Abstract
Recent research has demonstrated a relationship between fruit and vegetable intake and gastrointestinal cancers, but the causality of these associations remains uncertain. This investigation sought to elucidate whether there is a potential causal relationship between the intake of fruits and vegetables and the risk of gastrointestinal cancers. Employing two-sample Mendelian randomization (MR), this research explored the causative influence of consuming fruits (fresh and dried) and vegetables (salads/raw and cooked) on the susceptibility to gastrointestinal cancers. The UK Medical Research Council-Integrative Epidemiology Unit (MRC-IEU) provided the summary statistics for these exposure variables, while the summary statistics for outcome data came from four other data sources. Univariable and multivariable MR were performed using inverse variance-weighted (IVW), MR-Egger, weighted media (WM), and Lasso model methods. Besides, multiple methods were employed for sensitivity analyses to guarantee the robustness of the findings, including MR-Egger intercept, Cochran’s Q test, and MR-PRESSO. Furthermore, the Phenoscanner V2 database was employed to identify possible confounders. The main analysis of univariable MR found that dried fruit consumption provided protection against pancreatic and oral cavity/pharyngeal cancers. However, this correlation becomes non-significant when potential confounders such as smoking, drinking, and body mass index (BMI) are accounted for. Furthermore, neither univariate nor multivariate MR analyses revealed enough data to prove a causal relationship between the intake of fresh fruit, vegetables (including salad/raw and cooked), and gastrointestinal cancers. There is insufficient evidence for a causal association between the consumption of fruits and vegetables and the occurrence of gastrointestinal cancers. Further empirical research is needed to corroborate these dietary factors’ role in the etiology of gastrointestinal cancers.
Similar content being viewed by others
Introduction
Cancers that arise in the digestive system and associated organs are known as gastrointestinal cancers. This includes cancers in the mouth, throat, esophagus, stomach, small intestine, and colorectum. Additionally, cancers in accessory organs like the pancreas, gallbladder, and liver also fall under gastrointestinal cancers1. According to the International Agency for Research on Cancer2, gastrointestinal cancers account for 30% of all cancers and 39% of all deaths due to cancer in 2020 worldwide, which poses a large global disease burden. Consequently, it is crucial to explore the etiology of gastrointestinal cancers, especially the modifiable lifestyle factors, to carry out primary prevention.
Diet happens to be a potentially modifiable lifestyle factor that leads to gastrointestinal cancers3. In recent years, it has been found that the consumption of fruits and vegetables has a preventive effect against certain cancers, especially gastrointestinal cancers, due to their high content of fiber and antioxidants4,5,6,7. A meta-analysis incorporating 18 prospective studies reveals that increased consumption of fruits alone and in combination with vegetables corresponds to a decrease in the probability of gastric cancer by 13% and 25%, respectively8. Similarly, the findings of a meta-analysis encompassing 32 studies involving 10,037 patients diagnosed with esophageal squamous cell carcinoma (ESCC) indicated a substantial risk reduction in ESCC correlated with elevated consumption of vegetables and fruits9. A study based on the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort suggests that intensive intake of fruits and vegetables is linked to a decreased incidence of distal colon cancer10. Conversely, some studies present different results between fruits and vegetables. A comprehensive meta-analysis of 24 studies involving more than 2.4 million individuals (6,632 gastric cancer events) revealed a significant protective effect of fruit consumption against gastric cancer risk while vegetable intake did not11. Not coincidentally, a meta-analysis comprising 19 studies with involvement of 1,290,045 participants (3,912 patients with hepatocellular carcinoma) suggested that a rise in the consumption of vegetables, not fruits, has been associated with a decline in the risk of hepatocellular carcinoma12. In addition, a comprehensive review has shown that the data supporting the negative correlation between substantial fruit and vegetable consumption and the susceptibility to colorectal cancer is not enough13. In conclusion, the existing studies examining the association between fruit and vegetable intake and gastrointestinal cancers present conflicting findings. Most of these are conventional observational investigations, potentially susceptible to confounding variables and reverse causality, thereby yielding conclusions that may be deemed unreliable. Therefore, a more reliable method is needed to investigate whether there is a causal relationship between the intake of fruits and vegetables and the risk of gastrointestinal cancers.
