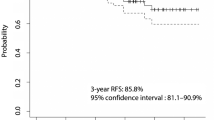
Abstract
Up to 80% of children/adolescents with Graves’ disease (GD) may require second-line treatment with either surgery or radioactive iodine (RAI) therapy after treatment with antithyroid drugs. These interventions aim to induce permanent hypothyroidism, but are not always successful. We aimed to evaluate the initial success rate (within the first year) of RAI treatment and its determining factors as second-line treatment in teenagers with GD. We also assessed the tolerability of RAI therapy and the onset speed of RAI-induced hypothyroidism. We conducted a retrospective chart review of children < 18 years treated with RAI (scaled fixed dose) for GD between January 2007 and December 2022 at the UZ Brussels. Fourteen teenagers treated with RAI were identified. Their ages at time of treatment ranged from 9.8 to 17.3 years, with administered I131 doses between 5.8 and 15.0mCi (median 7.9mCi). All but two patients responded within six months. Thyroxine treatment was started between 4 and 14 weeks (median 9 weeks) after RAI therapy. The time to thyroxine substitution correlated positively with age (Rho = 0.498; p = 0.099) and total I131-dose (Rho = 0.582; p = 0.047). One patient experienced transient RAI induced sialadenitis. None of the patients relapsed during a follow-up period of 1.2 to 13 years. A cure rate of 86% was observed in GD teenagers receiving a second-line RAI treatment, with no major complications. Most patients became hypothyroid within three months, underscoring the importance of early thyroid function monitoring.
Similar content being viewed by others
Introduction
Graves’ disease (GD) is an autoimmune disease characterized by stimulating anti-thyroid stimulating hormone receptor (TSHR) antibodies causing excessive production and release of thyroid hormone1,2,3,4. Although relatively rare in childhood, it is the most common cause of hyperthyroidism in children and adolescents1,3,4,5,6,7,8,9,10,11,12,13. The incidence of GD in Northern Europe is approximately 1:100 000 person-years in young children and 3:100 000 person-years in adolescents2,8,13. Moreover, it seems to be increasing in recent years3,8,14.
Most paediatric patients are female and are diagnosed after the age of 11 years, often after a delay of several months1,8,10,11,15. In the majority there is a positive family history of autoimmune thyroid disease or other autoimmune diseases1,3,8,16. Clinically, GD in childhood manifests itself most often with palpitations, weight loss, nervousness/irritability and restless sleep1,3,6,8. Upon physical examination, tachycardia, a diffuse goiter and sometimes exophthalmos may be present1,3,5,6,8,15. However, the signs and symptoms are often very subtle or even absent1,8,15. In the long-term, the disease can lead to persistent fatigue, worsening of school performances, and untoward effects on growth and pubertal development1,3,6,8,12,16.
Upon diagnosis, first-line treatment, at least in Europe, is the introduction of antithyroid drugs (ATD)1,2,4,7,8,11,12,13,14,15,17,18. Reported remission rates of ATD therapy, often given for 18 or 24 months, vary between 15–30%2,3,4,6,10,12,13,14,15,19, while some prolonged treatment courses report remission in up to 50–65% of the patients7,8,12,14,15,16. Several studies have investigated clinical and hormonal characteristics as predictive factors for remission. In most, but not all, younger age, a larger thyroid gland and higher levels of free thyroxine (FT4) and anti-TSHR antibodies at diagnosis were predictive of poor remission rates3,13,17.
Upon relapse, thyroidectomy or the administration of RAI is often proposed3,4,13,14,15,17,19. Other indications for considering a definitive therapy are persistent (often more than 2 years) need for ATD, poor compliance with ATD, ATD intolerance or patient preference3,4,8,11,13,15,16,19. In some centres, RAI treatment is preferred above thyroidectomy because it is considered safer, less expensive and is generally well tolerated1,5,9,10,11,14,19.
The current goal of RAI treatment in children is the induction of permanent hypothyroidism3,4,7,11,17,19. Cure rates of up to 97.5% have been reported in some studies, but others report much lower percentages of ~ 65%4,7,10,14. Differences in patient characteristics as well as differences in nuclear medicine practices (as there is no universally accepted guideline on I131-dosing, especially in the paediatric population) may explain this cure rate variability. Furthermore, an optimal schedule for monitoring the hypothyroidism onset after RAI therapy has not yet been established, and closer monitoring might be needed in those receiving higher RAI doses.
