP at low aeration rates with the rate of 97.69% before aeration and 95.93% after aeration, respectively. The CO2, CH4 and N2O emissions at 2.4 L min−1, 3.4 L min−1, 4.4 L min−1 aeration rates before aeration were 267.24 mg m−2,0.76 mg m−2 and 54.47 mg m−2, 97.55 mg m−2, 0.44 mg m−2 and 5.45 mg m−2 and 352.19 mg m−2, 0.30 mg m−2 and 8.26 mg m−2, respectively. After aeration, the CO2, CH4 and N2O emissions at 2.4 L min−1, 3.4 L min−1, 4.4 L min−1 aeration rates were 163.23 mg m−2, 0.39 mg m−2 and 40.60 mg m−2, 105.39 mg m−2, 0.32 mg m−2 and 8.13 mg m−2, and 67.28 mg m−2, 0.15 mg m−2, and 3.26 mg m−2, respectively (Fig. 6).
With respect to dredging, the CO2 and CH4 emissions after dredging was lower than those of before dredging, after dredging, CO2 and CH4 emissions were reduced by 51.73% and 92%, respectively, but GWP and N2O emissions were higher than those before dredging. After dredging, GWP and N2O emissions increased by 252% and 359.46%, respectively (Fig. 7).
Effect of aeration on CO2, CH4 and N2O emissions from the water-air interface.
Effect of dredging on CO2, CH4 and N2O emissions from the water-air interface.
The effect of aeration and dredging on GHGs fluxes at the water-air interface
At different aeration rates, the patterns of CO2 fluxes and its change rate were irregular. The maximum change rate was 79.03%, 3888.96% and 883.88% at 3 h, 3 h, and 1.5 h aeration stage at low, medium and high aeration rate, respectively. The mean CO2 flux before and after aeration and its change rate were 87.20 mg m−2 h−1, 54.06 mg m−2 h−1 and − 28.06%, 38.83 mg m−2 h−1, 25.01 mg m−2 h−1 and 623.99%, 83.18 mg m−2 h−1, 21.87 mg m−2 h−1 and 105.05% at low, medium and high aeration rate, respectively. In addition,, the CO2 flux before and after aeration ranged from 17.75 to 142.36 mg m−2 h−1, and from 3.12 to 142.27 mg m−2 h−1 at low aeration rate respectively, from 1.23 to 91.69 mg m−2 h−1 and from 2.70 to 59.78 mg m−2 h−1 at a moderate aeration rate respectively; from 7.26 to 159.69 mg m−2 h−1, and from 5.30 to 71.41 mg m−2 h−1 at high aeration rates respectively. Specifically, the CO2 fluxes after aeration were generally lower than those of before aeration. However, the CO2 fluxes after aeration were lower than those of before aeration between 0.5-h and 1.5 h aeration rates at low and high aeration rates, but was larger than those of before aeration between 1.5 h and 4 h aeration rate at medium aeration rates (Fig. 8). Compared with before dredging, the CO2 fluxes after dredging showed a trend of decreasing first and then increasing. The CO2 fluxes reached the lowest (8.92 mg m−2 h−1) at 0.5 h after dredging, then increased in a fluctuating manner, and reached the maximum (35.32 mg m−2 h−1) at 3 h after dredging (Fig. 9).
With respect to CH4, the patterns of CH4 fluxes demonstrated “W” and “M” shapes at low and high aeration rates respectively, but the contrary trend of CH4 fluxes change rate. In term of the medium aeration rate, CH4 fluxes before and after aeration.
