Abstract
We aimed to evaluate the incidence of residual shunt after patent foramen ovale (PFO) closure and analyze the anatomical features of PFO to determine the risk factors for significant residual shunt after PFO closure. Ninety-two patients who underwent PFO closure at our center between September 2021 and June 2022 were consecutively enrolled. Transthoracic saline contrast echocardiography was performed at 6 and 12 months postoperatively to evaluate the presence of a significant residual shunt. Preoperative measurements of the anatomical structures of PFO were performed using transesophageal echocardiography. Binary logistic regression analysis was performed to determine the risk factors for significant residual shunt at 12 months after PFO closure, and receiving operating characteristic curves were plotted to calculate the cutoff values. At the follow-up 12 months postoperatively, 22 (24%) of the 92 patients showed significant residual shunt. Thicker secondary septum and longer tunnel were independent risk factors for significant residual shunt after PFO closure. In particular, patients with secondary septum thickness > 6.55 mm or tunnel length > 10.10 mm were more susceptible to significant residual shunt. This study may guide clinicians in developing individualized PFO treatment strategies.
Similar content being viewed by others
Introduction
Patent foramen ovale (PFO) is a common structural heart disease with an incidence rate of approximately 25% in the adult population1. Currently, PFO closure is commonly used for the secondary prevention of PFO-associated stroke and for the treatment of refractory migraines in some patients2,3. PFO closure involves implanting a special metal occluder into the interatrial septum to eliminate the right-to-left shunt, thereby preventing stroke or treating migraine. Theoretically, the metal device will eventually achieve the complete elimination of right-to-left shunt through its mechanical effect and endothelialization. However, in reality, recent studies have shown that the incidence rate of residual shunt 1 year after PFO closure ranges from 10 to 30%4,5,6,7,8. However, the occurrence of residual shunts may be the result of one factor or the combination of multiple factors, including the anatomical features of the PFO channel, inappropriate occluder devices, and failure of endothelialization (different follow-up durations)4,5,9,10,11.
Here, we aimed to evaluate the effect of PFO anatomical features as measured by transesophageal echocardiography (TEE) on the presence of significant residual shunt after PFO closure.
Methods
Population
Ninety-two patients who underwent PFO closure at Yongchuan Hospital of Chongqing Medical University between September 2021 and June 2022 were enrolled consecutively. All patients underwent PFO closure under TEE guidance. To eliminate the impact of occluder device size on study results, only patients using a 25 mm occluder were included in this study cohort. The patient inclusion criterion was transthoracic saline contrast echocardiography (scTTE) showing significant shunting (≥ large shunt). The patient exclusion criteria were: (1) presence of other congenital cardiac anomalies, including atrial septal defect, ventricular septal defect, patent ductus arteriosus;(2) preoperative three-dimensional computed tomography (CT) pulmonary angiogram and scTTE showing shunting at the pulmonary level; (3) patients who failed to complete postoperative follow-up on time.
The study was approved by the Institutional Review Board of Yongchuan Hospital of Chongqing Medical University (IRB no.:2022-KLS-146), and we confirm that all methods were performed in accordance with the relevant guidelines. All patients or their guardians (for patients younger than 18 years) provided informed consent in writing prior to the procedure.
PFO diagnosis and TEE-guided PFO closure
PFO diagnosis
Patients with PFO were first screened and diagnosed using the transcranial Doppler (TCD) bubble test and then graded using ScTTE during the Valsalva maneuver. Specifically, a 10-ml syringe containing 1 ml of air and another 10-ml syringe containing 8 ml of saline were obtained. The syringes were connected to a 3-way stopcock, and 1 ml of venous blood was collected and mixed with them. After mixing, the resulting mixture was immediately injected via an elbow vein. Based on the number of bubbles observed in the left atrium via transthoracic echocardiography (TTE), PFO right-to-left shunt was divided into five grades: (1) no shunt: no bubbles observed in the left atrium; (2) small shunt: 1–10 bubbles; (3) medium shunt: 10–30 bubbles; (4) large shunt: > 30 bubbles but still countable; (5) extra-large shunt: the left atrium is turbid and filled with many bubbles that cannot be counted12. The study was approved by the Institutional Review Board of Yongchuan Hospital of Chongqing Medical University.
