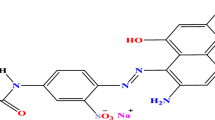
Abstract
Industrial wastewater treatment using UV irradiation in combination with oxidants or catalysts (TiO2) has attracted attention as a promising substitute for conventional methods. Studying the preparation and characterization of TiO2–SnO2 nanocomposites with different ratios, as well as their use in the enhanced photocatalytic degradation of acid red 37 dye in aqueous solution under UV irradiation as a model pollutant, are the goals of the research. The crystalline structures of the prepared nanomaterials were confirmed by XRD and the surface morphology of the samples was studied by TEM. The elemental compositions of the catalysts were confirmed by EDAX. The optical properties of the powder samples were analyzed with UV–Vis spectroscopy and their band gaps were estimated. The photocatalytic degradation was investigated using several advanced oxidation techniques using a batch photoreactor. The TiO2–SnO2 (90:10) nanocomposite showed the best degradation efficiency.
Similar content being viewed by others
Introduction
However, as we learn more about how finite the earth’s supply of fossil raw materials has sparked a flurry of initiatives to discover environmentally friendly and long-term replacements. Aside from attempting to employ more renewable as a fundamental chemical feedstock, one of the key difficulties for science and engineering is meeting the world’s expanding energy demand1. As a result, it is critical to create new efficient technologies for converting energy from renewable sources. Hydrogen is one of the most interesting energy resources since it allows for the effective use of a fuel cell to convert chemical energy into electrical energy with the only byproduct being water2. As a result, an obvious benefit of the enormous reduction in pollutants is a hydrogen economy.
Due to its many benefits, such as its simplicity of use, high efficiency, and absence of secondary pollutants, a lot of focus has lately been placed on photocatalysis as one of the emerging advanced oxidation methods. It is widely recognized as method of treating wastewater that shows the greatest promise as being eco-friendly to the environment3,4. Catalysts play a crucial part in the photocatalytic process, as we all know. Titanium dioxide, among the many photocatalysts, is of particular interest because of its chemical stability, peculiar physical characteristics, and lack of toxicity. It is frequently used in energy conversion, hydrogen production, air purification, and wastewater treatment5. Titanium dioxide, a semiconductor, helps electrons to go from the valence band to the conduction band when exposed to light.
Compared to more traditional chemical oxidation techniques, photocatalytic applications, and semiconductor-based photocatalysis in particular, stand out as the most attractive means for decomposing harmful substances to non-hazardous output. This is due to the fact that semiconductors are (i) cheap, (ii) are non-toxic, (iii) have a high surface area, (iv) have broad absorption spectra with high absorption coefficients, (v) exhibit tunable properties that can be modified through size reduction, doping, sensitizers, etc., (vi) afford the facility for multielectrons transfer process, and (vii) can be used for long periods of time without significantly losing its photocatalytic activity.
In order for a semiconductor to be photochemically active, its redox potential must be positive enough for the photogenerated valence band hole to produce adsorbed OH radicals that can oxidise organic pollutants and negative enough for the conduction band electron to reduce adsorbed O2 to superoxide.
The category of photocatalysts known as metal-oxide semiconductor photocatalysts includes TiO2. TiO2 is a good n-type semiconductor photocatalyst because of its high activity in Fig. 1, sustained resistance to photo- and chemical-corrosion during reaction circumstances, strong oxidizing power, stable substance, and nontoxic. TiO2nanostructures, on the other hand, are a potential semiconductor for a variety of applications, including batteries, solar cells, dye pollution catalysis, hydrogen generation, and water splitting6,7,8.
The three crystal phases of TiO2are rutile, brookite and anatase9. Although antase and rutile are the most commonly investigated phases, anatase has a greater photocatalytic degradation activity with organic pollutants than rutile due to its crystal structural arrangement10. TiO2 can only absorb ultraviolet light due to its high band gap energy 3.2 eV for anatase and 3.0 eV for rutile, respectively. This prevents TiO2from being used for visible light absorption11.
Schematic of the chemical reaction pathway of an n-type semiconductor such as TiO2.
Tin oxide (SnO2) has sparked a lot of scientific interest. SnO2 is an outstanding optical and electrical material that, at room temperature, is an of n-type semiconductor that has a wide band gap (3.6 eV), unusual optical transparency, low resistivity, and a high theoretical specific capacity. The electron mobility in SnO2 is quite high (between 100 and 200 cm2 V1 s1), which suggests that photoexcited electron transport happens more quickly. SnO2 nanoparticles (SnO2 NPs) have high band gap energies, high stability, and unusual structural and optical properties. Another distinctive property of SnO2 NPs is the blue shift of the band edge transition energy. Furthermore, as the wide surface area serves to increase photocatalytic reaction sites, the small size of SnO2 NPs may be a contributing factor in the considerable degradation rate of the organic dye on as-produced SnO2NPs with diameters smaller than 10 nm and increases electron-hole separation efficiency12.
Results and discussion
HR-TEM of TiO2–SnO2 wt% nanocomposites
TiO2-SnO2 nanocomposite samples were examined with HR-TEM to investigate the homogenety and the morphology of the samples. Prior to the investigation, the samples were coated with gold using sputtering coater (model: S 150 B, Edwards High Vacuum Ltd., England).
Figure 2 shows the HR-TEM picture of the synthesized TiO2–SnO2 (97:3) wt% nanocomposite. The particles had tetragonal shape with a diameter of 26.3–88 nm, according to a detailed analysis of the micrograph.
