Abstract
To determine diagnostic value of diffusion tensor imaging (DTI) in amyotrophic lateral sclerosis (ALS) patients and investigate the association between DTI and neurofilaments (NFs), including serum and cerebrospinal fluid (CSF) levels of neurofilament light chain (NFL) and phosphorylated neurofilament heavy chain (pNFH). Forty-three clinically diagnosed ALS patients and 32 control subjects without neurological disorders underwent routine MRI (magnetic resonance imaging) and DTI scans. DTI parameters (mean diffusivity [MD] and fractional anisotropy [FA]) at axial levels of internal capsules and cerebral peduncles along the corticospinal tract (CST) were measured. The study compared the differences of DTI parameters between ALS patients and controls using the Mann-Whitney U test. Diagnostic efficacy of each DTI metric was evaluated using the receiver operating characteristic (ROC) curve. NFs (NFL and pNFH levels in serum and CSF) were measured by enzyme-linked immunosorbent assay. Correlation analyses were conducted between DTI parameters and NFs. Capsule-MD and Peduncle-MD in ALS patients were higher than those in controls; whereas Capsule-FA and Peduncle-FA in ALS patients were lower than those in controls (all, p < 0.05). The area under curve (AUC) was 0.730 for Capsule-FA, 0.828 for Capsule-MD, 0.890 for Peduncle-FA, and 0.896 for Peduncle-MD. Capsule-FA was negatively correlated with CSF-NFL (r = − 0.813, p < 0.001), Serum-NFL (r = − 0.493, p = 0.001), CSF-pNFH (r = − 0.637, p < 0.001), and Serum-pNFH (r = − 0.672, p < 0.001); Peduncle-FA negatively with CSF-NFL (r = − 0.562, p < 0.001), CSF-pNFH (r = − 0.506, p = 0.001), and Serum-pNFH (r = − 0.488, p = 0.001); Peduncle-MD positively with CSF-NFL (r = 0.516, p < 0.001), CSF-pNFH (r = 0.494, p = 0.001). DTI had superior performance in identifying ALS patients and could serve as a reliable predictor. DTI parameters related to neurofilament markers, and Capsule-FA may become a robust surrogate biomarker indicating disease severity and progression rate for ALS patients.
Similar content being viewed by others
Introduction
ALS is a fatal neurodegenerative disease. The main pathological features include the loss of selective motor neurons and associated astrocytosis in the CST, brain stem nuclei and ventral roots of the spinal cord1. This results in progressive muscle weakness and atrophy, ultimately leading to paralysis and respiratory failure. Currently, there are only two FDA-approved treatments available: riluzole and edaravone. Most patients have a poor prognosis and die of respiratory failure within 3–5 years of clinical onset1. Early diagnosis and intervention are crucial for improving outcomes and quality of life for ALS patients.
Pathological examinations in ALS showed that the disease had invaded the white matter, such as CST, the corpus callosum and so forth, especially CST. Conventional MRI has achieved various findings on the involvement of CST in ALS. T2-weighted2 and FLAIR3 sequences have demonstrated a symmetrical high signal in the posterior limb of internal capsule. Nonetheless, only the patients in the advanced stage of ALS can be detected with above conventional MRI approach4.
With the application of brain functional MRI, disorders of the central nervous system (CNS) have been widely investigated4. DTI is considered as one of the most effective functional MRI techniques for assessing various pathological changes of white matter fiber tracts in the CNS. DTI has been employed to examine upper motor neuron pathology and assess disease progression in ALS patients5. FA and MD are widely used parameters in DTI. FA is a measure for fraction anisotropy of water molecular diffusion and indicates various characteristics of the number and size of axon fibers, and the density of crossing fibers6,7,8. MD represents the mean rate of water molecular motion and has been investigated to be related to cellularity, edema, and necrosis, which may reflect the microstructural abnormalities of white matter9,10. DTI has been used to investigate the severity of white matter impairment in ALS patients11,12. Sarro et al.13 have shown that FA values are decreased within white matter of focal brain regions in ALS patients.
