Abstract
To investigate the characteristics of acute visual dysfunction and fundus lesions in patients with COVID-19 pneumonia. A retrospective case series study was conducted. Data from 48 patients (96 eyes) with COVID-19 infection who presented to our ophthalmology department with acute onset visual disturbance between December 5, 2022, and February 28, 2023 were collected. Asymptomatic patients and those who had already recovered were excluded. Data collected included patient demographics, ophthalmic examinations, multicolor imaging (MCI), infrared autofluorescence (IR), spectral-domain optical coherence tomography (OCT), fundus fluorescein angiography (FFA). Of the 48 patients, 15 were male and 33 were female, with a mean age of 32 years. All patients had bilateral involvement. OCT showed hyperreflective signals in the outer plexiform layer and outer nuclear layer of the macular region in all 96 eyes of 48 patients (100%). Additionally, 66 eyes of 33 patients (68.8%) of eyes demonstrated abnormal reflectivity in the ellipsoid and interdigitation zones. MCI revealed petaloid or wedge-shaped hyperreflective areas in the macula in 46 (47.9%) of eyes, corresponding to hyporeflective areas on IR. Cotton-wool spots were observed in the peripapillary or posterior pole area in 54 (56.3%) of eyes. COVID-19 infection can lead to acute, bilateral, symmetric, and widespread retinal damage. Characteristic findings can be observed in ophthalmological examinations such as OCT, MCI, and IR.
Similar content being viewed by others
Introduction
The novel coronavirus disease 2019 (COVID-19), caused by the novel coronavirus SARS-CoV-2, has been present for three years now. It spreads rapidly and widely. The disease primarily manifests with respiratory symptoms, but it can also exacerbate underlying conditions or lead to multi-organ damage throughout the body. Ophthalmic complications in COVID-19 patients involve various aspects of the anterior and posterior segments of the eye, and there have been numerous reports on this topic in domestic and international literature [1, 2]. However, the focus of these reports is mainly on anterior segment diseases and ophthalmic epidemic prevention and control, with relatively fewer reports on complications related to the posterior segment of the eye. Recently, following adjustments to epidemic prevention and control policies, the number of patients with posterior segment eye diseases seeking treatment at our hospital has significantly increased. The following is a report on these cases.
Materials and methods
Paitents
Due to the retrospective nature of the study, the ethics committee of Henan Provincial People’s Hospital waived the need of obtaining informed consent’ in the manuscript. Based on the Declaration of Helsinki, a total of 48 COVID-19 patients (15 males and 33 females) with acute visual dysfunction were included in this study. The data were collected from December 5, 2022, to February 28, 2023, from patients who visited the ophthalmology department of our hospital for posterior segment eye diseases. Collecting patient data from the ophthalmology outpatient clinic database.
The inclusion criteria were as follows:
Confirmed diagnosis of COVID-19 with positive results from real-time reverse transcription-polymerase chain reaction (RT-PCR) or antigen testing for two consecutive times.
Onset of systemic symptoms, especially fever, fatigue, headache, and respiratory symptoms, after SARS-CoV-2 infection.
None of the patients received any topical ophthalmic medication during the study period.
Exclusion of patients with pre-existing underlying conditions such as diabetes, systemic hypertension, renal failure, proteinuria, or a history of dementia. Patients with a history of intraocular surgery or significant refractive errors (<-6.00D or > + 5.00D) were also excluded.
Ophthalmology examination
Ophthalmic routine examinations included visual acuity, corrected visual acuity, intraocular pressure measurement, slit-lamp examination, and fundus examination. The fundus examination was performed using the Heidelberg Retina Angiograph-2 (HRA-2) imaging system from Germany, which included MCI, infrared autofluorescence (IR), frequency-domain optical coherence tomography (OCT), and fundus fluorescein angiography (FFA).