Device Specifications and Closure Procedure
In this study, two brands of occlusion devices were used: the Amplatzer PFO Occluder manufactured by Abbott, USA, and a Cardi-O-Fix PFO occluder produced by Starway Medical Co., Ltd., China. Both devices consist of two self-expanding discs made of nickel-titanium alloy, filled with non-woven polyester sheets. This material not only blocks abnormal blood flow but also stimulates endothelialization. The Amplatzer PFO Occluder is available in four sizes: 9-PFO-1818, 9-PFO-2518, 9-PFO-3025, and 9-PFO-3525, while the Cardi-O-Fix PFO occluder is available in five sizes: PF1818, PF1825, PF2525, PF2535, and PF3030. In this study cohort, the selected devices were the Amplatzer PFO Occluder model 9-PFO-2518 and the Cardi-O-Fix PFO occluder by Starway Medical Co., Ltd. model PF2525 (Fig. 1).
size diagram of PFO Occluder Devices.
The selection of device size was based on the standards outlined in the Abbott PFO Occluder device manual, adjusted for local population differences, and determined by an experienced interventional physician. For simple PFOs, an occluder size of 25 mm was selected, whereas a 30 mm occluder was used if one or more complex PFO characteristics were present: (1) Atrial septal aneurysm (ASA, ≥ 10 mm excursion) with long tunnel (≥ 10 mm length); (2) ASA (≥ 10 mm excursion) with thickened septum secundum (≥ 10 mm thickness); (3) Prominent ASA with excessive mobility (≥ 20 mm total excursion); (4) Lipomatous hypertrophy of septum secundum (≥ 15 mm thickness).
The procedure was performed under general anesthesia. Figure 2 shows details of the TEE-guided PFO closure. All patients received antiplatelet therapy postoperatively, comprising 100 mg of aspirin for 6 months and 75 mg of clopidogrel for 3 months after the procedure.
The percutaneous intervention involved (a) Transesophageal ultrasound showing PFO; (b) guiding the contrast catheter to the interatrial septum with a soft-tip guidewire under transesophageal ultrasound monitoring; (C) passing the guidewire through the PFO; (d) placing the guidewire into the left superior pulmonary vein; (e) exchanging the contrast catheter for the occluder delivery sheath; (f) withdrawal of the soft-tip guidewire; (g) passing the occluder along the delivery sheath to the left atrium, releasing the left atrial umbrella; (h) retracting the delivery sheath to release the right atrial umbrella; (i) Traction testing was used to confirm good fixation of the occluder; (j) Release PFO occluder. All key steps in the diagram are pointed out with green arrows. PFO, patent foramen ovale; RA, right atrium; LA, left atrium; LSPV, left superior pulmonary vein.
TEE measurement of PFO anatomical features
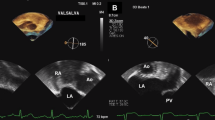
TEE is the gold standard for evaluating the anatomical features of PFO. The anatomical data of all patients were measured using TEE before the percutaneous intervention. The measurements included opening size on the left atrial aspect, opening size on the right atrial aspect, tunnel length and thickness of secondary septum, as well as the presence of comorbid ASA, Eustachian valve, Chiari network, and atrial septal defects. ASA was defined as a localized outpouching of the interatrial septum with a protrusion of more than 10 mm beyond the plane of the atrial septum, observed either towards the right atrium or the left atrium, or exhibiting bidirectional movement. The measurement methods are shown in Fig. 3. The PFO anatomical data of all patients were measured by the same experienced sonographer using the same TEE instrument (model no.: ACUSON NX3 Elite, Siemens Medical Solutions, Issaquah, WA, USA).