HR-TEM of TiO2–SnO2 (97:3) wt% nanocomposite.
Figure 3 shows the HR-TEM picture of the synthesized TiO2–SnO2 (90:10) wt% nanocomposite. The particles had a roughly tetragonal shape and semi round shape with a diameter of 27.6–54.5 nm, according to a detailed analysis of the micrograph.
HR-TEM of TiO2–SnO2 (90:10) wt% nanocomposite.
Figure 4 shows the HR-TEM picture of the prepared TiO2–SnO2 (80:20) wt% nanocomposite. The particles had tetragonal and semi round shaped with a diameter of 30.2–72.2 nm, according to a detailed analysis of the micrograph.
HR-TEM of TiO2–SnO2 (80:20) wt% nanocomposite.
It can be concluded from the previous Figs. 2, 3 and 4 that increasing of SnO2 (3 wt%) in TiO2–SnO2nanocomposite lead to an increase in the particle size due to nucleation and growth happen13. Further increase in SnO2 to 10 wt% results in increasing the particle size, whereas further increase in SnO2 up to 20 wt% results in decreasing the particle size of the nanocomposite due to agglomeration.
XRD of the prepared TiO2–SnO2 nanocomposites
The purity, crystallite size, and crystal structure of the generated nanoparticles are all examined using XRD analysis. The width of a certain XRD peak in a diffraction pattern connected with a particular planar reflection inside the crystal unit cell is used to calculate crystallite size. The size of the crystallites is therefore reflected in the peak broadening.
XRD of TiO2–SnO2 (97:3) wt% nanocomposite
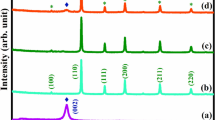
Figure 5 illustrates the XRD pattern of TiO2–SnO2 (97:3) wt% nanocomposite sample with diffraction peaks at 2θ = 25.30, 26.56, 27.42, 33.92, 36.09, 37.81, 38.65, 41.29, 47.99, 51.85, 54.03, 55.0, and 68.92. The crystal tetragonal structure’s allocated pattern, which indicates how it was formed, corresponds to TiO2–SnO2 (97:3) wt%. The measured diffraction peaks are consistent having a standard card issued by the Joint Committee of Powder Diffraction Standards (JCPDS card No 041-1445 and 001-0562) which showed a slight peak shift and an increase in the intensity of peak, with an average crystal size of 42.3 nm.
XRD of TiO2–SnO2 (90:10) wt% nanocomposite
Figure 5 depicts the XRD pattern of TiO2–SnO2 (90:10) wt% nanocomposite sample with diffraction peaks at 2θ = 25.33, 26.66, 27.43, 33.82, 36.08, 37.92, 41.24, 48.08, 51.75, 54.33, 56.68, 62.72, and 68.92. The tetragonal crystal structure’s allocated pattern, which indicates how it was formed corresponds to TiO2–SnO2 (90:10) wt%. The measured diffraction peaks are consistent having a standard card issued by the Joint Committee of Powder Diffraction Standard (JCPDS card No 041-1445 and 001-0562) which showed a slight peak shift and an increase in the intensity of peak, with an average crystal size of 37.09 nm.
XRD of TiO2–SnO2 (80:20) wt% nanocomposite
Figure 5 depicts the XRD pattern of TiO2–SnO2 (80:20) wt% nanocomposite sample with diffraction peaks at 2θ = 25.28, 26.57, 27.39, 33.87, 36.92, 35.92, 37.78, 41.29, 48.09, 51.70, 54.14, 62.77, and 68.86. The tetragonal crystal structure’s allocated pattern, which indicates how it was formed corresponds to TiO2–SnO2 (80:20) wt%. The measured diffraction peaks are consistent with the Joint Committee of Powder Diffraction standard (JCPDS card No 041-1445 and 001-0562) which showed a slight peak shift and an increase in the intensity of peak, with an average crystal size of 38.5 nm.
XRD of the prepared TiO2–SnO2 nanocomposites.
According to the results as shown Fig. 5 which obtained from XRD of TiO2-SnO2 nanocomposites, it could be concluded that the average crystal size by Scherrer Eq. (1) of TiO2–SnO2 (97:3) wt% was 42.3 nm and was found to decrease to 37.05 with increasing the w % of SnO2. Further increase of SnO2 wt% to 20 resulted in decreasing the average crystal size to 38.5 nm.
Peak width (B) is inversely proportional to crystallite size (L).
K (the Scherrer constant)—λ wavelength
According to the results as shown Figs. 5, 6 and 7 which obtained from XRD of TiO2-SnO2 nanocomposites, it could be concluded that the average crystal size of TiO2–SnO2 (97:3) wt% was 42.3 nm and was found to decrease to 37.05 with increasing the wt% of SnO2. Further increase of SnO2 wt% to 20 resulted in decreasing the average crystal size to 38.5 nm. respectively according to Table 1.
EDAX of the prepared TiO2–SnO2 nanocomposites
The obtained results from the EDAX of TiO2–SnO2 nanocomposites as shown in Figs. 6, 7 and 8 confirmed the elemental ratios of the prepared nanocomposites.
EDAX of TiO2–SnO2 (97:3) wt% nanocomposite.
EDAX of TiO2–SnO2 (90:10) wt% nanocomposite.
