Abstract
Deep brain stimulation is a well-established treatment for improving motor symptoms in Parkinson’s disease. However, persistent non-motor symptoms, such as excessive daytime sleepiness, remain a significant challenge and necessitate further investigation. In this study, we conducted repeated measurements of daytime sleepiness using a modified multiple sleep latency test in a healthy monkey (macaca fascicularis), which was later rendered parkinsonian through MPTP administration. Deep brain stimulation targeting the lateral hypothalamic area revealed frequency-dependent modulation of both sleepiness level and core body temperature. High-frequency stimulation (80 Hz) increased sleepiness in the healthy state, while low-frequency stimulation (20 Hz) promoted wakefulness in the parkinsonian state. These findings suggest a promising therapeutic approach for addressing sleep/wake disturbances, not only in Parkinson’s disease but also in other severe sleep disorders.
Similar content being viewed by others
Introduction
Excessive daytime sleepiness (EDS) poses a significant clinical challenge, characterized by an increase need for sleep and intrusive sleep episodes, severely impacting daily functioning and overall well-being1,2,3,4. Its prevalence notably intensifies in various neurodegenerative disorders, including Parkinson’s disease5,6. However, evidence for effective treatment options remains limited, deserving further investigation.
Deep brain stimulation (DBS), a proven neuromodulation therapy recognized for its efficacy in managing motor symptoms of Parkinson’s disease7, has also shown promise in targeting additional neural circuits to alleviate other symptoms. Our previous research demonstrated the capacity of DBS to regulate sleep/wake behavior, particularly through low-frequency stimulation (LFS) of the pedunculopontine nucleus, which improved EDS in parkinsonian monkeys8. Given the challenges of targeting the pedunculopontine nucleus—primarily due to its deep location, small size, and proximity to critical brainstem regions— we shifted our focus to a more accessible yet equally important structure involved in the regulation of sleep and wakefulness, the lateral hypothalamic area (LHA).
The LHA is a heterogeneous structure housing spatially intermingled but distinct cell populations crucial for controlling sleep/wake behavior and energy metabolism. In particular, the orexin (Ox), alternatively called hypocretin (Hcrt), neurons within the LHA play a pivotal role in arousal stabilization9,10 and core body temperature regulation11,12. Recent findings in mice exhibiting narcoleptic symptoms revealed that 15 Hz DBS of the LHA facilitated wakefulness consolidation13. Interestingly, postmortem analysis of brains from patients with advanced Parkinson’s disease, who can experience severe EDS, abnormal temperature sensations, and sweating, revealed a significant reduction in the number of Ox/Hcrt neurons within the LHA14.
Given the role of the LHA in energy metabolism regulation and arousal, DBS targeting this region holds promise for mitigating non-motor symptoms in Parkinson’s disease. In this study, we investigated the neuromodulatory effects of LHA-DBS on EDS and core body temperature in a Parkinson’s disease monkey model, known to exhibit EDS symptom15. Our findings demonstrate frequency-dependent control of wakefulness and core body temperature by LHA-DBS, offering valuable insights for advancing research and developing innovative therapeutics for sleep/wake disturbances.
Results
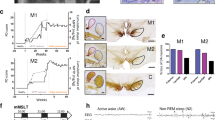
We implanted a monkey with a quadrupole electrode at the level of Ox/Hcrt neurons of the LHA, confirmed through postmortem immunohistology (Fig. 1a&b), which was connected to subcutaneous device, allowing continuous high (HFS, 80 Hz) and low-frequency stimulation (LFS, 20 Hz). The parkinsonian state was established through the chronic administration of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), which led to stable onset of motor symptoms (parkinsonian score: 14.6 ± 0.2, corresponding to bradykinesia, postural disorder, and decreased general activity) during 8 consecutive weeks (see methods section). Dopaminergic depletion was confirmed through postmortem immunohistology, which revealed markedly decreased tyrosine hydroxylase expression in the striatum and substantia nigra (Fig. 1c).