Mendelian randomization (MR), gaining popularity as a statistical approach, minimizes confounding variables and reverse causality by employing genetic variants, typically single nucleotide polymorphisms (SNPs), as instrumental variables (IVs). Since genetic variants are assigned at conception and unaffected by factors post-conception, their use as IVs in MR helps to reduce confounding variables and reverse causality14. Hence, this method enables the exploration of causal connections between the exposure and the outcome15. At this point, genetic variants need to meet three assumptions ( Fig. 1)16: Initially, genetic variants must be closely related to the exposure factors (Assumption 1). Subsequently, genetic variants must not be linked to any confounding factors (Assumption 2). Lastly, genetic variants can only affect gastrointestinal cancers through exposure factors (Assumption 3).
Schematic overview of the Mendelian randomization design. (By Figdraw). Abbreviations: SNP, single-nucleotide polymorphism; OC, oral cavity cancer; HC, pharyngeal cancer; EC, oesophageal cancer; LC, liver cancer; GC, gastric cancer; PC, pancreatic cancer; CRC, colorectal cancer.
Consequently, we intend to perform MR analysis to delve deeper into the relationship between the consumption of fruits and vegetables and the risk of six prevalent gastrointestinal cancers, with the ultimate aim of cancer prevention.
Materials and methods
Design of the study
To evaluate the potential causative effects of fruit and vegetable intake on six prevalent gastrointestinal cancers, initial univariable MR analysis were conducted. Given the vast variety of fruits and vegetables, analyzing the association between each specific type and gastrointestinal cancers would be impractical due to the extensive workload required. Therefore, we categorized fruits into fresh and dried, and vegetables into fresh and cooked, based on their nutritional differences and the dietary habits of different populations. This classification not only facilitates the design and implementation of the study but is also supported by relevant GWAS datasets from the UK Biobank, making it feasible for MR analysis.
In this analysis, the impacts of fresh and dried fruit, as well as cooked and salad/raw vegetable intake, were assessed separately. Additionally, recognizing a genetic correlation between fruit and vegetable consumption, subsequent multivariable MR analyses were employed to evaluate their independent effects simultaneously. Furthermore, to identify possible confounders that could induce pleiotropy, the Phenoscanner V2 database was employed. The variables including smoking, drinking, and body mass index (BMI) were selected for inclusion in the multivariable MR analyses. All the MR analyses utilized a two-sample method.
Source of data on fruit and vegetable consumption
The investigation incorporated factors such as the intake of fresh fruits, dried fruits, salads/raw vegetables, and cooked vegetables into its study of exposures. Genome-Wide Association Study (GWAS) summary statistics for these factors were sourced from The UK Medical Research Council-Integrative Epidemiology Unit (MRC-IEU) and can be accessed via the IEU Open GWAS project (https://gwas.mrcieu.ac.uk/). This prospective cohort study, which ran from 2006 to 2010, included around 500,000 participants. The primary aim is to explore diet-related genotypes. Comprehensive data on various phenotypes are collected through a standardized touch-screen questionnaire and anthropometric measurements.
Regarding fruit intake, participants were asked, “How many pieces of fresh or dried fruit do you consume daily?” They were instructed to respond with the mean (whole number) reflecting their intake over the preceding year. Non-consumers indicated “0,” while other options included “10,” “1,” or “3,” denoting quantities less than 1, uncertainty, and a choice not to answer, respectively. Finally, a total of 446,462 and 421,764 participants of European origin reported their fresh and dried fruit intake through the questionnaire. As for vegetable intake, the assessment was based on heaped tablespoons per day. Participants were asked, “On average, how many tablespoons of cooked (or salad/raw) vegetables do you consume daily?” The exposure to vegetable intake was established through the total reported average daily consumption of both cooked and salad/raw vegetables. This total daily amount in tablespoons was categorized into an ordinal scale: 0, 1–3, 4–6, and ≥ 7 tablespoons per day to reduce measurement bias. In total, 448,651 and 435,435 participants of European origin provided data on cooked vegetable intake and salad/raw vegetable intake through the questionnaire.
The questionnaire has undergone previous analyses to determine its effectiveness, with the results affirming its reliability and validity for data collection purposes17. Between August 2012 and June 2013, approximately 20,000 participants completed a comprehensive follow-up visit to the assessment center, using standardized questionnaire tools, about four years after their initial recruitment. The assessments demonstrated a robust 82% repeatability for cooked vegetables and fresh fruit, while salad/raw vegetables and dried fruits exhibited a slightly lower but respectable repeatability of 72%. These findings were consistent when compared to repeated assessments conducted four years later and a 24-hour dietary recall assessment.
