Abstract
X-irradiation of blood products is an alternative for gamma-ray to prevent post-transfusion GvHD. However, commercial X-irradiators are not widely available while little is known about their safety and efficacy for platelet products. This study introduces an efficient, accessible and cost-effective “X irradiation system” for platelet concentrates (PCs). By constructing a suitable radiation box (phantom) for a clinically available linear accelerator, an “X irradiation system” was designed specifically for PCs. PCs were divided into three equal bags either exposed to X- and gamma-irradiation or kept unirradiated (control). Irradiation-induced inhibition of T cells proliferation was examined by MTT and cell cycle assays on mononuclear cells (MNCs) obtained from PCs. The inhibitory effect of irradiation on allorecognition ability of MNCs was assessed by mixed lymphocyte reaction where MTT evaluated lymphocyte proliferation responses and flowcytometry examined CD8+T lymphocytes activity. Platelet activation was also examined with P-selectin expression and PAC-1 binding by flowcytometry. X- and gamma-irradiation reduced T cell proliferation while disturbing the cell-cycle with reduced entry of T-cells into the S phase and their G2 arrest. Both types of irradiations also effectively reduced “lymphocyte allorecognition responses” while inactivating CD8+T lymphocytes in platelet products but with no significant effect on platelet activity. This is the first study that showed “X irradiation system” effectively suppresses T cell proliferation and CD8+T lymphocyte activity in platelet products, with no effect to platelet quality and activation markers. This may suggest the LINAC-based “X irradiation system” with a dose of 30Gy as efficient and safe as gamma-irradiation for platelet products.
Similar content being viewed by others
Introduction
Platelet products are mainly administered in cases of thrombocytopenia and platelet dysfunction1,2. Platelet transfusions represent only 10% of the total transfused blood products to recipients, while they account for more than 25% of all post-transfusion adverse events reported by healthcare authorities. Transfusion-associated-graft versus host disease (TA-GvHD), a crucial adverse reaction of platelet transfusion with a high mortality rate is a rare but serious condition which predominantly occurs in immunocompromised patients3. In this context, a systematic review carried out by Kopolovic I et al.4, was found that platelet products were involved in 20 cases of TA-GVHD out of a total of 243 cases. This study clarified platelet products as the third most significant blood component associated with TA-GVHD. The most effective method to prevent TA-GVHD is irradiation using ionizing rays including x and γ, with a recommended dose of 25–50 Gy5,6. In recent years, gamma-irradiators have been gradually replaced by commercial X-irradiators as the preferred option for irradiating blood products. Currently, at least four major companies manufacture commercial FDA-approved X-irradiators. However, due to their high cost, these instruments are not easily accessible in all medical healthcare facilities. In situations where X-irradiators are not available, a linear accelerator (LINAC) as a device for cancer radiation therapy can be a cost-effective and accessible alternative to X-ray irradiation of blood products. Considering that the majority of large medical centers have LINAC, the use of this equipment for blood irradiation not only reduces costs, but also facilitates access to irradiated blood components in a shorter period of time7. So far, several researches have been performed to replace X-irradiators with the “blood products X irradiation system” consisting of a LINAC and a suitable radiation space specially a box called “phantom” adjusted for appropriate dose distribution of radiation. However, so far, no research has focused on designing such a system for X-irradiation of platelet products. In an earlier study, in 1997, Moroff et al. have suggested utilization of two sheets of thick plastic on the upper and lower sides of blood bags8, the design that provided a space for irradiation, ensuring consistent and uniform exposure. Few years later, employing Perspex sheets, Buston et al.9 presented an irradiation chamber generally called as “phantom” which further developed by other studies such as those of designing a special irradiation box named “thermal device” which consisted of a double acrylic wall filled by foam10 or a phantom that made of Polymethylmethacrylate7. Using these innovative designs, all studies not only ensure consistent and uniform irradiation, but also demonstrate high efficacy and quality for blood components. In this regard, LINACs which serve as the fundamental basis for modern commercial X-irradiators, continue to show a reliable ability to function as a viable alternative in scenarios where commercial irradiations are not are not accessible. Nevertheless, there is still insufficient evidence of specific effects of X-irradiation on platelet products. Hence, the study presented here, for the first time attempted to design and develop a suitable “X irradiation system” exclusively for platelet products. Following the confirmation of the irradiation dose, the effects of irradiation on lymphocyte and platelet activation were investigated. Two different approaches were utilized to examine the effects of irradiation on lymphocytes. The initial approach centered on evaluating the proliferative response of T cells to a mitogen, phytohemagglutinin (PHA). The second approach focused on assessing the activity and functional response of T cells through alloantigen stimulation. In order to measure the proliferative response of T cells to PHA, two assays were set up and conducted: a proliferation assay using MTT and a cell cycle assay using propidium iodide (PI). To investigate the functional response of T cells to alloantigen, a mixed lymphocyte reaction (MLR) assay is designed where subsequently the flow cytometry technique was also applied to examine the expression of CD8+T lymphocyte activity markers. Additionally, the impact of irradiation on activation of platelets was assessed by examining the P-selectin expression and PAC-1 binding (evaluating the activated form of GPIIb/IIIa) by flow cytometry. It is noteworthy that although the effect of X-irradiation on platelet phenotype and activation state in platelet products has been previously examined in at least one research, this is the first attempt to properly assess the inhibitory effect of X-irradiation on functional activity of CD8+T cells, as the main players in post transfusion GvHD, in platelet products.
Materials and methods
Reagents and chemicals
MTT (thiazolyl blue tetrazolium bromide), Mitomycin C, and propidium iodide (PI) obtained from Sigma (USA). RPMI 1640, FBS, and phytohemagglutinin (PHA)-M were from Invitrogen (USA). Pen/strep purchased from Biosera (China). Sodium citrate, DMSO, and triton X-100 were from Merck (Germany). Monoclonal antibodies against human CD8, CD25, CD62-P (conjugated with PE fluorochrome), CD69, PAC-1 (conjugated with FITC), and Mouse IgG1 Isotype control (PE and FITC conjugated), all of which purchased from BD Pharmingen (USA).
X irradiation system
Phantom design and construction
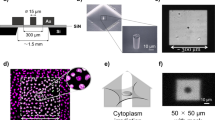
The first step in the development of a practical procedure for blood product exposure involves the designing and construction of an appropriate phantom. The phantom designed in the current study is specifically constructed to accommodate a single platelet product. It is important to note that the phantom was fabricated using Perspex which is a well-known material in phantom and accessories construction in radiotherapy departments due to its water equivalent material properties, physical resistance, low cost, and availability. The internal and external dimensions of the designed box as well as its wall thickness are 25 × 17 × 2cm3 and 29 × 21 × 6cm3 and 2 cm, respectively. A specific space allocated within the phantom as an ionization chamber to place a dosimeter (e.g., farmer) for online monitoring on demand. Additionally, the lid of the phantom is also made of Perspex with dimensions of 21 × 6cm2 and thickness of 2 cm to be sure that the platelet product is located behind the build-up region of the 6MV photons and keep the experiment more precise and reproducible (Fig. 1A).