Statistical analysis
SPSS statistics 25.0 software (IBM, USA) was used to analyze the data. Categorical data was described by frequencies and percentages. Using the Kolmogorov-Smirnov test to determine if a continuous variable follows a normal distribution. Continuous variables with a normal distribution are presented as the mean ± standard deviation (SD), and a t test was used for comparisons. Nonnormal variables are reported as medians (interquartile ranges). A P value less than or equal to 0.05 was considered to indicate a statistically significant difference.
Results
Among the 48 patients, There were 15 (31.2%) males and 33 (68.8%) females. Before the evaluation, 39 (81.3%) patients had received COVID-19 vaccines, while 9 had not. During their COVID-19 infection, 37 (77.1%) patients used antipyretic drugs such as acetaminophen and ibuprofen. The age of the patients ranged from 14 to 46 years, with the mean was 32.0 and the SD was 7.2. There was no significant difference in age between male and female patients (t = −0.757, P = 0.453).
All patients experienced acute visual dysfunction and developed systemic symptoms within one week of their COVID-19 diagnosis, followed by ocular presentation within one month. These patients sought medical attention 3–5 days after the onset of ocular symptoms. The median time from COVID-19 diagnosis to the onset of visual symptoms was 3 days, and IQR was [1, 4].
Visual acuity ranged from 0.01 to 1.0, and corrected visual acuity ranged from 0.05 to 1.0. The median of Corrected Visual Acuity was 0.5, and IQR was [0.3, 0.7]. Nine patients (15 eyes, 15.6%) had visual acuity below 0.3. All patients experienced bilateral onset, with only 2 cases (2.1%) showing a difference of more than 2 lines in visual acuity between the two eyes. For the rest of the patients, the difference in corrected visual acuity between both eyes was within 2 lines. (Table 1)
Special fundus examinations revealed the following findings:
OCT examination(Figure 1): A common characteristic was high reflectivity signals in the outer retinal layer (outer plexiform layer and/or outer granular layer) of the macular area, with some cases showing high reflectivity signals in the inner nuclear layer or even the entire layer. Abnormal changes in reflectivity signals were observed in the ellipsoid zone and interdigitation zone of 33 patients (66 eyes, 68.8%). Macular edema and retinal pigment epithelial detachment were present in 2 cases (4 eyes, 4.2%), and peripapillary nerve fiber layer thickening was observed in 1 case (2 eyes, 2.1%).
(a) Multicolor imaging (MCI): Petal-like deep red changes in the macular area. (b). IR (Infrared): Petal-like low reflective signal in the macular area. (c). OCT (Optical Coherence Tomography): High reflective signals in the outer plexiform and outer nuclear layers. (d)MCI: Wedge-shaped deep red changes in the macular area. (e)IR: Wedge-shaped low reflective signal in the macular area. (f)OCT: High reflective signal in the outer plexiform layer; cotton wool spots corresponding to high reflective signal in the nerve fiber layer. (g)MCI: Patchy deep red changes in the macular area. (h)IR: Patchy low reflective signal in the macular area. (i)OCT: High reflective signal in the outer nuclear layer; irregular reflective signal in the ellipsoid zone. (j)MCI: No obvious abnormalities in the macular area. (k)IR: No obvious abnormalities in the macular area. (l)OCT: High reflective signals visible in the outer nuclear, inner nuclear, and ganglion cell layers. (m)MCI: No obvious abnormalities in the macular area; numerous cotton wool spots. (n)IR: No obvious abnormalities in the macular area. (o)OCT: High reflective signal across the full thickness of the neurosensory retina.
Multicolor imaging (MCI): 23 cases (46 eyes, 47.9%) showed petaloid or wedge-shaped deep red changes in the macular area, while 25 cases (50 eyes, 52.1%) did not exhibit any apparent abnormalities.
Cotton-wool spots were found in 31 cases (54 eyes, 56.3%) in the peripapillary or posterior pole area, and 6 cases (12 eyes, 12.5%) showed a large number of cotton-wool spots. Additionally, 11 cases (19 eyes, 19.8%) presented with scattered hemorrhagic spots around the optic disc, and 1 case (2 eyes, 2.1%) showed optic disc edema.