Measurements made in the TEE mid-esophageal bicaval view (90–120°): (A) Size of right atrial opening: the widest distance between the primary septum and secondary septum was measured perpendicular to the interatrial septal plane. (B) Size of left atrial opening: the widest distance between the primary septum and secondary septum was measured perpendicular to the interatrial septal plane. (C) Tunnel length: the length of overlap between the primary septum and secondary septum was measured parallel to the interatrial septal plane. (D) Secondary septum thickness: the width of the thickest point on the middle to lower segment of the secondary septum was measured perpendicular to the secondary septum. PFO, Patent foramen ovale; LA, Left Atrium; RA, Right Atrium; SS, Secondary septum.
PFO closure postoperative follow-up
All patients underwent electrocardiogram (ECG) monitoring for 24 h after the percutaneous intervention. Complete blood count, ECG, and transthoracic electrocardiography were performed at 24 h. Echocardiography was performed at 1, 3, 6, and 12 months post-discharge, and scTTE was performed at 6 and 12 months for the quantitative assessment of residual shunt during the Valsalva maneuver. The presence of significant residual shunt was defined as ≥ medium shunt (10–30 bubbles).
Statistical analysis
Statistical analysis was performed using SPSS 26.0. Continuous data were expressed as the mean ± standard deviation. Count data were expressed as frequency and/or percentage. Using the presence of significant residual shunt (≥ medium residual shunt) as the dependent variable, univariate analysis was performed using logistic regression analysis. Variables showing statistical significance in the univariate analysis were subsequently included in the multivariate analysis to determine the risk factors and calculate the odds ratio with a 95% confidence interval (CI). Receiver operating characteristic (ROC) curves were plotted for all significant variables in the multivariate analysis, and the cutoff values were calculated. Differences with p ≤ 0.05 were considered statistically significant.
Results
Study population
Ninety-two patients were consecutively enrolled in this study, including 21 males (23%) and 71 females (77%), with a mean age of 40.5 ± 14.8 years. All patients completed follow-up at 6 and 12 months postoperatively. Table 1 presents the patients’ baseline characteristics and PFO anatomical characteristics. All patients underwent percutaneous intervention under TEE guidance with a 100% success rate of occluder implantation. Symmetrical occluders were used in 12 (13%) cases and asymmetrical occluders in 80 (87%) cases. Postoperative adverse events included 2 cases of delayed pericardial effusion occurring at 2 d and 4 d postoperatively, respectively, which recovered after puncture and drainage; 6 cases of arrhythmia,all of which were cases of paroxysmal atrial fibrillation that recovered completely with symptomatic treatment; and 2 cases of ecchymosis at the puncture site, which recovered after re-compression for hemostasis (Table 2).
Factors Influencing Residual Shunt Post-PFO Closure
At the follow-up 6 months postoperatively, 26 (28%) patients showed significant residual shunt. At 12 months, 22 (24%) patients showed significant residual shunt, including 1 (1%) case with extra-large residual shunt, 8 (9%) cases with large residual shunt, and 13 (14%) cases with medium residual shunt (Table 3). During the follow-up period between 6 and 12 months, the number of patients with significant residual shunt decreased by 4. All 4 patients showed medium residual shunt at 6 months; by 12 months, the residual shunt decreased to a small shunt in 1 patient and no shunt in 3 patients.
Using the presence of significant residual shunt (≥ medium shunt) at 12 months as the dependent variable, logistic regression analysis was used to perform univariate analysis on gender, age, size of the left atrial opening, size of the right atrial opening, tunnel length, secondary septal thickness, presence of comorbid ASA, presence of Eustachian valve, occluder type, and baseline shunt grade. The results indicated that the size of the left atrial opening (hazard ratio (HR): 1.36, 95% CI: 1.00–1.84, P = 0.047), tunnel length (HR: 1.23, 95% CI: 1.08–1.39, P = 0.002), and secondary septal thickness (HR: 4.53, 95% CI: 2.25–9.13, P < 0.001) were significant. These variables were included in the multivariate analysis, which showed that tunnel length (HR: 1.19, 95% CI: 1.01–1.40, P = 0.036) and secondary septal thickness (HR: 4.04, 95% CI: 1.97–8.26, P < 0.001) were independent risk factors for significant residual shunt at 12 months postoperatively (Table 4).