Therefore, we decided to analyse in our centre the efficacy and tolerance of RAI treatment given as a second-line therapy for GD in adolescents and the need for early monitoring afterward. We aimed to evaluate the failure rate (no hypothyroidism within the first year) after the first RAI treatment as well as the potential determinants of success or failure (such as age, sex, duration of disease, and ATD use before RAI, dose of I-131, tracer uptake at diagnostic I123 testing) in patients treated over the last 15 years. Safety and tolerance were analysed by evaluating complaints reported within the first year. We also assessed the follow-up regimen to identify the optimal timing for thyroxine replacement therapy.
Methods
Design
A monocentric retrospective study was set up to evaluate the cure/ failure rate and tolerance of RAI as a second-line treatment for GD in children.
Recruitment
We reviewed the hospital records of children and adolescents, who had received RAI treatment at the Department of Nuclear Medicine of the UZ Brussel (Brussels, Belgium) between January 2007 and December 2022.
Inclusion and exclusion
Inclusion criteria were an age below 18 years at the time of RAI therapy and a clinical/ biological follow-up of at least one year. Data on 14 teenagers were retrieved and all were included in the analysis, even when medical data were incomplete.
RAI procedure
RAI is administered orally as an out-patient procedure. Iodine overload is excluded by urinary spot analysis, being lower than 300 µg/L. A pregnancy test is performed in women of childbearing potential. Patients are instructed to stop ATD one week before RAI. A diagnostic 24 h I123 uptake test is first done except when patients refuse (e.g., due to transportation problems) or when problems with compliance are anticipated (e.g., needle fear, mental health problems). A scaled fixed dose based primarily on age ( a dose between 5 and 10mCi in the age group 10–13 and between 10 and 15mCi in the age group 14–18 years), with the highest dose given in those with increased thyroid volume (no thyromegaly or visible thyromegaly) and a lower fractional 24 h I123 uptake (< 50% and > 50%). Clinical visits after one and three months are routinely scheduled, allowing assessment of ongoing and past complaints, as well as thyroid function testing.
Definitions
Isolated hyperthyrotropinemia was defined as TSH 5-10mIU/L in combination with normal fT4, while hypothyroidism was defined as TSH over 10mIU/L independently from fT4 levels. Treatment failure was defined as persistent hyperthyroidism at 12 months and/or a relapse of hyperthyroidism after initial hypothyroidism. Theoretical thyroid volume was defined as the upper limit of thyroid volume in adolescents, as defined by the WHO and the International Council for Control of Iodine Deficiency Disorders (Bulletin of WHO, 1997).
Calculations
Total I131 activity administered per kg body weight, m2 body surface area, and theoretical thyroid volume were calculated.
Data collection
Clinical, hormonal, and imaging data pre- and post-RAI administration were extracted from the electronic medical records. The following clinical data were collected: sex, age, and clinical symptoms at diagnosis; duration of ATD treatment; observed complications of ATD; reason for RAI therapy; and age, complaints and body size measurements at the moment of RAI administration and hospital visits afterward. Results of thyroid function, including anti-TSHR antibodies, were collected at diagnosis and around the time of RAI treatment.
Statistical analysis
Statistical analysis was performed using the IBM SPSS Statistics software. Data are presented as median and range. For comparing categorical and continuous variables, the Chi-square test/ Fisher’s exact test and Mann–Whitney U test/ Kruskal-Wallis were used respectively. Spearman Rank correlation analysis was used for correlation between continuous parameters. A two-sided p-value ≤ 0.05 was considered significant.
Ethics approval
The study protocol was approved by the “Commissie Medische Ethiek” of UZ Brussel. Due to the retrospective nature of the study and in compliance with our institutional board review guidelines, informed consent and assent were not obtained from participants. All methods were conducted in accordance with relevant guidelines and regulations, and the need for informed consent was waived by the “Commissie Medische Ethiek” of UZ Brussel.