Cotton-wool spots appeared as areas of high reflectivity in the nerve fiber layer on OCT and as low-fluorescence spots in fundus fluorescein angiography (FFA).
IR examination (Fig. 1): The MCI revealed dark red lesions in the macular area, corresponding to low reflectivity signals. Among the patients without obvious dark red changes in the macular area, no abnormalities were observed in IR.
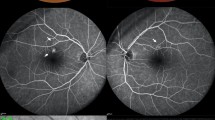
FFA findings (Fig. 2): In one patient (2 eyes, 2.1%) with macular edema and retinal pigment epithelial detachment, diffuse fluorescence leakage spots were observed. In two cases (3 eyes, 3.1%), there were occasional fluorescent spots around the optic disc. In one case (2 eyes, 2.1%) with optic disc edema, the optic disc showed mild fluorescence staining. No obvious fluorescence leakage was observed in the macular area. (Table 2)
(a) No significant abnormal fluorescein leakage in the macular area. (b)Scattered spots of retinal vascular fluorescein leakage. (c)Diffuse spots of retinal vascular leakage, subretinal blocking fluorescence spots, and faint fluorescein accumulation. (d)Thickening and edema of the neurosensory retina, high reflectivity in the outer plexiform layer, and neurosensory detachment (corresponding OCT of Fig. 2c).
Discussion
COVID-19 is a highly contagious disease caused by SARS-CoV-2, with a rapid transmission rate and high disability and mortality rates1. The virus can lead to primary infection or result in various systemic complications, especially those related to respiratory system infections. Eye symptoms are among the important systemic manifestations after SARS-CoV-2 infection2.
SARS-CoV-2 enters host cells through its spike protein interacting with the angiotensin-converting enzyme 2 (ACE-2) receptor when transmembrane serine protease 2 (TMPRSS2) is present3. Novel coronavirus eye disease refers to a series of eye disorders caused by SARS-CoV-2, including inflammatory lesions, vascular lesions, optic neuritis, cranial nerve lesions, or Miller Fisher syndrome, which can be the initial, accompanying, or sole lesions of SARS-CoV-2 infection. The disease involves various aspects of ophthalmology, with diseases affecting visual function, such as retinal diseases, including optic disc optic neuropathy, retinal lesions and macular lesions. SARS-CoV-2 enters cells by binding to the angiotensin-converting enzyme 2 (ACE2) receptor4,5, making cells containing ACE2 receptors susceptible to SARS-CoV-2 infection. In the retina, Müller glial cells, ganglion cells, retinal vascular endothelial cells (BRECs), and photoreceptors contain ACE2 receptors6,7.
Additionally, some studies have reported finding SARS-CoV-2 RNA in the retinas of patients who died from COVID-198. Due to adjustments in national epidemic prevention and control policies, the number of patients with diseases visiting our ophthalmology department had rapidly increased, and many cases of combined retinal diseases in patients with COVID-19 infection had been discovered in the short term.
All included patients had fundus lesions mainly in the posterior pole of the eye, concentrated in the macula and optic disc, and no patients with peripheral lesions have been found. There is no detailed literature reporting this phenomenon. Macular lesions appeared as patients with hyperreflective signals in the outer layer of the retina on OCT, with some patients showing hyperreflective signals in the inner nuclear layer and even the full layer. Some patients had local damage to the ellipsoid zone and interdigitation zone. Among them, 23 cases (46 eyes) showed abnormal locations in the macular area on OCT, corresponding to petal-shaped or wedge-shaped deep red changes seen in MCI, low reflectivity signals seen in IR, and paracentral scotomas seen in visual fields. These 46 cases were characteristic of acute macular neuroretinopathy (AMN). AMN is more common in reproductive-aged women, typically without systemic diseases, aged 21 to 43 years. This may lead to a relatively higher proportion of female patients among the included patients. Common triggers include oral contraceptives, excessive caffeine intake, treatment with adrenaline or ephedrine, viral infections, vascular spasms, use of adrenergic mimetics, dengue fever, and systemic lupus erythematosus9,10,11,12,13,14,15. It is characterized by the acute reddish-brown wedge-shaped lesion around the fovea, causing paracentral scotomas. On OCT, there are hyperreflective bands in the outer plexiform layer (OPL) and outer nuclear layer (ONL)16,17 with disruption of the ellipsoid zone; during the disease progression, the ellipsoid zone slowly recovers, ONL hyperreflectivity returns, and ONL becomes chronically thinner18.