EDAX of TiO2–SnO2 (80:20) wt% nanocomposite.
FTIR of the prepared TiO2–SnO2 nanocomposites
As shown from Figs. 9, 10 and 11 the FTIR of TiO2–SnO2 nanocomposites shows that the peaks of TiO2 & SnO2 were slightly shifted due to the change in SnO2 loading percentage.
FTIR of TiO2–SnO2 (97:3) wt% nanocomposite.
FTIR of TiO2–SnO2 (90:10) wt% nanocomposite.
FTIR of TiO2–SnO2 (80:20) wt% nanocomposite.
XPS of the prepared TiO2–SnO2 nanocomposites
The analytical technique (XPS), commonly known as electron spectroscopy for chemical analysis (ESCA), that uses X-rays to bombard a specimen and then analyses the energy of the released electrons (Figs. 12 and 13).
XPS of TiO2–SnO2 (90:10) wt% nanocomposite calcined at 500 °C for 3 h (Survey spectra).
XPS of TiO2–SnO2 (90:10) wt% nanocomposite calcined at 500 °C for 3 h (O 1s region).
XPS of TiO2–SnO2 (90:10) wt% nanocomposite
Figures 14, 15, 16 and 17 shows the results of XPS analysis of the elements’ chemical make up and content. Peaks of O 1s, Ti 2p, Sn 3d, and C1s were seen in the survey spectra. Figure 14 in the region of 1200-0 eV, showing that Ti, O made up the majority of the samples. Figure 15 displays the spectrum of asymmetric TiO2-SnO2 (90:10) wt% with an O 1s core level. The spectrum was fitted by two Gaussian peaks at around 531.44 eV (Ti-O-Sn) and 528.75 eV. (Sn–O). Although there were still some Tin ions dispersed on the surface of TiO2, it was hypothesized that Ti-O-Sn bonds connecting the composite sample were generated it in the majority. This increased the oxygen concentration on the surface. It could be fitted into 6 peaks in the spectrum in Ti 2p region (Fig. 16) with energies of 464.15, 457.94, 471.54, 459.98, 462.91, and 464.69 eV. 464.15 and 457.94 eV peaks were attributed to Ti4+2p1/2 and Ti4+2p3/2, respectively. Figure 17 displays the doublet Sn 3d spectrum, with binding energies of 495.03 and 486.49 eV, respectively, for Sn3+3d3/2 and Sn3+3d5/2lines14.
XPS of TiO2–SnO2 (90:10) wt% nanocomposite calcined at 500 °C for 3 h (Ti 2p region).
XPS of TiO2–SnO2 (90:10) wt% nanocomposite calcined at 500 °C for 3 h (Sn 3d region).
The band gap energy of TiO2–SnO2 (97:3) wt% nanocomposite.
The band gap energy of TiO2–SnO2 (90:10) wt% nanocomposite.
Finally, from the previous Figs. 12, 13, 14 and 15 it was found that the catalysts might have more lattice defects. The presence of Sn3+, could make it easier for electrons to be captured. As a result, the likelihood of photoinduced electrons and holes recombining was decreased14.
Calculations of band gap energy of TiO2–SnO2nanocomposites
Calculation of band gap energy of TiO2–SnO2(97:3) wt% nanocomposite
It was found that, increasing the amount of SnO2 in TiO2–SnO2 nanocomposite causes a red-shift to longer wavelengths. This has to do with the development of new energy levels in TiO2band gap6,15. Oxygen vacancies are the principal source of the tiny absorption edges in the visible area16,17.
The UV-Vis analysis is applied to investigate and calculate the band gap energy by using Tauc Eq. (1).
where n = 1/2 for direct band gap materials and n = 2 for indirect band gap materials, α is absorption coefficient, h is the Planck constant, υ is the wavenumber, A is a constant, and Egis the band gap energy18. The band gap energy (Eg) value of TiO2–SnO2 (97:3) wt% is determined from the plot of (αhυ)2 against photon energy in electron volts. The derived band gap energy from Figs. 16, 17 and 18 was obtained from the slope of the line. The calculated band gap energy of TiO2–SnO2 (97:3) wt% nanccomposite was found to be 2.91 eV which was a less value compared with TiO2 (3.2 eV) and SnO2(3.6 eV)19.
Figure 17 depicts the band gap energy (Eg) of TiO2–SnO2 (90:10) wt% nanocomposite. The band gap energy (Eg) of TiO2–SnO2 (93:7) wt% is determined from the plot of (αhυ)2 against photon energy in electron volts which was found to be 2.8 eV when compared with TiO2 (3.2 eV) and SnO2 (3.6 eV). So, it is decreased with increasing the wt% of SnO2in the nanocomposite19.
Figure 18 depicts the band gap energy (Eg) of TiO2–SnO2 (80:20) wt% nanocomposite. The calculated band gap energy was found to be 2.86 eV. So, it is increased again with increasing the wt% of SnO2to 20 in the nanocomposite19.
The band gap energy of TiO2–SnO2 (80:20) wt% nanocomposite.
From the previous results on band gap calculation, it was found according to band gap values as shown in Table 2 that the prepared nanocomposites have lower band gap energies than native TiO2 and SnO2.