Results
Clinical and biological characteristics at diagnosis of GD
The clinical characteristics of the patients at diagnosis are shown in Table 1. Ten patients were female. Median age at diagnosis was 11.5 (range 6–14) years. One patient had Down syndrome. In 9 out of 11 patients with a detailed history available, a positive family history of thyroid disease was present. One patient was asymptomatic and was diagnosed on a routine hormonal screening for tall stature. None of the patients were diagnosed with another auto-immune disease. All patients had elevated anti-TSHR antibodies, suppressed TSH values and increased fT4 concentrations.
ATD treatment characteristics
All patients have been treated with methimazole. The median starting dose was 0.5 mg/ kg bodyweight, while it ranged between 0.25 and 1 mg/ kg bodyweight. In total, 7 patients have received only one course of ATD, while RAI therapy was introduced after 2 ATD courses in 6 patients and 3 ATD courses in 1 patient (Table 1). In total, 9 patients received ATD treatment for a total duration of at least two years. The median total duration of the ATD treatment was 36 months and ranged from 12 to 50 months, after excluding 1 patient with intolerance for ATD. This patient had to discontinue the medication after 2 weeks because of a methimazole-related rhabdomyolysis. Arthralgia/myalgia and a pruritic rash were other side effects noted in the medical files during the ATD treatment. These were reported during the first course of ATD but did not necessitate discontinuation.
RAI treatment characteristics
In 6 patients, RAI therapy was introduced because of relapse after stopping ATD; in another 6 patients, because of persisting disease despite prolonged ATD treatment (chosen by the patient or physician); in 1 patient, because of non-compliance at the request of the treating physician; and in 1 adolescent because of severe ATD-related rhabdomyolysis. The median RAI dose administered was 7.9mCi and ranged between 5.8 and 15.0mCi. Calculated for body weight, RAI dose ranged between 79 and 205µCi/kg and per theoretical thyroid volume, doses ranged between 407 and 955µCi/ml. Total RAI increased exponentially with age, while no age effect on the RAI per theoretical thyroid volume can be seen (Fig. 1).
Scatterplots of total RAI dose in relation to age at time of RAI treatment (top) and I131 dose per estimated thyroid volume in relation to age at RAI treatment.
Clinical and biological characteristics at RAI administration
The median age at RAI administration was 14.5 (9.8–17.3) years. In all patients with available data, anti-TSHR antibodies were present. Two patients were prepubertal, 7 pubertal, and the remaining 5 were postpubertal (Tanner stage V). BMI varied between 18 and 27 kg/m2 (median 21.8 kg/m2), with one patient with a BMI > 25 kg/m2 (Table 1). In 3 patients slight and stable proptosis was present. In the 10 patients with a diagnostic study, the iodine uptake after 24 h ranged between 33 and 83% (median 62%). Urinary iodine excretion varied between 20.9 and 155 µg/l in the 11 patients with available urine collections.
Cure rate and relapse after RAI treatment
RAI therapy induced hypothyroidism in all but 4 patients after a median duration of 9 weeks (4–14 weeks). Isolated hyperthyrotropinemia was observed in two more patients. The time between RAI administration and the start of thyroxine treatment was positively related to the age of the patients (Rho = 0.498; p = 0.099) and the total I131 dose (Rho = 0.582; p = 0.047).
In the hypothyroid patients, a thyroxine dose of 2 µg/kg bodyweight was started with a maximum dose of 100 µg in those aged 13–16 years and 150 µg in those older than 16 years. The median thyroxine dose after one year was 1.9 µg/kg body weight, ranging between 1.3 and 2.5 µg/kg. The shortest period of available clinical follow-up after RAI administration was 1.2 years, and the longest was 13 years. No patients had a relapse of GD.
In one of two adolescents with persisting hyperthyroidism, a second higher I131 dose of 10mCi was given after 12 months (the first dose was 6mCi at the age of 14.6 years). Unfortunately, no diagnostic I123 uptake-study was performed at the initial RAI administration in this particular patient, but iodine overload was excluded by urinary measurement. He had an I131 treatment-resistant GD, ultimately requiring a complete thyroidectomy. In the other adolescent (age 13.6 years) with RAI therapy failure, a dose of 8mCi was given. Her 24-hour 123 uptake was 67%, and urinary iodine excretion was normal. After 3 years of follow-up, she is still receiving a low dose of methimazole to control her hyperthyroidism.
