Abstract
Due to the resistance of carbapenem-resistant Klebsiella pneumoniae (CRKP) to most antibiotics, CRKP treatment is challenging, which results a high mortality rate. CRKP infection poses a significant challenge for infection management and treatment, especially among neonates and premature infants. Therefore, it is important to understand the clinical characteristics of CRKP bloodstream infection (BSI) in premature infants and identify the related risk factors for death. This study aims to explore and analyze the clinical characteristics and risk factors affecting mortality of BSI caused by CRKP in premature infants. A retrospective study was conducted in a Children’s Hospital in Henan to analyze clinical data of premature infants with CRKP BSI admitted from January 2015 to December 2022. Univariate and multivariate logistic regression models were utilized to investigate risk factors affecting mortality. Receiver operating characteristic curves were employed to evaluate the predictive value of different indicators on mortality, and differences in area under the curve (AUC) were compared using Stata 17 SE. A total of 96 premature infants with CRKP BSI were enrolled, including 70 patients in the survival group and 26 in the death group. At the onset of infection, 69 (71.9%) patients exhibited persistent tachycardia (heart rate > 180 beats/min), 61 (63.5%) had fever, and 59 (61.4%) experienced apnea episodes. Concurrent meningitis (OR 9.588, 95% CI 1.401–57.613, P = 0.021), concurrent necrotizing enterocolitis (NEC) (OR 7.881, 95% CI 1.672–73.842, P = 0.032), and the maximum vasoactive-inotropic score (VIS) value within 48 h of onset (OR 1.467, 95% CI 1.021–1.782, P = 0.001) were identified as independent risk factors for mortality. The univariate analysis showed that ceftazidime-avibactam (CAZ/AVI) treatment and appropriate early antimicrobial treatment were significantly associated with survival (P < 0.05). The combined predictive AUC for mortality in premature infants with CRKP BSI using the maximum VIS value ≥ 52.5 points within 48 h of onset, concurrent NEC, and purulent meningitis was 0.931 (95% CI 0.856–1.000) with a sensitivity of 92% and specificity of 85.7%. CRKP BSI was a significant mortality risk in premature infants. It is crucial to administer proper antimicrobial therapy in order to increase survival rates of the patients. CAZ/AVI has the potential to improve outcomes in this particular population; however, further research is required to evaluate the effectiveness of specific treatment in premature infants.
Similar content being viewed by others
Introduction
Late-onset sepsis presents a significant challenge in the care of premature infants. Late-onset sepsis increased risks of infants suffering from in-hospital complications, mortality, and adverse neurodevelopmental outcome1,2. Klebsiella pneumoniae is the predominant pathogen causing bloodstream infection(BSI) in newborns in many developing countries, such as China3,4,5,6. The rise of carbapenem-resistant Enterobacteriaceae (CRE), particularly Klebsiella pneumoniae, has emerged as a serious threat to public health due to limited treatment options. The prevalence of carbapenem-resistant Klebsiella pneumoniae (CRKP) among pediatric patients in China has become a pressing clinical issue7. A study analyzing clinical isolates of multi-drug resistant bacteria from 11 tertiary children’s hospitals in China between 2016 and 2020 revealed that CRKP infection rates were 18.7% in newborns and 20.3% in non-newborns8. Premature infants, due to factors such as extended hospital stays, central venous catheter use, mechanical ventilation, prolonged total parenteral nutrition, and extensive antibiotic treatment; appear to be particularly vulnerable to CRKP infections9,10.
Moreover, CRKP BSI are related with higher mortality rate compared to other types of infections. The impact of CRKP infection is well studied in children, whereas there is limited information available regarding premature infants. An independent two-year study reported a high mortality rate of 40% for neonatal carbapenem-resistant Gram-negative bacterial sepsis patients11. A study conducted across three neonatal intensive care units (NICUs) in New Delhi found that 35% of Klebsiella and 15% of E. coli were carbapenem-resistant, with a case fatality rate exceeding 50%3. Due to the resistance of these organisms to most antibiotics, treatment is challenging, which results a high mortality rate. Therefore, early and accurate identification of premature infants with CRKP is crucial for timely appropriate treatment.