featured opposite trend, and CH4 fluxes change rate demonstrated similar pattern to that of CH4 flux after aeration. The maximum change rate of CH4 fluxes was1276.72%, 187.21% and 370.06% at 1.5-h, 2-h and 0.5-h aeration stage at low, medium and high aeration rate, respectively. The mean CH4 flux before and after aeration and its change rate were 0.27 mg m−2 h−1, 0.15 mg m−2 h−1 and 189.26%, 0.15 mg m−2 h−1, 0.08 mg m−2 h−1 and 17.92%, 0.073 mg m−2 h−1, 0.045 mg m−2 h−1 and 27.26% at low, medium and high aeration rate, respectively. Additionally, the CH4 flux before and after aeration ranged from 0.0078 mg m−2 h−1 to 0.50 mg m−2 h−1, from 0.032 mg m−2 h−1 to 0.44 mg m−2 h−at low aeration rate respectively, from 0.044 mg m−2 h−1to 0.38 mg m−2 h−1and from 0.016 mg m−2 h−1 to 0.15 mg m−2 h−1 at a moderate aeration rate respectively; from 0.011 mg m−2 h−1 to 0.13 mg m−2 h−1, and from 0.021 mg m−2 h−1 to 0.075 mg m−2 h−1 at high aeration rates respectively. At low and high aeration rates, the CH4 fluxes after aeration was generally lower than the CH4 fluxes before aeration. At medium aeration rates, the CH4 fluxes after aeration was lower than those of before aeration between 0.5 h and 1.5 h, but were larger than those of before aeration between 1.5 h and 3 h (Fig. 8). Compared with before dredging, the CH4 fluxes decreased significantly after dredging, and the CH4 fluxes after 0.5 h after dredging was only 2.90% of that before dredging. After dredging, it showed a continuous downward trend, reaching the maximum after 1.5 h (0.75 mg m−2 h−1), but it was only 17.79% of the CH4 fluxes before dredging (Fig. 9).
For N2O, the patterns of N2O fluxes after aeration and its change rates demonstrated “M” and “W” shapes at low and high aeration rates respectively, but the contrary trend of N2O fluxes before aeration and its change rate at medium aeration rates. The maximum change rate of N2O fluxes were 1049.66%, 1673.17% and 24.36% at 1.5-h, 2-h and 0.5-h aeration stage at low, medium and high aeration rate, respectively. The mean N2O flux before and after aeration and its change rate were 18.09 mg m−2 h−1, 9.27 mg m−2 h−1 and 192.34%, 2.15 mg m−2 h−1, 2.03 mg m−2 h−1 and 397.99%, 2.35 mg m−2 h−1, 1.21 mg m−2 h−1 and − 46.56% at low, medium and high aeration rate, respectively. Additionally, the N2O flux before and after aeration ranged from 1.09 mg m−2 h−1 to 86.86 mg m−2 h−1, from 0.26 mg m−2 h−1 to 13.12 mg m−2 h−at low aeration rate respectively, from 0.22 mg m−2 h−1to 4.64 mg m−2 h−1and from 0.83 mg m−2 h−1 to 3.93 mg m−2 h−1 at a moderate aeration rate respectively; from 0.89 mg m−2 h−1 to 5.13 mg m−2 h−1, and from 0.15 mg m−2 h−1 to 3.21 mg m−2 h−1 at high aeration rates respectively. Especially, the N2O fluxes after aeration was lower than those of before aeration only at high aeration rates (Fig. 8). Compared with before dredging, the N2O fluxes decreased first and then increased after dredging and reached the maximum (46.88 mg m−2 h−1) at 1.5 h after dredging, which was much higher than that before dredging. The N2O fluxes were 26.62 times that before dredging, but the N2O fluxes decreased rapidly to 0.11 mg m−2 h−1 at 4 h after dredging, which was only 6.23% of that before dredging (Fig. 9).
Effect of aeration on CO2, CH4 and N2O fluxes at the water-air interface. (a) aeration rate of 2.4 L min−1; (b) aeration rate of 3.4 L min−1 and (c) aeration rate of 4.4 L min−1.
Effect of dredging on CO2, CH4 and N2O fluxes at the water-air interface.
The influence of water and soil physical and chemical properties on GHGs fluxes
In our study, DO, ORP, AK and NH4+-N in the sediment influenced GHGs fluxes at the water-air interface in the aeration by affecting GHGs production conditions. CO2 fluxes were strongly correlated with ORP before aeration (r = 0.82, p < 0.05). However, no significant correlation was found between the CO2 fluxes and other environmental factors. The CH4 efflux was highly correlated with DO before aeration, NH4+-N in the sediment before aeration and AK after aeration (p < 0.01, r=-0.83, 0.86 and-0.84, respectively). We did not find any significant relationship between the CH4 efflux and other environmental variables.
We found that the N2O efflux in the lake was significantly correlated with DO after aeration, ORP at the low aeration rate before aeration and NH4+-N in the sediment before aeration (p < 0.05, r=-0.83, -0.82 and 0.86, respectively) (Table 4), ORP at medium aeration rate before aeration (p < 0.01, r=-0.95) (Table 4).
In addition, we made correlation analyses for three aeration rates between GHGs effluxes and environmental variables. But we did not observe any significant correlation between GHGs effluxes and environmental parameters. In terms of dredging, there was no correlation between environmental factors and GHGs fluxes (Table 5).