Tolerance and side effects of RAI treatment
One patient experienced a transient salivatory gland inflammation in the first month after the administration of 10mCi. No other side effects were noted.
Discussion
The choice for RAI treatment
RAI therapy as a second-line therapy in paediatric GD is increasingly proposed in Europe1. Clinicians are getting more comfortable with RAI treatment in young people, given the rising evidence of short-term tolerance and long-term safety3,7,20,21. In our centre, RAI therapy has been the preferred second-line therapy for GD in adolescents for the last 15 years. On the other hand, thyroidectomy is also considered a safe treatment option for paediatric GD with high success rates22,23.
In most European centres for paediatric endocrinology, ATD are proposed as a first-line treatment to all GD patients and are usually given, when well tolerated, for at least 18 months6,7,14,15,17,24. In our cohort, most patients were treated with an ATD course given for at least 2 years. However, the optimal duration of ATD therapy is controversial, and more prolonged ATD administration has been advocated, although the evidence is limited17,24,25,26. A recent meta-analysis reported an overall remission rate of 34.4% in children and adolescents27. While prolonged ATD treatment of up to 8 years increases the GD remission rates of GD to 50–65%8,17,28, many paediatric endocrinologists are convinced there is limited clinical benefit in an ATD therapy for longer than 2 years and propose a definite treatment when relapse is observed after ATD discontinuation4,6,10,13,15,17,29,30.
In 6 of our 14 (42%) patients, RAI treatment was given for a relapse of the disease after stopping ATD, while in 6 others (42%), RAI was administered because of the need for prolonged (> 2 years) ATD therapy. One patient was referred for RAI therapy by his physician because of non-compliance with ATD therapy. Rhabdomyolysis related to methimazole was the reason for an urgent RAI administration in 1 of our patients. Consistent with previous studies, the majority of our patients were female and in puberty at the moment of RAI treatment1.
The goal of RAI treatment
The current goal of RAI in paediatric Graves’ disease, as stated by the European Thyroid Association, is to achieve permanent hypothyroidism3,7,18. Therefore, RAI should target thyroid ablation to minimise the time of continued medical supervision, the relapse rate, and the eventual long-term risk of malignant transformation of persistent viable but radiation-damaged thyroid cells18. In our study, the median time to induce hypothyroidism was 9 weeks, but the range from 4 to 14 weeks was quite broad pointing to large individual differences in thyroid sensitivity to RAI. It is known that most children/teenagers will become hypothyroid within 6 months after RAI, typically between 2 and 3 months3,4,5,6,7,9,15,17,19. However, a variable time course to hypothyroidism has been reported, up to 18 months in some studies, especially at lower RAI doses9,31.
We found a significant positive correlation between the time interval from RAI administration to the biological diagnosis of hypothyroidism and the total RAI dose administered (Table 2; Fig. 1). We hypothesize that this apparent paradoxical association is mediated by a larger thyroid gland in patients who received a higher RAI dose, since our scaled dosing favoured higher RAI doses in patients with larger thyroids. This slower remission of hyperthyroidism in teenagers receiving a higher RAI dose has been observed in two previous studies32,33. On the contrary, in several studies no association with RAI dose was found9,34. This difference between studies might be explained by different policies of administering ATD after RAI administration and the timing of subsequent thyroid function testing. Earlier onset of hypothyroidism has been documented in those patients not taking ATD and can be expected in those having earlier thyroid function testing post-RAI administration31. In our cohort, no ATD were given after RAI administration and thyroid function tests were in most patients obtained after one and two months, while subsequent follow-up measurements were more variable, depending on the clinical and hormonal status of the patient. The early onset of hypothyroidism post-RAI and the large individual variability highlight the need for both early and frequent follow-up visits and thyroid function tests to permit an early and optimal introduction of thyroxine replacement therapy.