X-Irradiation setup. To irradiate the platelet products, an “X irradiation system” consisting of a LINAC treatment machine and a phantom was applied. (A) shows an irradiation box (phantom) designed and constructed in this project to accommodate a single platelet product. This phantom is fabricated using Perspex and the internal/ external dimensions and thickness are 25 × 17 × 2 cm3, 29 × 21 × 6 cm3 and 2 cm, respectively. The lid of the phantom is also made of Perspex with dimensions and thickness of 21 × 6 cm2 and 2 cm to be sure that the platelet product is located behind the build-up region of the 6 MV photons and keep the experiment more precise and reproducible. A specific space allocated within the phantom as an ionization chamber to place a dosimeter (e.g., farmer) for online monitoring on demand. (B) demonstrates a sample view of treatment planning results for the filled phantom, in which green isodose dedicates the regions which are expected to get at least 95% of the prescribed dose. (C) displays phantom set up at the treatment room on the LINAC table.
Treatment planning
After phantom construction, a platelet product bag was filled with water and positioned in the phantom. Three dedicated Computed tomography (CT) markers were placed on the phantom surface indicating the CT laser intersections which were used for accurate phantom setup later in the treatment room. Imaging of the phantom with the water-filled platelet product bag was done using a CT scanner (Philips Spectral CT 7500) with a slice thickness of 3 mm. The captured image series was then transferred to the treatment planning system (TPS; Monaco®, Elekta AB, Stockholm, Sweden). The filled bag was contoured as the target organ. Treatment planning and field arrangement were done by an experienced medical physicist to obtain a uniform dose distribution and deliver the prescribed dose. The final output of the TPS was assigned filed set up and the monitor units (MUs) of each field that should be exposed by the linear accelerator (LINAC; Elekta Synergy®, Elekta AB, Stockholm, Sweden) treatment machine. As a summary, a photon beam of 6 MV was selected with dose rate of 507MU/min (i.e., about 5 Gy/min) where an anterior-posterior (AP) field was defined with both gantry and collimator angle of zero and field size of 35 × 35cm2 (Fig. 1B). The exposure set-up was selected based on the source-to-surface distance (SSD) which is equal to 100 cm at LINAC-center. Finally, monitor unit (MU) was obtained by the three-dimensional (3D) dose distribution based on the prescription doses of 25, 30, and 35 Gy to the delineated bag. It should be noted that the LINAC undergo regular quality assurance (QA) procedures to ensure the accuracy and precision of its output which generally means that it’s 1MU be equal to 1 cGy under the dosimetry standard conditions (i.e., SSD = 100 cm, field size = 10 × 10cm2, and measurement depth = 10 cm). This will ensure the patients’ treatment precision and accuracy for their every treatment fraction. Additionally, using online dosimetry, a capability considered during the phantom construction can help to ensure the accuracy of bag irradiation in case of any debate.
Phantom irradiation
At the treatment room, the whole explained plan set up was adjusted (Fig. 1C). A platelet product bag was then subjected to X-ray photon irradiation for doses of 25, 30, and 35 Gy in subsequent steps. The non-irradiated (control) platelet product bags were kept out of the treatment room during the whole procedure. After irradiation, the irradiated and non-irradiated platelet bags were placed in the incubator until sampling for laboratory tests.
Platelet-rich plasma-platelet concentrates (PRP-PC) collection
The platelet product bags were prepared from eligible volunteer blood donors after obtaining informed consent by the Blood Transfusion Organization where each product met the standard protocol and subsequently divided into three bags with equal weight using a digital balance (Sartorius, Germany) before irradiation. One platelet product bag was subjected to X-ray using a “X irradiation system” consisting of a linear accelerator (LINAC) treatment machine and a phantom (an irradiation box designed and constructed in this project). At the same time, the second platelet product bag was irradiated with 30Gy gamma (6 min at a rate of 5Gy/min) using a blood irradiator (Bio Beam 8000) that uses a cesium-137 radiation source. The last platelet bag was kept as a non-irradiated control bag. The research received approval from a regional Ethics Committee of the High Institute for Research and Education in Transfusion Medicine, Tehran, Iran (Ethics approval number: IR.TMI.REC.1401.017& IR.TMI.REC.1402.001). Accordingly, it is also noteworthy that all methods were performed in accordance with the relevant guidelines and regulations.
Sample preparation
Under sterile conditions, two separate samples were collected from non-irradiated platelet bag and X- or gamma-irradiated platelet bags. The first sample was used to isolate MNCs by density gradient centrifugation. These MNCs were then suspended in a growth medium (RPMI-1640, FBS 10%, pen-strep 1%) with the concentration of 1 × 106/mL and after treatment with PHA 1% were incubated at 37 °C/5% CO2 for intervals of 12 to 24 h. The second sample was resuspended in Tyrode’s buffer11 with a concentration of 2 × 107/mL for platelet flow cytometry analysis.
T lymphocyte proliferation/cell cycle assays
Microscopic images of cell colony expansion
Microscopic images of MNCs obtained from irradiated and non-irradiated platelet products in different conditions were captured at intervals of 12 to 24 h of culture. Then total surface area covered by T cell colonies (µm2) was calculated using ImageJ software (Research Services Branch, National Institute of Mental Health, Bethesda, MD, USA).
Proliferation assay
To assess the impact of irradiation on lymphocyte viability and proliferation response, MTT assay was applied which is a colorimetric method based on the cleavage of a water-soluble dye, MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) into an insoluble Formazan in living cells as an estimate of the number of living cells or the rate of metabolic activity. Following 24-hour culture of MNCs obtained from irradiated and non-irradiated platelet products in the presence of PHA 1%, MTT solution (5 mg/mL) was added into each well and the plate was incubated for 4-hours at 37˚C / 5%CO2. The plate was then centrifuged at 4500RPM for 15 min, and the supernatant was subsequently removed. To dissolve the insoluble Formazan, DMSO was added and finally the optical density (OD) of blue-purple color solution produced was measured at 570 nm using a microplate-reader.
Cell cycle assay
To evaluate the impact of irradiation on T cell cycle perturbations, following 12 to 24-hour culture of MNCs obtained from irradiated and non-irradiated platelet products in the presence of PHA 1%, the cells were washed and resuspended in a dye solution containing PI (6 µg/mL), sodium citrate (0.01%), PBS, triton x-100, and RNase (100 µg/mL). The cells were then incubated at 37˚C/ 5%CO2 for 30 min and subsequently, cell cycle distribution was analyzed using a flow cytometer (CyFlowSpace, Partec GmbH, Germany). It is important to highlight that in order to analyze lymphocytes; the whole fraction of MNCs was examined based on the gating strategy. Although a small portion of B lymphocytes, monocytes, and natural killer (NK) cells may remain in the sample, all proliferating cells analyzed are anticipated to be T lymphocytes, since PHA specifically stimulates their proliferation.