Limited data available on CRKP BSI in premature infants highlights the necessity for a better understanding of mortality rates and risk factors within this population. This study seeks to analyze the clinical characteristics, antibiotic sensitivity profiles, and detailed antibiotic regimens for CRKP BSI in premature infants. Additionally, it aims to assess the impact of CRKP BSI on mortality rate and identify predictive factors that influence mortality outcomes.
Materials and methods
Study design and patients included
Premature infants admitted to the NICU between January 2015 and December 2022, diagnosed with CRKP BSI, were included in this retrospective cohort study. Infants with genetic metabolic diseases, congenital dysplasia, incomplete medical records, or co-infections with other bacterial or fungal BSI were excluded. The infants were categorized into a survival group and a death group based on their prognosis. The survival group exhibited resolved infection symptoms, stable spontaneous breathing, negative blood culture upon reexamination, body weight ≥ 2000 g before discharge, and stable vital signs during one-week follow-up post-discharge. The death group comprised of patients in-hospital deaths primarily due to CRKP BSI, as well as children who chose to leave the hospital due to difficulty in maintaining vital signs despite active treatment and rescue, and who passed away within one week indicated by the post-discharge follow-up.
Clinical data collection
Clinical data were collected through a review of electronic medical records, capturing details such as maternal pregnancy status, gestational age of premature infants, birth weight, gender, Apgar score at birth, mode of delivery, age at onset, length of hospital stay, preinfectious invasive procedures, antimicrobial exposure, clinical infection symptoms, occurrence of complications, and antibiotic treatment options. The observed complications included purulent meningitis, necrotizing enterocolitis (NEC), intraventricular hemorrhage (IVH), septic shock, and capillary leak syndrome. Blood tests were performed within 24 h before and after positive blood culture samples were obtained from premature infants, along with biochemical tests, C-reactive protein (CRP) levels, procalcitonin (PCT) levels, imaging data, and other relevant findings. Blood routine indicators, CRP, and PCT data will be averaged for statistics. The neonatal sequential organ failure assessment (nSOFA) score was calculated at the onset of the condition. Additionally, vasoactive drugs administered to infants within 48 h of illness onset were recorded, and the vasoactive inotropy score (VIS) was subsequently calculated.
Definitions
CRKP BSI refers to an infectionthat blood culture was positive for CRKP. The onset of BSI is determined by the date when the first positive blood culture is collected. Carbapenem exposure is defined as the administration of any carbapenem within 30 days prior to the BSI. Septic shock is characterized by sepsis accompanied by hypotension and perfusion abnormalities that are resistant to organ dysfunction. The calculation formula for the VIS is as follows: VIS = dopamine [μg/(kg·min)] + dobutamine [μg/(kg·min)] + 10 × milrinone [μg/(kg·min)] + 100 × adrenal gland vasopressin [μg/(kg·min)] + 100 × norepinephrine [μg/(kg·min)] + 10,000 × vasopressin [U/(kg·min)]12. The nSOFA score was developed as a score to assess sepsis severity in preterm neonates13. The nSOFA uses categorical scores with a total score ranging from 0 as best to 15 as worst, which were used to objectively describe dynamic changes in respiratory deterioration, hemodynamic compromise and hematologic dysfunction14.
Antibiotic susceptibility test
Blood samples were collected from infants showing symptoms of infection prior to the administration of antibacterial drugs. The FX400 fully automatic blood culture instrument from Becton Dickinson Company (USA) was utilized for blood sample detection, and bacterial strains were isolated following standard operating procedures. Bacterial identification and drug sensitivity testing were conducted using the VITEK-2 Compact fully automatic bacterial identification instrument from BioMérieux Company (France). Drug sensitivity testing was performed in accordance with the standards of the American Association for Clinical Laboratory Standards to determine sensitivity results15.
Statistical analysis
Continuous variables with normal distributions are reported as means ± standard deviation (SD), while those with non-normal distributions are reported as medians and interquartile ranges. Categorical variables are presented as frequencies and proportions. The t-test was employed for comparing normally distributed variables, whereas the Mann–Whitney U-test was used for non-normally distributed variables, and chi-squared were utilized for categorical variables. Univariate and multivariate logistic regression models were used to examine factors influencing mortality in premature infants with CRKP BSI. Receiver operating characteristic (ROC) curves were used to assess the predictive value of various indicators on mortality, with differences in area under the curve (AUC) compared using Stata 17 SE. The analyses were conducted using SPSS (version 21.0, IBM Corp., New York, USA). Statistical significance was set at P < 0.05.