The success rate of RAI treatment
Impressive cure rates of up to ≥ 95%, as well as very poor success rates of only 20%, have been reported in paediatric RAI treatment studies3,7,10,11,15,17,19,23,27,35. This diversity in cure rates has been explained by different goals of RAI treatment (either obtaining euthyroidism or hypothyroidism), different moments of evaluation (at 6 months or 12 months) and variability in RAI dosing between centres, even within the same country27. This great variability was clearly shown in a Belgian study exploring the responses of nuclear medicine to hypothetical adult cases of GD: although a mean dose of 6.8mCi would be given, in individual cases the difference between the lowest and highest dose was more than 17mCi in more than 50% of the cases36.
Currently, some nuclear medicine centres continue or have switched to using a fixed dose of RAI. While in others, a variable RAI dose is administered, based either on thyroid volume or age (also called scale fixed dosing) or a tailored dose is given, which is calculated based on a combination of thyroid volume, the 24 h I123 tracer uptake and the desired RAI activity per calculated weight of volume/weight of the thyroid5,7,17,37. A tendency to give higher RAI doses in the last decade has been observed38. In our study, where semi-calculated doses were given, mainly based on age, thyroid volume (visible goiter or no goiter) and I123 activity (< 50% and > 50%), the total I131 varied between 5.8 and 15.0mCi. These total RAI doses align with those administered in the most recent paediatric studies19,39,40.
Several investigators have suggested delivering doses of > 250µCi/g thyroid to be successful19,40. The 2022 European Thyroid Association even proposes a minimum dose of 400µCi/g thyroid18. Unfortunately, no thyroid volume estimations by ultrasound were performed in our patients to allow a calculation of the I131 dose/g thyroid tissue. When based on age-estimated theoretical upper limit thyroid volumes, a minimum delivered dose of 400µCi/ml thyroid volume was calculated in our cohort. However, since it is well known that in the majority of GD patients thyromegaly is present, most patients in our cohort probably received a dose below 400µgCi/g thyroid, but probably at least 250µCi/g thyroid, since severe thyromegaly is a contraindication for administering RAI at our centre41.
In many centres, including ours, failure to obtain hypothyroidism at 6 months is considered a therapeutic failure, and a second, often higher, dose of RAI is proposed3,42. In our study, treatment failures were observed in two older teenagers, who had received 6 and 10mCi respectively, suggesting that in post-pubertal teenagers doses above 10mCi might be more appropriate. A younger age, a larger thyroid gland, GD-associated ophthalmopathy, higher pretreatment thyroid hormone values, higher levels of anti-TSHR antibodies, longer use of ATD, and a high I123 uptake have been reported as risk factors for RAI treatment failure in adolescents with GD4,6,7,8,19,25,27,31,40. A thyroid volume of less than 45mL has been proposed as the best predictor for optimal success in one recent study43. Other studies, however, could not find any clinical or biological predictors for the induction of hypothyroidism within the first year7,9,11. Unfortunately, our cohort size is too small to look for predictors of success or failure.
Of note, in none of our patients who became hypothyroid within the first 6 months, a relapse was observed afterwards. Relapses are seldom seen after induction of hypothyroidism, but some long-term follow-up studies reported percentages between 1 and 2%32,40,44.
The side effects and risks of RAI treatment
Although the RAI procedure is considered to be relatively safe, some concerns about acute severe side effects, such as worsening of Graves’ ophthalmopathy and provocation of a thyroid storm, exist among paediatric endocrinologists4,12,13,17. However, in most paediatric studies as well as in our cohort, these complications were not seen7,8,10,13. Transient minor side effects such as neck tenderness and nausea in the first week of administration have been reported in some studies3,4,8. No such complaints were observed in our cohort, in accordance with the observations in several other studies4,14,32,44. However, one patient in our cohort did experience some transient complaints, which were suggestive of transient salivatory gland inflammation. In the literature, no other paediatric cases of salivary gland inflammation have been reported after RAI therapy given for hyperthyroidism, while this is a common complication after high-dose RAI for the treatment of thyroid cancer.