When SnO2 wt% increases from (3–10), the band gap decreases from 2.91 to 2.8 eV and then return to increase when SnO2 wt% increases from (15–20). This may be due to band gap energy of the coupled oxides should results from band gap overlap with the individual oxides. The band gap was found to shrink slightly as Sn concentration increased. Reportedly, SnO2 exhibits a narrow indirect band gap (3.4 eV) and a wide direct band gap (< 3.4 eV) (1.6 to 2.9 eV). The shift in the band gap energy with increasing the wt% of SnO2 is likely due to changes in crystal shifting on the plane, as indicated by XRD.
Also, doping of TiO2 with SnO2 creates Fermi level below the conduction band of the nanocomposite, so the band gap of TiO2–SnO2 nanocomposite decreases compared with native TiO2 and SnO2alone20.
Also, it was found that increasing the percentage of SnO2 from 3 to 10 wt% resulted in decreasing the Fermi level position and hence decreasing the band gap energy of TiO2-SnO2 nanocomposite from 2.91 to 2.8 eV. Further increasing in the wt% of SnO2 from 15 to 20 resulted in increasing the band gap from 2.82 to 2.86 eV respectively. This increase in the band gap energy is probably due to the formation of homojunction of SnO2–SnO2 on the expense of TiO2 -SnO2 nanocomposite. So, the prepared nanocomposites need lower energy for electron excitation from VB to CB than native TiO2 and SnO2.
It was concluded also that TiO2-SnO2 (90:10) wt% nanocomposite has the lowest band gap value (2.8 eV), so (90:10) wt% is the optimum ratio which means it needs lower energy for excitation as shown in Table 2.
Photocatalysis of acid red 37 dye
The photocatalytic degradation of aqueous acid Red 37 dye solution was studied with different AOPs using UV/TiO2, UV/SnO2, UV/TiO2–SnO2 (97:3), UV/TiO2–SnO2 (90:10), UV/TiO2–SnO2 (80:20), UV/TiO2–SnO2 (90:10)/H2O2, UV/TiO2–SnO2 (90:10)/S2O8 and UV/TiO2–SnO2 (90:10)/IO4−. In every experiment, the batch photoreactor was employed as the photoreactor.
Effect of native TiO2, SnO2 and TiO2–SnO2 nanocomposites as photocatalysts
Photodegradation was caused when UV light with a wavelength of 254 nm was used to irradiate slurry solutions of TiO2 and SnO2 (0.4 g L−1) containing acid red 37 dye. This can be represented as shown in Fig. 1as mentioned in the introduction1,21,22,23. A first order model for the degradation of dye was used to visualize the data. The slopes of the straight lines on plots of ln (C/Co) over time, where Co represents the starting concentration of acid red 37 dye and C is the dye concentration at time t, were used to calculate the values of the first-order rate constant (kapp). The time necessary for the reactants to deteriorate to half of their starting concentrations is known as the half-life time (t1/2) of the pseudo first order reaction. t1/2 and kapp are inversely proportional as shown by Eq. (2).
It was discovered that increasing the apparent rate constant (kapp) causes the t1/2 to decrease, as indicated in Table 3.
Irradiation of SnO2 (0.4 g L−1) with UV light of wavelength 254 nm resulted in a lower photodegradation of acid red 37 dye compared with UV/TiO2. But SnO2 exhibited good catalytic performance and stability. The superior photocatalytic activity of TiO2 over SnO2 may be attributed to smaller TiO2 particle size, quick e−/h+ pair recombination rate and poor quantum yield of SnO2in aqueous solutions24.
According to Fig. 19, the synthesized TiO2–SnO2 nanocomposite exhibits a higher photocatalytic activity compared with native TiO2 or SnO2. TiO2–SnO2 composite’s high photocatalytic activity can be attributed to the more effective separation of photoinduced electron–hole (e−/h+) pairs; specifically, the n-p heterojunction of TiO2–SnO2 causes the photogenerated holes to migrate toward the interface and the electrons to migrate toward the bulk.
Because the conduction band of SnO2 CBSnO2 is more positive than that of the conduction band of TiO2 CBTiO2, the excited electrons on TiO2 can also move to SnO2 whereas the photogenerated holes can also move to the valance band of SnO₂ loading VBSnO2 to the conduction band of TiO2 VBTiO2 (e−/h+) pairs separation as shown in Fig. 20. In comparison with native TiO2 or SnO2, TiO2–SnO2 nanocomposite demonstrated much better photocatalytic activity. Degradation of acid red 37 dye via photocatalysis depends significantly on the content of SnO2 present in TiO2–SnO2 nanocomposite. It was discovered that increasing the quantity of SnO2 from 3 to 10 wt% led to a stronger photocatalytic degradation of the dye. This may be due to decreasing in the band gap as shown in band gap calculation to a certain limit at TiO2–SnO2 (90:10) wt% nanocomposite, which is the lowest band gap that avoid the recombination and decreases the time needed to transfer the electrons and holes between CB and VB which gives the highest photocatalytic activity. The decreasing in photocatalytic degradation of the dye with increasing SnO2 content from 10 to 20 wt% may be due to the formation of SnO2–SnO2 homojunctions which decrease the number of TiO2–SnO2 heterojunctions and also increase the number of free SnO2particles, which possess low photocatalytic activity25.
Change of ln (C/Co) with time for photocatalytic degradation of acid red 37 dye using UV/TiO2, UV/SnO2, UV/TiO2–SnO2 (97:3), UV/TiO2–SnO2 (93:7), UV/TiO2–SnO2 (90:10), UV/TiO2–SnO2 (85:15) and UV/TiO2–SnO2 (80:20).