Ethics approval and consent to participate
All methods in this study were conducted in compliance with the ethical standards outlined in the 1964 Declaration of Helsinki and its subsequent amendments or similar ethical standards. The Life Science Ethics Committee of Children’s Hospital affiliated to Zhengzhou University approved the study (Number: 2023-k-076). Informed consent was exempt by Life Science Ethics Committee of Children’s Hospital affiliated to Zhengzhou University.
Results
Demographics and clinical characteristics of the study population
During the study period, a total of 6,556 premature infants were admitted to the NICU of Children’s Hospital Affiliated to Zhengzhou University, of which 196 were diagnosed with Klebsiella pneumoniae BSI. Among these 196 cases, 101 (51.5%) were confirmed as CRKP BSI. After a thorough review of the cases, two patients were excluded due to congenital dysplasia, two patients was excluded due to genetic metabolic diseases, and another patient was excluded due to incomplete medical records and co-infections with other bacterial BSI. Consequently, the remaining 96 cases were included in the analysis. Of these, 55 (57.3%) were male, and 41 (42.7%) were female. The mean gestational age was 30.0 ± 1.6 weeks (range: 27–34 weeks) and the mean birth weight was 1249 ± 174 g (range: 800–2010 g). At the onset of infection, 69 (71.9%) patients had persistent tachycardia (heart rate > 180 beats/min), 61 (63.5%) had fever, and 59 (61.4%) had apnea episodes. A total of 88 (91.7%) patients had septic shock, 35 (36.5%) had meningitis, and 91 (94.8) patients need vasoactive drug support. More than 50% of patients had been exposed to carbapenems prior to the onset of infection. The time from infection onset to pathogen identification in 96 premature infants was 35.4 ± 6.2 h, while the time from infection onset to the results of antibiotic susceptibility testing was 56.5 ± 6.5 h.
Seventy of 96 premature infants survived and the mortality rates caused by CRKP BSI was 27.1%. Except for 17 cases who received single ceftazidime-avibactam (CAI/AVZ) treatment, all other cases received combination therapy. Meropenem/cefoperazone- sulbactam combinations and meropenem/fosfomycin combinations were the most commonly used antibacterial regimens,with frequencies of 31.3% and 28.1%, respectively as illustrated in Table 1.
Antibiotic susceptibilities of carbapenem-resistant Klebsiella pneumoniae in premature infants
All tested CRKP isolates were resistant to piperacillin/tazobactam, ceftazidime, amtrazumab, imipenem and meropenem (n = 96). The most common resistances were to amikacin (71 of 96 strains tested [73.9%]) and ciprofloxacin (76 of 96 strains tested [79.2%]). None were resistant to tigecycline, two (2.3%) was resistant to polymyxin B and twenty (23.5%) were resistant to Trimethoprim/sulfamethoxazole (TMP/SMX), as shown in Table 2. Ceftazidime-avibactam (CAZ/AVI) susceptibility was not routinely tested at our hospital.
Comparison of clinical data of the two groups
In comparison to the survival group, a higher proportion of patients with a history of carbapenem exposure prior to bloodstream infection (BSI) was observed in the death group (P = 0.032). Additionally, the death group exhibited a greater proportion of infections complicated by necrotizing enterocolitis (NEC) and purulent meningitis (all P < 0.001). The duration of hospitalization was shorter in the death group compared to the survival group (P < 0.001), and the platelet count at the time of infection was significantly lower (P = 0.011). Furthermore, the death group had a higher proportion of patients who underwent central venous catheterization and surgical procedures than the survival group (P = 0.040 and P = 0.029, respectively). However, there was no statistically significant difference between the groups regarding the time from infection onset to pathogen identification and antibiotic susceptibility testing. Within 48 h of onset, the maximum vasoactive-inotropic score (VIS) in the death group was significantly higher than that in the survival group (P < 0.001) (Table 3).
Comparison of antibiotic treatment between the two groups
The univariate analysis indicated that CAZ/AVI treatment and and appropriate early antimicrobial treatment were significantly associated with survival (P < 0.05) (Table 4).
