Abstract
Nicotinamide adenine dinucleotide phosphate-diaphorase (NADPH-d) was used to detect neurodegenerations in aged monkeys. Our previous studies have shown that aging-related NADPH-d positive bodies (ANBs) and megaloneurites appeared in the lumbosacral spinal cord of aged rats and dogs, respectively. To determine the occurrence of megaloneurites and ANBs in non-human primates, we used NADPH-d histochemistry to perform an advanced study of aging-related alterations in aged male monkeys. We identified two distinct abnormal NADPH-d positive alterations, which were expressed as ANBs and megaloneurites, mainly distributed in the superficial dorsal horn, dorsal gray commissure, lateral collateral pathway (LCP) and sacral parasympathetic nucleus of the sacral spinal cord in aged monkeys. Meanwhile, large diameter punctate NADPH-d abnormalities occurred and scattered in the lateral white matter of the LCP and dorsal root entry zone at the same level of megaloneurites in the gray matter. Immunohistochemical results showed that megaloneurites and ANBs are two distinct abnormal alterations, with megaloneurites co-localizing with vasoactive intestinal peptide immunoreactivity, whereas ANBs were not co-localized. Both ANBs and megaloneurites provide consistent evidence that the anomalous NADPH-d alterations in the aged sacral spinal cord are referred to as a specialized aging marker in the pelvic visceral organs in non-human primates.
Similar content being viewed by others
Introduction
The lumbosacral spinal cord is unique among spinal segments due to its autonomic centers that regulate the pelvic visceral organs1. Within this region, the sacral parasympathetic nucleus (SPN)2 and the dorsal gray commissure (DGC) are critical for receiving projections from primary visceral and somatic afferent fibers originating from the colon3, bladder4,5,6, external urethral sphincter5,7, urethra8 and the pudendal nerve9,10. Somatic and visceral afferent fibers extend from the dorsal root entry zone (DREZ) through Lissauer’s tract (LT) to the region of the SPN and/or reach the DGC through the lateral collateral pathway (LCP) and medial collateral pathway (MCP)10,11,12,13,14. In addition, the gracile nucleus in the medulla oblongata plays a critical function in transmitting and processing visceral and somatosensory information15,16. Visceral and somatosensory signals ascend along the posterior funiculus of the sacral spinal cord to the gracile nucleus, which then emanates fibers that continue to ascend to higher parts of the brain15.
The nicotinamide adenine dinucleotide phosphate-diaphorase (NADPH-d) has been used as a histochemical marker in both central and peripheral nervous systems17. The NADPH-d activity has been used to study neuritic dystrophy in the aging condition18 as well as neurodegenerative changes19,20,21. The NADPH-d activity is widespread in visceral and somatosensory pathways and is associated with neuronal senility changes in the sacral spinal cord and gracile nucleus22,23,24. The large number of NADPH-d positive neurons in the spinal cord appears to be involved in visceral regulation, innervating most of the pelvic visceral organ25,26,27,28. Notably, NADPH-d positive reactivity in the sacral spinal cord is distributed in the visceral afferent pathway29,30,31, which is very similar to the central projections of afferent neurons innervating the pelvic viscera30,32.
Our previous studies have demonstrated that the aging-related NADPH-d positive bodies (ANBs) specifically appear in the sacral spinal cord of aged rats33. ANBs are clearly distinct from normal neural structures, with distinctive morphological and distributional characteristics, and are a distinctive age-related neuroanatomical change. ANBs present spherical or multi-angular shapes existed in the dorsal region of the aged sacral spinal cord. With increasing age, there is a gradual increase in the incidence and size of ANBs. The major distribution of ANBs indicates some anomalous neuronal changes that may account for the disturbance of autonomic and sensory pathways in the pelvic visceral organs in the aging condition.
Recently, we also found NADPH-d positive megaloneurites in the sacral spinal cord of aged dogs34. Megaloneurites are an aging-related aberrant structures characterized by abnormally enlarged diameter and length that are distinct from normal neural structures and are not found in young dogs. Megaloneurites are selectively present on dorsal sensory afferent pathways in the sacral spinal cord and are usually co-localized with vasoactive intestinal peptide (VIP) immunoreactivity, suggesting a potential role in the autonomic nervous system. Megaloneurites may disrupt sensory afferent pathways in the autonomic nervous system, leading to pelvic visceral dysfunction with aging, and serve as a biomarker for age-related neurodegenerative changes in the spinal cord.
ANBs are clearly distinguished from megaloneurites, but both forms of abnormal alterations may be associated with aging and potential neurodegenerative changes. These findings have prompted us to investigate the prevalence of aging-related neurodegenerations across different species, particularly in non-human primates, which are evolutionarily closer to humans. In the present study, we aimed to determine the occurrence and characterization of megaloneurites and/or ANBs in the sacral spinal cord of non-human primates. These degenerative changes, if present, may act as markers of aging in the pelvic visceral organs and could provide valuable information for understanding the neuroanatomical changes that accompany aging and potentially provide targets for therapeutic interventions in aging-related neurodegenerative diseases.