Sleepiness level was evaluated by performing a minimum of five modified multiple sleep latency tests (mMSLT), each consisting of three consecutive 20-minute light-OFF sessions conducted in the morning at 10:00, 11:00, and 12:00 (lights were turned ON between sessions). The animal was transferred to the behavioral cage at 9:20 to allow sufficient time for habituation. The mMSLT protocol was performed under three conditions: stim-OFF, LFS-ON, and HFS-ON, in both healthy and parkinsonian states (Fig. 1d). Electrodes coupled with a radiotelemetry transmitter, enabled the recording of EEG, EOG and EMG signals in freely moving conditions, allowing for the detection of wake and sleep stages. During the mMSLT protocol, the observed stages were limited to active wake, quiet wake, and light sleep stages 1 and 2 (Fig. 1e).
Surgery report and parkinsonian model. (a), X-ray of the final implantation of the electrode in the LHA (left), in both coronal and sagittal views with the internal landmarks AC-PC line determined via ventriculography (LV: lateral ventricle). (b), Cryostat photography of a coronal plane showing the trace and reconstruction of the electrode in the lateral hypothalamic area at -3 mm from AC at the level of OxA/Hcrt1 neurons. The framed image highlights the immunostaining of OxA/Hcrt1 neurons (scale bar = 100 μm). (c), Tyrosine hydroxylase (TH) immunostaining at the level of the striatum (scale bar = 2000 μm) and the substantia nigra (scale bar = 1000 μm) for a control animal and MPTP-treated animal, associated with graph showing the percent loss of TH staining in the caudate (Cd), the putamen (Pu) and the substantia nigra compacta (SNc) compared with that in a control animal. d, Design of the modified multiple sleep latency test (mMSLT) protocol with 3 consecutive 20-minute light-OFF sessions at 10:00 [1], 11:00 [2] and 12:00 [3], performed in the behavioral cage and under different conditions: stim-OFF or continuous LHA-DBS; LFS-ON (20 Hz) or HFS-ON (80 Hz). e, Example of 30 s epochs showing EEG, EOG and EMG signals during wake and sleep stages observed in this study; wake states were: active wake and quiet wake and sleep states were: light sleep stage 1 and stage 2. AC-PC line: Anterior Commissure-Posterior Commissure line, LV: Lateral Ventricle, Pu: Putamen, Cd: Caudate, SNc: Substantia nigra compacta.
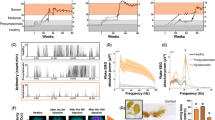
In the healthy state, the monkey spontaneously fell asleep in 20% of the light-OFF sessions in the stim-OFF condition; the incidence of at least one sleep episode per session did not significantly differ under the LFS condition (25%) and significantly increased under the HFS condition (83%) (Fig. 2a). In the stim-OFF condition, these sleep episodes occurred at a mean latency of 19.6 ± 0.2 min (all light-OFF sessions pooled) and was relatively consistent among the three successive lights-OFF sessions. In the LFS condition, sleep latency did not significantly differ from that in the stim-OFF condition. However, a gradual decrease in sleep latency was observed under the HFS condition, which significantly differed from that in the stim-OFF condition for the light-OFF sessions 2 and 3 (HFS//light-OFF session [1]: 16.6 ± 1.5 min, p = 0.0807; [2]: 11.4 ± 2.1 min, p = 0.0121; [3]: 9.7 ± 1.3 min, p = 0.0106) (Fig. 2b). In both the stim-OFF and LFS conditions, sleep episodes were essentially nonexistent and lasted only a few seconds after sleep onset (stim-OFF//light-OFF session [1]: 0.1 ± 0.1 min; [2]: 0.1 ± 0.1 min; [3]: 0.2 ± 0.1 min). In contrast, the duration of these sleep episodes increased in the HFS condition, and the sleep duration significantly differed from that in the stim-OFF condition for light-OFF session 3 (HFS//light-OFF session [1]: 0.6 ± 0.3 min, p = 0.3278; [2]: 1.6 ± 0.5 min, p = 0.1944; [3]: 2.3 ± 0.4 min, p = 0.0444) (Fig. 2c). In the healthy state, over the total 3 h period of the mMSLT procedure, mean core body temperature of the monkey was 37.7 ± 0.1 °C in the stim-OFF condition, increased to 38.1 ± 0.3 °C in the LFS condition, and decreased to 37.3 °C ± 0.1 °C in the HFS condition (Fig. 2d). In both LFS and HFS conditions, core body temperature changed gradually over time (Fig. 2e), but was significantly different from the stim-OFF condition in the last 5 min of the experiments in LFS-condition, while no difference was observed in the HFS condition (Fig. 2f).
