Abstract
D-dimer is a biomarker of coagulation and fibrinolytic system activation in response to the hypercoagulable state of the body. The research aimed to analyze the value of D-dimer in the prognosis of patients with dilated cardiomyopathy (DCM). Patients admitted to our center for the first time with DCM were enrolled consecutively. The clinical characteristics variables were obtained from the electronic medical record system, and the prognostic information was obtained using telephone return visits and a review of repeated hospitalization records. Univariate and multivariate Cox regression was used to explore the association of D-dimer with all-cause mortality. Smooth curve fitting, threshold saturation effect analysis, and subgroup analysis were performed. Ultimately, 534 patients were included. After a follow-up of the enrolled patients, 485 patients obtained prognostic information, of which 159 died from all causes, and the main cause of death was heart failure (89/159), the sudden death accounted for about 17%. The independent positive association between D-dimer and all-cause mortality remained unchanged in both unadjusted and adjusted Cox regression models. In the fully adjusted model, each standard deviation increase in D-dimer was associated with a 14% increase in all-cause mortality (HR = 1.14; 95% CI: 1.02 ~ 1.27; P < 0.05). Curve fitting and threshold effect analysis showed an inflection point in the relationship between D-dimer and all-cause mortality (non-linear test: P = 0.03). When D-dimer was equal to 362ng/ml, HR = 1; and as the value increased, the risk of all-cause mortality increased by 34.7% for every 2-fold increase in D-dimer gradually (HR = 1.347; 95% CI: 1.069 ~ 1.697; P = 0.012). In subgroup analysis, D-dimer and BMI had a significant interaction on all-cause mortality, with a significantly increased risk of all-cause mortality in subjects with BMI ≥ 25 kg/m2 (HR = 1.99; 95% CI: 1.34 ~ 2.97; P < 0.01). The ROC curve showed that D-dimer was a good predictor of all-cause mortality, and the areas under the curve at 1-, 3-, and 5-year were 0.71, 0.64, and 0.59, respectively. In addition, D-dimer improved the predictive performance of the MAGGIC heart failure score in patients with DCM. D-dimer is not only independently associated with all-cause mortality in DCM patients, but also has good predictive value, suggesting that D-dimer may be an early and useful marker for improving the management of DCM patients.
Similar content being viewed by others
Introduction
Dilated cardiomyopathy (DCM) is characterized as left ventricular dilatation and systolic dysfunction in the absence of coronary artery disease or abnormal loading proportional to the degree of left ventricular damage1. As one of the critical causes of heart failure (HF), left ventricular systolic dysfunction is generally progressive in patients with DCM. Historical survival data from tertiary referral centers for adults with DCM indicate that 1-year mortality in DCM is 25–30%, and 5-year survival is approximately 50%2. The incidence of sudden death was as high as 12%, accounting for 25 ~ 30% of all deaths2. Given the poor prognosis, timely and accurate assessment of the disease and the development of individualized therapy strategies are imperative.
D-dimer is a plasmin-derived soluble cross-linked fibrin degradation product3. It is not only widely used in the diagnosis of venous thromboembolism4,5, but also has been studied in some diseases associated with activation of the coagulation and fibrinolytic systems, such as disseminated intravascular coagulation6, acute aortic dissection7, pulmonary embolism8, coronary atherosclerosis9, acute myocardial infarction10, stroke11, aortic aneurysm12, etc.
Some recent studies have shown us that the role of D-dimer may not be limited to these situations. In a retrospective cohort study of coronavirus disease 2019 (COVID-19) in China in 2020, d-dimer greater than 1 µg/mL was found to be associated with a fatal outcome in COVID-1913. In another research, it was also discovered that D-dimer levels on admission could predict in-hospital mortality in patients with COVID-1914. Researchers conclude that elevated D-dimer may be connected to the hypercoagulable state in high-risk patients, which includes excessive thrombin generation due to endothelial cell dysfunction15, hypoxia-induced increase in blood viscosity, and activation of signaling pathways16, coagulation disorders caused by sepsis17, and enhanced risk factors for thrombosis such as advanced age, underlying diseases, prolonged bed rest, and invasive treatments. We know that in patients with DCM, ventricular dilatation and remodeling, cardiomyocyte fibrosis, progressive deterioration of cardiac function, blood stasis, combined pulmonary infection, and hemodynamic abnormalities caused by arrhythmias are also easily lead to hypercoagulability of the body. Therefore, D-dimer seems to have a non-negligible research value in these patients. Although there have been previous studies revealing the role of D-dimer levels in predicting adverse outcomes in hospitalized patients with acute, decompensated heart failure18, there are very few cohort studies in DCM. Accordingly, this research intensely analyzed the prognostic value of D-dimer in DCM in a retrospective cohort.