Results
NADPH diaphorase activity in the sacral spinal cord
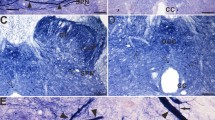
The NADPH-d positive reactivities were extensively distributed in the dorsal regions of the monkey’s spinal cord, and in the sacral spinal cord, where we focused our attention, a substantial amount of NADPH-d positive reactivities could be detected in the regions of the DREZ, LCP, SPN and DGC (Fig. 1A). Notably, we found abnormal megaloneurites and ANBs in these regions of the sacral spinal cord in aged monkeys, which were not present in young monkeys. The NADPH-d staining revealed a moderately dense fiber network of dendrites and axon terminals as well as moderately stained neurons in the dorsal horn, DREZ, LCP, and DGC of the sacral spinal cord of young monkeys (Fig. 1B, C). In the sacral spinal cord of aged monkeys, NADPH-d positive megaloneurites (Fig. 1D-G) were predominantly distributed in the DGC and LCP, and ANBs (black arrowheads in Fig. 1D, H, and I) were found largely in the SPN and superficial dorsal horn. Megaloneurites and ANBs were selectively detected in the sacral spinal cord but not in the lumbar or thoracic and cervical segments (Fig. 1J-L). The general location and selective segmental distribution of the NADPH-d positive megaloneurites and ANBs were related to the central projection of the primary pelvic visceral sensation, mostly located dorsal of the sacral spinal cord.
Microphotographs of NADPH-d positive reactivity in young and aged monkeys at the sacral spinal cord. All of the transverse sections are taken at the same levels. The illustration represents the magnification of the corresponding rectangular frame. A demonstrates the schematic diagram of the distribution NADPH-d staining in transverse sections at different spinal cord segments in monkeys. Black circles in the schematic diagram represent NADPH-d positive neurons, blue shapes represent megaloneurites, and green asterisks represent ANBs. NADPH-d positive reactivity in the dorsal horn and DGC of sacral spinal cord of young (B, C) and aged (D, E) monkeys. The megaloneurites and ANBs in the sacral spinal cord of aged monkeys are completely different from the surrounding normal fibers and neurons (F-I). The NADPH-d positive activity in the dorsal horn of cervical (J), thoracic (K) and lumbar (L) segments of aged monkeys. Frequency distribution of diameters of NADPH-positive fibers in the sacral spinal cord of aged (M) and young (N) monkeys. Comparison of the diameter of NADPH diphosphatase positive fibers in young and aged monkeys (O). The asterisks indicate vascular structures. Open arrowheads: NADPH-d positive neurons, black arrowheads: ANBs, open arrows: normal NADPH-d positive neurites, white arrowheads: megaloneurites. Scale bar in B, D, J-L = 100 μm, in C, E, F-I = 50 μm, ***P < 0.001.
Statistical data showed that in the sacral spinal cord of aged monkeys, the diameter of the majority of the NADPH-d megaloneurites ranged from 2 to 6 μm, with the thickest ones reaching 9 μm, whereas the diameter of normal fibers was typically less than 1 μm (Fig. 1M). In the sacral spinal cord of young monkeys, the diameter of most fibers was less than 1 μm, and no megaloneurites were detected (Fig. 1N). The average diameter of megaloneurites (3.718 ± 0.093 μm) was significantly thicker than the diameter of normal fibers (1.124 ± 0.017 μm) in aged monkeys; the diameter of normal fibers in aged monkeys was also significantly thicker than that of normal fibers in young (0.827 ± 0.013 μm) (Fig. 1O).
NADPH-d activity in the white matter and the DREZ of the sacral spinal cord
In transverse sections of the sacral spinal cord of aged monkeys, massive punctate NADPH-d aberrant reactivities (Fig. 2A) were detected in the lateral white matter of the LCP compared with that of young monkeys (Fig. 2B), and these abnormal alterations were dramatically different from normal nerve cells. It was confirmed to be similar to megaloneurites, which may be in close association with megaloneurites penetrating deeply into the white matter. Longitudinal sections of the sacral spinal cord indicated that the punctate NADPH-d alterations were longitudinally-arranged fibrous extending rostro-caudally and were greatly different from normal fiber bundles and vascular structures in the white matter (Fig. 2C). Numerous NADPH-d positive fibers and varicosities were evident in the lateral white matter of the LCP in young monkeys, and in some instances, these fibers could be traced along the whole length of the section (Fig. 2D). Given the same morphology and characteristics, we still refer to the aged and segmental related alterations in the white matter as megaloneurites.
The NADPH-d positive alterations in the white matter and DREZ of the sacral spinal cord. The location and morphology of the megaloneurites and ANBs in the white matter and LCP of the sacral spinal cord of aged monkeys (A). NADPH-d positive activity in the sacral spinal cord of young monkeys (B). In the longitudinal section, NADPH-d staining fibers in the white matter of the lateral fasciculus of the aged monkeys (C) and young monkeys (D). The distribution of NADPH-d activities in the DREZ of the sacral spinal cord of aged monkeys (E) and young monkeys (F). Longitudinal sections of aged monkeys (G) at the sacral segment showed aberrant NADPH-d positive megaloneurites in the DREZ, transitionally continuous with LT. The illustration represents the magnification of the corresponding rectangular frame. Asterisks indicate vascular structures in C and D. Open arrowheads: NADPH-d positive neurons, black arrowheads: ANBs, open arrows: normal NADPH-d positive neurites, white arrowheads: megaloneurites. Scale bar = 50 μm.
In the transverse sections of the sacral spinal cord of aged monkeys (Fig. 2E), showed numerous punctate intensely NADPH-d activities in the DREZ accompanied with the megaloneurites. These punctate NADPH-d abnormalities occurred specifically in the caudal spinal cord of the aged and were not present in young animals (Fig. 2F). In longitudinal sections, these punctate NADPH-d alterations had the same morphology as megaloneurites (Fig. 2G). The properties of the punctate anomalous structures in the longitudinal sections were that the afferent fibers from the dorsal rootlets underwent some pathological changes in the DREZ, forming NADPH-d megaloneurites that were continuous with Lissauer’s tract (Fig. 2G). Compared to the other segments of the spinal cords, the megaloneurites mainly occurred in the DREZ of the sacral segment in aged monkeys.