Mixed lymphocyte reaction (MLR) assay
Lymphocyte proliferative response
To evaluate lymphocyte proliferation response following any intervention such as irradiation, the ability of responder cells proliferation in response to allorecognition of inactivated stimulator cells obtained from a healthy donor was measured through the 36-hour12,13 culture of non-irradiated (control), either X- or gamma-irradiated responder cells (1 × 106 cells) obtained from platelet products (day 1 and 4 of storage) with an equal number of non-irradiated stimulator cells. Notably, stimulator cells were previously inactivated using 50 µg/mL mitomycin-C for 30 min at 37 °C in order to stop their proliferation14. The proliferation of responder cells against inactivated stimulator cells was then evaluated with MTT assay after 4-hour at 37 °C/5% CO2 where the optical density (OD) at 570 nm was measured using a microplate reader. Cell Stimulatory Index (SI) was also calculated to evaluate the inhibitory effect of irradiation on cell proliferation. For this purpose, OD of inactivated stimulator cells was subtracted from the OD of the mixture of inactivated stimulator cells with responder cells. Then, the OD of irradiated cells was divided into the OD of non-irradiated cells. In that case, if irradiation has an inhibitory effect on cell proliferation, the cell stimulatory index should be less than 1.
Lymphocyte activation response
To assess lymphocyte activation response following irradiation, the capability of responder cells activation in response to allorecognition of inactivated stimulator cells was evaluated by the co-culture of non-irradiated (control) and either X- or gamma-irradiated responder cells (1 × 106 cells) obtained from platelet products (day 1 of storage) with an equal number of inactivated stimulator cells. Notably, in order to stop the proliferation, the stimulator cells were previously inactivated using 50 µg/mL mitomycin-C for 30 min at 37 °C. For this purpose, 24-hour and 48-hour co-culturing of responder-stimulator cells at 37 °C/5% CO2 were applied to detect the expression of CD69 and CD25 respectively as early and late activation markers of lymphocytes on CD8+T lymphocytes by flow cytometry.
Assessment the quality markers of platelets
Platelet counts and swirling were analyzed as general quality markers of platelet products. pH also monitored to evaluate and compare metabolic state of each product15. For this purpose, the designated volumes of PRP obtained from both X- and gamma-irradiated as well as non-irradiated PCs were subjected to a pH meter (826 pH mobile/827 pH lab, Metrohm AG, Switzerland). The pH assessment was performed at a temperature of 22 °C.
Flow cytometry analysis to monitor platelet activation
Firstly, platelets were stained with anti-P-selectin as an activation marker and antibody against which recognizes an epitope on the glycoprotein IIb/IIIa (GPIIb/IIIa) complex of activated platelets (PAC-1 binding). Following a 30 min incubation period, the samples subjected to a flow cytometer to measure the expression levels of P-selectin as well as PAC-1 binding on platelets as already described16.
Statistical analysis
The data was analyzed using GraphPad Prism software (GraphPad Prism Software, Inc., San Diego, CA). The mean and standard deviation (SD) were calculated. The Wilcoxon matched-pair signed-ranks test was used to compare parameters between paired groups of each run that was irradiated in the same set, while the Mann-Whitney U test was used to compare the results obtained from independent runs of different groups. Significance was attributed to p-values that were less than 0.05.
Results
The impact of irradiation on T lymphocyte proliferation/cell cycle
Cell colony expansion
To evaluate T cell proliferation capability, isolated MNCs from irradiated and non-irradiated platelet products, were treated with PHA 1%. After 12- and 24-hour culture, when T cells started to proliferate and form colonies, microscopic images were captured. The data presented in Fig. 2 demonstrate that the colony area in the non-irradiated (control) cells is considerably greater in size when compared to the irradiated ones (P < 0.001). It is worth noting that there was no notable difference between the X- and gamma-irradiated cells either at 12- or 24-hours culture.
Microscopic images of colony expansion of radiation-exposed cells after PHA treatment. Microscopic images of T cell colonies following 12- and 24-hour culture of platelet product-derived MNCs in the presence of PHA 1% (a mitogen that specifically induces T cell expansion) were captured. Image (A) displays T cell colonies after 12-hour culture, whereas image (B) shows the colonies after 24-hours. Graphs (C) and (D) illustrate the significantly (P < 0.001) inhibitory effects of both gamma- and X-irradiations on T cell expansion and colony formation during both 12-hour and 24-hour culture, respectively. n = 5; ***, P < 0.001; ns, non-significant; MNC, mononuclear cell; PHA, phytohemagglutinin.
T lymphocyte viability
The lymphocyte viability was assessed using the MTT assay on MNCs obtained from X- or gamma irradiated non-irradiated platelet products. As depicted in Fig. 3A, neither X-irradiation nor gamma-irradiation affected lymphocyte viability when compared to non-irradiated lymphocytes.
T lymphocyte viability/ proliferation following irradiation. After exposure of platelet products to X- or gamma-irradiations, platelet product-derived MNCs alone were used to assess cell viability, as well as MNCs treated with PHA 1% for 24-hours was applied to the assay of proliferation. The cell viability did not show significant changes in irradiated cells compared to the non-irradiated ones (A). However, our results revealed that irradiations effectively inhibit (P < 0.01) the T cell proliferation response to PHA when compared to non-irradiated samples (B). n = 5; **, P < 0.01; ns, non-significant; MNC, mononuclear cell; PHA, phytohemagglutinin.
T lymphocyte proliferation
To investigate the effect of irradiation on T cell capacity to proliferate in response to PHA 1%, MTT assay was performed after a 24-hour culture of MNCs obtained from X- or gamma-irradiated and non-irradiated platelet products. As shown in Fig. 3B, irradiation significantly induces reduction in T cell proliferation response (P < 0.01), however, there were no significant difference observed between X- and gamma-irradiation.
T cell cycle perturbations
Cell cycle assay was conducted using PI staining to investigate the impact of X- and gamma-irradiation on the suppression of T cell proliferation capacity. For this purpose, the MNCs obtained from irradiated and non-irradiated platelet products was treated with PHA 1% for 12- and 24-hour then underwent PI staining. Representative figures demonstrate X- (Fig. 4G) and gamma (Fig. 4H) -irradiation-induced perturbations of T cell cycle when compared to the non-irradiated ones (Fig. 4F) after 24 h of culture with PHA 1%. As shown in Fig. 4A and E, irradiation has a notable disruptive effect on the T cell cycle, so that it not only hinders T cell entry into the S phase of the cell cycle in both intervals of 12 (P < 0.05) and 24-hours (P < 0.01) but also leads to G2 arrest in both intervals of 12- to 24-hours (P < 0.05). Noteworthy, there was no significant difference in the perturbations of T cell cycles between X-irradiation and gamma-irradiation.