Materials and methods
Patients
This retrospective study included DCM patients at first admission between October 2012 and May 2020 from the First Affiliated Hospital of Guangxi Medical University. A total of 860 patients met the diagnostic criteria of DCM according to the scientific statement established by the American Heart Association19. To ensure the accuracy of this definition, we performed coronary angiography on all patients suspected of having coronary artery disease to exclude those with related conditions. This approach ensures that our study population is distinct from patients with heart failure with reduced ejection fraction (HFrEF) due to ischemic heart disease. Exclusion criteria: (1) Acute pulmonary embolism; (2) Acute stroke; (3) Acute myocardial infarction; (4) Peripheral arterial and venous embolism; (5) Disseminated intravascular coagulation; (6) Major surgery, severe trauma, or thrombolytic therapy within three months; (7) Under the age of 16; (8) Pregnancy; (9) Malignant tumor; (10) Previous cardiac pacemaker or left ventricular assist device implantation; (11) Previous heart transplantation; (12) Previous valve surgery; (13) Inpatient echocardiography data is missing; (14) D-dimer data or critical baseline data are missing; (15) Individuals who received anticoagulation or antiplatelet therapy prior to admission. Finally, 534 patients enrolled in the study. The flow chart of the study population enrollment is shown in Supplementary Fig. 1.
Data collection
We collected and analyzed the following variables of the subjects: Population Characteristics, Medical history and comorbidities, Physical examination, Blood biochemistry, Electrocardiogram, Echocardiogram, Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) score scale20, Drug use, Discharge outcome, Return visit information (the details shown in Supplementary Table 1). All patients underwent the examination within the first two days of hospitalization. For repeated examinations, only the first results at the time of hospitalization were taken. Blood samples were collected within 24 h of admission, and all blood samples were sent to the inspection center of the First Affiliated Hospital of Guangxi Medical University for inspection in time. D-dimer was measured by immunoturbidimetric quantification (BECKMAN ACL-TOP, USA) with normal reference values of 0 ~ 450 µg/L. Prognostic information was collected on all selected patients through follow-up phone calls and by accessing their electronic medical records. All data were extracted from electronic medical records using a standardized data collection form, and all co-authors examined the data separately to ensure accuracy.
Relevant definitions
Length of medical history: symptoms associated with DCM and the time of their appearance. Pulmonary inflammation: acute and chronic inflammation of the lungs, including bronchitis, pneumonia, and chronic obstructive pulmonary disease. The estimated glomerular filtration rate (eGFR) was computed using the CKD-EPI equation21. Pulmonary hypertension was defined as the pulmonary artery pressure estimated at ≥ 30mmHg based on the tricuspid regurgitation pressure difference. Sudden death: accidental death within 1 h of cardiac symptoms without progressive cardiac deterioration; death in sleep; or death within 24 h of the last sighting alive22. Heart failure death: predictable death due to progressive deterioration of heart function.
Statistical analysis
(1) Grouped according to D-dimer tertiles, divided into T1, T2, and T3. The normality test was performed using the Shapiro-Wilk test. Normally distributed continuous variables were expressed as mean ± standard deviation (xˉ± s), and one-way analysis of variance was used for comparison between groups; skewed continuous variables were expressed as median (interquartile range) [Median (IQR)], and Kruskal-Wallis rank sum test (H test) was used for comparison between groups. Categorical variables were expressed as the number of cases and the corresponding percentages [n (%)], and the Pearson chi-square test or Fisher’s exact probability method was used for comparison between groups. (2) Histograms were drawn to compare plasma D-dimer levels in subjects with different follow-up outcomes. (3) The Kaplan-Meier survival curve was drawn, and the log rank test was used to compare the differences in survival rates between groups. (4) Independent correlations between D-dimer and all-cause mortality were analyzed using univariate and multivariate Cox regression models. The risk was quantified by the hazard ratios (HR) and its 95% confidence interval (CI). Covariate screening: potential covariates (variables with P < 0.1 in univariate analysis) were entered one by one in the basic model of the multivariate Cox regression model or removed one by one in the full model to identify potential covariates that changed the initial regression coefficient by more than 10%; appropriate variables were selected for adjustment in the final model by combining clinical analyses23. In addition, variables with high variance inflation factors in the multiple colinearity analysis were excluded. Extended Cox regression model: since D-dimer had a significantly skewed distribution, it was standardized transformed, log-transformed, and grouped by tertiles, respectively; the extended model approach was used to adjust for potential confounders to evaluate the stability of the relationship between D-dimer and all-cause mortality. (5) Smoothed curve fitting and threshold saturation effect analysis: A generalized additive model was used to assess the nonlinear relationship between serum D-dimer levels and all-cause mortality; (24) corresponding adjustments were made in the multivariate-adjusted model25. Inflection points were set according to the smoothing curve, and segmented regression models were constructed based on the inflection points. The D-dimer was log-transformed to give an approximately normal distribution. In addition, symmetric cropping at the 0.5% level (excluding observations that took values in the upper 0.5% and lower 0.5% intervals) was performed to minimize the effect of outliers23. (6) Subgroup analysis: we used some relevant effect covariates for stratification and drew forest plots, and P values for interactions were calculated by likelihood ratio tests. (7) The accuracy of D-dimer in predicting all-cause mortality at different times was assessed by the area under the curve (AUC) of the time receiver operating characteristic (ROC) curve. (8) The data from this study was used to validate the accuracy of the MAGGIC Heart Failure Rating Scale in the Chinese DCM population. The risk score of each patient was calculated by a network calculator (http://www.heartfailurerisk.org), and the performance of the scoring scale was verified by the ROC curve and calibration curve. D-dimer was added to the scale, and its performance was evaluated by plotting the ROC curve, calibration curve, and calculating the Net Reclassification Improvement (NRI) Index and Integrated Discrimination Improvement (IDI) Index26,27.