While many studies have utilized multiple DTI metrics to investigate differences between ALS patients and healthy controls, the reliability and consistency of these parameters across different patient populations and imaging protocols remain areas of ongoing research. Our study aimed to contribute to this body of knowledge by applying both FA and MD indices along bilateral CST in our specific cohort, demonstrating consistent diagnostic efficacy. Furthermore, the prior research focused on DTI of ALS patients on a single slice12. In contrast, this study examined various DTI indices on multiple slices of the CST. This approach can offer a more precise and comprehensive evaluation of the extent of white matter impairment in ALS patients and furnish quantitative markers for the early detection of ALS.
For another, experimental indicators are sensitive biomarkers for the diagnosis of ALS. An increasing body of evidence suggests that neurofilaments (NFs) levels, especially phosphorylated neurofilament heavy chain (pNFH) and neurofilament light chain (NFL), were elevated in serum and cerebrospinal fluid (CSF) of ALS patients14,15. NFs have been internationally recognized as biomarkers for ALS. NFs in CSF and serum hold significant importance in diagnosing ALS and assessing the progression of the disease14. However, few studies have linked NFs and DTI metrics, and the experimental indicators mentioned above have not been widely used in clinical practice. Through analyzing the relationships among various NFs and multiple DTI indices across different regions, we aim to uncover novel insights.
This study aims to determine the diagnostic utility of DTI in patients with ALS, explore the association between DTI and neurofilament markers including the serum and CSF levels of NFL and pNFH, and validate the significance of DTI parameters as potential biomarkers for early diagnosis and treatment of ALS.
Materials and methods
Subjects
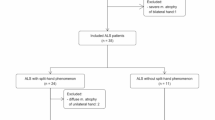
Forty-three patients (median age 59 years; range 25–81 years) with clinically confirmed ALS according to El Escorial criteria16 and 32 controls (median age 54.5; range 22–76 years) from First Hospital of Shanxi Medical University were recruited. This study was approved by the Ethics Committee of First Hospital of Shanxi Medical University and informed consent requirement was obtained in all patients. Inclusion criteria comprised: (1) newly diagnosed ALS; (2) underwent laboratory examination of NFL and pNFH; (3) underwent conventional MRI and DTI. Exclusion criteria included: (1) images with head motion artifacts; (2) claustrophobic or other contraindications of MRI examination; (3) history of other neurologic or psychiatric. Disease duration was quantified as the number of months from the initial presentation of symptoms to the collection of biofluid samples. The disease progression rate (DPR) was calculated as follows: (48 − ALSFRS-R score at the time of sampling)/disease duration (months). The ALSFRS-R is used to assess the functional status of ALS patients with a total score of 48; elevated scores signify superior functional ability.
MRI data acquisition
Participants underwent MRI examinations on a 3.0 T MRI scanner (MAGNETOM Skyra, Siemens Medical Solutions, Germany) equipped with a 20-channel array coil. The scanning protocol included conventional MRI sequences (T1-weighted images [T1WI], T2 fluid-attenuated inversion recovery [T2FLAIR] images, T2-weighted images [T2WI]) and DTI sequence. The scanning parameters of MRI were as follows: repetition time (TR) 220 ms and echo time (TE) 2.46 ms for T1WI; TR 3570 ms and TE 175 ms for T2WI; and TR 8000 ms, TE 103 ms, and interval time (TI) 2374 ms for FLAIR. Thickness and slice interval were 6.0/1.0 mm; field of view (FOV) was 220 mm × 220 mm. DTI was performed using an echo planar imaging (EPI) sequence with three b-values (0, 1000, and 2000 s/mm2) and 30 diffusion directions. DTI data were obtained using the following parameters: TR/TE, 4000/116 ms; FOV, 220 mm × 220 mm; matrix, 192 × 192; number of signals acquired, 1; Thickness and slice interval were 6.0/1.0 mm.