Relative energy position of the bands and the process of charge separation in theTiO2/SnO2 system.
Effect of addition of various inorganic oxidants to TiO2–SnO2 (90:10) nanocomposite
Effect of addition of H2O2 to TiO2–SnO2 (90:10) wt% nanocomposite
TiO2–SnO2 (90: 10) wt% nanocomposite with SnO2 (10 wt%) was utilized to explore the influence of several oxidants (H2O2, Na2S2O8 and NaIO4) on the photocatalytic breakdown of acid red 37 dye based on earlier findings. Figure 21 depicts the effect of adding different concentrations of hydrogen peroxide to TiO2–SnO2 (90:10) (0.4 g L−1) for photodegradation of acid red 37 dye under UV irradiation21. The data was plotted using a first-order dye destruction model. The addition low concentration of hydrogen peroxide (2 × 10−2 M) to UV/TiO2-SnO2 (90:10) wt% (0.4 g L−1) increases the rate of photocatalytic degradation of acid red 37 dye when compared with UV/TiO2-SnO2 (90:10) wt% alone. The rate of photodegradation increased when the concentration of hydrogen peroxide was increased to 6 × 10−2 M with kapp ranged from 789 × 10−4− 997 × 10−4 min−1, then decreased with further increasing of hydrogen peroxide concentration to 8 × 10−2 M with kapp of 698 × 10−2 M (Table 1).
A power law relationship describes the effect of rate-determining species:
where kapp and K are the apparent and true rate constants, respectively. Plotting the logarithm of kapp against the logarithm of the concentration of hydrogen peroxide in Fig. 22 produced a value of 2.3 for the exponent, n, the order of reaction with regard to the oxidant species (Table 1). According to the following reaction, the photogenerated conduction band electrons of TiO2–SnO2 are more efficiently trapped by H2O2 than they are by O2, which may explain why the rate of dye degradation is increased when using UV/TiO2–SnO2 (90: 10) wt%/H2O2 system as compared with UV/TiO2–SnO2(90:10) wt% alone22,23:
Change of ln (C/Co) with time for photocatalytic degradation of acid red 37 dye using 0.4 g/L of TiO2–SnO2 (90: 10) wt% with different concentrations of H2O2 (M).
Change of ln kapp (min−1) with ln [H2O2] (M) for photocatalytic degradation of acid red 37 dye.
Additional oxidizing species (·OH), can aid in the oxidative decay process as a whole, besides the scavenging action which slowing the recombination interaction between photogenerated carriers, (electrons and holes). Since the employed UV source’s main output was at 254 nm, significant photolysis of the H2O2would have taken place, creating additional hydroxyl radicals25,26. Thus,
By raising the H2O2 concentrations (2 × 10−2–6 × 10−2 M), dye oxidation and degradation are accelerated as well as the electron scavenging. Further increasing in H2O2 concentration to 8 × 10−2 M decreases the photocatalytic degradation rate due to the recombination of the resulting of OH radicals which occurs at high concentration of H2O226.
Effect of addition of S2O82− to TiO2–SnO2 (90:10) wt% nanocomposite
The oxygen on the surface of irradiated TiO2–SnO2 (90: 10) wt% suspension acts as a natural sink for the photogenerated conduction band electrons. The sorbed H2O or hydroxyl ions on TiO2–SnO2surface are subsequently oxidized by the remaining holes to produce the OH radicals. It could be advantageous to use an electron acceptor other than oxygen that is more effective27,28,29.
The impact of persulfate addition to TiO2–SnO2 (90:10) wt% (0.4 g L−1) on the photocatalytic degradation of acid red 37 dye is shown in Fig. 23. The rate of deterioration using TiO2–SnO2 (90:10) (0.4 g L−1) was found to be accelerated by addition of a little amount of persulfate (1.0 × 10−3 M). The dye degraded completely and quickly when the persulfate concentration was increased to 10 × 10−3 M (2.9 min). Figure 24 shows the reaction rate order with regard to persulfate, and it was determined to be 0.5 (Table 4). The persulfate anions may capture the photogenerated conduction band electrons of TiO2–SnO2 more than O2 or H2O2 and produce the powerful oxidizer SO4⋅−30.
Additionally at a wavelength of 254 nm, the sulphate radical anion can interact with the solvent and produces ·OH in the following processes6,31,32:
As a result, increasing the persulfate concentrations (1 × 10−3–10 × 10−3 M) improved both the trapping of photogenerated conduction band electrons and the production of SO4·− and ⋅OH which led to higher rate of photocatalytic degradation.
Change of ln (C/Co) with time for photocatalytic degradation of acid red 37 dye using 0.4 g/L of TiO2–SnO2 (90: 10) wt% with different concentrations of S2O82− (M).
Change of ln kapp (min−1) with ln [S2O82−] (M) for photocatalytic degradation of acid red 37 dye.
Effect of addition of IO4− to TiO2–SnO2 (90:10) wt% nanocomposite
The effect of addition of periodate to TiO2–SnO2 (90:10) wt% (0.4 g L−1) for photocatalytic degradation of the studied acid red 37 dye is shown in Fig. 25. It can be seen that the addition of very low concentration of periodate (1 × 10−4 M) to UV/TiO2–SnO2 (90:10) wt% (0.4 g L−1) resulted in higher photocatalytic degradation rate compared with UV/TiO2–SnO2 (90–10) wt% (0.4 g L−1) system alone. Also, increasing the periodate concentration to 1 × 10−3 resulted in higher photocatalytic degradation of the dye compared with UV/TiO2–SnO2 (90:10) wt% (0.4 g L−1)/H2O2 or UV/TiO2–SnO2 (90:10) wt% (0.4 g L−1)/S2O82− systems, indicating the effectiveness of periodate over peroxide or persulfate.