Source of data on gastrointestinal cancers
The study focused on six prevalent gastrointestinal cancers: Colorectal, Gastric, Liver, Oesophageal, Pancreatic, and Oral cavity/pharyngeal cancer. These were designated as the outcomes of the research. The information on statistical datasets is outlined in Table 1.
The summary data for pancreatic cancer in the study were obtained from the PanScan1 consortium and included 1,896 cases and 1,939 controls, originating from 12 prospective cohort studies18. Summary statistics for GWAS concerning oral/pharyngeal cancer originated from the Oncoarray Oral Cavity and Oropharyngeal Cancer Consortium. Included within this dataset were 2,342 instances of cases and 2,329 instances of controls19. Data summarizing Colorectal cancer statistics were secured from the FinnGen biobank repository, encompassing 3,022 instances of the condition alongside 215,770 control subjects. The compendium of esophageal cancer statistics, including 740 instances and 372,016 comparative controls, has been procured from the UK biobank. Summary statistics for gastric cancer, derived from the FinnGen biobank, encompassed 633 instances of the condition alongside 218,159 control subjects. Furthermore, participants in the liver cancer dataset were derived from the FinnGen biobank, totaling 304 cases and 218,560 controls.
All GWAS summary statistics concerning the consumption of fresh fruits, dried fruits, salads/raw vegetables, and cooked vegetables, along with the aforementioned cancer datasets, exclusively involved subjects with European ancestry20. Furthermore, neither during nor after data collection did the authors have access to information that may be used to identify individual individuals. Notably, all the original studies acquired the ethical approval and participation agreement. Besides, ethical review and approval were waived for this study due to the use of public available data of IEU Open GWAS project.
Selection of IVs
To guarantee the precision and efficacy of the causal association between fruit and vegetable intake and the risk of gastrointestinal cancers, we restricted the inclusion criteria for IVs. The selection criteria included: (i) The SNPs were strongly associated with exposure at a genome-wide significance level (P < 5 × 10− 8). (ii) Some undesirable SNPs (r2 > 0.001, window size < 10,000 kb) were excluded through linkage disequilibrium clumping. (iii) To align with the third assumption of MR, any IVs that exhibited a strong association with outcomes or confounding variables (P < 5 × 10− 8) were excluded. (iv) The datasets concerning both exposure and outcomes underwent a harmonization process, after which SNPs displaying palindromic structures and possessing allele frequencies of an intermediate nature were removed. (v) For instances where fewer than four instrumental SNPs associated with exposure appeared in the datasets of outcomes, proxy SNPs were identified by employing variants in linkage disequilibrium (r² > 0.8, distance < 500 kb).
For each SNP, the F-statistic was utilized to determine the strength of correlation and mitigate bias introduced by weak IVs, thereby guaranteeing a strong relationship between IVs and factors of exposure. A threshold of F > 10 was set to indicate no weak IVs21. The calculation of the F-statistic for each single nucleotide polymorphism (SNP) was conducted in accordance with the prescribed formula:
where ‘N’ denotes the size of the sample within the exposure dataset, ‘K’ stands for the count of SNPs, and ' R2’ signifies the fraction of variability accounted for by instrumental variables within said dataset; ' R2’ was calculated using the formula:
here, ‘MAF’ denotes the frequency of the minor allele, a figure corresponding to EAF in the computation of R2, β represents the magnitude of the allele effect, and SD refers to the standard deviation22. Moreover, for each SNP, the summary-level association statistics were systematically organized across different GWAS to ensure that the estimations of their impacts were consistently referenced to the same alleles. Furthermore, IVs selected for inclusion in this study underwent scrutiny using the PhenoScanner V2 database (http://www.phenoscanner.medschl.cam.ac.uk/) to identify potential horizontal pleiotropic effects. Should any SNP exhibit a correlation with either the outcomes or potential confounding variables, such as alcohol drinking, smoking, and BMI, it is imperative that these SNPs be removed from the IVs before conducting the MR analysis.
Statistical analysis
The inverse variance-weighted (IVW) method was performed as the principal analysis to estimate the associations between fruit and vegetable intake and six prevalent gastrointestinal cancers. Through a meta-analysis, the IVW method aggregated Wald estimates for every single nucleotide polymorphism to provide an overall estimate. The total estimate was represented by the weighted regression slope of the result’s influence on the exposure’s effect, with the intercept limited to zero. Concurrently, sensitivity analyses were conducted using alternative methodologies, such as the weighted median (WM) and MR-Egger methods23. The MR-Egger intercept served to assess the possibility of pleiotropy in the IVW model. The WM, on the other hand, can generate reliable and accurate estimations, with at least 50% of the variations being deemed useful instruments24. These methods are predicated on various assumptions, and the persuasiveness of results about causation is increased when effects are consistent across several methods.