Image processing and analysis
The original DTI maps were transmitted to post-processing workstation with Neuro 3D software to generate the MD and FA maps. Regions of interest (ROIs) were selected on the FA and MD maps. These ROIs were manually delineated on axial slices of the posterior limb of internal capsule and the cerebral peduncle along the left and right corticospinal tracts respectively. We calculated MD and FA values for bilateral ROIs. In clinical practice, we found that ALS patients typically exhibit symmetrical lesions on both sides in CST in T2FLAIR sequence of MRI. Using average measurements from both sides could reduce errors. Then, the average value of the bilateral ROIs at the same slice was calculated. Capsule-FA, Peduncle-FA, Capsule-MD and Peduncle-MD values were derived, representing the DTI parameters at the levels of the internal capsule and the cerebral peduncle. Figure 1A,B demonstrated T2FLAIR, MD, FA and FA pseudo-color maps of healthy control and ALS. A total of two blinded, board-certified radiologists performed the image analyses.
(A) Upper row: T2FLAIR (a), MD (b), FA (c) and FA peudo-color (d) maps of healthy control on the internal capsule level; Low row: T2FLAIR (e), MD (f), FA (g) and FA peudo-color (h) maps of healthy control on the cerebral peduncle level. (B) Upper row: T2FLAIR (a), MD (b), FA (c) and FA peudo-color (d) maps of patient with ALS on the internal capsule level; Low row: T2FLAIR (e), MD (f), FA (g) and FA peudo-color (h) maps of patient with ALS on the cerebral peduncle level.
Laboratory indexes of blood and CSF
Samples of blood in dry tubes and CSF in polypropylene tubes were collected and then centrifuged (blood samples at 1500 g for 10 min; CSF samples at 300 g for 5 min). The supernatants were immediately frozen and stored in − 80° C refrigerator. NFs concentrations were determined using commercial enzyme-linked immunosorbent assays (ELISAs) in accordance with the manufacturer’s instructions (pNFH; Abcam; NFL; Novus). The ELISA exhibited an analytical sensitivity of 9.38 pg/ml for NFL and 20 pg/ml for pNFH. The mean intra-assay coefficient of variation (CV) was less than 10%, whereas the inter-assay CV was less than 12% for both NFL and pNFH concentrations.
Statistical analysis
The data analyses were performed by using SPSS (version 23.0, Chicago, IL, USA) statistical software. Inter-group differences between ALS and control concerning MD and FA parameters on the posterior limb of the internal capsule and the cerebral peduncle slice were compared respectively using the Mann-Whitney U test, and p < 0.05 was considered statistically significant. Receiver operating characteristic (ROC) curves were analyzed for each DTI parameter (MD and FA value) to identify the optimal cut-off values. Z-scores were used to compare the performance of DTI in distinguishing ALS patients from healthy subjects. Associations between the DTI parameters and the NFs indexes were analyzed using Spearman’s correlation test.
Ethical approval
This study was conducted in accordance with the tenets of the 2013 revision of the Declaration of Helsinki and was approved by the Ethics Committee of First Hospital of Shanxi Medical University. This study complied with all relevant guidelines and regulations.
Informed consent
The authors declare that informed consent was obtained in all patients.
Results
Patients
The study included 43 patients with ALS (26 male and 17 female; median age 59 years, range 25–81 years) and 32 healthy controls (18 male and 14 female; median age 54.5 years, range 22–76 years). The demographic and clinical characteristics are summarized in Table 1. There was no significant difference in above features between ALS patients and healthy controls (all p > 0.05).
ALS amyotrophic lateral sclerosis, ALSFRS-R amyotrophic lateral sclerosis functional rating scale revised.
Comparison of DTI parameters between ALS and controls
Compared with healthy controls, patients with ALS showed significantly decreased Capsule-FA values (p = 0.001) on the posterior limb of the internal capsule slice, and Peduncle-FA values on the cerebral peduncle slice respectively (p < 0.001) (Supplementary Table S1). Capsule-MD and Peduncle-MD values were significantly higher in patients with ALS compared to healthy controls on the posterior limb of internal capsule and the cerebral peduncle slice respectively (p < 0.001) (Supplementary Table S1). The box-and-whiskers graphs for the DTI parameters between patients with ALS and healthy controls are shown in Fig. 2.