Discussion
The reduction of pollution and carbon emissions
In our current study, we found that aeration and dredging promoted the synergistic effect of pollutants and GHGs emission reduction in the aeration and dredging, similar to other studies in ponds26,27,28 and constructed wetlands29,30,31. In our study, after aeration and dredging the pollutants removal rate, GHGs emission reduction and the coefficient of synergistic effect met standard requirements of pollution and carbon reduction (Tables 2 and 3). In addition, Chen et al. (2021) reported that aeration increased the removal rate of pollutants (COD, TN and NH4+-N and reduced GHGs emissions (CH4 and N2O) in wastewater ecological soil infiltration systems. Meanwhile, Wan et al. (2021) found that aeration can reduce N2O emission on the basis of the constant N removal rate. That may be pollutants as substrate supply promoted higher GHGs conversion efficiency. Another reason may be the fact that DO increased pollutants removals32,34,35 and reduced ebullitive CH4, inhibited CH4 production, enhanced CH4 oxidation and N2O production26,30.
Specially, aeration at 3.4 L min−1 and 4.4 L min−1 aeration rates gave better synergistic effect than that of 2.4 L min−1 because three GHGs simultaneously reached synergistic effect in most aeration time. That may be due to the sufficient DO supply at 3.4 L min−1 and 4.4 L min−1 aeration rates. The intermittent aeration between aerobic and anaerobic conversion at high aeration rates may be influence biological uptake by microbes (microbial growth), low GHGs emissions and favoring pollutant removal in our study.
The effect of aeration on GHGs emissions
In our study, aeration reduced GHGs emissions except increased N2O emission, which was agreement with other studies36,37. Aeration in the 2.4 L min−1 and 4.4 L min−1 in the current study reduced the GWP of 65.79% and 81.15% respectively. For example, Fang et al. (2022) found that aeration can reduce the 71% of global CH4 emissions and 63% of CH4 emissions in the aquaculture. Yang et al. (2023) reported that aeration was able to decrease GWP of aquaculture ponds by 40%, while the GWP of 206 (mg CO2-eqm−2h−1-CH4 and N2O) in aerated aquaculture ponds was notably lower than that of in the aeration in non-aerated pond (342 mg CO2-eqm−2h−1). Ji et al. (2020) found that intermittent aeration reduced 43.9% GWP in the subsurface-flow constructed wetlands. The proportion of GWP we have reduced were substantially higher than previous studies above-mentioned, which may be due to intermittent aeration in our study32,38.
In addition, we found that aeration decreased CO2 and CH4 emissions, which was consistent with past studies5,34,39,40. Adequate oxygen after aeration reduced CH4 production as it benefits under anaerobic condition and increased CH4 oxidation39,40. Intermittent aeration could not supply enough oxygen that inhibited organic matter aerobic oxidation and reduced CO2 emissions32. On the contrary, aeration may increase N2O emission in our study because of its opposite trend under the oxygen-deficient condition34,41. For example, Yang et al. (2023) found that aeration increased N2O emission by 98%. In particular, the GHGs fluxes at different aeration rates demonstrated irregular patterns, which may achieve different gas exchange balance between the atmosphere and water.
The effect of aeration on pollutants removals
In the current study, the N and P pollutants removal rates were 11.76% at 3.4 L min−1 aeration rate and 28.23% and 9.88% at 3.4 L min−1 and 4.4 L min−1, respectively, which were in agreements with previous studies37,42,43. Liu et al. (2019) found that the COD removal efficiencies of the aerated systems from 93.32 to 94.71% were much higher than that in non-aerated systems (from 69.89 to 77.53%). Abundant oxygen supply can create aerobic conditions in constructed wetlands, which elevate the growing of aerobic microorganism and therefore enhance aerobic removal of organic matters45,46,47. Additionally, the low pollutants removals rates may be that there was no plant in the microcosms. Plant can absorb the N and P chemical compound as their growing nutrition. It should be noted that low pollutants removals may be the reason for the low efficiency of aeration as physical-treatment48.