Double staining with three neuropeptides
The double-staining of NADPH-d histochemistry combined with CGRP, Neuropeptide Y and VIP immunofluorescence were used to characterize megaloneurites and ANBs. In the DGC, no structures corresponding to NADPH-d positive megaloneurites were detected by Neuropeptide Y (Fig. 3A-C), CGRP immunofluorescence (Fig. 3D-F). For VIP immunoreactivity, VIP and NADPH-d megaloneurites co-localized positively in DGC (white arrowheads in Fig. 3G-I). Similarly, in the white matter, LCP and DREZ of the sacral spinal cord of aged monkeys, NADPH-d positive megaloneurites were not co-localized with Neuropeptide Y (Fig. 4A-C, J-L) or CGRP (Fig. 4D-F, M-O) immunofluorescence, whereas co-localized with VIP immunofluorescence (Fig. 4G-I, P-R). However, the ANBs (black arrowheads) distributed in the superficial dorsal horn and SPN were not co-localized with the VIP immunofluorescence, indicating that the composition of the ANB was different from the NADPH-d positive megaloneurites and they were two different degenerative structures appearing in the lumbosacral spinal cord.
The double-staining of NADPH-d histochemistry combined with Neuropeptide Y, CGRP and VIP immunofluorescence in the DGC of the sacral spinal cord from aged monkeys (n = 3). Neuropeptide Y (A-C) and CGRP (D-F) immunofluorescence failed to label the megaloneurites, whereas VIP immunofluorescence co-localized with the megaloneurites (G-I). Asterisks indicate vascular structures. Black/white arrows: megaloneurites, open arrowheads: normal NADPH-d positive neurons. Scale bar = 50 μm.
The double-staining of NADPH-d histochemistry combined with Neuropeptide Y, CGRP and VIP immunofluorescence in the white matter, LCP and DREZ of the sacral spinal cord from aged monkeys (n = 3). The megaloneurites were not co-localized with Neuropeptide Y (A-C, J-L) or CGRP (D-F, M-O) immunofluorescence in the white matter, LCP and DREZ but were labelled by VIP immunofluorescence (G-I, P-R). The ANBs (black arrows) distributed in the superficial dorsal horn and SPN were not co-localized with the VIP immunofluorescence. Black arrowheads: ANBs, black/white arrows: megaloneurites. Scale bar = 50 μm.
NADPH-d activity in the medulla oblongata
In the medulla oblongata of aged monkeys, primarily in the gracile nucleus and cuneate nucleus (Fig. 5A-D), a large number of spheroidal ANBs were detected compared with young monkeys (Fig. 5E-H). These results coincide with those obtained from previous studies in the gracile nucleus of aged rats35. ANBs in the gracile nucleus and cuneate nucleus were not co-labelled with VIP immunoreactivity (data not shown here).
Microphotographs of NADPH-d positive reactivity in the gracile nucleus and cuneate nucleus of age and young monkeys. Note abnormal spherical ANBs (black arrowheads) in the gracile nucleus (A-B) and cuneate nucleus (C-D) of aged monkeys. The regular NADPH-d positive neurons were observed in the gracile nucleus (E-F) and cuneate nucleus (G-H) of young monkeys. Open arrowheads: NADPH-d positive neurons, black arrowheads: ANBs. Scale bar in A, C, E, G = 100 μm, in B, D, F, H = 50 μm.
Discussion
Many studies have proven that NADPH-d reactivity occurs extensively in the neurons and sensory pathways in the spinal cord29,36,37,38,39,40. Our previous study demonstrated that ANBs occurred in the lumbosacral spinal cord of aged rats33. Recent studies have also shown that NADPH-d positive megaloneurites, a novel neurodegeneration, were found in the sacral spinal cord of aged dogs34. Although the normal and some experimental NADPH-d positivity were studied in the spinal cord of monkeys, few reports were available on the aging-related NADPH-d alterations in the sacral spinal cord41.
The major finding of this study was the identification of two forms of neurodegeneration, ANBs and megaloneurites, in the sacral spinal cord of aged monkeys, and the description of their basic morphological properties, segmental and laminar distribution. Aging neuropathological studies of NADPH-d positivity could be used to reveal the neuronal terminal-pathy of aged conditions23 and neurodegenerative animal models42. In our previous study, ANBs were detected in the gracile nucleus and lumbosacral spinal cord of aged rats36. We similarly detected spherical aberrant ANBs in the gracile nucleus and cuneate nucleus as well as in the dorsal horn of the sacral spinal cord of aged monkeys. The central-projecting axons of the first-order sensory neurons in DRG could configure bifurcation in the spinal cord. The bifurcating branches could terminate in the corresponding spinal segments and ascend to dorsal column nuclei respectively43. Besides somatic sensory inputs, dorsal column nuclei also receive visceral sensory inputs16. We considered that the ANBs in aged rats and monkeys were postulated as dying back in the lumbosacral spinal cord and gracile nucleus accompanied by aging33,35,44,45.
The NADPH-d positive megaloneurites with morphology and organization were first discovered in the sacral spinal cord in aged dogs34. The megaloneurites were different from the previously reported meganeurites, which occurred proximal to axonal initial segments of the somas in younger organisms46. The anatomical locations of the two structures are also strikingly different. The megaloneurites were considered to be swollen neuronal processes, which were enlarged-diameter formations much longer than meganeurites46. Normal NADPH-d positive fibers stained with clear punctate and considerable varicosities. Megaloneurites were intensively stained with less varicosities, and traceable for substantial distances in sacral segments, especially in the longitudinal sections.