The box-and-whiskers graphs for the DTI parameters. Capsule-FA (a), Capsule-MD (b), Peduncle-FA (c), and Peduncle-MD (d).
ROC analysis to identify the best predictor for differentiating ALS patients
The AUC was 0.828 for Capsule-MD and 0.896 for Peduncle-MD in differentiation of patients with ALS from healthy controls, and this difference was significant (all p < 0.001) (Fig. 3a). The cut-off values, accuracy, sensitivity and specificity are shown in Supplementary Table S2. The AUC was 0.730 for Capsule-FA and 0.890 for Peduncle-FA in differentiation of patients with ALS from healthy controls, and this difference was significant (p = 0.001 and p < 0.001) (Fig. 3b). The cut-off values, accuracy, sensitivity and specificity are shown in Supplementary Table S2.
Receiver operator curve analysis of DTI parameters for differentiating ALS patients from controls.
Correlations between DTI parameters and NFs
Correlation between DTI parameters and NFL levels were demonstrated in Fig. 4A). Capsule-FA was negatively correlated with CSF-NFL (r = − 0.813, p < 0.001) (a) and Serum-NFL (r = − 0.493, p = 0.001) (b). Peduncle-FA was negatively correlated with CSF-NFL (r = − 0.562, p < 0.001) (c) and weak negatively correlated with Serum-NFL (r = − 0.337, p = 0.027) (d). No association was found between Capsule-MD and CSF-NFL (r = 0.034, p = 0.827) (e), Capsule-MD and Serum-NFL (r = 0.024, p = 0.881) (f). Peduncle-MD was positively correlated with the CSF-NFL (r = 0.516, p < 0.001) (g) and weak positively correlated with the Serum-NFL (r = 0.309, p = 0.044) (h). ALL the correlations between DTI parameters and pNFH levels are shown in Supplementary Table S3.
Correlation between DTI parameters and pNFH levels were shown in Fig. 4B. Capsule-FA was negatively correlated with CSF-pNFH (r = − 0.637, p < 0.001) (a) and Serum-pNFH (r = − 0.672, p < 0.001) (b). Peduncle-FA was negatively correlated with CSF-pNFH (r = − 0.506, p = 0.001) (c) and Serum-pNFH (r = − 0.488, p = 0.001) (d). No association was found between Capsule-MD and CSF-pNFH (r = 0.168, p = 0.282) (e), Capsule-MD and Serum-pNFH (r = 0.118, p = 0.452) (f). Peduncle-MD was positively correlated with CSF-pNFH (r = 0.494, p = 0.001) (g), and weak positively correlated with Serum-pNFH (r = 0.372, p = 0.014) (h). ALL the correlations between DTI parameters and pNFH levels are shown in Supplementary Table S4.
(A) Correlation between DTI parameters and NFL level. Capsule-FA and CSF-NFL (a), Capsule-FA and Serum-NFL (b), Peduncle-FA and CSF-NFL (c), Peduncle-FA and Serum-NFL (d), Capsule-MD and CSF-NFL (e), Capsule-MD and Serum-NFL (f), Peduncle-MD and CSF-NFL (g), Peduncle-MD and Serum-NFL (h). (B) Correlation between DTI parameters and pNFH levels. Capsule-FA and CSF-pNFH (a), Capsule-FA and Serum-pNFH (b), Peduncle-FA and CSF-pNFH (c), and Peduncle-FA and Serum-pNFH (d), Capsule-MD and CSF-pNFH (e), Capsule-MD and Serum-pNFH (f). Peduncle-MD and CSF-pNFH (g), Peduncle-MD and Serum-pNFH (h).
Discussion
Early and accurate identification of ALS is crucial for determining the optimal clinical treatment strategy. In the present study, the utilization of FA and MD parameters based on the DTI sequence facilitated the discrimination between patients with ALS and healthy controls. The accuracy of the DTI parameters in differentiating ALS patients varies depending on the specific anatomical location. Moreover, Peduncle-MD showed superior diagnostic performance compared to other DTI parameters in distinguishing ALS patients from healthy controls.