T cell cycle perturbations following irradiation. Following 12- and 24-hour cultures of MNCs obtained from X- or gamma-irradiated and non-irradiated platelet products in presence of PHA 1%, propidium iodide (PI) staining was performed to evaluate the T cell cycle. Bar chart (A) illustrates the cell cycle distribution of X-irradiated (doses of 25, 30, and 35 Gy) and 30 Gy gamma-irradiated cells after a 12-hour culture in the presence of PHA 1%, whereas bar hart (B) depicts the cell cycle distribution of X- and gamma-irradiated cells at a dose of 30 Gy following a 24-hour culture in presence of PHA 1%. Although during 12- and 24-hour cultures in presence of PHA 1%, the G1 phase in X- and gamma-irradiated cells at a dose of 30 Gy increased compared to the non-irradiated ones, however this increase was not statistically significant (C) (P = 0.06). Conversely, the S phase of irradiated cells significantly diminished compared to the non-irradiated ones after 12-hour (P < 0.05) and 24-hour (P < 0.01) cultures (D). However, the G2 phase of irradiated cells significantly increased (P < 0.05) compared to the non-irradiated ones which means there was a G2 arrest following X- and gamma-irradiation in both 12- and 24-hour cultures in presence of PHA 1% (E). Figure 4G and H show the representative T cell cycle perturbations following X- and gamma-irradiation, respectively in compared to non-irradiated ones (4 F) after 24 h of culture with PHA 1%. n = 5; *, P < 0.05; **, P < 0.01; ns, non-significant; MNC, mononuclear cell; PHA, phytohemagglutinin; PI, propidium iodide.
The impact of irradiation on allogenic immune recognition
The impact of irradiation on lymphocyte proliferative response
MLR assay was used to determine the effect of irradiation on allogeneic immune recognition. In response to allorecognition of mitomycin C-inactivated MNCs (from a blood donor), the proliferation of both X-irradiated and gamma -irradiated MNCs (obtained from platelet products) was significantly decreased (Optical Density of mean ± SD) = 1.86 ± 0.22, 1.95 ± 0.21 on day 1 of storage and 1.59 ± 0.26, 1.64 ± 0.36 on day 4, respectively) compared to non-irradiated MNCs (2.7 ± 0.35, 2.15 ± 0.31, respectively) (Fig. 5A). No significant difference was observed between X- and gamma-irradiated responder cells indicating that both types of irradiations effectively reduce lymphocyte proliferative responses. Our results also showed that the stimulatory indexes (SI) of X- and gamma -irradiated cells were (0.36 ± 0.09), (0.44 ± 0.07) on day 1 and (0.57 ± 0.09), (0.60 ± 0.19) on day 4 of storage, respectively (Fig. 5B). Cell stimulatory indexes were lower than “1” suggesting that both types of irradiations successfully inhibited cell proliferation.
Lymphocyte proliferative/activation response following irradiation. Non-irradiated (control) and either X- or gamma-irradiated responder cells were co-cultured with mitomycin-C inactivated stimulator cells. In the MLR assay, the ability of responder cells to proliferate in response to allorecognition of inactivated stimulator cells was assessed and OD at 570 nm was measured with a microplate reader (A). To evaluate the inhibitory effect of irradiation on cell proliferation, the cell stimulatory index (SI) was also calculated (B). In addition, lymphocyte activation was evaluated following co-culture of non-irradiated or irradiated responder cells with mitomycin-C inactivated stimulator cells where the percentages of CD8+69+T lymphocytes after 24-hour culture (C) and CD8+25+T lymphocytes after 48-hour (D) were measured by flow cytometry. Data shown as mean ± standard deviation; n = 5; *, P < 0.05; ns, non-significant; MLR, mixed lymphocyte reaction; OD, Optical Density.
The impact of radiation on the lymphocyte activation response
Following 24- and 48-hour MLR assays, the activation of responder cells against inactivated stimulator cells was assessed by the expression of early and late lymphocyte activation markers CD69 and CD25 on CD8+T lymphocytes, respectively. After 24-hour co-culturing, the expression of early activation marker CD69 on CD8+T lymphocytes was significantly lower in X- (6.3 ± 1.8%) and gamma-irradiated (4.6 ± 1.6%) compared to non-irradiated samples (14 ± 1.7%; P < 0.05) (Fig. 5C). Here, the lack of significant difference between X-irradiated and gamma-irradiated cells indicates effective reduction of lymphocyte activity by both types of radiation. After 48-hour co-culturing, the expression of late activation marker CD25 on CD8+T lymphocytes was significantly lower in X- (5.1 ± 1.1%) and gamma-irradiated (4.7 ± 1.3%) compared to non-irradiated cells (15 ± 2%; P < 0.05) (Fig. 5D). Hence, since significant difference was not seen between X- and gamma-irradiated cells, both types of irradiations efficiently decrease lymphocyte activity. Representative Fig. 5E and F demonstrate X- and gamma -irradiation-induced suppression on CD8+T lymphocyte activity when compared to the non-irradiated ones.
The effect of irradiation on the quality markers of platelets
X- and gamma irradiation had no effect on the general quality markers of platelet products including platelet count and swirling as well as pH. In this context, the statistical analysis did not show significant differences between the values of abovementioned parameters in X- /gamma- irradiated products compared to control (non-irradiated product).
The impact of irradiation on platelet activity
After exposure to either X- or gamma-irradiation, the effects of irradiation on platelet activity were assessed using P-selectin expression and PAC-1 binding. As illustrated in Figs. 6 and 7, both types of X- and gamma -irradiations had no effect on platelet P-selectin expression and PAC-1 binding compared to non-irradiated ones.
The expression of platelet P-selectin upon irradiation. The representative histogram and dot plot obtained from flow cytometry analysis demonstrate the effect of irradiation on P-selectin expression (A). Charts (B) and (C) reveal both types of irradiations did not significantly affect P-selectin expression. n = 5. Table abbreviation; Cell: Unstained cell control, ISO: Isotype control, X30: X irradiated-30Gys, Gamma: Gamma irradiated, PE: Phycoerythrin, PSEL: P-selectin.
Platelet PAC-1 binding upon irradiation. The representative histogram and dot plot obtained from flow cytometry analysis show the influence of irradiation on PAC-1 binding (A). Charts (B) and (C) show that both types of irradiations did not significantly affect PAC-1 binding. n = 5. Table abbreviation; Cell: Unstained cell control, ISO: Isotype control, X30: X irradiated-30Gys, Gamma: Gamma irradiated, FITC: Fluorescein isothiocyanate.
Discussion
Gamma irradiation has conventionally been considered the most reliable approach to prevent TA-GvHD. However, X-irradiation has now emerged as a preferred choice for blood irradiation, mainly due to its lower biohazard risks17. At similar dosages applied (approximately 30 Gy) and with the same energy level (6 mV) adjusted (with comparable linear energy transfer; LET) studies showed comparative levels of dsDNA damage by either X- or gamma irradiation17,18. As a result, there has been a steady increase in the demand for commercial X-irradiators, which has led to the gradual replacement of gamma-irradiators in the marketplace. Nevertheless, this equipment might not be accessible in every healthcare setting, as a consequent of their high cost in the market. Therefore, the design and development of a suitable “X irradiation system” with LINAC has been considered as a cost-effective alternative to commercial X-irradiators, although this alternative system has not yet been used and investigated in platelet products.