Similar to the experimental results in the sacral spinal cord of aged dogs34, the dense hypertrophic megaloneurites extend from DREZ through LT along the lateral edge of the dorsal horn to the SPN and DGC in the sacral spinal cord of aged monkeys, and the fibers in this pathway might play a role in visceral reflex pathways11,14, as the DGC and the LCP receive terminations from the somatic and visceral afferents11,12,13,47. Functionally, the sacral spinal cord was known to be associated with bowel bladder, and sexual dysfunction48,49,50. Previous tracing experiments in rats have also demonstrated that a large percentage of afferent neurons projecting to pelvic visceral organs and specifically to the bladder and urethra exhibit NADPH-d activity6,30,51. The distribution of the megaloneurites overlapped both the efferent and afferent pathways of the autonomic system, which regulates the pelvic organs. Segmental specialized aged-related changes may be also considered for the segmental distribution of NADPH-d neurons in the spinal cord52. The neuron disorder of aging may be caused by environmental factors. For example, NADPH-d neurons in the white matter are also vulnerable to chronic heavy metal poisoning53.
The megaloneurites were characterized as longitudinally extending fiber bundles in both transverse and longitudinal sections of the sacral spinal cord. The morphology of megaloneurites was clearly distinguished from the vascular structures found under light microscopy. The megaloneurites occurred in the DREZ, which is the transitional location between the peripheral nervous system and CNS in the spinal cord and brain stem54. The pelvic visceral sensory information from the urogenital organs is routed to the sacral spinal cord through centrally-projecting axons that pass into the CNS via the DREZ and continue with LT. DREZ plays an important role in neurodegeneration55 and axon routing during development56. All these segmental selective deteriorations demonstrated that the sacral segments exited relevant adverse conditions more vulnerable to aging degenerations.
The double-staining experiment demonstrated that VIP immunoreactivity and NADPH-d megaloneurites positively co-localized in the DREZ, DGC, LCP and white matter of the sacral spinal cord of aged monkeys. VIP is involved in the information transmission and physiological regulation of many functions of the organism and is released in the central and peripheral nervous system to support neuronal survival under physiological and pathological conditions57,58. Studies have shown that VIP can be considered as a marker for parasympathetic nerves59 and that VIP-containing fibers and terminals are well distributed in the lumbosacral spinal cord and play an important role in the efferent and afferent pathways of the autonomic nervous system that innervate the pelvic organs60,61,62,63,64. In our previous study, megaloneurites in the sacral spinal cord of aged dogs similarly overlapped with the distribution of VIP immunoreactivity, suggesting that megaloneurites may affect sensory afferent pathways in the autonomic nervous system in the aging condition, leading to pelvic visceral dysfunction.
Conclusion
In summary, this study revealed a special occurrence of the megaloneurites and ANBs in the sacral spinal cord of aged monkeys. These results confirm our previous discovery in aged rats and dogs, respectively. Furthermore, NADPH-d megaloneurites could be co-labelled by VIP immunofluorescence, whereas ANBs were not, indicating that megaloneurites and ANBs were two distinct forms of neurodegeneration. Our results suggested that megaloneurites and ANBs are recognized as specific aging markers associated with progressive aging deterioration and aberrant structures.
Materials and methods
Animal and tissue preparation
Young (3.8 ± 0.7 years old, n = 4) and aged (23.8 ± 4.3 years old, n = 5) female cynomolgus monkeys, weighing 3–5 kg, were used in our experiments. All animals were housed under a 12:12-h light/dark cycle in the animal care facilities. The facilities are certified by the Council on Accreditation of the Association for Assessment and Accreditation of Laboratory Animals Care (International) and China National Accreditation Service for Conformity Assessment. The ambient temperature was maintained at 24 ± 2 ℃ and relative humidity at 65 ± 4%. Reverse osmosis water was available ad libitum. Normal food, fresh fruit and vegetables were supplied twice daily. Animals were purchased from Beijing Xierxin Biological Resources Research Institute (Permit Number: 11805300010425, Beijing, China), Wincon TheraCells Biotechnologies Co., Ltd. (Permit Number: WD-0312010, Nanning, China) and Hainan Jingang Biotech Co., Ltd (Permit Number: 46001200000114, Hainan, China). These animals did not show any neurological deficits before experiments and were humanely euthanized. The experimentations have been conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org). All experimental procedures followed the International Guiding Principles for Biomedical Research Involving Animals and the Laboratory Animal-Guideline for ethical review of animal welfare (GB/T 35892 − 2018) and were approved by the Ethics Committee in Animal and Human Experimentation of the Jinzhou Medical University (IACUC Number: 2014JCATBH0E-NNSF-0004).
The animals were anesthetized with sodium pentobarbital (Laboratory Animal-Guidelines for euthanasia, GB/T 39760 − 2018, 80 mg/kg i.v.) and perfused transcardially with saline followed by 4% paraformaldehyde in a 0.01 M phosphate buffer saline (PBS, pH 7.4). Following perfusion fixation, the spinal cords and brain were obtained and placed in 25% sucrose for 48 h. Following dehydration and fixation, the spinal cord and medulla oblongata tissues were divided into cervical, thoracic, lumbar, and sacral segments, as well as the gracile nucleus of the medulla. A subset of these tissues (from three aged and two young monkeys) was sectioned transversely into 40 μm slices using a cryostat. To observe the longitudinal morphology and distribution of NADPH-diaphorase positivity, additional monkeys (two aged and two young) were selected, and longitudinal sections of 40 μm in thickness were prepared from each spinal segment. To facilitate the comparison between transverse and longitudinal sections from the same tissue, a 5 mm length of each spinal segment was sectioned transversely during the preparation of longitudinal slices. Sections from each segment were divided equally into three sets and collected into well plates containing 0.3% Triton X-100 in PBS.