The present investigation indicated that patients with ALS exhibited a higher MD value compared to the healthy controls, while the FA value was lower than that of the healthy controls in two key regions of CST: the internal capsule and cerebral peduncle. These results support previous academic contributions to the field. DTI studies of ALS patients have consistently reported decreased FA and increased MD of the CST8,17. Increases in MD have been regarded as indicators of axonal loss and neurodegeneration18. Our finding of elevated MD in ALS patients supports this interpretation, though future investigations should explore whether the alterations in MD observed in ALS patients represent a disease-specific pathological process. FA is a major indicator of white matter fibers anisotropy, being related to myelin integrity, and density and parallelism of fibers19. A decrease in FA values is recognized as a sign of damage to the white matter fiber tract20. Our study revealed that ALS patients exhibited lower FA values compared to healthy controls, although the difference was not significant. Previous research suggested that the FA values in the CST exhibited a significant decrease at the brainstem level19. The current study demonstrated some differences from above study, possibly due to variations in magnetic field intensity of MRI equipments and potential deviation in the selection of ROIs. Moreover, FA values vary among different phenotypes of ALS patients, such as those with bulbar-onset and limb-onset ALS21,22,23. To sum up, changes in FA and MD values highlight the features of reduced anisotropy and accelerated water diffusion following CST degeneration in ALS patients.
CST anatomically represents the motor pathway extending from the cerebral cortex to the spinal cord. Our analysis focused on multiple DTI parameters, particularly at the cerebral peduncle and internal capsule levels in ALS patients. When considering diagnostic accuracy, for the same DTI index (MD or FA value), diagnosing ALS at the cerebral peduncle level proves more accurate than at the internal capsule level. Notably, Peduncle-FA and Peduncle-MD values showed higher accuracy in identifying ALS patients and healthy controls. Given the dense arrangement of tract fibers in the brainstem, alterations of DTI metrics at this specific location may be more pronounced compared to other regions, when pathological change occurs. Hence, DTI values at the level of the brainstem may exhibit heightened sensitivity to structural alterations. According to Prokscha et al.24, DTI-based data already provided 100% sensitivity and 92% specificity in distinguishing between 12 controls and 13 patients’ samples. FA could be defined to discriminate ALS patients from controls (sensitivity/specificity, 74%/72%)17. The variations in outcomes could potentially be attributed to disparities in sample size or DTI post-processing methods. A meta-analysis conducted by Foerster et al.25 reported a pooled sensitivity of 65% and a pooled specificity of 67% and assumed that the discriminatory power of DTI to diagnose ALS was only modest. This study found that multi-parameter studies of DTI (including FA and MD values on different levels of CST) have higher sensitivity and specificity in the diagnosis of ALS. Additionally, the study possesses a moderate sample size, rendering it valuable for clinical reference. However, there are inconsistent findings about the diagnostic performance of DTI. Schimrigk et al.26 found that significant disparities exist between healthy individuals and ALS patients on the internal capsule level with the largest discrepancies and the minimal variations. These results differ from our findings, possibly due to the small sample size, which included 10 cases. This limitation could have introduced sampling bias. In addition, in terms of research methods, there could be somewhat subjective variances in the delineation of ROIs. By measuring DTI parameters at different levels targeting this specific site of CST, our findings indirectly shed light on the link between upper motor neuron impairment and CST degeneration through changes in FA and MD values27.
This study also investigated the correlation between DTI parameters and laboratory indicators of ALS, NFs (including in CSF and serum). Currently, the majority of studies have primarily focused on the correlation between DTI and clinically relevant measures, such as ALSFRS-R28 and the summation of compound muscle action potential Z scores (CMAP Z sum score)29. A decreased FA value in the CST has been observed to exhibit correlations with disease severity and rate of disease progression23,30. However, these clinical indicators are somewhat subjective and are also non-quantitative measures. For example, the weak correlation between the DTI parameters and ALSFRS-R suggests that changes in ALSFRS-R may lack specificity and exhibit a limited association with the pathological processes in ALS11,19,27,31. This study conducted correlation analysis between DTI and quantitative laboratory markers, which could more precisely indicate ALS severity.