ROC curves and cutoff values of independent risk factors
ROC curve analysis was performed to obtain the optimal cutoff values for tunnel length and secondary septal thickness to determine the risk factors for significant residual shunt after PFO closure. For tunnel length > 10.1 mm, the maximum Youden index was 0.417 (area under the curve: 0.67), sensitivity was 82%, and specificity was 86%. For secondary septal thickness > 6.6 mm, the maximum Youden index was 0.675 (area under the curve: 0.853), sensitivity was 55%, and specificity was 87% (Fig. 4).
ROC curves of secondary septal thickness and tunnel length.
Discussion
Recent studies have demonstrated that adding PFO closure to drug therapy leads to better clinical outcomes for patients with PFO-associated strokes. Furthermore, multiple studies have also revealed that PFO closure can reduce the frequency and duration of migraine attacks in patients with PFO with comorbid migraine2,13. More extensive exploration of PFO closure has garnered growing interest regarding the incidence of residual shunts. The “effective closure” of PFO has been defined as ≤ small (0–10 microbubbles in the left atrium) right-to-left residual shunt on scTTE14. The occurrence of residual shunt after PFO closure is mainly affected by factors such as tunnel length, presence of ASA, selection of occluder device, and PFO size5,9,10. Numerous factors influence the occurrence of residual shunt after PFO closure. This study investigates the impact of PFO anatomical features on residual shunt after PFO closure, offering clinical insights to optimize PFO closure strategies.
In this study, anatomical structures of PFO were measured preoperatively using TEE, and residual shunt at 6 and 12 months postoperatively was quantitatively assessed using scTTE. We believe that this study benefitted from the following methodological advantages: 1. Residual shunt was assessed using scTTE. Although this was a semi-invasive test, it did not have the disadvantage of underestimating residual shunt as with the TCD bubble test. 2. All enrolled patients received PFO-specific occluder devices of the same size, which prevented bias caused by using occluder devices of different sizes in previous studies when determining the factors for predicting the effect of PFO anatomical features on residual shunt. 3. Patients were selected through consecutive enrollment, and a consecutive cohort can prevent selection bias to some extent.
Early studies defined complex PFO as cases with tunnel length > 8 mm and/or secondary septal thickness > 10 mm15. Excessive overlap between the primary and secondary septum can prevent occluder devices with a shorter waist from being fully deployed and, hence, fail to conform closely to the interatrial septum15. Our multivariate analysis revealed that tunnel length > 10.1 mm (HR: 1.19, 95% CI: 1.01–1.40) was an independent risk factor for significant residual shunt after PFO closure. We believe that when an occluder device is used to cover a PFO, a thinner primary septum will undergo deformation due to the tension at the waist of the device and bunch up between the occluder discs on both sides; a longer tunnel may result in more content between the two discs, which will generate greater closure tension in the interatrial septum, and prevent the full deployment of the occluder discs to cover the fossa ovalis. Both conditions can lead to the occurrence of residual shunt after PFO closure. The secondary septum is a blood-shunting ridge during the fetal period, and its thickness varies greatly between individuals15.
Our multivariate analysis revealed that secondary septal thickness > 6.6 mm (HR: 4.04, 95% CI: 1.97–8.26, P < 0.001) is an independent risk factor for residual shunt after PFO closure. This is a novel finding. We believe that when the secondary septum is thicker, its bulge will cause a blank space between the right atrial disc of the occluder device and the fossa ovalis, thereby forming a new shunt channel. This will prevent the device from conforming fully to the fossa ovalis and, hence, create a residual shunt channel (Fig. 5a). In fact, both secondary septal thickening and excessive tunnel length can increase the content between the two discs, thus giving rise to greater tension in the occluder device when covering the PFO. Greater tension can impede the full deployment of occluder devices with a shorter waist or harder texture and hence prevent them from fully conforming to the interatrial septum (Fig. 5b). Previously, patients with tunnel length > 8 mm or secondary septum thickness > 10 mm were defined as complex PFO. Furthermore, softer occluder devices with a longer waist were preferred for long-tunnel PFO, whereas smaller occluder devices were usually chosen for PFO with a thicker secondary septum to ensure better conformance to the fossa ovalis15. Both approaches are intended to reduce the tension of the occluder device when covering the PFO. Therefore, we suggest that individualized treatment plans should be designed for patients with PFO based on the anatomical characteristics of PFO. Emphasis should be placed on using anatomical features to guide the selection of occluder devices. For patients with the risk factors of thicker secondary septum and longer tunnel, we recommend the use of occluder devices with a longer waist and a softer texture.