In the parkinsonian state and stim-OFF condition, the monkey exhibited severe sleepiness, characterized by a higher incidence of at least one sleep episode per session (100%), faster sleep latency (stim-OFF//light-OFF session [1]: 11.0 ± 2.3 min; [2]: 8.4 ± 2.0 min; [3]: 10.5 ± 2.8 min; mean: 10.0 ± 1.3 min), and longer sleep durations than those in the healthy state (stim-OFF//light-OFF session [1]: 6.0 ± 1.6 min; [2]: 5.1 ± 1.1 min; [3]: 8.0 ± 1.7 min). The incidence of at least one sleep episode decreased significantly from 100% in the stim-OFF condition to 66% in the LFS condition but was essentially similar in the HFS condition (83%) (Fig. 2g). In the LFS condition, sleep latency progressively increased, with the increase reaching significance in light-OFF session 3 compared to the stim-OFF condition (LFS//light-OFF session [1]: 10.5 ± 1.5 min, p = 0.0998; [2]: 15.5 ± 1.3 min, p = 0.0643; [3]: 18.8 ± 0.8 min, p = 0.0422) (Fig. 2h). Furthermore, sleep duration progressively decreased in the LFS condition compared to the stim-OFF condition (LFS//light-OFF session [1]: 2.6 ± 0.4 min, p = 0.5232; [2]: 1.5 ± 1.0 min, p = 0.2522; [3]: 0.9 ± 0.9 min, p = 0.0012) (Fig. 2i). In the HFS condition, sleep latency and sleep duration were not significantly different from those in the stim-OFF condition. Compared to the healthy state (37.7 ± 0.1 °C), the monkey exhibited a slight decrease in core body temperature in the parkinsonian state (37.4 ± 0.1 °C); then core body temperature increased under LFS condition to 37.7 ± 0.1 °C and tended to decrease in the HFS condition to 37.3 ± 0.1 °C (Fig. 2j). Core body temperature changed gradually over time in LHS condition (Fig. 2k), and was significantly different from the stim-OFF condition in the last 5 min of the experiments, whereas no such difference was observed in the HFS condition (Fig. 2l).
LHA-DBS at low- and high-frequency on sleep/wake behavior and core body temperature. (a, g), Occurrence of at least one sleep episode, expressed as a percentage, during all light-OFF sessions combined in stim-OFF (n = 18), LFS-ON (n = 15), and HFS-ON (n = 15) conditions for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange), ǂ different from stim-OFF condition: Fisher’s exact test. (b, h), Sleep latency, expressed in minutes ± SEM, for each light-OFF session: (1) from 10:00 to 10:20, (2) from 11:00 to 11:20, (3) from 12:00 to 12:20, in stim-OFF (n = 6; solid line), LFS-ON (n = 5; bold dotted line), and HFS-ON (n = 5; thin dotted line), for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange). c, i, Sleep duration, expressed in minutes ± SEM, for each light-OFF session: (1) from 10:00 to 10:20, (2) from 11:00 to 11:20, (3) from 12:00 to 12:20, in stim-OFF (n = 6; solid line), LFS-ON (n = 5; bold dotted line), and HFS-ON (n = 5; thin dotted line), for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange). (d, j), Mean core body temperature, expressed in °C ± SEM, during all light-OFF sessions combined in stim-OFF (n = 6), LFS-ON (n = 5), and HFS-ON conditions (n = 5), for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange). (e, k), Example of core body temperature over time, from 9:30 am to 12:30 pm, in stim-OFF, LFS-ON, and HFS-ON conditions, for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange). Black rectangles represent the light-OFF sessions. (f, l), Mean core body temperature ± SEM, expressed in °C, during the first 5 min and the last 5 min of the experiments in stim-OFF (n = 6), LFS-ON (n = 5), and HFS-ON conditions (n = 5), for the healthy state (indicated in gray) and the Parkinsonian state (indicated in orange). *Significance was assessed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test (p < 0.05). SEM: Standard Error of the Mean, Stim-OFF: Stimulation Off, LFS-ON: Low-Frequency Stimulation On, HFS-ON: High-Frequency Stimulation On.