Currently, few studies have sought to establish a connection between NFs and MRI measures. We observed a complex relationship between DTI parameters and NFs levels in CSF and serum. Notably, FA in both the capsule and peduncle regions showed consistent negative correlations with NFL and pNFH levels in both CSF and serum. This aligns with the conclusion of Menke et al.32 that higher NFs levels are associated with lower FA values in ALS patients. A noteworthy aspect of our finding is the stronger negative correlation between Capsule-FA and CSF-NFL (r = - 0.813, p < 0.001). Our findings suggested that the capsule area of the CST may be more severely affected in ALS patients. This is evidenced by the fact DTI metrics were sensitive to Wallerian-type myelin degeneration in this region32. Meanwhile, NFs are associated with the disease duration of ALS and the rate of disease progression33,34,35,36. CSF-NFL levels reflect a more accurate correlation with axonal and motor neuronal injuries in ALS patients compared to blood samples15,37,38. Accordingly, we could infer that the Capsule-FA index may serve as a proxy for CSF-NFL concentrations.
Interestingly, MD showed variable correlations, with peduncle-MD positively correlating with NFs, while capsule-MD showed no significant associations. These results, combined with the previous observation of increased radial diffusivity in CST32, indicated that different DTI parameters may reflect distinct aspects of neurodegeneration. From another perspective, it can be inferred that the accuracy of the MD metric in reflecting CST disruption in ALS patients is comparatively lower than that of the FA metric. This finding emphasizes the importance for medical practitioners to pay more attention to the changes in FA values among ALS patients, in order to better understand the progression of ALS.
The clinical implications of these findings are substantial. Firstly, NFs tests remain largely inaccessible in the majority of hospitals across China. Capsule-FA, as a noninvasive and easily accessible parameter appears promising. Our study further underscores the potential of the Capsule-FA index in the early diagnosis and clinical assessment of ALS, offering a valuable surrogate biomarker for gauging motor neuron degeneration and disease progression in ALS patients, especially in longitudinal monitoring or evaluating treatment efficacy. Furthermore, for hospitals that are equipped to assess the NFs, the potential is significantly higher utilizing the integration of DTI and NF analysis as a robust methodology for monitoring ALS pathology and its progression.
The present research encountered certain constraints. Initially, there existed a trend that ALS patients being older than the controls. This may slightly impact the comparative results of DTI indices between the two groups. Furthermore, the examination of DTI parameters was restricted to only two MRI slices, lacking a more extensive assessment of CST damage. Finally, while ALS pathologically affects both upper and lower motor neurons, this study solely focused on upper motor neuron damage in the DTI analysis, without exploring their interrelation in ALS pathogenesis.
Conclusion
The present study investigated the diagnostic value of DTI parameters and their relevance with NFs in ALS patients. Our findings suggested that DTI parameters in CST could provide useful clues for the clinical diagnosis of ALS. Additionally, Capsule-FA might serve as a reliable surrogate biomarker for assessing disease severity and progression rate in ALS patients. In the future, we hope to conduct prospective studies on suspected ALS patients, confirmed ALS patients, and symptomatic predisposed individuals to fully explore the potential of combined laboratory markers NFs and MRI-DTI as biomarkers.
Data availability
The datasets used and analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- MRI:
-
Magnetic resonance imaging
- DTI:
-
Diffusion tensor imaging
- FA:
-
Fractional anisotropy
- MD:
-
Mean diffusivity
- NFs:
-
Neurofilaments
- NFL:
-
Neurofilament light chain
- pNFH:
-
Phosphorylated neurofilament heavy chain
- CSF:
-
Cerebrospinal fluid
- ALSFRS-R:
-
Amyotrophic lateral sclerosis functional rating scale revised
References
Brown, R. H. & Al-Chalabi, A. Amyotrophic lateral sclerosis. N Engl. J. Med. 377 (2), 162–172 (2017).
Hofmann, E., Ochs, G., Pelzl, A. & Warmuth-Metz, M. The corticospinal tract in amyotrophic lateral sclerosis: an MRI study. Neuroradiology. 40 (2), 71–75 (1998).