All statistical analyses were performed using R software (http://www.R-project.org); P < 0.05 was considered statistically significant.
Results
Follow-up outcomes
We followed up with the included patients, and the information was available after 30.6 ± 24.7 months, there were 49 patients lost to follow-up, and ultimately 485 patients were given prognostic information. Among 159 all-cause deaths, 89 died of heart failure (4 heart transplant patients included in this category), 27 died of sudden death, and 43 died of other causes (including multiple organ failure, sepsis, severe pneumonia, cerebral hemorrhage, cerebral infarction, and unidentified causes).
Baseline characteristics and clinical outcomes in different groups
According to the D-dimer tertiles from small to large, the patients were divided into three groups: T1, T2, and T3. Compared with T1, patients in T3 had a lower mean age, a higher proportion in the New York Heart Association (NYHA) class IV, and more critically ill cases. In terms of complications, the proportion of pulmonary hypertension and pulmonary inflammation in T3 was higher than that in T1 and T2 (P < 0.01). During the admission physical examination, the proportion of patients with edema of both lower extremities and the average heart rate progressively increased from T1 to T3, while the systolic pressure and pulse pressure gradually decreased (P < 0.01). In terms of biochemical tests, the levels of white blood cell count (WBC), Neutrophils (NE), Neutrophil to Lymphocyte Ratio (NLR), Red blood cell distribution width (RDW-SD), Creatine kinase (CK), Creatine kinase isoenzyme (CK-MB), Lactic dehydrogenase (LDH), Lactic dehydrogenase-1 (LD1), Alpha-hydroxybutyrate dehydrogenase (α-HBD), Homocysteine (Hcy), Fibrinogen (FIB), Prothrombin time (PT), International normalized ratio (INR), Thrombin time (TT), Aspartate aminotransferase (AST), Alanine aminotransferase (ALT), UREA, Creatinine (SCr), Cystatin C (CysC), Uric acid (UA) and N terminal pro B type natriuretic peptide (NT-proBNP) in T3 were markedly higher than T1; while the levels of Platelet (PLT), Lymphocyte (LY), Total cholesterol (TC), Triglycerides (TG), High-density lipoprotein (HDL), Serum sodium (Na+), Serum chlorine (Cl−), Albumin (ALB), Albumin to Globulin Ratio (A/G), and eGFR were lower (P < 0.05). On echocardiography, Left atrium anteroposterior dimension (LAD), Left ventricular fractional shortening (LVFS), and Left ventricular ejection fraction (LVEF) were lower in T3, and LVFS and LVEF gradually decreased from T1 to T3 (P < 0.05). In terms of drug treatment, the use of spironolactone and β-blockers did not differ among the three groups, and the use ratio of angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker (ACEI/ARB) gradually decreased from T1 to T3 (P < 0.05). In terms of hospitalization outcomes, the proportion of dead and unhealed patients in T3 was the highest. In the follow-up outcomes, the percentage of all-cause mortality progressively increased from T1 to T3 (20.9%, 32.7%, and 45.2%, respectively); among them, the proportion of heart failure deaths exhibited a synchronous upward trend (11.2%, 18%, 20.8%). (P < 0.01). (Supplementary Table 1)
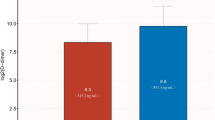
D-dimer levels for various follow-up outcomes
As can be seen from the histogram, patients with different follow-up outcomes had different levels of plasma D-dimer. Although specific causes of death varied, patients who died had higher average plasma concentrations of D-dimer than patients who survived. Among them, the average plasma D-dimer concentration in the patients who died of heart failure was the highest, and the difference between the two groups was statistically significant compared with the patients who survived (P < 0.05) (Supplementary Fig. 2).
Kaplan-Meier survival curve of T1, T2, and T3
Through the Kaplan-Meier survival curve, it was further found that among the tertiles, T3 had the highest all-cause mortality, followed by T2, and T1 had the lowest (P < 0.001 by log-rank test) (Fig. 1).
Kaplan-Meier survival curve of T1, T2, and T3. Notes: Lines represent different groups of populations, and shading represents 95% confidence intervals.