Discussion
Although the mechanisms underlying the effects of DBS are not yet fully known, when interpreting our findings, we considered the knowledge that LFS has an excitatory effect, while HFS may cause depolarization blockade and relative inhibition of neurons16,17. Building on this existing knowledge and our findings, we applied these insights to the flip-flop switch model, as described by Saper et al.18, to explore the potential mechanisms by which stimulation frequencies modulate sleep/wake regulatory structures (Fig. 3).
Proposal of the mechanisms of lateral hypothalamic deep brain stimulation to modulate sleep and wakefulness, according to the flip-flop switch model of Saper and colleagues. The figure illustrates the functional interaction among LHA neurons, wake-active (W-A) centers, and sleep-active (S-A) centers in healthy (top panel) and parkinsonian (bottom panel) states. Arrows indicate excitatory input, while T-shape lines represent inhibitory input. The thickness of arrows and lines reflects the relative strength of input. Circle sizes denote the relative activities of each group of neurons where LHA sends excitatory input to W-A neurons. In the healthy state, robust homeostatic and circadian pressures maintain optimal wakefulness through the high activity of W-A neurons. In the parkinsonian state, dopamine depletion alters these pressures, leading to reduced W-A neurons activity and increased S-A neurons activity, which in turn causes increased sleepiness. In both conditions, LHA-DBS modulates these neural balances: low-frequency stimulation (LFS) activates the LHA to promote wakefulness, while high-frequency stimulation (HFS) inhibits LHA activity to promote sleep, leading to differential behavioral effects depending on the health state.
In the healthy state, a high level of arousal, which may already be present because of good health condition and the time of day (mMSLT experiments from 9:20 to 12:20), could prevent any awakening effect of LFS. Moreover, the increase in core body temperature may be due to the activation of Ox/Hcrt neurons, as it has already been shown in a rat study using a local microinjection of bicuculline19. Indeed, Ox/Hcrt neurons innervate the rostral raphe pallidus, which regulates body temperature20, especially brown adipose tissue thermogenesis21. In contrast, LHA-HFS induced an increase in sleepiness and a slight decrease in core body temperature. These effects, similar to those observed in a murine LHA lesion model, suggest that HFS inhibits Ox/Hcrt neurons, inducing sleep-promoting effects.
However, LHA-HFS did not significantly change sleep latency or sleep duration in the parkinsonian state compared to those observed in the healthy state. Indeed, LHA-HFS, which had a sleep-promoting effect in the healthy state, did not trigger more sleep episodes in the animal when its state was already characterized by a severe form of sleepiness. In contrast, the awakening effect of LHA-LFS, which we did not observe in the healthy state, became evident in the Parkinsonian state, as the animal had a low level of arousal. Therefore, further arousal is challenging to induce when this state has reached its maximum level (e.g. in the healthy state); similarly, additional sleep episodes are challenging to induce once a state of sleepiness is firmly established (e.g. in the parkinsonian state). These outcomes suggest that LHA neurons operate within the limits of endogenous thresholds, implying that optimal values cannot be surpassed.
Moreover, under both LHA-DBS conditions and both health conditions, the effects manifested progressively over time. Specifically, during the 3 h mMSLT, the intensity of these effects increased from the first to the last light-OFF session in each mMSLT. These trends suggest that a cascade of events is necessary to induce changes in sleep/wake states and core body temperature, especially when triggered at an inappropriate time of the day.
The combined evolution of the core body temperature and sleep/wake behavior is in agreement with physiological considerations. Indeed, a decrease in core body temperature is necessary for sleep onset and maintenance, whereas an increase in core body temperature, a characteristic of arousal22, could make sleep onset more difficult. It is challenging to determine whether changes in core body temperature influence sleep/wake behavior or vice versa; however, these phenomena are closely interconnected.