Hecht, M. J. et al. MRI-FLAIR images of the head show corticospinal tract alterations in ALS patients more frequently than T2-, T1- and proton-density-weighted images. J. Neurol. Sci. 186 (1–2), 37–44 (2001).
Carrara, G. et al. A distinct MR imaging phenotype in amyotrophic lateral sclerosis: correlation between T1 magnetization transfer contrast hyperintensity along the corticospinal tract and diffusion tensor imaging analysis. AJNR. 33 (4), 733–739 (2012).
Agosta, F. et al. Assessment of white matter tract damage in patients with amyotrophic lateral sclerosis: a diffusion tensor MR imaging tractography study. AJNR. 31 (8), 1457–1461 (2010).
Roberts, R. E., Anderson, E. J. & Husain, M. White matter microstructure and cognitive function. Neuroscientist. 19 (1), 8–15 (2013).
Prell, T. et al. Diffusion tensor imaging patterns differ in bulbar and limb onset amyotrophic lateral sclerosis. Clin. Neurol. Neurosurg. 115 (8), 1281–1287 (2013).
Tang, M. et al. Quantitative assessment of amyotrophic lateral sclerosis with diffusion tensor imaging in 3.0T magnetic resonance. Int. J. Clin. Exp. Med. 8 (5), 8295–8303 (2015).
Alexander, A. L. et al. Characterization of cerebral white matter properties using quantitative magnetic resonance imaging stains. Brain Connect. 1 (6), 423–446 (2011).
Vos, S. B., Jones, D. K., Jeurissen, B., Viergever, M. A. & Leemans, A. The influence of complex white matter architecture on the mean diffusivity in diffusion tensor MRI of the human brain. Neuroimage. 59 (3), 2208–2216 (2012).
Geraldo, A. F. et al. Beyond fractional anisotropy in amyotrophic lateral sclerosis: the value of mean, axial, and radial diffusivity and its correlation with electrophysiological conductivity changes. Neuroradiology. 60 (5), 505–515 (2018).
Du, X. Q. et al. Brain white matter abnormalities and correlation with severity in amyotrophic lateral sclerosis: an atlas-based diffusion tensor imaging study. J. Neurol. Sci. 405, 116438 (2019).
Sarro, L. et al. Cognitive functions and white matter tract damage in amyotrophic lateral sclerosis: a diffusion tensor tractography study. AJNR. 32 (10), 1866–1872 (2011).
Brodovitch, A. et al. Combination of serum and CSF neurofilament-light and neuroinflammatory biomarkers to evaluate ALS. Sci. Rep. 11 (1), 703 (2021).
Sugimoto, K., Han, Y., Song, Y. & Gao, Y. Correlational analysis of ALS Progression and serum NfL measured by Simoa Assay in Chinese patients. Front. Neurol. 11, 579094 (2020).
Chieia, M. A., Oliveira, A. S., Silva, H. C. & Gabbai, A. A. Amyotrophic lateral sclerosis: considerations on diagnostic criteria. Arq. Neuropsiquiatr. 68 (6), 837–842 (2010).
Rosskopf, J. et al. Ex post facto assessment of diffusion tensor imaging metrics from different MRI protocols: preparing for multicentre studies in ALS. Amyotroph. Lateral Scler. Frontotemporal Degener. 16 (1–2), 92–101 (2015).
Aung, W. Y., Mar, S. & Benzinger, T. L. Diffusion tensor MRI as a biomarker in axonal and myelin damage. Imaging Med. 5 (5), 427–440 (2013).
Baek, S. H. et al. Usefulness of diffusion tensor imaging findings as biomarkers for amyotrophic lateral sclerosis. Sci. Rep. 10 (1), 5199 (2020).
Turner, M. R. et al. Towards a neuroimaging biomarker for amyotrophic lateral sclerosis. Lancet Neurol. 10 (5), 400–403 (2011).
Cardenas-Blanco, A. et al. Central white matter degeneration in bulbar- and limb-onset amyotrophic lateral sclerosis. J. Neurol. 261 (10), 1961–1967 (2014).