NADPH diaphorase histochemistry
NADPH-d staining was performed with transverse and longitudinal sections from one-third-well plates of each young and aged animal, and free-floating sections were used for NADPH-d staining33,34. The tissue sections were stained by NADPH-d histochemistry, with incubation in 0.01 M PBS (PH 7.4), 0.3% Triton X-100 containing 1.0 mM reduced-NADPH (Sigma, St. Louis, MO, USA) and 0.2 mM nitro blue tetrazolium (NBT, Sigma), at 37℃ for 2 to 3 h. The reaction was stopped by washing the sections with 0.01 M PBS.
Double immunofluorescence staining
Following NADPH-d histochemical staining of the sacral spinal cord sections, a selection of 30 slices of adjacent sections with typical megaloneurites from each aged animal (three aged animals included) were subjected to double-staining of NADPH-d with calcitonin gene-related peptide (CGRP), vasoactive intestinal peptide (VIP) or neuropeptide Y (NPY) immunofluorescence, respectively. The sections were collected in 24-well plates containing 0.01 M PBS and washed three times. Then sections were incubated for 1 h at room temperature in blocking solution (0.01 M PBS) at pH 7.4 with 1% BSA and processed for free-floating immunofluorescence using primary polyclonal antibodies that label CGRP (mouse; 1:100, Sigma, C7113, USA), VIP (rabbit, 1:1000, Sigma, HPA017324, USA), NPY (rabbit; 1:2000, Sigma, N9528, USA). The sections were incubated with the primary antibody in PBS containing 1% BSA for 24 h at 4 °C, respectively. For a negative control, a few of sections were incubated without primary antibody in each immunofluorescence test. The sections were then washed three times with PBS. Fluorescent-conjugated secondary antibodies (Goat anti-Rabbit IgG (H + L), Alexa Fluor 594 [1:800, Life] and Goat anti-Mouse IgG, Alexa Fluor 488 [1:800, Life]), were applied to the sections for 1.5 h at 37 °C in the dark. Finally, after several washes with PBS, the sections were counterstained with DAPI for 10 min. The sections were mounted onto slides and cover-slipped. For controls of immunofluorescence staining omitted the primary antibody, no specified immunostaining was detected in the control experiments.
Image acquisition and measurement of fibers
Images were captured with a DP80 camera in an Olympus BX53 microscope (Olympus, Japan). Sections were observed under the light microscope and 20 sections from the sacral spinal cord in each animal were quantitated using Olympus image analysis software (Cellsens Standard, Olympus). The diameter of 500 NADPH-d megaloneurites and normal fibers were also measured with Neurolucida 360 (MBF Bioscience, Inc, USA). Unbiased rating was set by the examiners who were not aware of the grouped labelling in the sections.
Statistics analysis and figure edition
GraphPad Prism 9.0 (GraphPad Software, La Jolla, CA) software was used to analyze the frequency distribution and the differences in the diameters of NADPH-d positive fibers in the sacral spinal cord of young and aged monkeys. To quantify and characterize the distribution of NADPH-d positive fibers, we statistically analyze the frequency distribution of the diameters of megaloneurites and normal fibers in the sacral spinal cord of aged and young monkeys. In addition, to analyze the differences in the diameter of NADPH-d positive normal fibers and megaloneurites within the sacral spinal cord across different age groups, for consistently normally distributed data, we used unpaired t-tests to compare the diameters of NADPH-d positive fibers in young and aged monkeys. Data are expressed as mean ± standard error, and the significance level was set at P < 0.05. Figures were edited with CorelDRAW Graphics Suite 2020 (Ottawa, Canada).
Data availability
The data used to support the findings of this study are available from the corresponding author upon request. The original manuscript as a preprint has been posted to BioRxiv (bioRxiv 527358; https://doi.org/10.1101/527358).
References
De Groat, W. C., Booth, A. M., Milne, R. J. & Roppolo, J. R. Parasympathetic preganglionic neurons in the sacral spinal cord. J. Auton. Nerv. Syst. 5, 23–43. https://doi.org/10.1016/0165-1838(82)90087-x (1982).
Nadelhaft, I., Roppolo, J., Morgan, C. & de Groat, W. C. Parasympathetic preganglionic neurons and visceral primary afferents in monkey sacral spinal cord revealed following application of horseradish peroxidase to pelvic nerve. J. Comp. Neurol. 216, 36–52. https://doi.org/10.1002/cne.902160105 (1983).
Palecek, J. & Willis, D. W. The dorsal column pathway facilitates visceromotor responses to colorectal distention after colon inflammation in rats. Pain. 104, 501–507. https://doi.org/10.1016/s0304-3959(03)00075-7 (2003).
Wang, H. F., Shortland, P., Park, M. J. & Grant, G. Retrograde and transganglionic transport of horseradish peroxidase-conjugated cholera toxin B subunit, wheatgerm agglutinin and isolectin B4 from Griffonia simplicifolia I in primary afferent neurons innervating the rat urinary bladder. Neuroscience. 87, 275–288. https://doi.org/10.1016/s0306-4522(98)00061-x (1998).
Nadelhaft, I. & Vera, P. L. Separate urinary bladder and external urethral sphincter neurons in the central nervous system of the rat: simultaneous labeling with two immunohistochemically distinguishable pseudorabies viruses. Brain Res. 903, 33–44. https://doi.org/10.1016/s0006-8993(01)02349-6 (2001).
McNeill, D. L., Traugh, N. E. Jr., Vaidya, A. M., Hua, H. T. & Papka, R. E. Origin and distribution of NADPH-diaphorase-positive neurons and fibers innervating the urinary bladder of the rat. Neurosci. Lett. 147, 33–36. https://doi.org/10.1016/0304-3940(92)90768-3 (1992).