Within the scope of multivariable MR analyses, it employed both the IVW-random effects model and the Lasso model in a multivariable context to evaluate a causal association between the consumption of fruits and vegetables and the incidence of six common types of gastrointestinal cancers after adjusting for potential confounding phenotypes25.
Furthermore, Cochran’s Q test was applied to assess heterogeneity (a potential indicator of pleiotropy)26. The MR-Egger intercept test served to reveal directional pleiotropy. A proximate zero value of the intercept term (P < 0.05) suggested a minimal impact from genetic pleiotropy27. The MR-PRESSO was applied to evaluate horizontal pleiotropy by identifying and excluding outlier instrumental variables (NbDistribution = 1000, SignifThreshold = 0.05)28. Leave-one-out analyses were conducted to determine if a single SNP was responsible for the overall estimates29. Ultimately, scatter plots, funnel charts, and forest diagrams were used to visualize the findings from MR analysis.
The study was carried out using the R packages “MendelianRandomization” and “TwoSampleMR” in R software (version 4.3.1). A significant two-sided P-value for an univariable MR analysis was set at 0.05. Given the 408 MR estimations, a Bonferroni-corrected P-value of 0.05/408 (1.22 × 10− 4) was determined for multivariable MR analyses, whereas P < 0.05 was considered nominally significant.
Results
Instrumental variables
After screening, we identified 46, 41, 19, and 16 SNPs associated with fresh fruit, dried fruit, salad/raw vegetable, and cooked vegetable intake, respectively (Supplementary Tables 1–4). These SNPs had genome-wide significance (P < 5 × 10− 8) and were independent of each other (R² < 0.001). For our MR analyses on gastric cancer, colorectal cancer, esophageal cancer, liver cancer, pancreatic cancer, and oral cavity/pharyngeal cancer, the number of SNPs selected as IVs is listed below: 44, 44, 41, 44, 6, and 40 SNPs in fresh fruit intake; 37, 37, 35, 37, 8, and 32 SNPs in dried fruit intake; 16, 16, 15, 16, 5, and 15 SNPs in salad/raw vegetable intake; and 16, 16, 16, 16, 8, and 16 SNPs in cooked vegetable intake. Among them, only 3 and 1 instrumental SNPs for salad/raw vegetable and cooked vegetable intake were identified in relation to pancreatic cancer. Therefore, proxies (r² > 0.8, distance < 500 kb) were subsequently used. Furthermore, the F-statistic of all SNPs is > 10, indicating that there are no weak IVs (Supplementary Tables 1–4).
Univariable MR analysis
A univariable MR analysis was conducted to explore the association between fruit (fresh and dried) and vegetable (salad/raw and cooked) intake and six common types of gastrointestinal cancers (gastric, colorectal, oesophageal, liver, pancreatic, and oral cavity/pharyngeal cancer). All results are shown in Figs. 2 and 3.
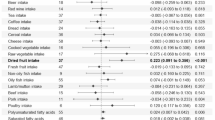
Forest plot of the causal association between fruit (fresh and dried) intake and six prevalent. Abbreviations: nSNP, number of SNPs; OR, odds ratio; IVW, inverse variance weighted; WM, weighted median.
Forest plot of the causal association between vegetable (salad/raw and cooked) intake and six prevalent. Abbreviations: nSNP, number of SNPs; OR, odds ratio; IVW, inverse variance weighted; WM, weighted median.
The IVW approach confirmed the causal association between dried fruit consumption and pancreatic cancer as well as oral cavity/pharyngeal cancer. The risk of cancer incidence decreases with increasing consumption of dried fruit. For every one standard deviation increase in dried fruit intake, the risk of pancreatic cancer was lowered by 97.3% (OR = 0.027, 95% CI: 0.001–0.678, P = 0.028); Similarly, the risk of oral cavity/pharyngeal cancer was lowered by 84.8% (OR = 0.152, 95% CI: 0.037–0.627, P = 0.09). The WM method likewise supported the link between dried fruit intake and pancreatic cancer risk (OR = 0.013, 95% CI 0.000-0.789, P = 0.04). However, the MR-Egger method found no significant connection (P = 0.32). Moreover, fresh and dried fruit intake levels were related to liver cancer (OR = 0.000, 95% CI 0.000-0.179, P = 0.03; OR = 0.000, 95% CI 0.000-0.308, P = 0.04) in the MR analysis with the MR-Egger method, but no significant connection was found in the other two MR methods. In addition, the three MR methods found no significant relationship between genetically predicted salad/raw and cooked vegetable intake levers and any of the six types of gastrointestinal cancer.