Conclusions
Our study demonstrates synergic effects between reduction of pollution and GHGs emissions in aeration and dredging. According to our results, the mean CO2, CH4 and N2O effluxes after aeration were lower 50% than those of before aeration. Furthermore, different pollutants and GHGs reached collaborative emission reduction at different aeration rates and at different time stages for aeration and dredging. Especially, the N2O contributed the most to the GWP at low aeration rates over 90% before aeration and after aeration. Aeration had no influence on DO, pH and ORP, but dredging influenced DO concentration and pH. The medium aeration rate has the best pollutant- removal effect. The GHGs effluxes were related to DO, ORP, AK and NH4+-N in the sediment in the aeration. Specifically, the CO2 fluxes after aeration were generally lower than those of before aeration. the CO2 fluxes after dredging showed a trend of decreasing first and then increasing.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Mishra, R. K. Fresh water availability and its global challenge. Sustainability 4(3), 1–78. https://doi.org/10.37745/bjmas.2022.0207 (2023).
IPCC. Climate Change 2021–The Physical Science Basis: Working Group I Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. https://doi.org/10.1017/9781009157896 (2023).
Tian, H. et al. A comprehensive quantification of global nitrous oxide sources and sinks. Nature 586(7828), 248–256. https://doi.org/10.1038/s41586-020-2780-0 (2020).
Zheng, B. et al. Global atmospheric carbon monoxide budget 2000–2017 inferred from multi-species atmospheric inversions. Earth Syst. Sci. Data 11(3), 1411–1436. https://doi.org/10.5194/essd-11-1411-2019 (2019).
Li, S. et al. Large greenhouse gases emissions from China’s lakes and reservoirs. Water Res. 147, 13–24. https://doi.org/10.1016/j.watres.2018.09.053 (2018).
Drake, T. W., Raymond, P. A. & Spencer, R. G. M. Terrestrial carbon inputs to inland waters: A current synthesis of estimates and uncertainty. Limnol. Oceanogr. Lett. 3(3), 132–142. https://doi.org/10.1002/lol2.10055 (2017).
Zheng, Y. et al. Global methane and nitrous oxide emissions from inland waters and estuaries. Global Change Biol. 28(15), 4713–4725. https://doi.org/10.1111/gcb.16233 (2022).
Li, Y. et al. Increased nitrous oxide emissions from global lakes and reservoirs since the pre-industrial era. Nat. Commun. 15(1), 942. https://doi.org/10.1038/s41467-024-45061-0 (2024).
Yao, Y. et al. Increased global nitrous oxide emissions from streams and rivers in the Anthropocene. Nat. Clim. Change 10(2), 138–142. https://doi.org/10.1038/s41558-019-0665-8 (2019).
Li, S., Luo, J., Wu, D. & Jun, Xu. Y. Carbon and nutrients as indictors of daily fluctuations of pCO2 and CO2 flux in a river draining a rapidly urbanizing area. Ecol. Indic. https://doi.org/10.1016/j.ecolind.2019.105821 (2020).
Wang, G. et al. Intense methane ebullition from urban inland waters and its significant contribution to greenhouse gas emissions. Water Res. 189, 116654. https://doi.org/10.1016/j.watres.2020.116654 (2021).
Cao, J. X. et al. A critical review of the appearance of black-odorous waterbodies in China and treatment methods. J. Hazard. Mater. 385, 121511. https://doi.org/10.1016/j.jhazmat.2019.121511 (2020).
Polrot, A., Kirby, J. R., Birkett, J. W. & Sharples, G. P. Combining sediment management and bioremediation in muddy ports and harbours: A review. Environ. Pollut. 289, 117853. https://doi.org/10.1016/j.envpol.2021.117853 (2021).
Xu, Q., Zhou, Z. & Chai, X. Micro- and nano-bubbles enhanced the treatment of an urban black-odor river. Sustainability https://doi.org/10.3390/su152416695 (2023).
Yin, H., Wang, J., Zhang, R. & Tang, W. Performance of physical and chemical methods in the co-reduction of internal phosphorus and nitrogen loading from the sediment of a black odorous river. Sci. Total Environ. 663, 68–77. https://doi.org/10.1016/j.scitotenv.2019.01.326 (2019).
Suriasni, P. A., Faizal, F., Panatarani, C., Hermawan, W. & Joni, I. M. A review of bubble aeration in biofilter to reduce total ammonia nitrogen of recirculating aquaculture system. Water https://doi.org/10.3390/w15040808 (2023).
Liu, L. et al. Dredging technology and its effect on the treatment of polluted water. J. Environ. Eng. Technol. 10(1), 63–71. https://doi.org/10.12153/j.issn.1674-991X.20190045 (2020).
Liu, L. et al. Research progress on the effect of aeration on urban black-odor water ecosystem. Res. Environ. Sci. 33(4), 932–939. https://doi.org/10.13198/j.issn.1001-6929.2020.01.02 (2020).