This condition is frequently differentiated from acute parafoveal middle maculopathy (PAMM). PAMM predominantly affects the elderly, aged 54–65, and is often associated with systemic conditions like hypertension, diabetes, carotid stenosis, immunodeficiency, and sickle cell disease. Both PAMM and AMN exhibit similar hyporeflective changes in the macular region on infrared imaging, accompanied by corresponding visual scotomas16. Funduscopic examination often reveals a faint, grayish-white parafoveal lesion. OCT demonstrates hyperreflective bands within the inner plexiform layer (IPL) and inner nuclear layer (INL). As the disease progresses, the INL thins without involvement of the outer retinal layers19. Thus, OCT can be used to distinguish between AMN and PAMM.
All these 23 patients of 46 eyes in this study fit the characteristics of AMN rather than PAMM. The exact pathogenesis of AMN is not yet fully understood, but studies using OCT have suggested that it is an acute injury of the outer layer of the retina secondary to ischemia of the retinal deep capillary network, mainly affecting the outer plexiform layer and outer nuclear layer10,20. Additionally, Georges Azar et al. found a significantly increased incidence of AMN during the COVID-19 pandemic21, suggesting a potential association between the pathogenesis of AMN and COVID-19 infection. However, Studies reported coincident AMN and PAMM in patients with Purtscher retinopathy, retinal vein occlusion, central retinal artery occlusion, and retinal vasculitis, suggesting Müller cell impairment as a common underlying pathology22,23.
In addition to the mechanism of SARS-CoV-19 infection of retinal cells caused by the expression of ACE2 receptors in the retina, COVID-19 has also been found to cause choroidal blood flow disorders. Since the retinal oxygen and nutrient supply depend on choroidal blood flow, choroidal blood flow disorders can lead to outer retinal damage. Other mechanisms may include abnormal activation of microglia cells caused by the super inflammatory response induced by SARS-CoV-2, leading to outer retinal damage, coagulation abnormalities, and endothelial damage mediated by SARS-CoV-2 viruses. Ten patients in this study presented with cotton-wool spots in the posterior pole and peripapillary regions, with a few cases accompanied by hemorrhages. Previous studies have reported microhemorrhages, flame-shaped hemorrhages, cotton-wool spots, venous dilation, vascular tortuosity, central retinal artery occlusion, and sector retinal pallor in patients with SARS-CoV-2 infection24,25,26,27,28. These changes are believed to be caused by retinal ischemia due to thromboembolic events29.
Two patients in this study were found to have optic disc edema.SARS-CoV-2 can infect the nervous system through hematogenous and neuronal pathways, with the invasion mechanism possibly mainly involving the ACE2 receptors present in the circulatory and nervous systems30. Infection-induced hypoxia and the “cytokine storm” can cause neurological damage31,32, and thus may also be the cause of optic nerve damage. Two patients in this study exhibited both macular edema and detachment of the retinal neuroepithelial layer. Macular edema post-COVID-19 can arise from uveitis, retinal vein occlusion, and inflammatory responses related to the infection33. Macular edema can also be observed without any additional findings in COVID-19, which is likely due to a parainfectious or a post-viral manifestation of COVID-1934.