Nadelhaft, I. & Vera, P. L. Neurons in the rat brain and spinal cord labeled after pseudorabies virus injected into the external urethral sphincter. J. Comp. Neurol. 375, 502–517. https://pubmed.ncbi.nlm.nih.gov/8915845/ (1996).
Vizzard, M. A., Erickson, V. L., Card, J. P., Roppolo, J. R. & de Groat, W. C. Transneuronal labeling of neurons in the adult rat brainstem and spinal cord after injection of pseudorabies virus into the urethra. J. Comp. Neurol. 355, 629–640. https://doi.org/10.1002/cne.903550411 (1995).
Dobberfuhl, A. D., Oti, T., Sakamoto, H. & Marson, L. Identification of CNS neurons innervating the levator ani and ventral bulbospongiosus muscles in male rats. J. Sex. Med. 11, 664–677. https://doi.org/10.1111/jsm.12418 (2014).
McKenna, K. E. & Nadelhaft, I. The organization of the pudendal nerve in the male and female rat. J. Comp. Neurol. 248, 532–549. https://doi.org/10.1002/cne.902480406 (1986).
Morgan, C., Nadelhaft, I. & de Groat, W. C. The distribution of visceral primary afferents from the pelvic nerve to Lissauer’s tract and the spinal gray matter and its relationship to the sacral parasympathetic nucleus. J. Comp. Neurol. 201, 415–440. https://doi.org/10.1002/cne.902010308 (1981).
Thor, K. B., Morgan, C., Nadelhaft, I., Houston, M. & De Groat, W. C. Organization of afferent and efferent pathways in the pudendal nerve of the female cat. J. Comp. Neurol. 288, 263–279. https://doi.org/10.1002/cne.902880206 (1989).
Bansal, U. et al. Lumbosacral spinal segmental contributions to tibial and pudendal neuromodulation of bladder overactivity in cats. Neurourol. Urodyn. 36, 1496–1502. https://doi.org/10.1002/nau.23159 (2017).
Nadelhaft, I. & Booth, A. M. The location and morphology of preganglionic neurons and the distribution of visceral afferents from the rat pelvic nerve: a horseradish peroxidase study. J. Comp. Neurol. 226, 238–245. https://doi.org/10.1002/cne.902260207 (1984).
Ding, Y. Q. et al. Primary afferent fibers of the pelvic nerve terminate in the gracile nucleus of the rat. Neurosci. Lett. 272, 211–214. https://doi.org/10.1016/s0304-3940(99)00599-6 (1999).
Willis, W., Al-Chaer, E., Quast, M. & Westlund, K. A. Visceral pain pathway in the dorsal column of the spinal cord. Proc. Natl. Acad. Sci. U.S.A. 96, 7675–7679. https://doi.org/10.1073/pnas.96.14.7675 (1999).
Hope, B. T., Michael, G. J., Knigge, K. M. & Vincent, S. R. Neuronal NADPH diaphorase is a nitric oxide synthase. Proc. Natl. Acad. Sci. U.S.A. 88, 2811–2814. https://doi.org/10.1073/pnas.88.7.2811 (1991).
Lukácová, N. et al. Peripheral axotomy affects nicotinamide adenine dinucleotide phosphate diaphorase and nitric oxide synthases in the spinal cord of the rabbit. J. Neurosci. Res. 71, 300–313. https://doi.org/10.1002/jnr.10470 (2003).
Benzing, W. C. & Mufson, E. J. Increased number of NADPH-d-positive neurons within the substantia innominata in Alzheimer’s disease. Brain Res. 670, 351–355. https://doi.org/10.1016/0006-8993(94)01362-l (1995).
Morton, A. J., Nicholson, L. F. B. & Faull, R. L. M. Compartmental loss of NADPH diaphorase in the neuropil of the human striatum in huntington’s disease. Neuroscience. 53, 159–168. https://doi.org/10.1016/0306-4522(93)90294-P (1993).
Kowall, N. W. & Beal, M. F. Cortical somatostatin, neuropeptide Y, and NADPH diaphorase neurons: normal anatomy and alterations in Alzheimer’s disease. Ann. Neurol. 23, 105–114. https://doi.org/10.1002/ana.410230202 (1988).
Ma, S. X. et al. Impaired expression of neuronal nitric oxide synthase in the gracile nucleus is involved in neuropathic changes in Zucker Diabetic Fatty rats with and without 2,5-hexanedione intoxication. Neurosci. Res. 106, 47–54. https://doi.org/10.1016/j.neures.2015.10.007 (2016).
Ma, S., Cornford, M. E., Vahabnezhad, I., Wei, S. & Li, X. Responses of nitric oxide synthase expression in the gracile nucleus to sciatic nerve injury in young and aged rats. Brain Res. 855, 124–131. https://doi.org/10.1016/s0006-8993(99)02379-3 (2000).
Ma, S. X., Holley, A. T., Sandra, A., Cassell, M. D. & Abboud, F. M. Increased expression of nitric oxide synthase in the gracile nucleus of aged rats. Neuroscience. 76, 659–663 (1997).
Tamura, M. et al. Coexistence of nitric oxide synthase, tyrosine hydroxylase and vasoactive intestinal polypeptide in human penile tissue–a triple histochemical and immunohistochemical study. J. Urol. 153, 530–534. https://doi.org/10.1097/00005392-199502000-00077 (1995).
Chakder, S. & Rattan, S. Evidence for VIP-induced increase in NO production in myenteric neurons of opossum internal anal sphincter. Am. J. Physiol. 270, G492–497. https://doi.org/10.1152/ajpgi.1996.270.3.G492 (1996).