In this regard and as the first setup for platelet product, the study presented here initially attempted to design and develop a standard LINAC-based “X irradiation system” to generate a uniform and accurate dose of X-ray while ensuring irradiation in the field can be easily monitored. This enabled us to predict the irradiation configuration, aiming to minimize dose discrepancy and enhance the accuracy of irradiation. This “X irradiation system” was completed by a unique phantom designed specifically to accommodate a single platelet product bag. In this regard, after establishing and confirming the “X irradiation system”, the study proceeded to investigate the effects of this designed method of irradiation on ability of lymphocytes proliferation, their viability and activation, as well as platelet activation compared to the standard gamma irradiation method. This was a necessary approach to validate the efficacy of X irradiation in preventing the risk of TA-GvHD in platelet products. Our results revealed that both X- and gamma-irradiation equally suppress lymphocyte functional activity, such as allorecognition capacity and proliferation response to mitogen, while lymphocyte viability remains largely unaffected. Additionally, exposure to X- and gamma-irradiation does not affect platelet activation, as there are no significant differences in the platelet activation markers such as P-selectin expression and PAC-1 binding.
A conventional method for stimulation of T cells to proliferate is their culturing in the presence of PHA, a mitogen that specifically induces T cell expansion19,20,21. Following T cell expansion in response to PHA, various methods have been employed to assess T cell response including limited dilution assay (LDA)10, Carboxyfluorescein diacetate succinimidyl ester (CFSE) labeling assay22, and Lymphocyte transformation test (LTT)23. In our study, after X-ray irradiation with doses of 25, 30, and 35 Gy to the platelet product bag, their isolated MNCs were first cultured in the presence of PHA for expansion and colony formation. Prior to any intervention, microscopic images were captured to observe the reaction of T cells to PHA. It is noteworthy that at both intervals of 12- to 24-hours, the analysis of microscopic images indicates a significant reduction of lymphocyte proliferative responses to PHA stimulation in X- and gamma-irradiated compared to the non-irradiated samples. This was evident from the fact that the total colony area in the irradiated samples was significantly smaller than that observed in the non-irradiated (control) group. Following this preliminary examination, among conventional methods assessing cellular proliferation, MTT assay as a metabolic activation-based technique was employed. One of the advantages of this assay over the aforementioned methods is that in addition to checking cell proliferation, MTT assay can also be used to analyze cellular viability where its setup is in the absence of any mitogen compound such as PHA. Therefore, MTT assay was also performed in the absence of PHA as a method to assess cell viability. Notably, here our findings on proliferative capacities were consistent with microscopic observations, where the cell proliferation of irradiated cells was significantly inhibited compared to the non-irradiated (control) without showing any significant differences between various doses, as well as different types of irradiations. However, both types of irradiation and different doses of X-irradiation did not induce any significant effect on lymphocyte viability. In line with our findings but in blood bags, Góes EG et al. employed the LDA by which they demonstrated that cobalt-60 gamma-irradiation with a dose of 25 Gy reduces the proliferation potency of T cells treated with PHA10. Furthermore, Janatpour K et al. also showed that a 25 Gy dose of both X- and gamma-irradiation can effectively inhibit T cell proliferation in response to PHA in blood units17. In another study, Olivo RA et al. established an “X irradiation system” and administered a 25 Gy dose to blood products. In this study, following irradiation and isolation of PBMCs, both irradiated and non-irradiated PBMC were labeled with CFSE and cultured in presence of PHA and the proliferation rate was subsequently determined based on the expansion of labeled T cells. Their findings revealed that irradiation effectively inhibits the T cell proliferation capacity22. Notably, although most of the methods mentioned above used a radiation dose of 25 Gy, here, for further comparison, this study used three different X-ray doses of 25, 30, and 35 Gy, in order to choose the optimal dose by comparing different parameters. It is important to note that the Association for the Advancement of Blood & Biotherapies (AABB) allows the implementation of X- or gamma-irradiation at doses ranging from 25 to 50 Gy. However, several lines of studies indicate that irradiation even at a dose of 30 Gy (which is conventionally applied in most of centers), can lead to some changes in platelets including the induction of ROS generation, pro-inflammatory and pro-apoptotic phenotypes24,25,26,27. This is why in our research we limited the dose to a maximum of 35 Gy, while in the most recent studies doses of 30–33 Gy have also been used for X- or gamma irradiation of platelet products18,28. In this regard, it should be noted that the main purpose of blood irradiation is to deactivate lymphocytes, not to induce their apoptosis. This is why the radiation dose is usually adjusted to a level that only inactivates the lymphocytes involved in TA-GvHD, as the use of higher doses can negatively affect platelets, whose proper function is essential for post-transfusion efficacy.
One practical method to demonstrate the impact of irradiation on the suppression of T cell proliferative capacity is cell cycle assay29. This assay is typically employed to determine the cell cycle phase of proliferative cells after a particular intervention such as drug treatment or radiation exposure30. Several studies have employed cell cycle assays to assess the impact of irradiation on the cancer cell cycle31. However, there are no specific studies investigating the effects of radiation on T cell cycle in blood products, while the limited studies that exist have only used low doses of X-irradiation on peripheral blood32. Hence, our study is the first attempt to use cell cycle assay to analyze the effect of radiation at doses of 25, 30, and 35 Gy on the cell cycle of T lymphocytes obtained from platelet product bag treated with PHA. In this study, the impact of X- and gamma-irradiation on the cell cycle of T cells was analyzed through PI staining. After 12- and 24- hours of cell culture in presence of PHA, the PI staining determined the distribution of T cells in various phases of the cell cycle. It is worth noting that both of X- and gamma-irradiations effectively disrupt the T cell cycle. Intriguingly, our findings demonstrate that both types of irradiations lead to peaks in G1 and G2 phases, which could be primarily due to ds-DNA damage that triggers the G1 and G2 checkpoints activation33. The G1 checkpoint prevents the majority of T cells from entering S phase, while the remaining cells that enter S phase are arrested in the G2 phase, where G2 checkpoints detect their DNA damage34. These data are in agreement with those of Duthoo E et al.32, who conducted a comprehensive study on the proliferative capacity of T cells obtained from peripheral blood (but not blood product) following X-irradiation to investigate the effect of low dose X-ray on proliferation and cell cycle distribution of T cells. In their study, the progression of T cell cycle was tracked using the EdU pulse-chase method in lymphocytes exposed to 2 and 4 Gy of X-irradiation, which previously stimulated with PHA. Their findings indicated that the radiation induces T cell cycle perturbation and G2 arrest. However, due to their relatively low dose of X-ray, T cells were still able to successfully progress their cell cycle over time, albeit at a significantly slower rate compared to non-irradiated samples. However, in our study, by the use of the higher dose of 30 Gy, X-irradiation effectively suppressed the cell cycle progression of PHA-treated T lymphocytes. Actually, this was illustrated by a greater population in the G1 phase and a significantly lower population of T cells that successfully entered to S phase while ultimately arresting in G2. Consequently, these data confirm that X-irradiation can successfully suppress T cell expansion to a similar extent as that achieved by standard gamma irradiation. However, proliferation and cell cycle assays per se may not provide sufficient data to fully validate the effectiveness of X-irradiation to eliminate the risk of TA-GvHD.