Univariate and multivariate Cox regression analysis
Unadjusted association between baseline variables and all-cause mortality. Univariate Cox regression analysis indicated that complicated with intracardiac thrombus, longer medical history, heart failure classification (NYHA), pulmonary inflammation, edema of both lower extremities, WBC, NE, NLR, RDW-SD, CK, LD1, α-HBD, Hcy, D-dimer, PT, INR, UREA, SCr, CysC, NT-proBNP, UA, LAD, LVDd, LVDS was associated with increased risk of all-cause mortality (P < 0.05); while systolic pressure, diastolic pressure, pulse pressure, RBC, PLT, TC, TG, HDL, LDL, Na+, Cl−, ALB, A/G, eGFR, LVFS, LVEF, spironolactone, beta-blocker, ACEI/ARB was associated with a reduced risk of all-cause mortality (P < 0.05) (Supplementary Table 2). Variables with P < 0.1 in univariate Cox regression analysis were included in the screening range, and the selected potential covariates were: medical history, heart failure class (NYHA), smoking history, pulmonary inflammation, pulse pressure difference, WBC, RBC, PLT, NLR, RDW-SD, CK, TG, HDL, Hcy, Na+, Cl−, INR, ALB, UREA, NT-proBNP, UA, LVEF, Spironolactone, ACEI/ARB. Combined with clinical analysis, the final selected covariate combination was: age, gender, smoking history, medical history, heart failure class (NYHA), pulmonary hypertension, atrial fibrillation, diabetes, pulmonary inflammation, spironolactone, beta-blockers, ACEI/ARB and pulse pressure, WBC, RDW-SD, CK, TG, Hcy, Na+, ALB, UREA, NT-proBNP (Supplementary Table 3).
Unadjusted and adjusted associations between D-dimer and all-cause mortality. In the extended Cox regression model (Table 1), we constructed five distinct models to evaluate the pre-adjusted and post-adjusted associations between D-dimer and all-cause mortality. In the unadjusted model, the risk of all-cause mortality increased progressively with increasing D-dimer plasma concentrations. Each standard deviation increase in D-dimer was associated with a 21% increased risk of all-cause mortality (HR = 1.21; 95% CI: 1.1–1.34; P < 0.01). Each 2-fold increase in D-dimer was associated with a 27% increased risk (HR = 1.27; 95% CI: 1.16–1.39; P < 0.01). In the comparison of tertile groups, the mortality risk of T3 was considerably higher than that of T1 (HR = 2.46; 95% CI: 1.64–3.71; P < 0.01), and the mortality risk showed a gradual rising trend from T1 to T3. The trend test indicated a mean increase in risk of 57% between adjacent high and low tiers (HR = 1.57; 95% CI: 1.28–1.92; P < 0.01). Adjustments for population characteristics, comorbidities, and medication use in Model b, Model c, and Model d did not significantly weaken the association between D-dimer and all-cause mortality in the extended model. Based on model d, model e further adjusted pulse pressure difference, WBC, RDW-SD, CK, TG, Hcy, Na+, ALB, UREA, NT-proBNP (these variables were grouped by tertile before inclusion), and ultimately, in the sufficiently adjusted model, each standard deviation increase in D-dimer was independently associated with a 14% increase in all-cause mortality (HR = 1.14; 95% CI: 1.02–1.27; P < 0.05); a 2-fold increase in D-dimer was independently associated with a 20% increase in all-cause mortality (HR = 1.2; 95% CI: 1.06–1.36; P < 0.05); and in the tertile groups, the risk of T3 was higher than that of T1 Significant increase, HR = 1.7 (95% CI: 1.04–2.76; P < 0.05), and the trend test displayed a statistically significant difference (HR = 1.29; 95% CI: 1.02–1.64; P < 0.05).
Smoothing curve fitting and threshold saturation effect analysis
The curve fitting after adjusting for covariates indicates that there may be an inflection point in the relationship between D-dimer and all-cause mortality. When log2(D-dimer) is equal to 8.5, HR = 1; and as the value rises, the risk of death progressively increases (Fig. 2). According to the figure, we set an inflection point: log28.5, and constructed a two-stage regression model. When log2(D-dimer) ≥ 8.5 (i.e. D-dimer ≥ 362ng/ml), every 2-fold increase in D-dimer was associated with a 34.7% increased risk of all-cause mortality (HR = 1.347; 95% CI: 1.069–1.697; P = 0.012); and when log2(D-dimer) < 8.5, every 2-fold increase in D-dimer was associated with a 5.8% increased risk of all-cause mortality (HR = 1.058; 95% CI: 0.678–1.651; P = 0.803). The non-linear test demonstrated a statistically significant difference between the two regression models (P = 0.03), indicating an inflection point in the association between D-dimer and all-cause mortality (Table 2).
Smooth curve fitting of the relationship between log2(D-dimer) and all-cause mortality. Notes: The blue solid and dashed lines represent estimates and their corresponding 95% confidence intervals. Only 95% of the data is displayed. Adjusted for all covariates of model e in the expanded model.