When considering the long-term risks, special interest in the paediatric population goes to the theoretical oncogenic and genetic damage potential of irradiation by the RAI procedure. Of note, an adolescent undergoing RAI therapy will receive a total body exposure equivalent to around 5–15 years of background radiation or 3–4 body CT scans, while the dose received by the gonads is the same as during a barium study6,12. To date, no increased risk for either thyroid cancer, gastric cancer or leukaemia has been reported, nor have congenital defects in children from mothers who had been treated in their own childhood been observed in long-term studies of more than 30 years follow-up4,6,7,10,17. In our study, the longest follow-up in one of our patients is only 13 years, which is not an optimal period for detecting malignancy after irradiation. In addition, no ultrasound examinations were performed as part of the follow-up to appreciate the eventual occurrence of a nodular benign transformation.
Recommendations
Based on our findings, we advise starting hormonal follow-up at one month post-RAI treatment with subsequent thyroid hormone evaluations every four weeks. However, if symptoms of hyper- or hypothyroidism are noticed by the patients in the meantime, they should have easy access to the laboratory. When falling FT4 concentrations are noticed, thyroxine treatment might be started before the patient becomes severely hypothyroid.
We also advocate the set-up of a European prospective comparative multi-centre study analysing the cure rate after 400µCi/g thyroid tissue not only in teenagers, but also in younger children (aged between 5 and 10 years), since GD is becoming also more frequent at younger ages. Furthermore, the need for a higher dose of 500µCI/g thyroid might be studied in teenagers with large goiters (> 45 ml).
Strengths and limitations
This study is the first study reporting on the use of RAI as second-line treatment for GD in children in Belgium. Comparison with outcomes after thyroidectomy as second-line treatment was not possible as only two paediatric patients underwent surgery at our hospital during the study period. Our study has several further limitations since it has a retrospective design, includes only a small number of patients, and spans a period of 15 years. However, national data on RAI outcomes are needed, since the iodine status, potentially influencing the uptake of RAI to some extent, differs between countries. We also agree that administering RAI in an outpatient setting limits the ability to capture less severe short-term complications.
Conclusion
This single-centre retrospective study on the use of RAI in Belgian teenagers with GD, given as a second-line treatment, confirms its good tolerance and an acceptable success rate of 86%. Our sample size is too small to reliably detect success predictors. Larger studies are thus needed to optimize the choice of the RAI dose and select the best candidates for RAI treatment.
Data availability
The data that support the findings of this study are available upon request to the corresponding author. The data are not publicly available due to restrictions e.g. their containing information that could compromise the privacy of research participants.
Abbreviations
- GD:
-
Graves’disease
- ATD:
-
Anti-thyroid drugs
- RAI:
-
Radioactive iodine
- BMI:
-
Bodymass index
- TSHR:
-
Thyroid stimulating hormone receptor
References
Ma, C., Kuang, A., Xie, J. & Liu, G. Radioiodine treatment for pediatric Graves’ disease. Cochrane Database Syst. Rev. 3, CD006294 (2008).
Marques, O., Antunes, A. & Oliveira, M. J. Treatment of Graves’ disease in children: The Portuguese experience. Endocrinol. Diabetes Nutr. (Engl Ed). 65 (3), 143–149 (2018).
Król, A., Czarniecka, A. & Jarząb, B. Definitive treatment of Graves’ disease in children and adolescents. Endokrynol Pol. 72 (6), 661–665 (2021).
Cohen, R. Z., Felner, E. I., Heiss, K. F., Wyly, J. B. & Muir, A. B. Outcomes analysis of radioactive iodine and total thyroidectomy for pediatric Graves’ disease. J. Pediatr. Endocrinol. Metab. 29 (3), 319–325 (2016).
Cemeroglu, A. P., Kleis, L., Wood, M. A. & Davis, A. T. Reliability of early iodine 123 uptake for treatment of Graves disease in children. Endocr. Pract. 17 (4), 541–545 (2011).
Rivkees, S. A. The treatment of Graves’ disease in children. J. Pediatr. Endocrinol. Metab. 19 (9), 1095–1111 (2006).
Pinto, T., Cummings, E. A., Barnes, D. & Salisbury, S. Clinical course of pediatric and adolescent Graves’ disease treated with radioactive iodine. J. Pediatr. Endocrinol. Metab. 20 (9), 973–980 (2007).
Léger, J., Oliver, I., Rodrigue, D., Lambert, A. S. & Coutant, R. Graves’ disease in children. Ann. Endocrinol. (Paris). 79 (6), 647–655 (2018).