Although studies examining the impact of irradiation on blood products have demonstrated that irradiation significantly inhibits the proliferation of T lymphocytes in response to PHA, the pro-survival properties of PHA per se may also prevent the apoptosis of T cells35. This evidence, which is mechanistically distinct from the mitogenic nature of PHA, may slightly interfere with PHA-dependent proliferative approaches in the presence of irradiation. As an alternative, monitoring the dynamics of DNA repair in irradiated cells by assessing the phosphorylation of histone-2AX (γ-H2AX), a well-known marker for DNA double-strand breaks, could be considered a suitable method36. As an another approach, in this study, we also attempted to evaluate cell cycle analysis in a 4-day stored platelet product in the absence of PHA. The rationale of this unique setup stems from several lines of evidence suggesting that platelets and their released growth factors and cytokines can act as potent stimuli for cell proliferation and expansion37, particularly for CD8 + T cells38. The storage of platelets is associated with a considerable release of cytokines and growth factors, which have a substantial capacity to promote cell proliferation39,40,41,42. Therefore, to eliminate the necessity for PHA, samples obtained from X- /gamma-irradiated and control bags that had been stored for four days were subjected to cell cycle analysis. In these conditions, lymphocytes exhibited natural proliferation due to the mediators released by platelets during the platelet storage lesion (PSL)40,43. In the control bags, cellular proliferation in products stored for 4-days was examined under microscopic fields and compared to day 0 of storage. The irradiated bags obviously displayed reduction in the colony formation of nucleated cells. Isolated PBMCs from each bag were subsequently analyzed using cell cycle assays. Notably, cell cycle progression was exclusively observed in the control sample on day 4, whereas the irradiated samples demonstrated inhibited cell cycle progression (see supplementary Fig. 1). The cell cycle assay relies on the interaction of PI with DNA. In this context, irradiated T cells exhibiting reduced DNA replication indicate a state of cell cycle arrest, a finding comparable to the γ-H2AX assay, which was similarly observed in the absence of PHA.
Given the fact that the purpose of irradiation is not limited to suppression of T cell proliferation, the post-irradiation function of T cell was also evaluated in this study. Hence, to ensure that irradiation successfully impairs the functional activity of T cells, we assessed the allogenic immune recognition capability of T cell using the mixed lymphocyte reaction (MLR) assay and flow cytometry technique. The MLR assay serves as a simple platform for evaluating interventions that alter the interaction between the antigen-presenting cells and T cells in order to either stimulate or inhibit the lymphocyte response44. Therefore, this method enables us to assess the effectiveness of different immunomodulators45 where the proliferation of lymphocytes from various donors is examined in an allogeneic response. This is a proper technique for evaluating lymphocyte activity following any intervention such as irradiation. In our study, the T cells activity was first assessed based on their allogenic immune recognition capabilities. Allorecognition potency of T cells was measured through two distinct methods, the MLR followed by either MTT assay or flow cytometry technique which detects changes in the expression of CD69 and CD25 as the activation markers on CD8+T cells. Our results showed that the proliferation of T cells in response to inactivated stimulator cells was markedly decreased in both X- and gamma-irradiated responder cells when compared to non-irradiated ones, indicating that both types of X- and gamma-irradiations comparably and effectively suppressed T cell responses. This elucidation was based on the stimulatory index for evaluating the inhibitory effect of radiation on cell proliferation, which showed a marked decrease in samples exposed to both types of radiation compared to the non-irradiated ones. In addition, flow cytometry analysis indicated that X- and gamma-irradiation successfully hindered the expression of CD69 on CD8+T cells, as an early marker of T cell activity. Our findings also revealed that in comparison with non-irradiated cells, both types of irradiations significantly inhibit the expression of CD25 as the late T cell activation indicator, on CD8+T cells after co-culturing. These results confirmed that X- and gamma-irradiations successfully suppress allorecognition ability of CD8+T cells in MNCs obtained from platelet products. Now, considering the lack of significant difference in T cell allorecognition ability between X- and gamma-irradiated samples, and while, both X- and gamma-irradiation were able to effectively inhibit response of T cells especially CD8+T cells as the main player in TA-GvHD, it seems that the recommended dose of 30 Gy of X-ray could also effectively eliminate the chance of TA-GvHD. In line with these findings, using a commercial X- and gamma irradiators, Davis AM et al.28 also employed the MLR assay on PBMCs mixed with plasma/ SSP + in a blood bag, to evaluate T cell allorecognition activity after exposure to X- and gamma-irradiation with doses of 30 and 33.6 Gy, respectively. Their results indicated that both types of irradiations successfully inhibit T cell response, whereas no significant difference was observed between X- and gamma-irradiation in terms of T cell activation. Furthermore, in their study, the lymphocytes were treated with Phorbol 12-myristate 13-acetate (PMA) to trigger their activation. Following that, the activation of T cells was evaluated by examining the expression of T cell activation markers by flow cytometry analysis. Their findings showed that irradiated T cells significantly express lower levels of CD69. Although, they employed a synthetic T cell stimulator, PMA, their findings are in line with our results in which a more physiological stimulation was applied to investigate T cell functionality in a real platelet product. They finally suggested that dose of ~ 30 Gy of X- and gamma-irradiation for blood product not only inhibits the proliferation capability and response of T cells but also impairs their functional activation, thereby supporting their potential use in the prevention of TA-GvHD. However, it should be noted that their setup did not meet the factual conditions of blood or platelet products, as they only simulated the conditions by adding PBMC plus plasma in a platelet bag rather than using the original blood components. Considering the limitation of above-mention research, our study is the first attempt to investigate the effect of X-irradiation on platelet products through the evaluating of MLR. While our research confirms the study which conducted by Davis et al., it is also the only one that shows such results obtained by either commercial X-irradiator or LINAC-based “X irradiation system” in platelet products.
In the continuation of our study, the impact of the designated dose for X-ray on platelets was investigated to show that X-ray is safe for the platelet product and does not have any negative effect on the quality and function of platelets. For this purpose, alongside with evaluating of general marker of platelet quality, we also examined two platelet activation markers of P-selectin and PAC-1 binding. The findings showed that neither X- nor gamma-irradiation had the significant effect on platelet activation markers and quality, confirming efficacy and safety of our LINAC-based “X-irradiation system” for platelet products. Regardless of the different methods of X-ray generation, our results are comparable to a previous study by Johnson L et al.46 on platelet products where they examined the impact of X-irradiation generated with a commercial device on platelet activation and showed that X-ray does not have a significant effect on the surface expression of platelet markers. However, again one important limitation of Johnson’s study is the lack of any T cell proliferation and activation assessment to rule out the risk of TA-GvHD in platelet products.