Subgroup analysis and forest plot
In the subgroup analysis, we further analyzed the stability of the association between D-dimer and all-cause mortality in various subgroups and produced a forest plot (Fig. 3). After full model adjustment, the results indicated that in subgroups of ethnicity (Han, minority), age (< 60, ≥ 60), eGFR (< 60, ≥ 60), and LVEF (< 35%, ≥ 35%), Serum D-dimer levels (every 2-fold change) were all positively associated with all-cause mortality risk and were relatively stable. In the separate subgroups by gender, D-dimer levels were negatively associated with the risk of all-cause mortality in female patients, unlike the positive association in the male subgroup. However, this was not statistically significant (P = 0.379). Notably, there was a significant interaction between D-dimer and BMI on all-cause mortality (The adjusted P-value for interaction was 0.027); in subjects with BMI < 25 kg/m2, each 2-fold increase in serum D-dimer was associated with an 8% increased risk of all-cause mortality (HR = 1.08; 95% CI: 0.94–1.25; p = 0.27); while this risk was more pronounced in subjects with a BMI ≥ 25 kg/m2 (HR = 1.99; 95% CI: 1.34–2.97; P < 0.01).
Association of D-dimer with all-cause mortality in different subgroups. Notes: Each stratification adjusted for all covariates of model e in the expanded model except the stratification factor itself. HR, hazard ratio; CI, confidence interval.
Accuracy of D-dimer in predicting all-cause mortality
The time ROC curve indicates that although the accuracy of D-dimer’s forecast of all-cause mortality slowly declines over time, it can still retain a high level. The AUCs at 6-month, 1-year, 2-year, 3-year, and 5-year were: 0.74, 0.71, 0.67, 0.64, and 0.59, respectively (Fig. 4).
ROC analysis of D-dimer in predicting all-cause mortality. Notes: A represents the ROC curve at each time point, and B represents the changing trend of the area under the curve with time (the unit of time in months).
Optimization of MAGGIC heart failure rating scale
The ROC curve showed that the predictive accuracy of the MAGGIC Heart Failure Rating Scale for all-cause mortality was 0.68, 0.69, and 0.68 at 1-year, 3-year, and 5-year, respectively, and the overall performance was comparatively reliable (Supplementary Table 4). After adding log2(D-dimer) to the model, the discrimination and calibration of the model are improved, and the areas under the ROC curve are 0.73, 0.71, and 0.69, respectively; the calibration curve displayed excellent calibration (Fig. 5); both NRI and IDI were greater than zero (Fig. 6), indicating that D-dimer enhanced the predictive performance of the MAGGIC Heart Failure Score in patients with DCM.
Comparison of ROC curve and calibration curve before and after adding D-dimer to the MAGGIC score scale. Notes: Model 1 represents the MAGGIC score scale, and Model 2 represents the “MAGGIC score scale + D-dimer”. (A, B, and C) are the ROC curves of the two models corresponding to 1-year, 3-year, and 5-year, respectively; (D, E, and F) are the calibration curves of the two models corresponding to 1 year, 3-year, and 5-year, respectively.
Comparison of IDI and NRI before and after adding D-dimer to MAGGIC score scale. Notes: Standard Model is the MAGGIC score scale model, New Model is the “MAGGIC score scale + D-dimer” model. (A, B, and C) are the IDI corresponding to 1-year, 3-year, and 5-year, respectively; (D, E, and F) are the NRIs corresponding to 1-year, 3-year, and 5-year, respectively.
Discussion
As a marker of coagulation and fibrinolysis activation3,2829, D-dimer is a reaction to the body’s hypercoagulable state, and it is also an indicator that is easy to obtain clinically. It provides a rapid assessment of thrombotic activity. It plays an essential role in the diagnosis of venous thromboembolism30. In cardiovascular disease, previous studies have uncovered that D-dimer is not only associated with all-cause mortality after hospitalization in patients with acute myocardial infarction31, but it is also independently associated with poor long-term prognosis in patients with end-stage heart failure due to idiopathic DCM32. On this basis, the present study conducted a more comprehensive and detailed analysis and found that higher plasma D-dimer levels were independently associated with an increased risk of all-cause mortality after adjusting for relevant covariates in a multivariate regression analysis.
The prognosis of DCM is a matter of great concern. Similar to the conclusions of previous studies2, we observed that in the follow-up patients, the proportion of all-cause death was 32.8%, heart failure death accounted for 56.0%, and sudden death accounted for 17.0%. In different follow-up outcomes, the plasma D-dimer average level of the dead patients was higher than that of the surviving patients. Among them, the average level of D-dimer in the patients who died of heart failure was the highest. Survival curves showed higher D-dimer levels higher mortality. Our research also discovered that higher D-dimer levels on admission were associated with poorer cardiac function, liver and kidney function, nutritional status, higher levels of inflammation, more severe myocardial damage, abnormal coagulation function, and adverse outcomes of hospitalization and follow-up. The independent positive association between D-dimer and all-cause mortality remained consistent in both unadjusted and adjusted models; which indicates that the hypercoagulable state of the body is affected by multiple factors, and the association between D-dimer and all-cause mortality in DCM is independent of other factors. It also further demonstrated that the body’s hypercoagulable state is closely related to poor cardiovascular prognosis, which is consistent with the previous study32. The difference is that our research is not limited to end-stage heart failure DCM patients. In addition, we also analyzed the nonlinear relationship between D-dimer and all-cause mortality, and we discovered that there was a threshold effect between the two. Below the threshold, the risk of death did not increase or decrease markedly with the increase of D-dimer; when D-dimer ≥ 362ng/ml, the risk of all-cause mortality increased by 34.7% for every 2-fold increase in D-dimer.