Prell, T. & Grosskreutz, J. The involvement of the cerebellum in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Frontotemporal Degener. 14 (7–8), 507–515 (2013).
van der Graaff, M. M. et al. Upper and extra-motoneuron involvement in early motoneuron disease: a diffusion tensor imaging study. Brain. 134 (Pt 4), 1211–1228 (2011).
Blain, C. R. et al. A longitudinal study of diffusion tensor MRI in ALS. Amyotroph. Lateral Scler. 8 (6), 348–355 (2007).
Foerster, B. R. et al. Diagnostic accuracy using diffusion tensor imaging in the diagnosis of ALS: a meta-analysis. Acad. Radiol. 19 (9), 1075–1086 (2012).
Schimrigk, S. K. et al. Diffusion tensor imaging-based fractional anisotropy quantification in the corticospinal tract of patients with amyotrophic lateral sclerosis using a probabilistic mixture model. AJNR. 28 (4), 724–730 (2007).
Hsueh, S. J. et al. Brain imaging signatures in amyotrophic lateral sclerosis: correlation with peripheral motor degeneration. Ann. Clin. Transl Neurol. 10 (8), 1456–1466 (2023).
Goutman, S. A. et al. Recent advances in the diagnosis and prognosis of amyotrophic lateral sclerosis. Lancet Neurol. 21 (5), 480–493 (2022).
Mazón, M., Vázquez Costa, J. F. & Ten-Esteve, A. Martí-Bonmatí, L. Imaging biomarkers for the diagnosis and prognosis of neurodegenerative diseases. The example of amyotrophic lateral sclerosis. Front. Neurosci. 12, 784 (2018).
Ciccarelli, O. et al. Investigation of white matter pathology in ALS and PLS using tract-based spatial statistics. Hum. Brain Mapp. 30 (2), 615–624 (2009).
Borsodi, F. et al. Multimodal assessment of white matter tracts in amyotrophic lateral sclerosis. PLoS One. 12 (6), e0178371 (2017).
Menke, R. A. et al. CSF neurofilament light chain reflects corticospinal tract degeneration in ALS. Ann. Clin. Transl Neurol. 2 (7), 748–755 (2015).
Shi, J. et al. Neurofilament markers in serum and cerebrospinal fluid of patients with amyotrophic lateral sclerosis. J. Cell. Mol. Med. 26 (2), 583–587 (2022).
Khalil, M. et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 14 (10), 577–589 (2018).
Gaiottino, J. et al. Increased neurofilament light chain blood levels in neurodegenerative neurological diseases. PLoS One. 8 (9), e75091 (2013).
Gaiani, A. et al. Diagnostic and prognostic biomarkers in amyotrophic lateral sclerosis: neurofilament light chain levels in definite subtypes of Disease. JAMA Neurol. 74 (5), 525–532 (2017).
Gong, Z. Y. et al. Neurofilament subunit L levels in the Cerebrospinal Fluid and serum of patients with amyotrophic lateral sclerosis. Neurodegener Dis. 18 (2–3), 165–172 (2018).
Verde, F. et al. Neurofilament light chain in serum for the diagnosis of amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry. 90 (2), 157–164 (2019).
Acknowledgements
This work was supported by grants from the Basic Research Project of Shanxi Province (202103021224405) and the Key Research and Development Project Plan of Shanxi Province concerning social advancement (201903D321049).
Funding
This study was supported by grants from the Basic Research Project of Shanxi Province (202103021224405) and the Key Research and Development Project Plan of Shanxi Province concerning social advancement (201903D321049).
Author information
Authors and Affiliations
Contributions
Q. conceived and designed the study, and wrote the manuscript. W. reviewed the manuscript. F. performed data analysis of NFs. Z. collected the clinical cases. W. performed statistic analysis. W. performed image of analysis. L. designed the study, reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qin, J., Wang, X., Fan, G. et al. Identifying amyotrophic lateral sclerosis using diffusion tensor imaging, and correlation with neurofilament markers. Sci Rep 14, 28110 (2024). https://doi.org/10.1038/s41598-024-79511-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79511-y