Conclusion
Taken together, study presented here showed that at levels comparable to gamma irradiation as a gold standard method, our introduced LINAC-based “X irradiation system” with the dose of 30 Gy can also successfully inactivate CD8+T cells, as the main player in TA-GvHD, without causing platelet activation. Therefore, this study is considered the first attempt to propose LINAC-based “X irradiation system” as a safe and efficient approach to irradiate platelet products, a cost-effective method of platelet irradiation that can be widely accessible by healthcare providers. On the other hand, our study also fulfills previous research that did not achieve to properly address all the irradiation specification criteria that support both quality and safety of X-irradiated platelet products.
Data availability
The corresponding author can make available some dataset upon reasonable request.
Abbreviations
- AABB:
-
Association for the Advancement of Blood & Biotherapies
- CFSE:
-
Carboxyfluorescein diacetate succinimidyl ester
- LDA:
-
Linear energy transfer
- LET:
-
Limited dilution assay
- LINAC:
-
Linear accelerator
- LTT:
-
Lymphocyte transformation test
- γ-H2AX:
-
Histone-2AX
- MLR:
-
Mixed lymphocyte reaction
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide
- PBMCs:
-
Peripheral blood mononuclear cells
- PDGF:
-
Platelet-derived growth factor
- PF4:
-
Platelet factor 4
- PHA:
-
Phytohemagglutinin
- PI:
-
Propidium iodide
- PMA:
-
Phorbol 12-myristate 13-acetate
- PSL:
-
Platelet storage lesion
- TA-GvHD:
-
Transfusion-associated-graft versus host disease
- TGF-β:
-
Transforming growth factor-β
References
Squires, J. E. Indications for platelet transfusion in patients with thrombocytopenia. Blood Transfus. 13(2), 221–226 (2015).
Estcourt, L. J. et al. Guidelines for the use of platelet transfusions. Br. J. Haematol. 176(3), 365–394 (2017).
Garraud, O. et al. Improving platelet transfusion safety: biomedical and technical considerations. Blood Transfus. 14(2), 109–122 (2016).
Kopolovic, I. et al. A systematic review of transfusion-associated graft-versus-host disease. Blood 126(3), 406–414 (2015).
Foukaneli, T. et al. Guidelines on the use of irradiated blood components. Br. J. Haematol. 191(5), 704–724 (2020).
Treleaven, J. et al. Guidelines on the use of irradiated blood components prepared by the British Committee for standards in haematology blood transfusion task force. Br. J. Haematol. 152(1), 35–51 (2011).
Pinnarò, P. et al. Implementation of a new cost efficacy method for blood irradiation using a non dedicated device. J. Exp. Clin. Cancer Res. 30(1), 7 (2011).
Moroff, G. & Luban, N. L. The irradiation of blood and blood components to prevent graft-versus-host disease: Technical issues and guidelines. Transfus. Med. Rev. 11(1), 15–26 (1997).
Butson, M. J., Cheung, T., Yu, P. K. & Stokes, M. J. Blood irradiation with accelerator produced electron beams. Phys. Med. Biol. 45(11), N139–N142 (2000).
Góes, E. G. et al. Quality control of blood irradiation: determination T cells radiosensitivity to cobalt-60 gamma rays. Transfusion 46(1), 34–40 (2006).
Kulkarni, S., Woollard, K. J., Thomas, S., Oxley, D. & Jackson, S. P. Conversion of platelets from a proaggregatory to a proinflammatory adhesive phenotype: Role of PAF in spatially regulating neutrophil adhesion and spreading. Blood J. Am. Soc. Hematol. 110(6), 1879–1886 (2007).
van Stipdonk, M. J., Lemmens, E. E. & Schoenberger, S. P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat. Immunol. 2 (5), 423–429 (2001).
Prlic, M., Hernandez-Hoyos, G. & Bevan, M. J. Duration of the initial TCR stimulus controls the magnitude but not functionality of the CD8 + T cell response. J. Exp. Med. 203 (9), 2135 (2006).
Hosseini, E., Ghasemzadeh, M., Kamalizad, M. & Schwarer, A. P. Ex vivo expansion of CD3(depleted) cord blood-MNCs in the presence of bone marrow stromal cells; an appropriate strategy to provide functional NK cells applicable for cellular therapy. Stem Cell. Res. 19, 148–155 (2017).
Hosseini, E., Solouki, A., Haghshenas, M., Ghasemzadeh, M. & Schoenwaelder, S. M. Agitation-dependent biomechanical forces modulate GPVI receptor expression and platelet adhesion capacity during storage. Thromb. J. 20(1), 3 (2022).
Ahmadi, J., Hosseini, E., Kargar, F., Maghsudlu, M. & Ghasemzadeh, M. Platelet transfusion enhances pro-aggregatory status shortly after coronary artery bypass grafting (CABG while modulating platelet pro‐inflammatory state 1‐week post‐surgery. J. Cell. Mol. Med. 28(15), e18573 (2024).
Janatpour, K. et al. Comparison of X-ray vs. gamma irradiation of CPDA-1 red cells. Vox Sang 89(4), 215–219 (2005).
Marks, D. C. et al. X- and gamma-irradiation have similar effects on the in vitro quality of stored red cell components. Transfusion 61(11), 3214–3223 (2021).
Gualde, N. & Goodwin, J. S. Effect of irradiation on human T-cell proliferation: Low dose irradiation stimulates mitogen-induced proliferation and function of the suppressor/cytotoxic T-cell subset. Cell. Immunol. 84(2), 439–445 (1984).
Pelszynski, M. M., Moroff, G., Luban, N. L. C., Taylor, B. J. & Quinones, R. R. Effect of γ irradiation of red blood cell units on T-Cell inactivation as assessed by limiting dilution analysis: Implications for preventing transfusion-Associated graft-versus-host disease. Blood 83(6), 1683–1689 (1994).
Akiyama, M. et al. Peripheral lymphocyte response to PHA and T cell population among atomic bomb survivors. Radiat. Res. 93(3), 572–580 (1983).
Olivo, R. A. et al. Evaluation of the effectiveness of packed red blood cell irradiation by a linear accelerator. Rev. Bras. Hematol. Hemoter 37(3), 153–159 (2015).
Pourfathollah, A. A., Shaiegan, M., Namiri, M. & Babae, G. R. Effect of gamma irradiation on lymphocyte proliferation and IL-8 production by lymphocytes isolated from platelet concentrates. Arch. Med. Res. 39(6), 590–593 (2008).
Tynngård, N., Studer, M., Lindahl, T. L., Trinks, M. & Berlin, G. The effect of gamma irradiation on the quality of apheresis platelets during storage for 7 days. Transfusion 48(8), 1669–1675 (2008).