He, Y., Yao, L., Li, W., Huang, M. & Zhang Y. Responses of bacterial community structure in malodorous river sediments to aeration by DGGE. J. East China Normal Univ. Nat. Sci. (2), 84–90. http://doi.org/1000-5641(2015)02-0084-07 (2015).
Pan, M., Wu, J., Heng, S., Zhen, S. & Zhao, J. Effects of the combination of aeration and biofilm technology on transformation of nitrogen in black-odor river. Water Sci. Technol. 74(3), 655–662. https://doi.org/10.2166/wst.2016.212 (2016).
Xu, X. & Cao, J.S. Study on combined process of river aeration and biological carriers for treatment of polluted river water. 24 (15), 36–39. http://doi.org/1000-4602(2008)15-0036-04 (2008).
Zhou, W. et al. Invasive water hyacinth (Eichhorniacrassipes) increases methane emissions from a subtropical lake in the Yangtze River in China. Diversity https://doi.org/10.3390/d14121036 (2022).
Shao, R. et al. Land use legacies and nitrogen fertilization affect methane emissions in the early years of rice field development. Nutr. Cycl. Agroecosyst. 107(3), 369–380. https://doi.org/10.1007/s10705-017-9838-x (2017).
Liu, L. X., Xu, M., Lin, M. & Zhang, X. Spatial variability of greenhouse gas effluxes and their controlling factors in the Poyang Lake in China. Pol. J. Environ. Stud. 22(3), 749–758 (2013).
Sun, W. et al. Effects of influent nitrogen composition on nitrogen removal and N2O emissions in surface flow constructed wetland with micro-polluted water. China Environ. Sci. 43(8), 4013–4023. https://doi.org/10.19674/j.cnki.issn1000-6923.2023.0134 (2023).
Fang, X. et al. Ebullitive CH4 flux and its mitigation potential by aeration in freshwater aquaculture: Measurements and global data synthesis. Agric. Ecosyst. Environ. https://doi.org/10.1016/j.agee.2022.108016 (2022).
Wu, J. et al. Effects of dredging wastewater input history and aquaculture type on greenhouse gas fluxes from mangrove sediments along the shorelines of the Jiulong River Estuary China. Environ. Pollut. 346, 123672. https://doi.org/10.1016/j.envpol.2024.123672 (2024).
Yang, P. et al. Contrasting effects of aeration on methane (CH4) and nitrous oxide (N2O) emissions from subtropical aquaculture ponds and implications for global warming mitigation. J. Hydrol. https://doi.org/10.1016/j.jhydrol.2022.128876 (2023).
Chen, G. et al. Increased nitrous oxide emissions from intertidal soil receiving wastewater from dredging shrimp pond sediments. Environ. Res. Lett. https://doi.org/10.1088/1748-9326/ab93fb (2020).
Jia, L., Jiang, B., Huang, F. & Hu, X. Nitrogen removal mechanism and microbial community changes of bioaugmentation subsurface wastewater infiltration system. Bioresour. Technol. 294, 122140. https://doi.org/10.1016/j.biortech.2019.122140 (2019).
Nijman, T. P. A. et al. Phosphorus control and dredging decrease methane emissions from shallow lakes. Sci. Total Environ. 847, 157584. https://doi.org/10.1016/j.scitotenv.2022.157584 (2022).
Chen, J. et al. Pollutants removal, greenhouse gases emission and functional genes in wastewater ecological soil infiltration systems: influences of influent surface organic loading and aeration mode. Water Sci. Technol. 83(7), 1619–1632. https://doi.org/10.2166/wst.2021.087 (2021).
Wan, X., Laureni, M., Jia, M. & Volcke, E. I. P. Impact of organics, aeration and flocs on N2O emissions during granular-based partial nitritation-anammox. Sci. Total Environ. 797, 149092. https://doi.org/10.1016/j.scitotenv.2021.149092 (2021).
Li, Y. et al. The role of freshwater eutrophication in greenhouse gas emissions: A review. Sci. Total Environ. 768, 144582. https://doi.org/10.1016/j.scitotenv.2020.144582 (2021).
Wang, X., Sun, T., Li, H., Li, Y. & Pan, J. Nitrogen removal enhanced by shunt distributing wastewater in a subsurface wastewater infiltration system. Ecol. Eng. 36(10), 1433–1438. https://doi.org/10.1016/j.ecoleng.2010.06.023 (2010).