We recognize the limitations inherent in this single-case animal study, but the stability of the responses during repeated trials in each condition and the precision of the implantation guarantee sufficiently robust results. We did not include an additional animal for this pilot study to comply with the 3R rule and thus reduce the number of animals used. We also recognize the difficulty of translation to the clinic, particularly in this Parkinson’s disease context where such an implantation approach seems inappropriate. Nevertheless, our findings provide new insights into the effects of LHA-DBS on sleep/wake behavior and core body temperature in an animal model exhibiting EDS. This study revealed that LHA-DBS enables frequency-dependent changes in sleep/wake behavior and core body temperature, with LFS acting in favor of wake state and HFS acting in favor of sleep state. This highlights the circadian potential of LHA-DBS, LFS to promote wakefulness during the day and HFS to promote sleep at night. Further investigations into the long-term effects of LHA-DBS, including experiments conducted during both daytime and nighttime periods, could contribute to the development of novel treatments for sleep/wake disturbances associated with various sleep disorders—such as narcolepsy, central hypersomnia, and insomnia—as well as in many neurodegenerative diseases.
Methods
Animal
In accordance with the Grenoble Alpes University, Grenoble Institut of Neurosciences (B3851610008) policies and with French legislation, experiments were performed in compliance with the European Community Council Directive of 2010 (2010/63/UE) for care of laboratory animals and in accordance with ARRIVE guidelines. All procedures were reviewed and validated by the local etic committee n˚004 and were authorized by the Direction Départementale des Services Vétérinaires de l’Isère – Ministère de l’Agriculture et de la Pêche, France. This study was performed on one adult male macaque (Macaca fascicularis-CRP Port Louis, Mauritius), 10 kg and 10 years old. The monkey was housed with another monkey and kept under controlled conditions, 12-hour light/dark cycles [off at 19:00], 23 ± 2 °C, 50 ± 5% humidity, and had access ad libitum to food and water.
Apparatus
The monkey was chronically implanted with polysomnographic equipment, a radio-telemeter transmitter (L04, Data Science International, France) allowing recordings in freely moving condition. The transmitter included a three-channel biopotential device for recording electroencephalogram (EEG), electroocculogram (EOG), and electromyogram (EMG) signals at sampling rate of 500 Hz, along with a continuous core body temperature sensor. As it has already been done and described15,23, EEG was recorded using two electrodes screwed unilaterally (one frontal and one parietal 10 mm lateral to the midline at right) into the skull, EOG was acquired from two electrodes affixed at the level of the right orbital arch bone unilaterally (one at the top and one at the external side), EMG was monitored from two leads sutured into the right neck musculature at 10 mm apart, and finally core body temperature was recorded at the level of the transmitter implanted within the abdominal muscle layers. These signals were acquired via two receivers mounted in a behavioral cage and then forwarded to a data exchange matrix connected to a computer for data recording, storage and off-line analysis (Dataquest A.R.T., Data Sciences International, France). EEG and EOG was bandpass-filtered in the range of 0.3 to 35 Hz and EMG was bandpass-filtered in the range of 10 to 100 Hz. The monkey was also chronically implanted with a deep brain electrode within the LHA (quadrupole electrode: contact length 1.5 mm, spaced 0.5 mm apart, outer diameter of electrode 1.27 mm, Medtronic 3389) connected to a neurostimulator (Activa®PC + S, Medtronic, Minneapolis, USA) with a lead extension (37086, 40 cm, Medtronic, USA). The stimulator could be turned on and off using a pulse generator programmer (Medtronic, 8840 Programmer) by placing the transmitter against the implanted stimulator. The monkey was trained to this manipulation in their home cage without any sedation.