Nebesio, T., Siddiqui, A., Pescovitz, O. & Eugster, E. Time course to hypothyroidism after fixed-dose radioablation therapy of Graves. J. Pediatr. 141 (1), 99–103 (2002).
Rivkees, S. A. & Dinauer, C. An optimal treatment for pediatric Graves’ disease is radioiodine. J. Clin. Endocrinol. Metab. 92 (3), 797–800 (2007).
Namwongprom, S., Unachak, K., Dejkhamron, P., Ua-apisitwong, S. & Ekmahachai, M. Radioactive iodine for thyrotoxicosis in childhood and adolescence: Treatment and outcomes. J. Clin. Res. Pediatr. Endocrinol. 5 (2), 95–97 (2013).
Cheetham, T. & Bliss, R. Treatment options in the young patient with Graves’ disease. Clin. Endocrinol. (Oxf). 85 (2), 161–164 (2016).
West, J. D., Cheetham, T. D., Dane, C. & Natarajan, A. Should radioiodine be the first-line treatment for paediatric Graves’ disease? J. Pediatr. Endocrinol. Metab. 28 (7–8), 797–804 (2015).
Azizi, F. & Amouzegar, A. Management of thyrotoxicosis in children and adolescents: 35 years’ experience inin 304 patients. J. Pediatr. Endocrinol. Metab. 31 (2), 159–165 (2018).
Bauer, A. J. Approach to the pediatric patient with Graves’ disease: When is definitive therapy warranted? J. Clin. Endocrinol. Metab. 96 (3), 580–588 (2011).
Chao, M. et al. Radioiodine treatment for pediatric hyperthyroid grave’s disease. Eur. J. Pediatr. 168 (10), 1165–1169 (2009).
Bahn, R. S. et al. Hyperthyroidism and other causes of thyrotoxicosis: management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Endocr. Pract. 17 (3), 456–520 (2011).
Mooij, C. F. et al. European Thyroid Association Guideline for the management of pediatric Graves’ disease. Eur. Thyroid J. 11 (1), e210073 (2022).
Kaplowitz, P. B., Jiang, J. & Vaidyanathan, P. Radioactive iodine therapy for pediatric Graves’ disease: A single-center experience over a 10-year period. J. Pediatr. Endocrinol. Metab. 33 (3), 383–389 (2020).
Turner, N., Driver, I., Salotti, J. A., Pearce, M. S. & Cheetham, T. Increasing use of radioiodine in young people with thyrotoxicosis in Great Britain. Eur. J. Endocrinol. 167 (5), (2012).
Tonge, J. & Soundararajan, K. Is radioiodine a better therapy than antithyroid drugs for achieving remission in those with juvenile Graves’ hyperthyroidism?. Arch. Dis. Child. 107 (1), 97–99 (2022).
Zaat, A. S., Derikx, J. P. M., Zwaveling-Soonawala, N., van Trotsenburg, A. S. P. & Mooij, C. F. Thyroidectomy in pediatric patients with Graves’ Disease: A systematic review of postoperative morbidity. Eur. Thyroid J. 10 (1), 39–51 (2021).
Engelbrecht-Wiggans, E. A. et al. Long-term retreatment outcomes after definitive management of Graves’ disease with radioactive iodine versus surgery. Surgery. S0039–6060 (24), 00699–00698 (2024).
van Lieshout, J. M., Mooij, C. F., van Trotsenburg, A. S. P. & Zwaveling-Soonawala, N. Methimazole-induced remission rates in pediatric Graves’ disease: A systematic review. Eur. J. Endocrinol. 185 (2), 219–229 (2021).
Song, A. et al. Long-term antithyroid drug treatment of Graves’ disease in children and adolescents: A 20-year single-center experience. Front. Endocrinol. (Lausanne). 12, 687834 (2021).
Azizi, F., Abdi, H., Amouzegar, A. & Habibi Moeini, A. S. Long-term thionamide antithyroid treatment of Graves’ disease. Best Pract. Res. Clin. Endocrinol. Metab. 37, 101631 (2023).
Puttawong, D. et al. Long-term outcomes of anti-thyroid drug treatment in childhood-onset Graves’ disease. Clin. Endocrinol. (Oxf). 98 (6), 823–831 (2023).