Risk factors for mortality
The univariate analysis showed that concurrent meningitis, previous carbapenems exposure, concurrent NEC, surgery prior to infection, central lines prior to infection, PLT < 50 × 109/L after onset of illness and the maximum VIS value within 48 h of onset were associated with mortality (P < 0.05). In the multivariate logistic analysis, after adjustment for potential confounders,concurrent meningitis (OR 9.588, 95% CI1.401–57.613, P = 0.021), concurrent NEC (OR 7.881, 95% CI 1.672–73.842, P = 0.032) and the maximum VIS value within 48 h of onset (OR 1.467, 95% CI 1.021–1.782, P = 0.001) were identified as independent risk factors (Table 5).
The clinical value of various indicators in predicting mortality in premature infants with CRKP BSI
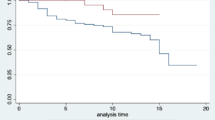
ROC curve analysis revealed that the maximum VIS value within 48 h of onset served as a reliable predictor of mortality in premature infants with CRKP BSI (Fig. 1). The area under the curve (AUC) was 0.840 (95% CI 0.734–0.946, P < 0.001), with a sensitivity of 72% and specificity of 91.4%, corresponding to a cutoff value of 52.5. Additionally, the AUC for concurrent NEC was 0.784 (95% CI 0.664–0.905) with a sensitivity of 64% and specificity of 92.9%, while the AUC for concurrent purulent meningitis was 0.711 (95% CI 0.590–0.833) with a sensitivity of 68% and specificity of 74.3%. Notably, there was no significant difference in AUC between the maximum VIS value within 48 h of onset and concurrent NEC and purulent meningitis (P = 0.401, P = 0.065).
Receiver operating characteristic (ROC) curve of various indicators for predicting the prognosis of CRKP BSI. NEC necrotizing enterocolitis; VIS vasoactive-inotropic score.
The combined predictive AUC for mortality in premature infants with CRKP BSI was 0.931 (95% CI 0.856–1.000) with a sensitivity of 92% and specificity of 85.7%. Its predictive performance was significantly superior to that of concurrent NEC and purulent meningitis (P = 0.002, P < 0.001).
Discussion
Due to the resistance of carbapenem-resistant Klebsiella pneumoniae (CRKP) to most antibiotics, CRKP treatment is challenging, which poses a significant challenge for infection management and treatment, especially among neonates and premature infants. This retrospective study seeks to assess the clinical characteristics and efficacy of antibiotic regimens for CRKP BSI in premature infants. It also aims to investigate predictors of patient mortality, identify and treat risk factors early, and improve outcomes for premature infants with CRKP BSI. The study aims to offer insights for clinical antibiotic selection and early intervention strategies to decrease mortality rates and enhance outcomes.
In this study, a mortality rate of 27% associated with CRKP BSI was reported, which is lower than previously reported rates of 45.2% and 50%16,17, as well as lower than mortality rates caused by other carbapenem-resistant Gram-negative pathogens18. The lower mortality rates in this study may be due to the high rate of appropriate early antimicrobial treatment (83.3%). It was observed that premature infants with persistent tachycardia, fever, respiratory arrest symptoms, shock, and declining white blood cell and platelet counts should be considered for CRKP BSI, and prompt and suitable antibiotic treatment should be administered. Another study involving 32,100 patients with BSI in the US hospitals demonstrated that receiving appropriate empirical antimicrobial treatment was linked to lower in-hospital mortality compared to inappropriate treatment19. The study also highlighted that appropriate antibiotic therapy within 24 h of blood culture collection was an independent predictor of reduced 30-day mortality rates in BSI patients20. Therefore, early identification of at-risk patients, and initiation of effective antibiotics against CRKP isolates may aid in reducing mortality rates in premature infants.