Surgery
The surgery was performed under aseptic conditions and general anaesthesia as previously described15. The neurostimulator was implanted in a subcutaneous pocket in the back, the lead extension was tunneled subcutaneously to the skull and the electrode was stereotactically implanted into the right LHA. For this purpose, a preoperative MRI (3D T1 sequence performed at Grenoble MRI facility IRMaGE) merged into the stereotaxic atlas of the macaque fascicularis24 allowed to define the coordinates of the target relative to internal anatomical landmarks: the reference line connecting the anterior commissure and the posterior commissure (AC-PC line). The target was determined at -3 mm from AC, at 3 mm from the midline, and 5 mm below the AC-PC line. Then, using ventriculographical X-ray control, using a cannula placed in the left lateral ventricular through which 2 ml of ventricular contrast (Iopamiron 200, iodine 200 mg/ml, Bracc) was injected, the electrode could be anchored in situ with acrylic dental and then connected to the stimulator. Analgesic/anti-inflammatory therapies (Ketoprofen, 2 mg/kg i.m.) and antibiotic (Clamoxyl, 20 mg/kg i.m.) were provided during the one-week post-operative period. The monkey was weighed, in its home cage without sedation, weekly before and after the surgery to detect any abnormal changes due to the surgery or DBS protocol. A notable reduction in feeding leading to weight loss was observed during the first postoperative week (10.7 ± 0.03 kg vs. 10.18 ± 0.06 kg, p = 0.002). However, a subsequent progressive recovery indicates that this effect was temporary, and feeding behavior remained essentially unaffected by the mechanical implantation of the electrode nor the DBS protocol. These findings align with certain studies exploring LHA-DBS for weight management, which have shown varied and inconsistent results25,26,27.
LHA stimulation setting
LHA-DBS was applied unilaterally with parameters selected from stimulation ranges carried out on awake animal, from 5 Hz to 130 Hz for each contact of the electrode. The two deepest contacts were considered as the optimal contacts within the LHA through examination of ventriculography. Saccadic eye movements were consistently observed when stimulating these contacts at all frequencies, with voltages greater than 3.8 V. However, discerning optimal stimulation frequencies based on additional behavioral changes proved elusive. Consequently, frequency parameters were selected in accordance with the innovative frequencies tested in the existing literature28, exploring a low and a high frequency: LFS at 20 Hz and HFS at 80 Hz. Both were applied in bipolar mode, with pulse width of 60 µs and a voltage set at 80% of the threshold value for inducing saccadic eye movements, i.e. 3 V. At these settings, no abnormal behavior indicative of discomfort was observed, nor any abnormal feeding behavior.
MPTP treatment and motor score evaluation
After collecting the baseline data, the monkey was treated with intramuscular injections of MPTP. A protocol consisting in chronic daily administration of small doses of MPTP (0.3 mg/kg, in NaCl 0.9%) during 5 consecutive days was used. The monkey received 1.5 mg/kg to achieve stable parkinsonian state. A rating scale of the parkinsonian syndrome was used before and after MPTP treatment in the home cage, as in our previous studies8,15. This scale includes 8 clinical symptoms (general activity, frequency of each arms movements, posture, bradykinesia, tremor, feeding, freezing, and vocalization), rated between 0 (normal) and 2–3 (depending on the degree of disability), with a total score out of 25. Assessments were performed by the same observer at 14:00 for 15 min, every day of the injections, and then once a week to ensure the syndrome stability.
Daytime sleepiness evaluation
Daytime sleepiness was evaluated using a mMSLT, as used in clinic but adapted to monkeys15, performed minimum 2 h after waking up. The animal was transferred to a behavioral cage and placed in a quiet room at 9:20 to allow sufficient time for habituation, and lights were turned off 3 times (light-OFF sessions at 10:00, 11:00, and 12:00) for a duration of 20 min. Between light-OFF sessions, lights were turned ON and every effort was made to keep the monkey awake. mMSLT was performed twice a week, alternating Stim-OFF, LFS-ON and HFS-ON conditions. Stimulation were applied continuously during 3 h (corresponding to the mMSLT protocol duration). A minimum interval of two days was maintained between experiments. Testing continued until a total of 6 mMSLT were completed under stim-OFF condition, and 5 mMSLT each were conducted under both LFS-ON and HFS-ON conditions. This protocol was uniformly applied in both health and parkinsonian state. The following parameters were calculated for each 20 min period of light-OFF: occurrence of at least one 30 s epoch of sleep, dichotomized measure (sleep versus no sleep during the light-OFF sessions) expressed in %, sleep latency and sleep duration both expressed in minutes. Sleep latency was determined if a 30 s epoch of any scorable sleep stage was observed. If no sleep onset was observed, sleep latency was designated to be 20 min. Sleep scoring was performed offline on a software (NeuroScore, Data Science International, France). Wake and sleep stages were manually scored according to the American Academy of Sleep Medicine criteria and were performed in 30 s epochs. The different stages identified were active wake and quiet wake (pooled together), and light sleep stage 1 characterized by θ waves and vertex sharp waves, and light sleep stage 2 characterized by θ waves and K complex/spindles (stage 1 and 2 pooled together).