a. Due to the irregular thickening of the secondary septum, the surface of the right atrial disc could not conform fully to the fossa ovalis and, hence, formed a new shunt channel on the side of the occluder device. b. Thickened secondary septum and/or overly long tunnel can lead to excessive tension between the two discs on the occluder device and poor conformance to the PFO. LA, Left Atrium; RA, Right Atrium.
Previous studies have demonstrated that the presence of ASA is associated with the occurrence of residual shunt after PFO closure5,9. Among the 92 patients enrolled in this study, only 2 patients had comorbid ASA, both of which were relatively small. Hence, we did not use a larger occluder device to cover the ASA, as a larger occluder device is an independent risk factor for residual shunt occurrence. The outcomes were as expected: at 12 months postoperatively, PFO was successfully closed in both cases, and no significant residual shunt was observed.
Deng et al.16 provided important insights into the long-term impact of residual shunt after PFO closure on the recurrence of stroke and TIA. Their findings indicate that residual shunt, particularly moderate or large residual shunt, significantly increases the risk of recurrent ischemic events. Specifically, patients with residual shunt had a higher recurrence rate of stroke or TIA compared to those with complete closure (2.32 vs. 0.75 events per 100 person-years)16. Currently, for patients with recurrent residual shunt, the main management strategy is re-closure with a larger occluder device. However, only a handful of studies have demonstrated the effectiveness of second closure, and the strategy for second closure remains controversial17,18,19,20. Therefore, identifying the risk factors for residual shunting after PFO closure and implementing individualized treatment plans are of significant importance to patients with PFO. Previous studies have shown that preventing residual shunt is crucial through careful patient selection, appropriate device selection, and ensuring complete closure during the procedure. Customized occluder devices that better match the anatomical characteristics of individual patients may help reduce the incidence of residual shunt. Additionally, rigorous follow-up using scTTE can help detect and manage residual shunts early, potentially improving long-term outcomes21.
In this study, we examined the application of double-disc occluder devices for PFO closure and their association with residual shunt occurrence. The double-disc design generally provides enhanced stability, offering improved support during the closure procedure and minimizing the risk of device displacement22. However, in certain anatomical conditions, using a double-disc device may increase the incidence of residual shunt, particularly in patients with longer tunnels or a thicker septum secundum23. These anatomical characteristics can hinder the full conformity of the two discs to the atrial septum, thus compromising closure efficacy. When both of these anatomical features are present, the likelihood of postoperative residual shunt significantly increases. Therefore, it is crucial to consider alternative closure approaches, including bioabsorbable occluders or percutaneous suture-based closure techniques for PFO.
This study has several limitations. Firstly, all PFO measurements were taken solely in the bicaval view. The tunnel length and anatomical PFO opening depend on the TOE probe angle. This approach may miss the largest PFO dimensions, sometimes seen in the short-axis 45° (SAX 45°) view. Different TEE angles can reveal varying PFO dimensions, leading to discrepancies in anatomical assessment. This reduces the reproducibility of our study. Future studies should use multiple echocardiographic views and standardize or incorporate multiple TEE angles for a more accurate assessment of PFO anatomy. Secondly, this study only included patients with large shunts, possibly explaining the high incidence of residual shunts. Future research should include a broader range of shunt sizes for a more comprehensive evaluation. Additionally, further follow-up at 2 years would help assess long-term outcomes of PFO closure and the persistence of residual shunts. Finally, all ultrasound measurements were performed by a single physician, which may introduce bias. Future research should involve multiple independent physicians to improve data reliability and generalizability. Nevertheless, given the enrollment of a consecutive cohort based on strict inclusion and exclusion criteria, as well as the timely preoperative measurement of precise anatomical features using TEE, we believe that our study is of crucial clinical significance.