Scatter plots illustrate the distributions of individual ratio estimates for the consumption of fruits (fresh and dried) and vegetables (salad/raw and cooked) concerning their association with six prevalent gastrointestinal cancers. The trend lines obtained from the three MR approaches mentioned above imply a causal association, and the plot’s increasing slope implies a negative correlation. The multiple lines show the estimation of causal effects using various approaches. Funnel plots were used as visual tools to exhibit potential heterogeneity among effect estimations. The symmetry observed in these funnel plots, specifically within the IVW method, suggested that there was no heterogeneity. Notably, although the MR-Egger method revealed a significant association, the funnel plots for fresh and dried fruit intake levels on liver cancer were asymmetrical, indicating the presence of heterogeneity. All scatter plots and funnel plots are illustrated in Figs. 4 and 5.
The scatter plot displaying the causative estimate for various MR of fruit intake on and the funnel plot from single SNP analysis for fruit intake on. (A–F and M–R) scatter plot; (G–L and S–X) funnel plot. (A, G, M and S) gastric cancer; (B, H, N and T) colorectal cancer; (C, I, O and U) oesophageal cancer; (D, J, P and V) liver cancer; (E, K, Q and W) pancreatic cancer; (F, L, R and X) oral cavity/pharyngeal cancer; (A–L) fresh fruit intake; (M–X) dried fruit intake.
The scatter plot displaying the causative estimate for various MR of vegetable intake on and the funnel plot from single SNP analysis for vegetable intake on. (A–F and M–R) scatter plot; (G–L and S–X) funnel plot. (A, G, M and S) gastric cancer; (B, H, N and T) colorectal cancer; (C, I, O and U) oesophageal cancer; (D, J, P and V) liver cancer; (E, K, Q and W) pancreatic cancer; (F, L, R and X) oral cavity/pharyngeal cancer; (A–L) salad/raw vegetable intake; (M–X) cooked vegetable intake.
Multivariable MR analyses
Multivariable MR analyses were carried out to address potential confounding variables. Smoking, drinking, and BMI have been identified as risk factors for gastrointestinal cancers. Furthermore, the intake of fruits and vegetables interacts with smoking, drinking, and BMI. For example, individuals with higher fruit and vegetable intake tend to have a relatively lower BMI. Besides, some SNPs we selected correlated with these three factors through the PhenoScanner v2 database (Supplementary Table 5). Based on the reasons stated above, we looked through the IEU open GWAS for every trait associated with drinking, smoking, and BMI. Ultimately, three GWAS summary statistics (Supplementary Table 6) with a greater number of SNPs and relatively newer data sets were included in the multivariable MR analyses.
Using the IVW approach, the multivariable MR analysis indicated that fresh fruit intake had a nominally protective effect on liver cancer (OR = 0.052, 95% CI 0.003–0.940, P = 0.045); however, the results were not statistically significant following lasso regression. Moreover, this result did not meet the Bonferroni adjustment p-value criterion (1.22 × 10− 4). In addition, the multivariable MR analyses showed fresh fruit intake was not statistically significant for the remaining five gastrointestinal cancers. The dried fruit, raw/salad vegetables, and cooked vegetable intake were all insignificant for six prevalent gastrointestinal cancers (Figs. 6 and 7). In summary, there is not enough evidence to indicate a causal association between fruit and vegetable intake and gastrointestinal cancers from the multivariable MR analyses.
Multivariable MR analyses forest plot of the causal association between fruit (fresh and dried) intake and. Abbreviations: nSNP, number of SNPs; OR, odds ratio; IVW, inverse variance weighted; BMI, body mass index.
Multivariable MR analyses forest plot of the causal association between vegetable (salad/raw and cooked) intake and. Abbreviations: nSNP, number of SNPs; OR, odds ratio; IVW, inverse variance weighted; BMI, body mass index.