The reaction rate order with respect to periodate was found to be 2.9 (Fig. 26; Table 4). The enhancement of the dye degradation may be due to the scavenging of the photogenerated conduction band electrons of the excited TiO2–SnO2 by periodate which is more efficient than trapping with O2, H2O2 or S2O82−as follows7:
In addition, under UV irradiation (254 nm), a variety of highly reactive radical- and non-radical intermediates (IO3⋅, ⋅OH, and IO4⋅) are produced during the photolytic degradation of periodate which enhanced the degradation rate8:
Change of ln (C/Co) with time for photocatalytic degradation of acid red 37 dye using 0.4 g/L of TiO2–SnO2 (90:10) wt% with different concentrations of IO4− (M).
Change of ln kapp (min−1) with ln [IO4−] (M) for photocatalytic degradation of acid red 37 dye.
The formation of IO3⋅, O⋅−, ⋅OH, and IO4⋅ as well as the trapping of the photogenerated conduction band electrons of TiO2 and SnO2 (90:10) wt% are both enhanced by an increase in periodate concentration.
Qamar et al.33 have shown that the degradation of chrysoidine R and Acid Red 29 dyes was hastened when peroxide or periodate was added to UV light containing TiO2. It was discovered that the response rate followed the sequence:
-
UV/TiO2/S2O82− > UV/TiO2/H2O2 > UV/TiO2.
Similarly, Qamar et al.34 observed higher rates of chromotrope 2B and amido black 10 B photocatalytic degradation in the presence of H2O2, KBrO3, and (NH4)2S2O8. The pace of deterioration was about:
-
UV/TiO2/BrO3− > UV/TiO2/S2O82− > UV/TiO2/H2O2 > UV/TiO2.
Finally, we can showed the total results that for three addition of inorganic antioxidants H2O2,SO4− and IO4− in shown Table 4 and compare rate of reaction with TiO2 and SnO2 alone then after addition inorganic antioxidant.
Materials and methods
Artificial polluted wastewater preparation
An aqueous solution of Acid Red 37 dye (1.04 × 10–4 M) were prepared in double distilled water as a model for wastewater pollutant. The pH of TiO2–SnO2/dye suspension was 6.1. Addition of H2O2, NaIO4 or Na2S2O8 to TiO2–SnO2/dye suspension decreases the pH below 6.1. A few drops of an alkali were added to adjust the pH to its original value 6.1.
Preparation of TiO2–SnO2 nanocomposites
TiO2–SnO2 nanocomposites were prepared through a solid-state reaction route. The starting material was TiO2 (99.58%). 3, 7, 10, 15 and 20 wt% of SnO2 powder have been added to TiO2. The powders have been mixed uniformly by grinding in mortar with pestle for 3 h to get fine powders. The resultant powders have been annealed in air at 500 °C for 3 h in an electric muffle furnace35.
Schematic diagram of batch photoreactor
The reactor used for studying the photodegradation of the dye in all experiments was a batch photoreactor as shown Fig. 27. To increase solution fluidization and access oxygen for Eq. (13)
air was blown into the reaction solution using an air pump at a flow rate of 10 m3/h. Blowing cooled air into the solution eliminated the lamp’s heat effect and kept the temperature at around 25 °C36. It is consists of a glass container (250 ml). The contents (TiO2/dye, SnO2/dye, TiO2–SnO2/dye, or TiO2–SnO2/dye/oxidant) of the container were agitated by a magnetic stirrer and kept purged with air at a rate of 3000 ml min−1. The dye solution was stirred with TiO2–SnO2 nanocomposite in the dark for 30 min before irradiation with UV to obtain equilibrium adsorption. Irradiation was carried out with a tubular low pressure mercury lamp (total rating 43 W, total UV output at 513 nm 13.4 W, and length 120 cm, Voltarc Tubes Inc., USA) was located 10 cm from the surface of the dye solution. The total intensity reaching the slurry solution was measured using a UVX radiometer (UV Products Ltd., Cambridge) equipped with a sensor with peak sensitivity at 254 nm was 4 mW cm−2. Samples of dye solution (1.0 × 10−4 mol/L) which prepared in distilled water as a model for wastewater pollutants were removed from the container via a sample port periodically and measured after filtration through 0.2 μm polyether sulfone membrane. All photodegradation experiments were done in a temperature of 22 ± 2 °C.
Schematic diagram of the batch photoreactor.
Measurement of photocatalytic activity
The photocatalytic performance of native TiO2, SnO2 nanoparticles and TiO2–SnO2nanocomposites was investigated by the decomposition of acid red 37 dye in an aqueous solution under UV light (254 nm). Absorbance was readily described by Beer-Lambert law37
Where A is absorbance, ε is the molar extinction coefficient, L is the light path length (1.00 cm) and C is the concentration. The absorbance which given at each irradiation time was related to a specific dye concentration using a calibration curve for acid red 37 dye solutions with different concentrations. The percent of degradation of the dye could be calculated according to equation (15) :
Where Co = initial concentration of dye solution, C = concentration of dye solution after photoirradiation at time t.