Risk factors for mortality of CRKP BSI in infants include intensive care admission, intubation, inotropic support, monocyte count, and a higher Pitt bacteremia score21,22. However, there are no published studies on premature infants. This study suggests that concurrent meningitis, concurrent NEC, and the maximum VIS value within 48 h of onset are associated with significantly higher mortality risk in premature infants with CRKP BSI. NEC, a common condition in premature neonates, is influenced by factors like intestinal barrier immaturity, hyperinflammatory immune cells, and dysbiosis23. Sepsis is considered a risk factor for NEC, with an incidence of 32.3% in this study, aligning with previous literature24,25. The mortality rate of NEC can range from 20 to 30%, with a significant portion requiring surgical treatment26. However, the timing of surgical intervention for NEC infants is challenging27, leading to some infants missing the opportunity for surgery due to rapid deterioration and multiple organ failure. Neonates with sepsis and NEC also experience more severe anemia and hypoproteinemia, worsening the disease severity and increasing the risk of death28. Meningitis is a serious disease that is more frequently observed in the neonatal period compared to other age groups. A multi-center study conducted in China, involving 13 children’s hospitals, highlighted the influences of drug resistant pathogens causing bacterial meningitis in children, particularly the high prevalence of CRKP (54.5%) meningitis29. The infection caused by drug resistant pathogens poses a significant challenge in the treatment of pediatric bacterial meningitis. Previous studies have shown that CRKP is an independent risk factor for mortality in adult patients with Klebsiella pneumoniae meningitis30. In this study, 17 cases resulted in death among 35 infants with meningitis, serving as a stark reminder. It is crucial to consider the antibiotic agent’s ability to penetrate the blood–brain barrier and achieve the minimum inhibitory concentration (MIC) in the brain for effective treatment of intracranial infections, in addition to the antibacterial spectrum and drug susceptibility tests31. The limited options of antimicrobial drugs available for treating CRKP in newborns, particularly premature infants, have also contributed to the mortality rate of infants with CRKP BSI-related meningitis. Research suggests that for newborns with CRKP BSI-related purulent meningitis, a combination of fluoroquinolones and other drugs susceptible to Klebsiella pneumoniae can reduce neonatal mortality32, emphasizing the need for attention. Therefore, premature infants with CRKP BSI complicated by NEC and meningitis require increased vigilance, and these infants are in urgent need of new treatment strategies.
The Vasopressor Inotropic Score (VIS) is a composite score that combines various vasopressors and inotropes to measure cardiovascular support. VIS and similar metrics are commonly used to assess illness severity in neonates and infants undergoing congenital heart surgery, pediatric patients with sepsis in the pediatric intensive care unit (PICU), and adult patients with sepsis in the medical intensive care unit (MICU)33,34,35,36. Premature infants, compared to children and adults, have lower basal myocardial contractility and contractile reserve due to reduced myofibril content and fewer mitochondria in their heart muscle. Infections in premature infants, such as CRKP BSI, can lead to cardiovascular dysfunction and progress rapidly to septic shock due to the activation of inflammatory mediators that affect vascular tone and cardiac function. The VIS can be a useful tool to quantify cardiovascular support in premature infants requiring hemodynamic support. Identifying the maximum VIS within 48 h of treatment initiation may help predict adverse outcomes in premature infants37. Aziz’s study on extremely low birth weight infants showed a correlation between the maximum VIS and mortality rate38. It is recommended that premature infants with CRKP BSI undergo early and thorough assessments of hemodynamic and cardiac function, with prompt intervention to reverse disease progression and reduce mortality. It is important to note that predicting the risk factors for death is only meaningful after the identification of the pathogen, as this study found that the average time from the onset of infection to pathogen identification was nearly 36 h. Prior to the diagnosis of CRKP BSI, identifying risk factors cannot fully predict mortality. Thus, only preliminary predictions can be made based on the clinical characteristics associated with the onset of infection.
Considering the poor outcomes of patients with CRKP BSI, early initiation of an active therapy is crucial. However, it is important to acknowledge that due to the underdeveloped organs in premature infants, most drugs sensitive to CRKP lack sufficient safety and efficacy data. Moreover, compared to adults and children, there is a scarcity of clinical experience and trials investigating CRKP-active drugs in premature infants, leading to limited effective antibiotic therapy options39. Our research indicates that tigecycline and polymyxin B are the two most sensitive drugs in CRKP drug sensitivity testing. Nevertheless, tigecycline is not recommended for use in children due to its side effect profile, including concerns about increased mortality rates40. Our current study observed frequent use of carbapenem-based combination therapy for treating CRKP infections in premature infants. Carbapenem- based combination therapy has shown lower mortality rates compared to non- carbapenem combination therapy41. Regardless of the presence or absence of carbapenem resistance genes, combinations like meropenem/polymyxin B and meropenem/fosfomycin have demonstrated high synergistic activity against a significant percentage of CRKP strains41. However, the univariate analysis in our study revealed that only treatment based on ceftazidime/avibactam (CAZ/AVI) was associated with a reduced risk of mortality. There are some studies on the pharmacokinetics and safety profiles of CAZ/AVI in pediatric patients aged 3 months to 18 years, whereas there is limited information on its efficacy and safety in newborns, particularly premature infants. Nonetheless, CAZ/AVI has shown efficacy and tolerability as a salvage therapy in neonates and children under 5 years old with CRE causing BSI42. Currently, the application of CAZ/AVI in premature infants is limited to a few case reports43,44,45. In this study, 17 infants were treated with CAZ/AVI, resulting in only one fatality. The clinical outcomes were positive, with no apparent adverse reactions observed. Therefore, premature infants could potentially benefit from CAZ/AVI therapy against CRKP isolates, potentially lowering mortality rates. However, due to the limited number of patients included in this study, further research is necessary to validate the efficacy and safety of CAZ/AVI for CRKP BSI in premature infants through larger clinical trials.