Sensitivity analysis
The present study performed thorough sensitivity analyses to validate the preliminary results derived from MR. Horizontal pleiotropy was identified using the MR-Egger intercept test and MR-PRESSO Global test to evaluate the robustness of the aforementioned analysis results. Additionally, heterogeneity was examined using Cochran’s Q test. As depicted in Supplementary Tables 7–10, all intercept terms were close to zero (P > 0.05), suggesting minimal susceptibility to potential biases. Concurrently, the results of the MR-PRESSO Global test are consistent with the MR-Egger regression, indicating the lack of gene pleiotropy. The application of Cochran’s statistical test indicated a lack of heterogeneity (Q p-value > 0.05). The impact of each IV was then evaluated using leave-one-out and MR-PRESSO analysis to see how robust the MR data were. As shown in Supplementary Figs. 1–4, the results of the leave-one-out testing showed that the MR findings were not significantly changed by the removal of any SNP. Moreover, the MR-PRESSO analysis revealed no outliers (Supplementary Tables 7–10). In conclusion, the sensitivity analyses confirmed the reliability and appropriateness of our MR analysis in examining the relationship between fruit and vegetable intake and six prevalent gastrointestinal cancers.
Discussion
As far as we know, this is the first MR analysis to thoroughly examine the causal connection between fruit and vegetable intake and gastrointestinal cancers. According to the main MR analysis, eating dried fruit provided protection against pancreatic and oral cavity/pharyngeal cancers (geographic region: Europe). Nevertheless, the causal effects lost statistical significance after accounting for smoking, drinking, and BMI and applying the Bonferroni correction. Thus, our study does not provide adequate evidence to support the proposition that the intake of fruits and vegetables delivers a protective effect against gastrointestinal cancers.
There is mounting evidence suggesting that dietary factors contribute to cancer development. While healthy diets are often advocated for cancer prevention, a universally accepted definition of “healthy diets” remains elusive. Nevertheless, the consensus acknowledges that increasing fruit and vegetable consumption is beneficial for health. This is because different kinds of fruits and vegetables offer various nutrients and bioactive compounds, such as phytochemicals, vitamins, minerals, and fibers30. The health benefits associated with the intake of fruits and vegetables stem from the synergistic interactions among bioactive compounds and various nutrients inherent in whole foods31. Furthermore, current research indicates that bioactive compounds found in fruits and vegetables exhibit diverse mechanisms of action, including antioxidant and antiproliferative activities32.
Fruit and vegetable consumption has been extensively recognized to have anticancer benefits. Previous research has shown that fruit consumption has a protective effect against pancreatic cancer and oral/pharyngeal cancer6,7. Our preliminary findings from univariable MR analysis support this observation. Nonetheless, the protective effect became statistically insignificant after the multivariate MR analysis adjusted for confounders. Additionally, previous research suggests that vegetable consumption may lower the risk of liver cancer, gastric cancer, ESCC, and colorectal cancer8,9,10,12, whereas those in our studies were not verified. Notably, prior research has noted that the inverse association between total fruit intake and pancreatic cancer risk was particularly pronounced among never smokers7. Thus, we speculate that this phenomenon may be attributed to the susceptibility of conventional observational studies to confounding factors and reverse causality, potentially resulting in unreliable conclusions.
There were also previous studies that are consistent with our current results. A large cohort research by Mirjam M. et al. found no correlation between the intake of fruits and vegetables and pancreatic cancer33. A pooled Analysis of 14 cohort studies indicated fruit and vegetable intakes were not strongly associated with colon cancer risk overall10. Based on this result, we speculate that specific foods and food groups may reveal particular components of fruits and vegetables associated with risk. However, this effect could be diluted in an analysis of total fruit and vegetable consumption. Notably, there also have been a few studies focusing on individual fruits or vegetables. Research has shown that spinach intake has been linked to a reduced risk of colon cancer, while other studies also have reported a lower risk of colon cancer associated with banana consumption34,35,36. The findings of this study challenge the conventional belief that increased consumption of fruits and vegetables can prevent tumors. Moreover, these results can provide valuable insights for the improvement and revision of dietary guidelines. Lastly, this study may encourage researchers and healthcare professionals to shift their focus. Many observational studies currently focus on the total intake of fruits and vegetables, rather than examining specific types of fruits or vegetables. We believe that when studying overall fruit and vegetable consumption, the effects of individual varieties may be overlooked, which could diminish their clinical value. Therefore, these findings also provide some direction for future related research.