Mallhi, R. S., Biswas, A. K., Philip, J. & Chatterjee, T. To study the effects of gamma irradiation on single donor apheresis platelet units by measurement of cellular counts, functional indicators and a panel of biochemical parameters, in order to assess pre-transfusion platelet quantity and quality during the shelf life of the product. Med. J. Armed Forces India. 72(1), 19–26 (2016).
Nodeh, F. K., Hosseini, E. & Ghasemzadeh, M. The effect of gamma irradiation on platelet redox state during storage. Transfusion 61(2), 579–593 (2021).
Hosseini, E., Nodeh, F. K. & Ghasemzadeh, M. Gamma irradiation induces a pro-apoptotic state in longer stored platelets, without progressing to an overt apoptosis by day 7 of storage. Apoptosis 28(7–8), 1141–1153 (2023).
Davis, A. M. et al. X-irradiation and gamma-irradiation inactivate lymphocytes in blood components. Transfusion 61(11), 3081–3086 (2021).
Tell, R. et al. Comparison between radiation-induced cell cycle delay in lymphocytes and radiotherapy response in head and neck cancer. Br. J. Cancer. 77(4), 643–649 (1998).
Lonati, L., Barbieri, S., Guardamagna, I., Ottolenghi, A. & Baiocco, G. Radiation-induced cell cycle perturbations: A computational tool validated with flow-cytometry data. Sci. Rep. 11(1), 925 (2021).
Otani, K. et al. Cell-cycle-controlled radiation therapy was effective for treating a murine malignant melanoma cell line in vitro and in vivo. Sci. Rep. 6(1), 30689 (2016).
Duthoo, E., Vral, A. & Baeyens, A. An updated view into the cell cycle kinetics of human T lymphocytes and the impact of irradiation. Sci. Rep. 12(1), 7687 (2022).
Barnum, K. J. & O’Connell, M. J. Cell cycle regulation by checkpoints. Methods Mol. Biol. 1170, 29–40 (2014).
Liu, C., Nie, J., Wang, R. & Mao, W. The cell cycle G2/M block is an indicator of cellular radiosensitivity. Dose Response. 17(4), 1559325819891008 (2019).
Carloni, M., Meschini, R., Ovidi, L. & Palitti, F. PHA-induced cell proliferation rescues human peripheral blood lymphocytes from x-ray-induced apoptosis. Mutagenesis 16(2), 115–120 (2001).
Mah, L. J., El-Osta, A. & Karagiannis, T. C. γH2AX: A sensitive molecular marker of DNA damage and repair. Leukemia 24(4), 679–686 (2010).
Tan, S. et al. Platelets enhance CD4 + central memory T cell responses via platelet factor 4-dependent mitochondrial biogenesis and cell proliferation. Platelets 33(3), 360–370 (2022).
Chen, C. et al. Effects of platelets on characteristics of lymphocytes cultured in vitro and optimization of adoptive immunotherapy. BIOCELL 47, 2661–2669 (2023).
Zhu, L., Huang, Z., Stålesen, R., Hansson, G. K. & Li, N. Platelets provoke distinct dynamics of immune responses by differentially regulating CD4 + T-cell proliferation. J. Thromb. Haemost. 12(7), 1156–1165 (2014).
Torres Chavez, A. et al. Expanding CAR T cells in human platelet lysate renders T cells with in vivo longevity. J. Immunother. Cancer. 7(1), 330 (2019).
Mo F, Tsai C-T, Zheng R, Cheng C, Heslop HE, Brenner MK, et al. Human platelet lysate enhances <em>in vivo</em> activity of CAR-V; 2 T cells by reducing cellular senescence and apoptosis. Cytotherapy 26(8), 858–868 (2014).
Peterson, L. et al. Immunotherapy: platelets induce regulatory T-cell expansion during car-T cell manufacturing. Cytotherapy 25, E13 (2023).
Ng, M. S. Y., Tung, J-P. & Fraser, J. F. Platelet Storage lesions: What more do we know now? Transfus. Med. Rev. 32(3), 144–154 (2018).
Barra, G. et al. The immunoregulatory effect of the TREM2-agonist sulfavant A in human allogeneic mixed lymphocyte reaction. Front. Immunol. 14, 1050113 (2023).
Heidari, N. et al. Up-regulation of CD200/CD200R1 immunomodulatory axis of allogenic peripheral blood mononuclear cells in a co-culture with adipose-derived mesenchymal stem cells. Iran. J. Allergy Asthma Immunol. 19(5), 484–496 (2020).
Johnson, L. et al. The in vitro quality of X-irradiated platelet components in PAS-E is equivalent to gamma-irradiated components. Transfusion 61(11), 3075–3080 (2021).
Acknowledgements
Parts of this work were presented as a dissertation for doctoral student Jalal Naghinezhad. The authors declare no conflict of interest. The authors thank Ali Hasani who helped with X-irradiation with linear accelerator device.
Funding
This work is a part of the project of Dr. Hosseini and Dr. Ghasemzadeh, which was approved and supported by the High Institute for Research and Education in Transfusion Medicine.
Author information
Authors and Affiliations
Contributions
J. N: Data curation (equal); formal analysis (supporting); investigation (supporting); writing – original draft (supporting). E. H: Conceptualization (equal); data curation (equal); formal analysis (lead); investigation (equal); methodology (equal); software (equal); supervision (equal); validation (equal); visualization (equal); writing – original draft (equal) . Z.S: Conceptualization (supporting); investigation (supporting); methodology (supporting). M. H: formal analysis (supporting); methodology (supporting). B.K: formal analysis (supporting); methodology (supporting). M .G: Conceptualization (equal); data curation (supporting); formal analysis (supporting); funding acquisition (lead); investigation (lead); methodology (equal); project administration (lead); resources (lead); software (equal); supervision (lead); validation (lead); visualization (lead); writing –review and editing (lead). All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was approved by the Ethics Committee of the High Institute for Research and Education in Transfusion Medicine, Tehran, Iran (Ethic approval numbers: IR.TMI.REC.1401.017& IR.TMI.REC.1402.001).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Naghinezhad, J., Hosseini, E., Siavashpour, Z. et al. Designing a linear accelerator-based “X irradiation system” for platelet products: an efficient, safe, accessible and cost-effective alternative for conventional X- or gamma irradiators. Sci Rep 14, 28363 (2024). https://doi.org/10.1038/s41598-024-80118-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-80118-6
Keywords
This article is cited by
-
Nanodrug-based modulation of platelet–leukocyte interactions in ovarian cancer: a new frontier in targeted therapy
Cancer Nanotechnology (2025)
-
Platelet apheresis with additive solution and plasma rinseback affects the cellular composition of LRS chamber products
Scientific Reports (2025)
-
The Cardiovascular Burden of Gonadotropin-Releasing Hormone (GnRH) Therapy: Mechanisms, Risks, and Interventions
Cardiovascular Toxicology (2025)
-
Interactions Between Platelets and Leukocytes in Ovarian Cancer: A New Approach to Cancer Treatment: A Review
Indian Journal of Gynecologic Oncology (2025)