This study delved into the clinical significance of identified risk factors in predicting mortality among premature infants with CRKP BSI. The ROC curve analysis indicated that the AUC for predicting mortality in infants with both concurrent NEC and purulent meningitis was > 0.700, while the AUC for predicting mortality in infants with a maximum VIS value ≥ 52.5 points within 48 h of onset was > 0.800. These findings suggest that these three factors play a crucial role in CRKP infections in premature infants and can effectively forecast BSI mortality. Furthermore, the study explored the combined clinical utility of these three indicators in assessing mortality among premature infants with CRKP BSI, resulting in an AUC of 0.931. The combined assessment of indicators demonstrated a marked enhancement in clinical predictive capability compared to assessing concurrent NEC and purulent meningitis individually.
The current study has several limitations. It was conducted in a children’s hospital with a high prevalence of CRKP and included only premature infants. Additionally, the retrospective nature of the study raises the possibility that some clinical and demographic variables that could have influenced the outcomes were not fully recorded. Furthermore, the small sample size may have limited the ability to identify risk factors and outcomes. When utilizing the ROC curve to assess the predictive value of each indicator, the small sample size raises concerns about potential overfitting of the predictive model, necessitating caution in the interpretation and analysis of these results. Moreover, multi-center studies with larger sample sizes are required to validate and enhance the robustness of the findings.
Conclusions
To sum up, CRKP BSI in preterm infants poses a significant challenge for infection management and treatment, which may lead to higher mortality rate and poor prognosis. Effective predictive factors help precise treatment and improve the outcomes of CRKP BSI in premature infants. This study revealed a notable mortality risk in premature infants with CRKP BSI. A VIS value ≥ 52.5 points within 48 h of onset, along with concurrent meningitis and NEC, showed promise in predicting mortality in these infants, particularly when combined with other indicators. Early administration of new agents like CAZ/AVI could potentially enhance outcomes in this population. Thus, more attention should be paid to CRKP BSI in preterm infants.
Data availability
Data are provided within the manuscript. The raw datasets generated during the study are not publicly available due to an ethical restriction but are available from the corresponding author on reasonable request.
References
Sung, T. J., Sohn, J. A., Oh, S. & Lee, J. A. The influence of the variation in sepsis rate between neonatal intensive care units on neonatal outcomes in very-low-birth-weight infants. Sci. Rep. 10, 6687 (2020).
Mukhopadhyay, S. et al. Neurodevelopmental outcomes following neonatal late-onset sepsis and blood culture-negative conditions. Arch. Dis. Child. Fetal Neonatal Ed. 106, 467–473 (2021).
Chaurasia, S. et al. Characterisation and antimicrobial resistance of sepsis pathogens in neonates born in tertiary care centres in Delhi, India: A cohort study. Lancet 4, e752–e760 (2016).
Yadav, N. S. et al. Bacteriological profile of neonatal sepsis and antibiotic susceptibility pattern of isolates admitted at Kanti Children’s Hospital, Kathmandu, Nepal. BMC Res. Notes 11, 301 (2018).
Zou, H. et al. Emerging threat of multidrug resistant pathogens from neonatal sepsis. Front. Cell. Infect. Microbiol. 11, 694093 (2021).
Fang, P. et al. Prevalence of multidrug-resistant pathogens causing neonatal early and late onset sepsis, a retrospective study from the tertiary referral children’s hospital. Infect. Drug Resist. 16, 4213–4225 (2023).
Qiu, Y. et al. Invasive Klebsiella pneumoniae infections in community-settings and healthcare settings. Infect. Drug Resist. 14, 2647–2656 (2021).