In this study, two-sample MR analyses were used to evaluate the causal link between fruit and vegetable intake and gastrointestinal cancers. Our study offers several advantages over earlier research. Firstly, the MR analysis significantly reduces the impact of reverse causality and confounding variables. Subsequently, many kinds of sensitivity analyses were performed to fortify the solidity of our findings. Furthermore, to eliminate bias, we applied non-overlapping exposure and outcome summary statistics (oesophageal cancer being the exception) derived from different consortiums. Lastly, multivariable MR analyses were employed to avoid associated pleiotropy caused by possible confounding variables. Even with a robust analysis, a few fundamental limits need to be noted. First, the use of genetic variants as IVs in MR analyses introduces the possibility of different allele frequencies and effects across diverse populations. In our current study, the individuals were of European ancestry. Our results regarding gastrointestinal cancers may not be reinforced by corroborative evidence from varied datasets. Therefore, one should treat carefully when applying these results to different populations. In addition, our study was on the intake of fruits and vegetables in general but not on specific types of fruits or vegetables. Consequently, the associations between specific food and gastrointestinal cancers could be diluted. Future research might benefit from distinguishing between different types of fruits and vegetables as well as cooking methods. Moreover, the outcome summary statistics of oesophageal cancer we used are from the UK biobank. There is some sample overlap with our exposure data. Therefore, the results of oesophageal cancer may be somewhat biased and do not take advantage of the MR study, which is free from confounding factors and may have served a similar function to a traditional observational study.
Conclusions
To summarize, a thorough study was conducted into the potential causative link between the consumption of fruits and vegetables and the incidence of gastrointestinal cancers. There is insufficient data to establish a causal relationship between fruit and vegetable intake and gastrointestinal cancers. Our research offers novel perspectives on the risk factors and mechanisms underlying gastrointestinal cancers, necessitating subsequent corroboration via further observational and experimental investigations.
Data availability
The original data are available through the IEU open GWAS project (https://gwas.mrcieu.ac.uk/). The phenotypes of all SNPs were obtained from Phenoscanner V2 (http://www.phenoscanner.medschl.cam.ac.uk/).
References
Keum, N. et al. Association of physical activity by type and intensity with digestive system cancer risk. JAMA Oncol. 2, 1146–1153 (2016).
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Kim, J. et al. Plant-based dietary patterns and the risk of digestive system cancers in 3 large prospective cohort studies. Eur. J. Epidemiol. 38, 617–627 (2023).
Slavin, J. L. & Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 3, 506–516 (2012).
Wang, T., Masedunskas, A., Willett, W. C. & Fontana, L. Vegetarian and vegan diets: benefits and drawbacks. Eur. Heart J. 44, 3423–3439 (2023).
Freedman, N. D. et al. Fruit and vegetable intake and head and neck cancer risk in a large United States prospective cohort study. Int. J. Cancer 122, 2330–2336 (2008).
Yamagiwa, Y. et al. Fruit and vegetable intake and pancreatic cancer risk in a population-based cohort study in Japan. Int. J. Cancer 144, 1858–1866 (2019).
Naemi Kermanshahi, M. et al. Fruit and vegetable intake in relation to gastric cancer risk: a comprehensive and updated systematic review and dose-response meta-analysis of cohort studies. Front. Nutr. 10, 973171 (2023).
Liu, J., Wang, J., Leng, Y. & Lv, C. Intake of fruit and vegetables and risk of esophageal squamous cell carcinoma: A meta-analysis of observational studies. Int. J. Cancer 133, 473–485 (2013).
Koushik, A. et al. Fruits, vegetables, and colon cancer risk in a pooled analysis of 14 cohort studies. J. Natl. Cancer Inst. 99, 1471–1483 (2007).
Wang, Q. et al. Consumption of fruit, but not vegetables, may reduce risk of gastric cancer: Results from a meta-analysis of cohort studies. Eur. J. Cancer 50, 1498–1509 (2014).
Yang, Y. et al. Increased intake of vegetables, but not fruit, reduces risk for hepatocellular carcinoma: a meta-analysis. Gastroenterology 147, 1031–1042 (2014).
Wiseman, M. & The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc. Nutr. Soc. 67, 253–256 (2008).
Smith, G. D. et al. Clustered environments and randomized genes: A fundamental distinction between conventional and genetic epidemiology. PLoS Med. 4, e352 (2007).
Smith, G. D. & Ebrahim, S. Mendelian randomization: Can genetic epidemiology contribute to understanding environmental determinants of disease? Int. J. Epidemiol. 32, 1–22 (2003).
Larsson, S. C., Burgess, S. & Michaëlsson, K. Association of genetic variants related to serum calcium levels with coronary artery disease and myocardial infarction. JAMA 318, 371–380 (2017).
Bradbury, K. E., Young, H. J., Guo, W. & Key, T. J. Dietary assessment in UK Biobank: An evaluation of the performance of the touchscreen dietary questionnaire. J. Nutr. Sci. 7, e6 (2018).