We compared the relationship between D-dimer and all-cause mortality in different subgroups and found that the relationship between the two was stable in different ethnic groups, age, cardiac function, and renal function. Nonetheless, in obese (BMI ≥ 25 kg/m2) subjects, this risk was enhanced, and D-dimer had a significant interaction with BMI on all-cause mortality. We know that D-dimer comes from the degradation of fibrin, and its production requires the continuous activity of three enzymes: thrombin, activated factor XIII (factor XIIIa), and plasmin33. Previous studies have found elevated levels of fibrinogen34, thrombin, and thrombin-antithrombin complex35 in obese individuals, and obesity is a key factor in thrombin generation36. In contrast, weight loss in morbidly obese patients significantly diminished the thrombin-generating potential37. Therefore, enhanced fibrinogen, thrombin, and other factors in obese patients may lead to higher levels of D-dimer. In addition, multiple previous studies have demonstrated a strong association between obesity and cardiovascular mortality38,39,40, and the increased risk of cardiovascular disease may be partly due to the prethrombotic state inherent in patients with abdominal obesity41. It is worth noting that in the female subgroup D-dimer was a protective factor, but it was not statistically significant; the possible reason was considered to be the protective effect of female hormones on cardiovascular existence. Previous studies have demonstrated that female patients with DCM have better cardiovascular outcomes than males, which is an important independent risk factor42,43. Of course, the chance results caused by the small sample size of female patients cannot be entirely ruled out. Finally, we also found that D-dimer was an excellent prognostic marker for DCM, especially in predicting early mortality. Additionally, D-dimer enhanced the predictive performance of the MAGGIC heart failure score.
In summary, in this study, we analyzed the predictive value of D-dimer in all-cause mortality in DCM. D-dimer is an objective indicator of coagulation and fibrinolysis, and its increase signifies that the body is in a hypercoagulable state. Patients with DCM are usually in a hypercoagulable state due to the presence of heart failure, myocardial damage, susceptibility to atrial fibrillation, and pulmonary infection, which has a tremendous impact on disease prognosis. This state can be well reflected by D-dimer, which is of great significance for the risk stratification of DCM. It is worth noting that the association of D-dimer with all-cause mortality has an interaction in different subgroups of BMI, which also reminds us to pay special attention to the risk assessment of obese patients with DCM.
Since this is an observational study and is prone to various confounding factors, strict statistical adjustments were used to minimize such effects. Nonetheless, the research still has certain limitations. First, our research may suffer from selection bias as it is a single-center, retrospective study, and despite our attempts to include all eligible patients, some patients were excluded from the study due to a lack of D-dimer levels at admission. As a result, the included sample size was small, and the number of end-point events observed after stratification was tiny, which limited the statistical power of this exploratory study to a certain extent. Nonetheless, after multi-level, multi-model analysis, we discovered that the association between D-dimer and end-point events was still robust; accordingly, we believe that the conclusions of this research are reliable. Second, since our study is a single-center study, the results may not be directly applicable to other races, so multicenter data is still required for validation. Third, this study cannot demonstrate causality between the hypercoagulable state and mortality. The high D-dimer concentration may merely be a surrogate marker for poor cardiac function and severe tissue damage. Fourth, Left atrial (LA) disease is known to be a strong predictor of outcomes in patients with DCM. Specifically, a reduction in the left atrial volume index (LAVI) has been associated with a lower risk of death, heart transplantation, or hospitalization for heart failure44. Unfortunately, due to the retrospective nature of our study and data constraints, we did not collect LA volume measurements. In future studies, we will consider incorporating LA volume data to provide a more comprehensive analysis. Fifth, the absence of myocardial biopsy and cardiac MRI limits our ability to fully exclude secondary cardiomyopathies, such as cardiac sarcoidosis or the dilated phase of hypertrophic cardiomyopathy (HCM). Sixth, D-dimer was only measured at admission, and it is unclear whether there were changes in D-dimer levels after treatment or if any changes were associated with prognosis. Lastly, although we have adjusted for various probable confounders, there may still be other under-accounted and unmeasured confounders, so large-scale prospective studies are still warranted.