Léger, J., Gelwane, G., Kaguelidou, F., Benmerad, M. & Alberti, C. Positive impact of long-term antithyroid drug treatment on the outcome of children with Graves’ disease: National long-term cohort study. J. Clin. Endocrinol. Metab. 97 (1), 110–119 (2012).
Rivkees, S. A. Approach to the patient: Management and the long-term consequences of Graves’ disease in children. J. Clin. Endocrinol. Metab. 107 (12), 3408–3417 (2022).
Rabon, S., Burton, A. M. & White, P. C. Graves’ disease in children: Long-term outcomes of medical therapy. Clin. Endocrinol. (Oxf). 85 (4), 632–635 (2016).
McCormack, S. et al. Radioactive iodine for hyperthyroidism in children and adolescents: Referral rate and response to treatment. Clin. Endocrinol. (Oxf). 71 (6), 884–891 (2009).
Rivkees, S. A., & Cornelius, E. A. Influence of iodine-131 dose on the outcome of hyperthyroidism in children. Pediatrics 11 (4 Pt 1), 745–749 (2003).
Nakatake, N., Fukata, S. & Tajiri, J. Prediction of post-treatment hypothyroidism using changes in thyroid volume after radioactive iodine therapy in adolescent patients with Graves’ disease. Int. J. Pediatr. Endocrinol. 2011 (1), 14 (2011).
Clark, J. D., Gelfand, M. J. & Elgazzar, A. H. Iodine-131 therapy of hyperthyroidism in pediatric patients. J. Nucl. Med. 36 (3), 442–445 (1995).
Lutterman, S. L. et al. The efficacy and short- and long-term side effects of radioactive iodine treatment in pediatric Graves’ disease: A systematic review. Eur. Thyroid J. 10 (5), 353–363 (2021).
Tondeur, M., Glinoer, D., Sand, A., Verelst, J. & Ham, H. Variability of the administered radioiodine doses for the treatment of hyperthyroidism in Belgium. Clin. Endocrinol. (Oxf). 65 (2), 206–209 (2006).
Jawwad, M. et al. Long-term outcome of treatments for Graves disease in the children and adolescent population. Endocrine 81 (2), 223–230 (2023).
Cury, A. N. et al. Clinical experience with radioactive iodine in the treatment of childhood and adolescent Graves’ disease. Endocr. Connect. 2 (1), 32–37 (2012).
Namwongprom, S., Dejkhamron, P. & Unachak, K. Success rate of radioactive iodine treatment for children and adolescent with hyperthyroidism. J. Endocrinol. Invest. 44 (3), 541–545 (2021).
Trout, A. T. et al. Radioiodine treatment of pediatric Graves disease: A multicenter review. Pediatr. Radiol. 53 (1), 1–27 (2023).
Hegedüs, L., Hansen, J. M. & Karstrup, S. High incidence of normal thyroid gland volume in patients with Graves’ disease. Clin. Endocrinol. (Oxf). 19 (5), 603–607 (1983).
Rivkees, S. A. Pediatric Graves’ disease: Controversies in management. Horm. Res. Paediatr. 74 (5), 305–311 https://doi.org/10.1159/000320028 (2010).
Sheremeta, M. et al. Thyroid volume is the key predictor of hyperthyroidism remission after radioactive iodine therapy in pediatric patients. Eur. J. Pediatr. 182 (11), 4931–4937 (2023).
Ward, L. et al. Outcome of pediatric Graves’ disease after treatment with antithyroid medication and radioiodine. Clin. Invest. Med. 22 (4), 132–139 (1999).
Author information
Authors and Affiliations
Contributions
LC, JDS and EN wrote the original draft. HE supervised the writing of the manuscript. BS and HE treated patients. WS, IG, JV and CE were involved in the follow up of the patients. All authors helped revise the article critically for important intellectual content and have given final approval.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chielens, L., Nauwynck, E., Bourgeois, S. et al. A Belgian single centre outcome study of radioiodine treatment in adolescents with Graves’ disease. Sci Rep 14, 31063 (2024). https://doi.org/10.1038/s41598-024-82052-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82052-z