Conclusions
The photocatalytic degradation of the investigated acid red 37 dye using TiO2 nanoparticles was found to be higher than that of SnO2. The photocatalytic activity results showed that the couple oxide TiO2–SnO2 exhibited much higher photocatalytic activity than the native SnO2 and native TiO2.Heterogeneous photocatalytic degradation of the investigated acid red 37 dye using TiO2–SnO2 nanocomposite with different wt% (3–20) indicated that: UV/TiO2–SnO2 (90:10) wt% performing the higher photocatalytic degradation of the dye than other ratios. Heterogeneous photocatalytic degradation using UV/TiO2–SnO2 (90:10)/Oxidant (H2O2, S2O82−, or IO4−) systems was found to be more effective than UV/SnO2 or UV/TiO2 and follows the following order:
-
UV/SnO2 < UV/TiO2 < TiO2–SnO2 (90:10) < UV/TiO2–SnO2 (90:10)/H2O2 < UV/TiO2–SnO2 (90:10)/Na2S2O8 < UV/TiO2–SnO2 (90:10)/NaIO4.
Data availability
All data generated or analyzed during this study are included in this article.
References
Kadem, A. J., Tan, Z. M., Suntharam, N. M., Pung, S. Y. & Ramakrishnan, S. Synthesis of CuO, ZnO and SnO2 coupled TiO2 photocatalyst particles for enhanced photodegradation of rhodamine B dye. Bull. Chem. React. Eng. Catal. 18(3), 506–520 (2023).
.Raub, A. A. M. et al. Photocatalytic activity enhancement of nanostructured metal-oxides photocatalyst: a review. Nanotechnology 1, 1 (2024).
Fatimah, I. et al. Ultrasound-assisted phyto-mediated synthesis of SnO2 nanoparticles as photocatalyst in tetracycline photocatalytic oxidation. Inorg. Chem. Commun. 1, 112096 (2024).
Yang, L. R. et al. Functionalizing slag woolfibers with photocatalytic activity by anatase TiO2 and CTAB modification. Ceram. Int. 44, 5842–5847 (2018).
Gandolfo, A. et al. Unexpectedly high levels of organic compounds released by indoor photocatalytic paints. Environ. Sci. Technol. 52, 328–11337 (2018).
Panwar, S., Upadhyay, G. K. & Purohit, L. P. Gd-doped ZnO: TiO2 heterogenous nanocomposites for advance oxidation process. Mater. Res. Bull. 145, 111534 (2022).
Obotey Ezugbe, E. & Rathilal, S. Membrane technologies in wastewater treatment: a review. Membranes 10(5), 89 (2020).
Fernandes, A., Makoś, P., Wang, Z. & Boczkaj, G. Synergistic effect of TiO2 photocatalytic advanced oxidation processes in the treatment of refinery effluents. Chem. Eng. J. 391, 123488 (2020).
Kumar, P. S. S., Raj, M. R. & Anandan, S. Nanoporous Au–TiMCM-41—An inorganic hybrid photocatalyst toward visible photooxidation of methyl orange. Sol. Energy Mater. Sol. Cells 94(10), 1783–1789 (2010).
Vinu, R., Akki, S. U. & Madras, G. Investigation of dye functional group on the photocatalytic degradation of dyes by nano-TiO2. J. Hazard. Mater. 176(1–3), 765–773 (2010).
Sadik, W. A., El-Demerdash, A. G. M., Nashed, A. W., Mostafa, A. A. & Hamad, H. A. Highly efficient photocatalytic performance of Cu2O@ TiO2 nanocomposite: influence of various inorganic oxidants and inorganic anions. J. Mater. Res. Technol. 8(6), 5405–5414 (2019).
Wang, H. et al. Engineering of SnO2/TiO2 heterojunction compact interface with efficient charge transfer pathway for photocatalytic hydrogen evolution. Chin. Chem. Lett. 34(1), 107125 (2023).
Thanh, N. T., Maclean, N. & Mahiddine, S. Mechanisms of nucleation and growth of nanoparticles in solution. Chem. Rev. 114(15), 7610–7630 (2014).
Kaur, N. & Singh, V. Current status and future challenges in ionic liquids, functionalized ionic liquids and deep eutectic solvent-mediated synthesis of nanostructured TiO2: a review. N. J. Chem. 41, 2844–2868 (2017).
Wang, Z. et al. Rapid preparation of terbium-doped titanium dioxide nanoparticles and their enhanced photocatalytic performance. R. Soc. Open. Sci. 6(10), 191077 (2019).
Tangale, N. P. et al. Synthesis of Sn-containing anatase (TiO2) by sol-gel method and their performance in catalytic water splitting under visible light as a function of tin content. Mater. Lett. 171, 50–54 (2016).
Molaei, M. J. Principles, mechanism, and identification of S-scheme heterojunction for photocatalysis: a critical review. J. Am. Ceram. Soc. 1, 1 (2024).
Suriya, P., Prabhu, M., Ezhilselvi, V. & Jagannathan, K. Improvement of power conversion efficiency by tailoring of energy band gaps in Ag doped TiO2–SnO2 nanocomposites. Phys. B Condens. Matter 670, 415359 (2023).
Saleh, T. A., Mustaqeem, M. & Khaled, M. Water treatment technologies in removing heavy metal ions from wastewater: a review. Environ. Nanatechnol. Monit. Manag. 17, 100617 (2022).