Conclusions
Precise measurements of the anatomical structures of PFO by standardized TEE revealed that a thicker secondary septum and longer tunnel are independent risk factors for significant residual shunt after PFO closure. In particular, patients with secondary septum thickness > 6.55 mm or tunnel length > 10.10 mm were more susceptible to significant residual shunt. In view of the current high incidence of residual shunts after PFO closure, this study may provide clinicians with a basis for the development of individualized treatment strategies for PFO.
Data availability
The datasets used and analysed during the current study are available from the corresponding author upon reasonable request. The requests to access the datasets should be directed to Yulin Hu, Department of Nephrology and Rheumatology, Yongchuan Hospital of Chongqing Medical University; email: huyl4949@126.com.
References
Homma, S. et al. Patent foramen ovale. Nat. Rev. Dis. Primers 2, 15086. https://doi.org/10.1038/nrdp.2015.86 (2016).
Mac Grory, B. et al. Advances in the management of cardioembolic stroke associated with patent foramen ovale. BMJ 376, e063161. https://doi.org/10.1136/bmj-2020-063161 (2022).
Farjat-Pasos, J. I. et al. Transcatheter patent foramen ovale closure in stroke patients with thrombophilia: Current status and future perspectives. J. Stroke 24, 335–344. https://doi.org/10.5853/jos.2022.01697 (2022).
Sorensen, S. G., Spruance, S. L., Smout, R. & Horn, S. Transcranial Doppler quantification of residual shunt after percutaneous patent foramen ovale closure: Correlation of device efficacy with intracardiac anatomic measures. J. Interv. Cardiol. 25, 304–312. https://doi.org/10.1111/j.1540-8183.2011.00714.x (2012).
Marchese, N. et al. Residual shunt after percutaneous closure of patent foramen ovale with AMPLATZER occluder devices—influence of anatomic features: A transcranial Doppler and intracardiac echocardiography study. EuroIntervention 9, 382–388. https://doi.org/10.4244/EIJV9I3A61 (2013).
Sorensen, S. G., Aguilar, H., McKnight, W. K., Thomas, H. & Muhlestein, J. B. Transcranial Doppler quantification of residual shunt after percutaneous patent foramen ovale closure. Comparison of two devices. J. Interv. Cardiol 23, 575–580. https://doi.org/10.1111/j.1540-8183.2010.00587.x (2010).
Braun, M. U. et al. Transcatheter closure of patent foramen ovale in patients with cerebral ischemia. J. Am. Coll. Cardiol. 39, 2019–2025. https://doi.org/10.1016/s0735-1097(02)01904-6 (2002).
Mi, Z. et al. Efficacy and safety of transesophageal ultrasound-guided patent foramen ovale closure for migraine in adolescents. Front. Pediatr. 11, 1296825. https://doi.org/10.3389/fped.2023.1296825 (2023).
von Bardeleben, R. S. et al. Long term follow up after percutaneous closure of PFO in 357 patients with paradoxical embolism: Difference in occlusion systems and influence of atrial septum aneurysm. Int. J. Cardiol. 134, 33–41. https://doi.org/10.1016/j.ijcard.2008.02.031 (2009).
Vitarelli, A. et al. Echocardiographic findings in simple and complex patent foramen ovale before and after transcatheter closure. Eur. Heart J. Cardiovasc. Imaging 15, 1377–1385. https://doi.org/10.1093/ehjci/jeu143 (2014).
Shafi, N. A. et al. Determinants and clinical significance of persistent residual shunting in patients with percutaneous patent foramen ovale closure devices. Int. J. Cardiol. 137, 314–316. https://doi.org/10.1016/j.ijcard.2009.06.045 (2009).