Zhou, Y., Tan, C. K. & Ling, E. A. Distribution of NADPH-diaphorase and nitric oxide synthase-containing neurons in the intramural ganglia of guinea pig urinary bladder. J. Anat. 190 (Pt 1), 135–145. https://doi.org/10.1046/j.1469-7580.1997.19010135.x (1997).
Zhou, Y. & Ling, E. A. Colocalization of nitric oxide synthase and some neurotransmitters in the intramural ganglia of the guinea pig urinary bladder. J. Comp. Neurol. 394, 496–505 (1998).
Vizzard, M. A., Erickson, K. & de Groat, W. C. Localization of NADPH diaphorase in the thoracolumbar and sacrococcygeal spinal cord of the dog. J. Auton. Nerv. Syst,. 64, 128–142. https://doi.org/10.1016/s0165-1838(97)00025-8 (1997).
Vizzard, M. A., Erdman, S. L. & de Groat, W. C. Localization of NADPH diaphorase in bladder afferent and postganglionic efferent neurons of the rat. J. Auton. Nerv. Syst,. 44, 85–90. https://doi.org/10.1016/0165-1838(93)90382-5 (1993).
Vizzard, M. A., Erdman, S. L. & de Groat, W. C. The effect of rhizotomy on NADPH diaphorase staining in the lumbar spinal cord of the rat. Brain Res. 607, 349–353. https://doi.org/10.1016/0006-8993(93)91530-6 (1993).
Gonzalez, E. J., Merrill, L. & Vizzard, M. A. Bladder sensory physiology: neuroactive compounds and receptors, sensory transducers, and target-derived growth factors as targets to improve function. Am. J. Physiol. Regul. Integr. Comp. Physiol. 306, R869–878. https://doi.org/10.1152/ajpregu.00030.2014 (2014).
Tan, H. et al. Age-related NADPH-diaphorase positive bodies in the lumbosacral spinal cord of aged rats. Arch. Histol. Cytol. 69, 297–310. https://doi.org/10.1679/aohc.69.297 (2006).
Li, Y. et al. De novo aging-related NADPH diaphorase positive megaloneurites in the sacral spinal cord of aged dogs. Sci. Rep. 13, 22193. https://doi.org/10.1038/s41598-023-49594-0 (2023).
Wei, Z. et al. Aging-related NADPH-diaphorase Body (Tan body): a morphological marker of aging neurodegenerative damage (2016).
Hou, W. et al. NADPH diaphorase neuronal dystrophy in gracile nucleus, cuneatus nucleus and spinal trigeminal nucleus in aged rat. bioRxiv. http://doi.org/10.1101/2019.12.21.885988 (2019).
Marsala, J. et al. Reduced nicotinamide adenine dinucleotide phosphate diaphorase in the spinal cord of dogs. Neuroscience. 85, 847–862. https://doi.org/10.1016/s0306-4522(97)00690-8 (1998).
Anderson, C. R. NADPH diaphorase-positive neurons in the rat spinal cord include a subpopulation of autonomic preganglionic neurons. Neurosci. Lett. 139, 280–284. https://doi.org/10.1016/0304-3940(92)90571-n (1992).
Valtschanoff, J. G., Weinberg, R. J. & Rustioni, A. NADPH diaphorase in the spinal cord of rats. J. Comp. Neurol. 321, 209–222. https://doi.org/10.1002/cne.903210204 (1992).
Marsala, J., Marsala, M., Vanický, I. & Taira, Y. Localization of NADPHd-exhibiting neurons in the spinal cord of the rabbit. J. Comp. Neurol. 406, 263–284. https://pubmed.ncbi.nlm.nih.gov/10096610/ (1999).
Torres-da-Silva, K. R. et al. Neurochemistry study of spinal cord in non-human primate (Sapajus spp). Eur. J. histochemistry: EJH. 60, 2623. https://doi.org/10.4081/ejh.2016.2623 (2016).
Quinn, J., Davis, F., Woodward, W. R. & Eckenstein, F. Beta-amyloid plaques induce neuritic dystrophy of nitric oxide-producing neurons in a transgenic mouse model of Alzheimer’s disease. Exp. Neurol. 168, 203–212. https://doi.org/10.1006/exnr.2000.7598 (2001).
Réthelyi, M. & Szentágothai, J. Somatosensory system (eds D. Albe-Fessard et al.) 207–252 (Springer Berlin Heidelberg, 1973).
Blakemore, W. F. & Cavanagh, J. B. Neuroaxonal dystrophy occurring in an experimental dying back process in the rat. Brain. 92, 789–804. https://doi.org/10.1093/brain/92.4.789 (1969).
Oda, K., Yamazaki, K., Miura, H., Shibasaki, H. & Kikuchi, T. Dying back type axonal degeneration of sensory nerve terminals in muscle spindles of the gracile axonal dystrophy (GAD) mutant mouse. Neuropathol. Appl. Neurobiol. 18, 265–281. https://doi.org/10.1111/j.1365-2990.1992.tb00789.x (1992).
Walkley, S. U. & Pierok, A. L. Ferric ion-ferrocyanide staining in ganglioside storage disease establishes that meganeurites are of axon hillock origin and distinct from axonal spheroids. Brain Res. 382, 379–386. https://doi.org/10.1016/0006-8993(86)91348-x (1986).
Al-Chaer, E. D., Lawand, N. B., Westlund, K. N. & Willis, W. D. Visceral nociceptive input into the ventral posterolateral nucleus of the thalamus: a new function for the dorsal column pathway. J. Neurophysiol. 76, 2661–2674. https://doi.org/10.1152/jn.1996.76.4.2661 (1996).