Fu, P. et al. Bacterial epidemiology and antimicrobial resistance profiles in children reported by the ISPED program in China, 2016 to 2020. Microbiol. Spectrum 9, e00283-e321 (2021).
Zarras, C. et al. Neonatal Bloodstream Infection with Ceftazidime-Avibactam-Resistant bla KPC-2-Producing Klebsiella pneumoniae Carrying bla VEB-25. Antibiotics 12, 1290 (2023).
Mijac, V. et al. Intestinal colonization of preterm neonates with carbapenem resistant Enterobacteria at hospital discharge. Antibiotics 12, 284 (2023).
Nour, I. et al. Risk factors and clinical outcomes for carbapenem-resistant Gram-negative late-onset sepsis in a neonatal intensive care unit. J. Hosp. Infect. 97, 52–58 (2017).
Gaies, M. G. et al. Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr. Crit. Care Med. 11, 234–238 (2010).
Kurul, Ş et al. Inflammation, sepsis severity and neurodevelopmental outcomes of late-onset sepsis in preterm neonates. Pediatr. Res. 94, 2026–2032 (2023).
Fleiss, N. et al. Evaluation of the neonatal sequential organ failure assessment and mortality risk in preterm infants with late-onset infection. JAMA Netw. Open 4, e2036518 (2021).
Humphries, R., Bobenchik, A. M., Hindler, J. A. & Schuetz, A. N. Overview of changes to the clinical and laboratory standards institute performance standards for antimicrobial susceptibility testing, M100. J. Clin. Microbiol. 59, e0021321 (2021).
Magobo, R. E. et al. Outbreak of NDM-1-and OXA-181-producing klebsiella pneumoniae bloodstream infections in a neonatal unit, South Africa. Emerg. Infect. Dis. 29, 1531–1539 (2023).
Ghaith, D. M. et al. Genetic diversity of carbapenem-resistant Klebsiella Pneumoniae causing neonatal sepsis in intensive care unit, Cairo, Egypt. Eur. J. Clin. Microbiol. Infect. Dis. 39, 583–591 (2020).
Ohnuma, T. et al. Association of appropriate empirical antimicrobial therapy with in-hospital mortality in patients with bloodstream infections in the US. JAMA Netw. Open 6, e2249353 (2023).
Falcone, M. et al. Time to appropriate antibiotic therapy is a predictor of outcome in patients with bloodstream infection caused by KPC-producing Klebsiella pneumoniae. Crit. Care 24, 29 (2020).
Meng, H. et al. Risk factors and clinical outcomes of carbapenem-resistant Klebsiella pneumoniae bacteraemia in children: A retrospective study. Int. J. Antimicrob. Agents 62, 106933 (2023).
Nabarro, L. E. et al. Clinical and bacterial risk factors for mortality in children with carbapenem-resistant Enterobacteriaceae bloodstream infections in India. Pediatr. Infect. Dis. J. 36, e161–e166 (2017).
Burge, K. Y. & Markel, T. A. Neonatal microbiome, intestinal inflammation, and necrotizing enterocolitis. Microorganisms 10, 1382 (2022).
Lu, Q., Cheng, S., Zhou, M. & Yu, J. Risk factors for necrotizing enterocolitis in neonates: A retrospective case-control study. Pediatr. Neonatol. 58, 165–170 (2017).
Gane, B. et al. Risk factors and outcome in neonatal necrotising enterocolitis. Indian J. Pediatr. 81, 425–428 (2014).
Neu, J. & Walker, W. A. Necrotizing enterocolitis. N. Engl. J. Med. 364, 255–264 (2011).
Kang, C. et al. Simple scoring system that predicts the need for surgical intervention in infants with necrotizing enterocolitis. Arch. Med. Res. 54, 37–44 (2023).
Wang, Z. L. et al. Risk factors of necrotizing enterocolitis in neonates with sepsis: A retrospective case-control study. Int. J. Immunopathol. Pharmacol. 34, 1–18 (2020).
Peng, X. et al. Prevalence and antimicrobial resistance patterns of bacteria isolated from cerebrospinal fluid among children with bacterial meningitis in China from 2016 to 2018: A multicenter retrospective study. Antimicrob. Resist. Infect. Control 10, 1–10 (2021).