Mannaa, M. A., Hassan, S. M. & Ahmed, A. I. Enhancement the photocatalytic activity of the SnO2/TiO2 nanocrystals under UV-visible light. Int. J. Mod. Chem. 9(1), 84–92 (2017).
Rajput, R. B., Jamble, S. N. & Kale, R. B. A review on TiO2/SnO2 heterostructures as a photocatalyst for the degradation of dyes and organic pollutants. J. Environ. Manag. 307, 114533 (2022).
Mousa, S. A., Abdallah, H., Ibrahim, S. S. & Khairy, S. A. Enhanced photocatalytic properties of graphene oxide/polyvinylchloride membranes by incorporation with green prepared SnO2 and TiO2 nanocomposite for water treatment. Appl. Phys. A 129(12), 831 (2023).
Hussein, F. M., Alani, R. R. & AL-MOKARAM, A. L. I. Synthesis and photocatalytic activity of TiO2-coupled SnO2 nanoparticles prepared by sol–gel technique. Egypt. J. Chem. 65(10), 551–559 (2022).
Enesca, A. Enhancing the photocatalytic activity of SnO2-TiO2 and ZnO-TiO2 tandem structures toward indoor air decontamination. Front. Chem. 8, 583270 (2020).
Messaadi, C. et al. Synthesis and characterization of SnO2-TiO2 nanocomposites photocatalysts. Curr. Nanosci. 15(4), 398–406 (2019).
Negishia, N. et al. Photocatalytic detoxification of aqueous organophosphorus by TiO2 immobilized silicagel. Appl. Catal. B 128, 105–118 (2012).
Zhao, X., Liu, X., Yu, M. M., Wang, C. & Li, J. The highly efficient and stable Cu, Co, Zn-porphyrinTiO2 photocatalysts with heterojunction by using fashioned one-step method. Dyes Pigm. 138, 648–656 (2017).
Gomeza, S., Marchenaa, C. L., Pizziob, L. & Pierella, L. Preparation and characterization of TiO2/HZSM-11 zeolite for photodegradation of dichlorvos in aqueous solution. J. Hazard. Mater. 258–259, 19–26 (2013).
Kuo, C. S., Lin, C. F. & Hong, P. K. Photocatalytic degradation of methamphetamine by UV/TiO2—kinetics, intermediates, and products. Water Res. 74, 1–9 (2015).
Putluru, S. R. & Schill, L. H. Mn/TiO2 and Mn-Fe/TiO2 catalysts synthesized by deposition precipitation—promising for selective catalytic reduction of NO with NH3 at low temperatures. Appl. Catal. B 165, 628–635 (2015).
Nam, Y., Li, L., Lee, J. Y. & Prezhdo, O. V. Size and shape effects on charge dynamics of TiO2 nanoclusters. J. Phys. Chem. C122, 5201–5208 (2018).
Kaur, N. & Singh, V. Current status and future challenges in ionic liquids, functionalized ionic liquids and deep eutectic solvent-mediated synthesis of nanostructured TiO2: a review. N. J. Chem. 41, 2844–2868 (2017).
Alosfur, F. K. M., Ridha, N. J., Jumali, M. H. H. & Radiman, S. One-step formation of TiO2 hollow spheres via a facile microwave-assisted process for photocatalytic activity. Nanotechnology 29(14), 145707 (2018).
Verma, A., Prakash, N. T. & Toor, A. P. An efficient TiO2 coated immobilized system for the degradation studies of herbicide isoproturon: Durability studies. Chemosphere 109, 7–13 (2014).
Hamad, H. et al. The superior photocatalytic performance and DFT insights of S-scheme CuO@ TiO2 heterojunction composites for simultaneous degradation of organics. Sci. Rep. 12(1), 2217 (2022).
Abdollahi, Y., Abdullah, A. H., Zainal, Z. & Yusof, N. A. Photodegradation of m-cresol by zinc oxide under visible-light irradiation. Int. J. Chem. 3(3), 31 (2011).
Gomez, S., Marchena, C. L., Pizzio, L. & Pierella, L. Preparation and characterization of TiO2/HZSM-11 zeolite for photodegradation of dichlorvos in aqueous solution. J. Hazard. Mater. 258, 19–26 (2013).
Acknowledgements
Thanks for Journal , I hope publish in our journal.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Wagih Sadik is head of the research group.Abdelghaffar Eldemerdash , Adel William Nashed, Elsayed Lamie is Investigated this paper.Amr Ahmed Mostafa is worked Methodology, investigation, formal analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sadik, W., M. El-Demerdash, A., Nashed, A.W. et al. Synthesis and investigation of optical properties and enhancement photocatalytic activity of TiO2–SnO2 semiconductor for degradation of organic compounds. Sci Rep 14, 27846 (2024). https://doi.org/10.1038/s41598-024-78755-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-78755-y
Keywords
This article is cited by
-
A comparative study on the effect of thickness in sol–gel deposited ZnO, SnO₂, and TiO₂ thin films and their photocatalytic performance in methylene blue decolorization
Journal of Sol-Gel Science and Technology (2026)
-
Enhanced rates photocatalytic degradation of acid red 37 dye by TiO2–SnO2 in solid state under UV-irradiation light
Discover Nano (2025)
-
SnO₂@ZIF-8 Core–shell structured nanocrystallites: Structural and Electrochemical investigation
Journal of Nanoparticle Research (2025)