Cohen, B. A., Major, M. R. & Huizenga, B. A. Pudendal nerve evoked potential monitoring in procedures involving low sacral fixation. Spine (Phila Pa. 1976). 16, S375–378 (1991).
Ogiwara, H. & Morota, N. Pudendal afferents mapping in posterior sacral rhizotomies. Neurosurgery. 74, 171–175. https://doi.org/10.1227/neu.0000000000000235 (2014).
Barbe, M. F. et al. Clarification of the innervation of the bladder, external urethral sphincter and clitoris: a neuronal tracing study in female mongrel hound dogs. Anat. record (Hoboken N J. : 2007). 301, 1426–1441. https://doi.org/10.1002/ar.23808 (2018).
Vizzard, M. A., Erdman, S. L. & de Groat, W. C. Localization of NADPH-diaphorase in pelvic afferent and efferent pathways of the rat. Neurosci. Lett. 152, 72–76. https://doi.org/10.1016/0304-3940(93)90486-5 (1993).
Freire, M. A. et al. Histochemical characterization, distribution and morphometric analysis of NADPH diaphorase neurons in the spinal cord of the agouti. Front. Neuroanat. 2, 2. https://doi.org/10.3389/neuro.05.002.2008 (2008).
Oliveira, R. B., Gomes-Leal, W., do-Nascimento, J. L. & Picanço-Diniz, C. W. Methylmercury intoxication and histochemical demonstration of NADPH-diaphorase activity in the striate cortex of adult cats. Brazilian J. Med. Biol. Res. = Revista brasileira de pesquisas medicas e biologicas. 31, 1157–1161. https://doi.org/10.1590/s0100-879x1998000900009 (1998).
Fraher, J. P. The CNS-PNS transitional zone of the rat. Morphometric studies at cranial and spinal levels. Prog. Neurobiol. 38, 261–316. https://doi.org/10.1016/0301-0082(92)90022-7 (1992).
Wu, D. et al. Expressing constitutively active rheb in adult dorsal root ganglion neurons enhances the integration of sensory axons that regenerate across a chondroitinase-treated dorsal root entry zone following dorsal root crush. Front. Mol. Neurosci. 9, 49. https://doi.org/10.3389/fnmol.2016.00049 (2016).
Smith, C. J. et al. TNFa/TNFR2 signaling is required for glial ensheathment at the dorsal root entry zone. PLoS Genet. 13, e1006712. https://doi.org/10.1371/journal.pgen.1006712 (2017).
Gozes, I. & Brenneman, D. E. VIP: molecular biology and neurobiological function. Mol. Neurobiol. 3, 201–236. https://doi.org/10.1007/bf02740606 (1989).
White, C. M., Ji, S., Cai, H., Maudsley, S. & Martin, B. Therapeutic potential of vasoactive intestinal peptide and its receptors in neurological disorders. CNS Neurol. Disord. Drug Target. 9, 661–666. https://doi.org/10.2174/187152710793361595 (2010).
Muraoka, K. et al. Site-dependent differences in the composite fibers of male pelvic plexus branches: an immunohistochemical analysis of donated elderly cadavers. BMC Urol. 18, 47. https://doi.org/10.1186/s12894-018-0369-9 (2018).
Rosati, L., Andreuccetti, P. & Prisco, M. Vasoactive intestinal peptide (VIP) localization in the epididymis of two vertebrate species. C.R. Biol. 340, 379–385. https://doi.org/10.1016/j.crvi.2017.07.001 (2017).
Barry, C. M. et al. Morphological and neurochemical differences in peptidergic nerve fibers of the mouse vagina. J. Comp. Neurol. 525, 2394–2410. https://doi.org/10.1002/cne.24214 (2017).
Yoshiyama, M. & de Groat, W. C. The role of vasoactive intestinal polypeptide and pituitary adenylate cyclase-activating polypeptide in the neural pathways controlling the lower urinary tract. J. Mol. Neurosci. 36, 227–240. https://doi.org/10.1007/s12031-008-9090-6 (2008).
Gibson, S. J. et al. A VIP/PHI-containing pathway links urinary bladder and sacral spinal cord. Peptides 7(Suppl 1), 205–219. https://doi.org/10.1016/0196-9781(86)90188-9 (1986).
Anand, P. et al. A VIP-containing system concentrated in the lumbosacral region of human spinal cord. Nature. 305, 143–145. https://doi.org/10.1038/305143a0 (1983).
Acknowledgements
We are grateful to all members of our study team for their whole-hearted cooperation. We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Funding
This work was supported by grants from National Natural Science Foundation of China (81471286), Undergraduate Training Programs for Innovation and Entrepreneurship of Liaoning (201410160007) and Research Start-Up Grant for New Science Faculty of Jinzhou Medical University (173514017).
Author information
Authors and Affiliations
Contributions
YHL and ZCW conceived and performed the experiments as well as analyzed data. YGJ, WH, YW, SY, GMS, CXR, XMX, HL, and JJS assisted YHL and ZCW in the experimentation. GHD and HBT provided important experimental guidance. HBT, SY and GHD discussed the results. YHL and HBT wrote the manuscript. HBT supervised the project and coordinated the study. All authors discussed the manuscript and approved the submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Wei, Z., Jia, Y. et al. Aging-related NADPH diaphorase positive neurodegenerations in the sacral spinal cord of aged non-human primates. Sci Rep 14, 27168 (2024). https://doi.org/10.1038/s41598-024-77974-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-77974-7
Keywords
This article is cited by
-
Biomarkers of ageing of humans and non-human primates
Nature Reviews Molecular Cell Biology (2025)