Yang, X. et al. Clinical characteristics and prognosis of Klebsiella pneumoniae meningitis in adults. Heliyon 10, e28010 (2024).
Huang, N. et al. Hypervirulent carbapenem-resistant Klebsiella pneumoniae causing highly fatal meningitis in southeastern China. Front. Public Health 10, 991306 (2022).
Wang, F., Jiang, J., Shi, G., Wang, J. & Zhou, S. Anti-infective treatment of purulent meningitis caused by carbapenem-resistant Klebsiella pneumoniae in a newborn: A case report. Transl. Pediatr. 9, 713–719 (2020).
Butts, R. J. et al. Comparison of maximum vasoactive inotropic score and low cardiac output syndrome as markers of early postoperative outcomes after neonatal cardiac surgery. Pediatr. Cardiol. 33, 633–638 (2012).
Song, J. et al. Vasoactive-inotropic score as an early predictor of mortality in adult patients with sepsis. J. Clin. Med. 10, 495 (2021).
Kallekkattu, D. et al. Threshold of inotropic score and vasoactive-inotropic score for predicting mortality in pediatric septic shock. Indian J. Pediatr. 89, 432–437 (2022).
Gaies, M. G. et al. Vasoactive-inotropic score is associated with outcome after infant cardiac surgery: An analysis from the Pediatric Cardiac Critical Care Consortium and Virtual PICU System Registries. Pediatr. Crit. Care Med. 15, 529–537 (2014).
Kharrat, A. et al. Validity of the vasoactive-inotropic score in preterm neonates receiving cardioactive therapies. Early Human Dev. 173, 105657 (2022).
Aziz, K. B. et al. Maximum vasoactive-inotropic score and mortality in extremely premature, extremely low birth weight infants. J. Perinatol. 41, 2337–2344 (2021).
Mukherjee, S., Mitra, S., Dutta, S. & Basu, S. Neonatal sepsis: The impact of carbapenem-resistant and hypervirulent Klebsiella pneumoniae. Front. Med. 8, 634349 (2021).
Mastrolia, M. V., Galli, L., De Martino, M. & Chiappini, E. Use of tigecycline in pediatric clinical practice. Expert Rev. Anti Infect. Ther. 15, 605–612 (2017).
Trecarichi, E. M. & Tumbarello, M. Therapeutic options for carbapenem-resistant Enterobacteriaceae infections. Virulence 8, 470–484 (2017).
Adaleti, R. et al. Investigation of in vitro efficacy of meropenem/polymyxin B and meropenem/fosfomycin combinations against carbapenem resistant Klebsiella pneumoniae strains. Acta Microbiol. Immunol. Hung. 70, 155–160 (2023).
Iosifidis, E. et al. Use of ceftazidime-avibactam for the treatment of extensively drug-resistant or pan drug-resistant Klebsiella pneumoniae in neonates and children< 5 years of age. Pediatr. Infect. Dis. J. 38, 812–815 (2019).
Nascimento, A. S. et al. Off-label use of ceftazidime-avibactam in a premature infant with multidrug-resistant Klebsiella pneumoniae infection: A case report. J. Pharm. Pract. 36, 1020–1025 (2023).
Pu, W. et al. Carbapenem-resistant Klebsiella pneumoniae osteoarthritis in two preterm infants treated with ceftazidime-avibactam. Pediatr. Infect. Dis. J. 42, 1124–1127 (2023).
Coskun, Y. & Atici, S. Successful treatment of pandrug-resistant Klebsiella pneumoniae infection with ceftazidime-avibactam in a preterm infant: A case report. Pediatr. Infect. Dis. J. 39, 854–856 (2020).
Funding
This study was supported by grants from the Scientific and Technological Projects of Henan Province (242102311201), the Medical Science and Technology Projects of Henan Province (LHGJ20230556) and the China Postdoctoral Science Foundation (2024M752951).
Author information
Authors and Affiliations
Contributions
Concept and study design: S.X. Data acquisition and analysis: Z.Y., H.Z., P.C. and L.L. Drafting of the manuscript and figures: P.C. and Z.Y. All authors approved the final manuscript as submitted and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, Z., Li, L., Cheng, P. et al. Clinical characteristics and mortality risk factors of premature infants with carbapenem-resistant Klebsiella pneumoniae bloodstream infection. Sci Rep 14, 29486 (2024). https://doi.org/10.1038/s41598-024-80974-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-80974-2