Immunohistochemistry
The animal was deeply anesthetized with ketamine and pentobarbital (10 mg/kg and 25 mg/kg i.m.), then transcardially perfused with 0.9% saline solution followed by 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. The brain was removed from the skull, post-fixed for 24 h and cryoprotected in sucrose density gradient (from 10 to 30%). 50 μm free-floating sections were cut, using a cryotome, and store at -20 °C in a cryoprotective solution until immunochemistry. To assess dopamine depletion, brain sections containing the substantia nigra (AC-6 mm) and striatum (AC + 2 mm), including the caudate nucleus and putamen, were selected treated with rabbit anti-tyrosine hydroxylase (TH) (AB152, Sigma-Merck, France) and optical density was quantified (as previously described8). To visualize Ox/Hcrt neurons, brain sections containing the electrode trace and the LHA (AC-3 mm) were selected and treated with mouse anti-OxA (Orexin-A KK09, SantaCruz).
Statistical analysis
Standard statistical methods using GraphPad Prism 8 software were applied. A Kruskal–Wallis test followed by Dunn’s multiple comparisons test was used for the comparison of sleep parameters in the healthy state, during LHA-LFS and -HFS conditions and after MPTP intoxication in the parkinsonian state. A fisher’s exact test was performed to determine if the occurrence of at least one sleep episode was depending on the LHA-DBS conditions. Data are presented as mean ± standard error of the mean (SEM) and the statistical significance was considered at a probability (p) value ≤ 0.05.
Data availability
All data reported in this article can be shared on reasonable request from qualified investigators by contacting the corresponding author.
References
Ohayon, M. M. From wakefulness to excessive sleepiness: What we know and still need to know. Sleep. Med. Rev. 12, 129–141 (2008).
Young, T. B. Epidemiology of daytime sleepiness: Definitions, symptomatology, and prevalence. J. Clin. Psychiatry 65(Suppl 1), 12–16 (2004).
Slater, G. & Steier, J. Excessive daytime sleepiness in sleep disorders. J. Thorac. Dis. 4, 608–616 (2012).
Gandhi, K. D., Mansukhani, M. P., Silber, M. H. & Kolla, B. P. Excessive daytime sleepiness: A clinical review. Mayo Clin. Proc. 96, 1288–1301 (2021).
Titova, N. & Chaudhuri, K. R. Non-motor parkinson disease: New concepts and personalised management. Med. J. Aust 208, 404–409 (2018).
Yoo, S. W., Kim, J. S., Oh, Y. S., Ryu, D. W. & Lee, K. S. Excessive daytime sleepiness and its impact on quality of life in de novo Parkinson’s disease. Neurol. Sci. Off J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 40, 1151–1156 (2019).
Pycroft, L., Stein, J. & Aziz, T. Deep brain stimulation: An overview of history, methods, and future developments. Brain Neurosci. Adv. 2 (2018).
Davin, A. et al. Excessive daytime sleepiness in a model of Parkinson’s disease improved by low-frequency stimulation of the pedunculopontine nucleus. NPJ Park Dis. 9, 9 (2023).
Mileykovskiy, B. Y., Kiyashchenko, L. I. & Siegel, J. M. Behavioral correlates of activity in identified hypocretin/orexin neurons. Neuron 46, 787–798 (2005).
Lee, M. G., Hassani, O. K. & Jones, B. E. Discharge of identified orexin/hypocretin neurons across the sleep-waking cycle. J. Neurosci. 25, 6716–6720 (2005).
Mochizuki, T., Klerman, E. B., Sakurai, T. & Scammell, T. E. Elevated body temperature during sleep in orexin knockout mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 291, R533–R540 (2006).
Martin, T. et al. Dual orexin receptor antagonist induces changes in core body temperature in rats after exercise. Sci. Rep. 9, 1–9 (2019).
Rogers, A. A. et al. Brain stimulation deep brain stimulation of hypothalamus for narcolepsy-cataplexy in mice. Brain Stimul 13, 1305–1316 (2020).