Amundadottir, L. et al. Genome-wide association study identifies variants in the ABO locus associated with susceptibility to pancreatic cancer. Nat. Genet. 41, 986–990 (2009).
Lesseur, C. et al. Genome-wide association analyses identify new susceptibility loci for oral cavity and pharyngeal cancer. Nat. Genet. 48, 1544–1550 (2016).
Zhu, G. L. et al. Causal relationship between genetically predicted depression and cancer risk: a two-sample bi-directional mendelian randomization. BMC Cancer 22, 353 (2022).
Burgess, S., Thompson, S. G. & CRP CHD Genetics Collaboration. Avoiding bias from weak instruments in mendelian randomization studies. Int. J. Epidemiol. 40, 755–764 (2011).
Davey Smith, G. & Hemani, G. Mendelian randomization: Genetic anchors for causal inference in epidemiological studies. Hum. Mol. Genet. 23, R89–98 (2014).
Sanderson, E., Davey Smith, G., Windmeijer, F. & Bowden, J. An examination of multivariable mendelian randomization in the single-sample and two-sample summary data settings. Int. J. Epidemiol. 48, 713–727 (2019).
Bowden, J., Davey Smith, G., Haycock, P. C. & Burgess, S. Consistent estimation in mendelian randomization with some Invalid instruments using a weighted median estimator. Genet. Epidemiol. 40, 304–314 (2016).
Rees, J. M. B., Wood, A. M. & Burgess, S. Extending the MR-Egger method for multivariable mendelian randomization to correct for both measured and unmeasured pleiotropy. Stat. Med. 36, 4705–4718 (2017).
Cohen, J. F. et al. Cochran’s Q test was useful to assess heterogeneity in likelihood ratios in studies of diagnostic accuracy. J. Clin. Epidemiol. 68, 299–306 (2015).
Bowden, J., Davey Smith, G. & Burgess, S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int. J. Epidemiol. 44, 512–525 (2015).
Verbanck, M., Chen, C. Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from mendelian randomization between complex traits and diseases. Nat. Genet. 50, 693–698 (2018).
Hemani, G. & Bowden, J. Davey Smith, G. evaluating the potential role of pleiotropy in mendelian randomization studies. Hum. Mol. Genet. 27, R195–R208 (2018).
Liu, R. H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 4, 384S–92S (2013).
Liu, R. H. Potential synergy of phytochemicals in cancer prevention: mechanism of action. J. Nutr. 134, 3479S–3485S (2004).
Liu, R. H. & Finley, J. Potential cell culture models for antioxidant research. J. Agric. Food Chem. 53, 4311–4314 (2005).
Heinen, M. M., Verhage, B. A. J., Goldbohm, R. A. & van den Brandt, P. A. Intake of vegetables, fruits, carotenoids and vitamins C and E and pancreatic cancer risk in the Netherlands cohort study. Int. J. Cancer 130, 147–158 (2012).
Wu, Z. Y., Chen, J. L., Li, H., Su, K. & Han, Y. W. Different types of fruit intake and colorectal cancer risk: a meta-analysis of observational studies. World J. Gastroenterol. 29, 2679–2700 (2023).
Deneo-Pellegrini, H., De Stefani, E. & Ronco, A. Vegetables, fruits, and risk of colorectal cancer: A case-control study from Uruguay. Nutr. Cancer 25, 297–304 (1996).
Deneo-Pellegrini, H. et al. Plant foods and differences between colon and rectal cancers. Eur. J. Cancer Prev. 11, 369–375 (2002).
Acknowledgements
We are grateful for the work of the IEU open GWAS project and Phenoscanner V2 database.
Funding
This study was supported by grants from the D&R Program of Liaoning Province (2022-BS-256).
Author information
Authors and Affiliations
Contributions
Conceptualization, J.W.; Data curation, Q.F. and H.L.; Formal analysis, J.W.; Funding acquisition, K.Z.; Investigation, J.W.; Methodology, J.W.; Project administration, L.L.; Resources, Y.X.; Software, Y.X., Q.F. and H.L.; Supervision, L.Li.; Validation, L.L.; Visualization, Y.X. and Q.F.; Writing—original draft, J.W.; Writing—review & editing, K.Z. and L.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wei, J., Xiao, Y., Feng, Q. et al. The association between fruit and vegetable intake and gastrointestinal cancers risk from Mendelian randomization analysis. Sci Rep 14, 28860 (2024). https://doi.org/10.1038/s41598-024-79650-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79650-2