Conclusions
This retrospective cohort study in a DCM population indicated that D-dimer, a marker of activation of the body’s coagulation and fibrinolytic systems, was independently associated with all-cause mortality in DCM. It is a valuable biomarker for DCM and is anticipated to be a potential tool to improve the risk stratification of cardiovascular disease. In addition, the prevention and management of the body’s hypercoagulable state are particularly critical for patients with DCM.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ALB:
-
Albumin
- ALT:
-
Alanine aminotransferase
- α-HBD:
-
Alpha-hydroxybutyrate dehydrogenase
- AST:
-
Aspartate aminotransferase
- AUC:
-
Area under the curve
- BMI:
-
Body mass index
- CK-MB:
-
Creatine kinase isoenzyme
- DCM:
-
Dilated cardiomyopathy
- eGFR:
-
Estimated glomerular filtration rate
- HB:
-
Hemoglobin
- HCT:
-
Hematocrit
- INR:
-
International normalized ratio
- LD1:
-
Lactate dehydrogenase isoenzyme
- LDH:
-
Lactate dehydrogenase
- LVEF:
-
Left ventricular ejection fraction
- LVFS:
-
Left ventricular short-axis shortening rate
- LVT:
-
Left ventricular thrombus
- NT-proBNP:
-
N terminal pro B type natriuretic peptide
- PT:
-
Prothrombin time
- RDW-SD:
-
Red blood cell distribution width
- ROC:
-
Receiver operating characteristics
- CL− :
-
Serum chloride
- NA+ :
-
Serum sodium
- UA:
-
Uric acid
- WBC:
-
White blood cell count
References
Elliott, P. Cardiomyopathy. Diagnosis and management of dilated cardiomyopathy. Heart. 84 (1), 106–112 (2000).
Dec, G. W. & Fuster, V. Idiopathic dilated cardiomyopathy. N Engl. J. Med. 331 (23), 1564–1575 (1994).
Adam, S. S., Key, N. S. & Greenberg, C. S. D-dimer antigen: current concepts and future prospects. Blood. 113 (13), 2878–2887 (2009).
Wells, P. S. et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl. J. Med. 349 (13), 1227–1235 (2003).
de Bastos, M., de Bastos, M. R., Bogutchi, T., Carneiro-Proietti, A. B. & Rezende, S. M. Duration of symptoms and D-dimer testing in the ruling-out of venous thromboembolism. J. Thromb. Haemost. 4 (9), 2079–2080 (2006).
Carr, J. M., McKinney, M. & McDonagh, J. Diagnosis of disseminated intravascular coagulation. Role of D-dimer. Am. J. Clin. Pathol. 91 (3), 280–287 (1989).
Sodeck, G. et al. D-dimer in ruling out acute aortic dissection: a systematic review and prospective cohort study. Eur. Heart J. 28 (24), 3067–3075 (2007).
Le Gal, G. et al. Prediction of pulmonary embolism in the emergency department: the revised Geneva score. Ann. Intern. Med. 144 (3), 165–171 (2006).
Danesh, J. et al. Fibrin D-dimer and coronary heart disease: prospective study and meta-analysis. Circulation. 103 (19), 2323–2327 (2001).
Lippi, G., Filippozzi, L., Montagnana, M., Salvagno, G. L. & Guidi, G. C. Diagnostic value of D-dimer measurement in patients referred to the emergency department with suspected myocardial ischemia. J. Thromb. Thrombolysis. 25 (3), 247–250 (2008).
Di Castelnuovo, A. et al. Elevated levels of D-dimers increase the risk of ischaemic and haemorrhagic stroke. Findings from the EPICOR Study. Thromb. Haemost. 112 (5), 941–946 (2014).
Golledge, J., Muller, R., Clancy, P., McCann, M. & Norman, P. E. Evaluation of the diagnostic and prognostic value of plasma D-dimer for abdominal aortic aneurysm. Eur. Heart J. 32 (3), 354–364 (2011).
Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 395 (10229), 1054–1062 (2020).
Zhang, L. et al. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J. Thromb. Haemost. 18 (6), 1324–1329 (2020).
Levi, M. & van der Poll, T. Coagulation and sepsis. Thromb. Res. 149, 38–44 (2017).
Gupta, N., Zhao, Y. Y. & Evans, C. E. The stimulation of thrombosis by hypoxia. Thromb. Res. 181, 77–83 (2019).
Iba, T. et al. Diagnosis and management of sepsis-induced coagulopathy and disseminated intravascular coagulation. J. Thromb. Haemost. 17 (11), 1989–1994 (2019).
Minami, Y. et al. Elevated D-dimer levels predict an adverse outcome in hospitalized patients with acute decompensated heart failure. Int. J. Cardiol. 204, 42–44 (2016).
Correction to. Current diagnostic and treatment strategies for specific dilated cardiomyopathies: A Scientific Statement from the American Heart Association. Circulation. 134 (23), e652 (2016).
Pocock, S. J. et al. Predicting survival in heart failure: a risk score based on 39 372 patients from 30 studies. Eur. Heart J. 34 (19), 1404–1413 (2013).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150 (9), 604–612 (2009).
Hicks, K. A. et al. 2014 ACC/AHA Key Data Elements and Definitions for Cardiovascular Endpoint Events in Clinical Trials: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (Writing Committee to Develop Cardiovascular Endpoints Data Standards). Circulation ;132(4):302–361. (2015).
Li, Z. Y. et al. Dose-response association between Adiposity and Liver Cancer incidence: a prospective cohort study among non-smoking and non-alcohol-drinking Chinese women. Cancer Epidemiol. Biomarkers Prev. 30 (6), 1200–1207 (2021).