Thannickal, T. C., Lai, Y. Y. & Siegel, J. M. Hypocretin (orexin) cell loss in Parkinson’s disease. Brain 130, 1586–1595 (2007).
Davin, A. et al. Early onset of sleep/wake disturbances in a progressive macaque model of Parkinson’s disease. Sci. Rep. 12, 1–14 (2022).
McIntyre, C. C., Savasta, M., Kerkerian-Le Goff, L. & Vitek, J. L. Uncovering the mechanism(s) of action of deep brain stimulation: Activation, inhibition, or both. Clin. Neurophysiol. Off J. Int. Fed. Clin. Neurophysiol. 115, 1239–1248 (2004).
Benabid, A. L. Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol. 24, 69–76 (2009).
Saper, C. B., Scammell, T. E. & Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 437, 1257–1263 (2005).
Cerri, M. & Morrison, S. F. Activation of lateral hypothalamic neurons stimulates brown adipose tissue thermogenesis. Neuroscience 135, 627–638 (2005).
Nakamura, K. & Morrison, S. F. Central efferent pathways for cold-defensive and febrile shivering. J. Physiol. 589, 3641–3658 (2011).
Tupone, D., Madden, C. J., Cano, G. & Morrison, S. F. An orexinergic projection from perifornical hypothalamus to raphe pallidus increases rat brown adipose tissue thermogenesis. J. Neurosci. 31, 15944–15955 (2011).
Cagnacci, A., Soldani, R., Laughlin, G. A. & Yen, S. S. Modification of circadian body temperature rhythm during the luteal menstrual phase: Role of melatonin. J. Appl. Physiol. 80, 25–29 (1996).
Belaid, H. et al. Sleep disorders in Parkinsonian macaques: Effects of L -Dopa Treatment and Pedunculopontine Nucleus Lesion. J. Neu 34, 9124–9133 (2014).
Martin, R. F. & Bowden, D. M. A stereotaxic template atlas of the macaque brain for digital imaging and quantitative neuroanatomy. Neuroimage 4, 119–150 (1996).
Sani, S., Jobe, K., Smith, A., Kordower, J. & Bakay, R. A. E. Deep brain stimulation for treatment of obesity in rats. J. Neurosurg. 107, 809–813 (2007).
Welkenhuysen, M., Van Kuyck, K., Das, J., Sciot, R. & Nuttin, B. Electrical stimulation in the lateral hypothalamus in rats in the activity-based anorexia model. Neurosurg. Focus 25, 29–32 (2008).
Whiting, D. M. et al. Lateral hypothalamic area deep brain stimulation for refractory obesity: A pilot study with preliminary data on safety, body weight, and energy metabolism. J. Neurosurg. 119, 56–63 (2013).
Arnulf, I. et al. Sleep induced by stimulation in the human pedunculopontine nucleus area. Ann. Neurol. 67, 546–549 (2010).
Acknowledgements
The authors thank Medtronic Inc (for providing the Activa PC+S devices used in this study), Luc Loveiko from CHUGA as well as Jessy Hugues Dit Ciles for technical and animal care assistance.
Author information
Authors and Affiliations
Contributions
BP and AD wrote the main manuscript text; AD prepare figures and did the experiments; BP found fundings, supervised and conceptualized the study; SC supervised the study and reviewed the manuscript; NT was the administrative supervisor and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.Chabardès declares that he has received consultation fees from Medtronic and Boston Scientific in the last 5 years. The other authors declare no competing interests. We used AI to improve readability and language of the work.
Ethics approval
In accordance with the Grenoble Alpes University, Grenoble Institut of Neurosciences (B3851610008) policies and with French legislation, experiments were performed in compliance with the European Community Council Directive of 2010 (2010/63/UE) for care of laboratory animals and in accordance with the ARRIVE guidelines. All procedures were reviewed and validated by the local ethic committee n˚004 and were authorized by the Direction Départementale des Services Vétérinaires de l’Isère – Ministère de l’Agriculture et de la Pêche, France.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Davin, A., Chabardès, S., Torres-Martinez, N. et al. Opposite effects of low and high frequency deep brain stimulation of lateral hypothalamus on arousal and temperature in a monkey pilot study. Sci Rep 14, 28318 (2024). https://doi.org/10.1038/s41598-024-79508-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79508-7