In conclusion, acute visual impairment in patients caused by SARS-CoV-2 infection is generally bilateral and relatively symmetric, with most cases presenting with mild to moderate impairment. On OCT, macular neuroepithelium in the macular area shows hyperreflective signals in the outer layer (outer plexiform layer and/or outer nuclear layer), and severely impaired patients may show significant damage to the ellipsoid zone. On MCI, the typical dark red wedge-shaped or petal-shaped changes in the macular area, cotton wool spots in the posterior pole and disc, and low reflection spots in the macular lesion area on IR can be seen. There is no obvious fluorescein leakage in the macular area on FFA.
Data availability
The datasets generated during the current study are not publicly available due the protection of patient privacy but are available from the corresponding author on reasonable request.
References
Diagnosis Treatment protocol for COVID-19. Chin. Med. 17, 481–487 (2022).
Ceran, B. B. & Ozates, S. Ocular manifestations of coronavirus disease 2019. Graefes Arch. Clin. Exp. Ophthalmol. 258, 1959–1963 (2020).
Gupta, A. et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 26, 1017–1032 (2020).
Zhou, P. et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 579, 270–270 (2020).
Hoffmann, M. et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 181, 271–271 (2020).
Choudhary, R., Kapoor, M. S., Singh, A. & Bodakhe, S. H. Therapeutic targets of renin-angiotensin system in ocular disorders. J. Curr. Ophthalmol. 29, 7–16 (2017).
Menuchin-Lasowski, Y. et al. SARS-CoV-2 infects and replicates in photoreceptor and retinal ganglion cells of human retinal organoids. Stem Cell. Rep. 17, 789–803 (2022).
Reinhold, A. et al. Ocular Pathology and occasionally detectable intraocular severe Acute Respiratory Syndrome Coronavirus-2 RNA in five fatal coronavirus Disease-19 cases. Ophthalmic Res. 64, 785–792 (2021).
Douglas, I. S. & Cockburn, D. M. Acute macular neuroretinopathy. Clin. Experimental Optometry. 86, 121–126 (2003).
Bhavsar, K. V. et al. Acute macular neuroretinopathy: a comprehensive review of the literature. Surv. Ophthalmol. 61, 538–565 (2016).
Liu, J. C., Nesper, P. L., Fawzi, A. A. & Gill, M. K. Acute macular neuroretinopathy associated with influenza vaccination with decreased flow at the deep capillary plexus on OCT angiography. Am. J. Ophthalmol. case Rep. 10, 96–100 (2018).
Shah, P., Zaveri, J. S. & Haddock, L. J. Acute Macular Neuroretinopathy following the administration of an influenza vaccination. Ophthalmic Surg. Lasers Imaging Retina. 49, 165–168 (2018).
Leys, M., Van Slycken, S., Koller, J. & Van de Sompel, W. Acute macular neuroretinopathy after shock. Bull. Soc. Belge Ophtalmol. 241, 95–104 (1991).
Munk, M. R. et al. New associations of classic acute macular neuroretinopathy. Br. J. Ophthalmol. 100, 389–394 (2016).
Lee, D. H., Lee, S. C. & Kim, M. Acute macular neuroretinopathy associated with systemic lupus erythematosus. Lupus. 25, 431–435 (2016).
Chen, X. J. et al. Spectrum of retinal vascular diseases Associated with Paracentral Acute Middle Maculopathy. Am. J. Ophthalmol. 160, 26–34 (2015).
Sridhar, J. et al. Optical coherence Tomography Angiography and En Face Optical Coherence Tomography features of Paracentral Acute Middle Maculopathy. Am. J. Ophthalmol. 160, 1259–1268 (2015).
Azar, G., Wolff, B., Cornut, P. L., Sahel, J. A. & Mauget-Faysse, M. Spectral domain optical coherence tomography evolutive features in acute macular neuroretinopathy. Eur. J. Ophthalmol. 22, 850–852 (2012).
Sarraf, D. et al. Paracentral Acute Middle Maculopathy A New variant of Acute Macular Neuroretinopathy Associated with Retinal Capillary Ischemia. Jama Ophthalmol. 131, 1275–1287 (2013).