Liao, S. et al. Association between perfluoroalkyl acids and the prevalence of hypertension among US adults. Ecotoxicol. Environ. Saf. 196, 110589 (2020).
Su, N. J. et al. Association between baseline LH/FSH and live-birth rate after fresh-embryo transfer in polycystic ovary syndrome women. Sci. Rep. 11 (1), 20490 (2021).
Pencina, M. J., D’Agostino, R. B., Sr., D’Agostino, R. B. Jr. & Vasan, R. S. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat. Med. 27 (2), 157–172 (2008). discussion 207 – 12.
Alba, A. C. et al. Discrimination and calibration of clinical prediction models: users’ guides to the Medical Literature. JAMA. 318 (14), 1377–1384 (2017).
Linkins, L. A. & Takach Lapner, S. Review of D-dimer testing: good, bad, and Ugly. Int. J. Lab. Hematol. 39 (Suppl 1), 98–103 (2017).
Favresse, J. et al. D-dimer: Preanalytical, analytical, postanalytical variables, and clinical applications. Crit. Rev. Clin. Lab. Sci. 55 (8), 548–577 (2018).
Wells, P. S. Integrated strategies for the diagnosis of venous thromboembolism. J. Thromb. Haemost. 5 (Suppl 1), 41–50 (2007).
Zhang, X. et al. D-dimer and the incidence of heart failure and mortality after acute myocardial infarction. Heart. 107 (3), 237–244 (2021).
Huang, B. et al. D-dimer level and long-term outcome in patients with end-stage heart failure secondary to idiopathic dilated cardiomyopathy. J. Geriatr. Cardiol. 16 (8), 621–629 (2019).
Weitz, J. I., Fredenburgh, J. C. & Eikelboom, J. W. A test in Context: D-Dimer. J. Am. Coll. Cardiol. 70 (19), 2411–2420 (2017).
De Pergola, G. & Pannacciulli, N. Coagulation and fibrinolysis abnormalities in obesity. J. Endocrinol. Invest. 25 (10), 899–904 (2002).
Siklar, Z. et al. Evaluation of hypercoagulability in obese children with thrombin generation test and microparticle release: effect of metabolic parameters. Clin. Appl. Thromb. Hemost. 17 (6), 585–589 (2011).
van Paridon, P. C. S. et al. Thrombin generation in cardiovascular disease and mortality - results from the Gutenberg Health Study. Haematologica. 105 (9), 2327–2334 (2020).
Ay, L. et al. Thrombin generation in morbid obesity: significant reduction after weight loss. J. Thromb. Haemost. 8 (4), 759–765 (2010).
Coutinho, T. et al. Central obesity and survival in subjects with coronary artery disease: a systematic review of the literature and collaborative analysis with individual subject data. J. Am. Coll. Cardiol. 57 (19), 1877–1886 (2011).
Yusuf, S. et al. Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a case-control study. Lancet. 366 (9497), 1640–1649 (2005).
Li, C., Sun, X. N., Zhao, S. & Scherer, P. E. Crosstalk between adipose tissue and the heart: an update. J. Transl Int. Med. 10 (3), 219–226 (2022).
Faber, D. R., de Groot, P. G. & Visseren, F. L. Role of adipose tissue in haemostasis, coagulation and fibrinolysis. Obes. Rev. 10 (5), 554–563 (2009).
Cannata, A. et al. Sex differences in the long-term prognosis of dilated cardiomyopathy. Can. J. Cardiol. 36 (1), 37–44 (2020).
Halliday, B. P. et al. Sex- and age-based differences in the natural history and outcome of dilated cardiomyopathy. Eur. J. Heart Fail. 20 (10), 1392–1400 (2018).
Nuzzi, V. et al. Left Atrial Reverse Remodeling in Dilated Cardiomyopathy. J. Am. Soc. Echocardiogr. 36 (2), 154–162 (2023).
Funding
This work was supported by Innovative Research Team Project of Guangxi Natural Science Foundation (Grant No. 2018GXNSFGA281006) and Guangxi Natural Science Foundation Project (Grant No. 2020GXNSFDA297014).
Author information
Authors and Affiliations
Contributions
Authors’ contributions(I) Conception and design: Y.H., C.G., Q.S. (II) Administrative support: C.G. (III) Provision of study materials or patients: C.G., Y.H., Q.S., L.-H.Y. (IV) Collection and assembly of data: Y.H., H.C., Y.-X.L. (V) Data analysis and interpretation: Y.H., H.-B.S., J.-H.L., L.-H.Y. (VI) Manuscript writing: all authors. (VII) Final approval of manuscript: all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was conducted following the Declaration of Helsinki (as revised in 2013), and approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University. Written informed consent was obtained from the patient himself or his close relatives.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, Y., Yang, LH., Li, YX. et al. The value of D-dimer in the prognosis of dilated cardiomyopathy: a retrospective cohort study. Sci Rep 14, 26806 (2024). https://doi.org/10.1038/s41598-024-76716-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-76716-z