Ji, B. et al. Roles of biochar media and oxygen supply strategies in treatment performance, greenhouse gas emissions, and bacterial community features of subsurface-flow constructed wetlands. Bioresour. Technol. 302, 122890. https://doi.org/10.1016/j.biortech.2020.122890 (2020).
Pang, J. et al. How do hydraulic load and intermittent aeration affect pollutants removal and greenhouse gases emission in wastewater ecological soil infiltration systems?. Ecol. Eng.. https://doi.org/10.1016/j.ecoleng.2020.105747 (2020).
Zeng, J. et al. Oxygen dynamics, organic matter degradation and main gas emissions during pig manure composting: Effect of intermittent aeration. Bioresour. Technol. 361, 127697. https://doi.org/10.1016/j.biortech.2022.127697 (2022).
Jacinthe, P. A., Filippelli, G. M., Tedesco, L. P. & Raftis, R. Carbon storage and greenhouse gases emission from a fluvial reservoir in an agricultural landscape. Catena 94, 53–63. https://doi.org/10.1016/j.catena.2011.03.012 (2012).
West, W. E., Creamer, K. P. & Jones, S. E. Productivity and depth regulate lake contributions to atmospheric methane. Limnol. Oceanogr. https://doi.org/10.1002/lno.10247 (2015).
Soued, C., del Giorgio, P. A. & Maranger, R. Nitrous oxide sinks and emissions in boreal aquatic networks in Québec. Nat. Geosci. 9(2), 116–120. https://doi.org/10.1038/ngeo2611 (2015).
Sun, Y. et al. Confirmation the optimal aeration parameters for nitrogen removal and nitrous oxide emission in wastewater ecological soil infiltration systems with brown earth. Water Sci. Technol. 80(1), 144–152. https://doi.org/10.2166/wst.2019.260 (2019).
Zhang, L. et al. How do aeration mode and influent carbon/nitrogen ratio affect pollutant removal, gas emission, functional genes and bacterial community in subsurface wastewater infiltration systems?. Water Sci. Technol. 88(11), 2793–2808. https://doi.org/10.2166/wst.2023.383 (2023).
Liu, F. F. et al. Intensified nitrogen transformation in intermittently aerated constructed wetlands: Removal pathways and microbial response mechanism. Sci. Total Environ. 650(Pt 2), 2880–2887. https://doi.org/10.1016/j.scitotenv.2018.10.037 (2019).
Dong, H., Qiang, Z., Li, T., Jin, H. & Chen, W. Effect of artificial aeration on the performance of vertical-flow constructed wetland treating heavily polluted river water. J. Environ. Sci. (China) 24(4), 596–601. https://doi.org/10.1016/s1001-0742(11)60804-8 (2012).
Sun, Y. et al. Organics removal, nitrogen removal and N2O emission in subsurface wastewater infiltration systems amended with/without biochar and sludge. Bioresour. Technol. 249, 57–61. https://doi.org/10.1016/j.biortech.2017.10.004 (2018).
Zheng, F. et al. Does influent surface organic loading and aeration mode affect nitrogen removal and N2O emission in subsurface wastewater infiltration systems?. Ecol. Eng. 123, 168–174. https://doi.org/10.1016/j.ecoleng.2018.09.015 (2018).
Jahangir, M. M. R. et al. Carbon and nitrogen dynamics and greenhouse gas emissions in constructed wetlands treating wastewater: A review. Hydrol. Earth Syst. Sci. 20(1), 109–123. https://doi.org/10.5194/hess-20-109-2016 (2016).
Acknowledgements
we thank the staff of Chinese Research Academy of Environmental Sciences, and State Key Laboratory of Environmental Criteria and Risk Assessment for their support and help.
Funding
This work was supported by the national natural science foundation of China (41907392).
Author information
Authors and Affiliations
Contributions
Conceptualization: L.L.X., Y.K., L.L.Z., Z.Y.P. Methodology: L.L.X., Y.H.R., J.Y.J. Investigation: Y.K., L.L.Z., L.W.W., Z.E.X., Z.Y.P., J.Y.J. Project administration: L.L.X. Supervision: Y.H.R., H.Y.w., J.Y.J. Writing—original draf: L.L.X., Y.K. Writing—review & editing: L.L.X., L.L.Z., Z.E.X.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, L., Yang, K., Li, L. et al. The aeration and dredging stimulate the reduction of pollution and carbon emissions in a sediment microcosm study. Sci Rep 14, 26172 (2024). https://doi.org/10.1038/s41598-024-75790-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-75790-7