Turbeville, S. D., Cowan, L. D. & Gass, J. D. M. Acute macular neuroretinopathy: a review of the literature. Surv. Ophthalmol. 48, 1–11 (2003).
Azar, G. et al. Did the COVID-19 pandemic increase the incidence of Acute Macular Neuroretinopathy? J. Clin. Med. 10, 5038 (2021).
Çebi, A. Y., Kiliçarslan, O. & Uçar, D. Coincident Acute Macular Neuroretinopathy and Paracentral Acute Middle Maculopathy in COVID-19. Turk. Oftalmoloji Dergisi-Turkish J. Ophthalmol. 53, 120–123 (2023).
Iovino, C. et al. Coincident PAMM and AMN and insights into a common pathophysiology. Am. J. Ophthalmol. 236, 136–146 (2022).
Jevnikar, K., Mekjavic, P. J., Valentincic, N. V., Petrovski, G. & Petrovic, M. G. An update on COVID-19 Related Ophthalmic manifestations. Ocul Immunol. Inflamm. 29, 684–689 (2021).
Invernizzi, A. et al. Retinal findings in patients with COVID-19: results from the SERPICO-19 study. Eclinicalmedicine 27, 100550 (2020).
Pereira, L. A. et al. Retinal findings in hospitalised patients with severe COVID-19. Br. J. Ophthalmol. 106, 102–105 (2022).
Bertoli, F. et al. Ocular Findings in COVID-19 Patients: A Review of Direct Manifestations and Indirect Effects on the Eye. Journal of Ophthalmol. (2020).
Acharya, S., Diamond, M., Anwar, S., Glaser, A. & Tyagi, P. Unique case of central retinal artery occlusion secondary to COVID-19 disease. Idcases 21, e00867 (2020).
Hayreh, S. S. Acute retinal arterial occlusive disorders. Prog Retin Eye Res. 30, 359–394 (2011).
Lima, M. et al. Unraveling the possible routes of SARS-COV-2 Invasion into the Central Nervous System. Curr. Treat. Options Neurol. 22 (2020).
Casagrande, M. et al. Detection of SARS-CoV-2 in human retinal biopsies of deceased COVID-19 patients. Ocul Immunol. Inflamm. 28, 721–725 (2020).
Huang, Y. H., Jiang, D. & Huang, J. T. SARS-CoV-2 detected in Cerebrospinal Fluid by PCR in a case of COVID-19 Encephalitis. Brain Behav. Immun. 87, 149–149 (2020).
Al-Abri, M., Al-Musalami, A., Al-Rawahi, B. & Al-Hinai, A. Al-Fadhil, N. Central retinal vein occlusion in a young healthy COVID-19 patient: a case report and literature review. Middle East. Afr. J. Ophthalmol. 28, 199–202 (2021).
Kilicarslan, O., Yilmaz Cebi, A., Ucar, D., Senturk, F. & Aras, C. Bilateral Cystoid Macular Edema after COVID-19: 1 year follow up. Ocul Immunol. Inflamm. 31, 1236–1239 (2023).
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
J W and ZL W conceived of the presented idea,X C and LH W collected the clinical data,J W wrote the manuscript with support from ZH W,JM L performed data analysis,All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
Due to the retrospective nature of the study, the ethics committee of Henan Provincial People’s Hospital waived the need of obtaining informed consent’ in the manuscript.
All methods were carried out in accordance with relevant guidelines and regulations.
All experimental protocols were approved by Henan Provincial People’s Hospital.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wei, J., Chen, X., Wang, L. et al. A cross-sectional study of fundus lesion characteristics in patients with acute visual impairment caused by COVID-19 infection. Sci Rep 14, 28134 (2024). https://doi.org/10.1038/s41598-024-79509-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79509-6
Keywords
This article is cited by
-
The characteristics of multimodal fundus imaging in AMN patients following COVID infection
Scientific Reports (2025)