Bernard, S., Churchill, T. W., Namasivayam, M. & Bertrand, P. B. Agitated saline contrast echocardiography in the identification of intra- and extracardiac shunts: Connecting the dots. J. Am. Soc. Echocardiogr. https://doi.org/10.1016/j.echo.2020.09.013 (2020).
Ahmed, Z. & Sommer, R. J. Reassessing the PFO-migraine trials: Are we closer to closure?. J. Am. Coll. Cardiol. 77, 677–679. https://doi.org/10.1016/j.jacc.2020.12.017 (2021).
Wang, S. B. et al. Prevalence and extent of right-to-left shunt on contrast-enhanced transcranial Doppler in Chinese patients with migraine in a multicentre case-control study. Cephalalgia 38, 690–696. https://doi.org/10.1177/0333102417708203 (2018).
Rana, B. S., Shapiro, L. M., McCarthy, K. P. & Ho, S. Y. Three-dimensional imaging of the atrial septum and patent foramen ovale anatomy: Defining the morphological phenotypes of patent foramen ovale. Eur. J. Echocardiogr. 11, i19-25. https://doi.org/10.1093/ejechocard/jeq122 (2010).
Deng, W. et al. Residual shunt after patent foramen ovale closure and long-term stroke recurrence: A prospective cohort study. Ann. Intern. Med. 172, 717–725. https://doi.org/10.7326/M19-3583 (2020).
Majunke, N., Wallenborn, J., Baranowski, A., Wunderlich, N. & Sievert, H. Device closure of residual shunt after percutaneous closure of patent foramen ovale. EuroIntervention 5, 833–837. https://doi.org/10.4244/eijv5i7a139 (2010).
Schwerzmann, M. et al. Implantation of a second closure device in patients with residual shunt after percutaneous closure of patent foramen ovale. Catheter. Cardiovasc. Interv. 63, 490–495. https://doi.org/10.1002/ccd.20221 (2004).
Diaz, T. et al. Management of residual shunts after initial percutaneous patent foramen ovale closure: A single center experience with immediate and long-term follow-up. Catheter. Cardiovasc. Interv. 76, 145–150. https://doi.org/10.1002/ccd.22475 (2010).
Butera, G. et al. Residual shunting after percutaneous PFO closure: How to manage and how to close. Catheter. Cardiovasc. Interv 82, 950–958. https://doi.org/10.1002/ccd.25097 (2013).
Hammerstingl, C. et al. Risk and fate of residual interatrial shunting after transcatheter closure of patent foramen ovale: A long term follow up study. Eur. J. Med. Res. 16, 13–19. https://doi.org/10.1186/2047-783x-16-1-13 (2011).
Khan, A. R. et al. Device closure of patent foramen ovale versus medical therapy in cryptogenic stroke: A systematic review and meta-analysis. JACC Cardiovasc. Interv. 6, 1316–1323. https://doi.org/10.1016/j.jcin.2013.08.001 (2013).
Verolino, G., Calderone, D., Gavazzoni, M., Sala, D. & Sganzerla, P. Clinical performance of the gore septal occluder in patent foramen ovale closure in different septal anatomies: 1-year results from a single-center experience. J. Clin. Med. https://doi.org/10.3390/jcm12185936 (2023).
Author information
Authors and Affiliations
Contributions
These authors contributed equally: Zeyu Mi and Chao Li. Z.Y.M., C.L., Y.L.H., G.H., D.Y.Y and D.X.H. conceived and designed the study. All the authors assessed and diagnosed the patients. All the authors were involved in the acquisition, analysis, and interpretation of the data, with Z.Y.M. and C.L. taking the primary role in the statistical analysis. Z.Y.M., C.L., H.L.G. and X.W.Z. drafted the manuscript. Y.L.H. detected any errors in the whole process and had primary responsibility for the final content. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was approved by the Institutional Review Board of Yongchuan Hospital of Chongqing Medical University (IRB no.:2022-KLS-146).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mi, Z., Li, C., He, G. et al. Relation between anatomical features of patent foramen ovale and residual shunt based on transesophageal echocardiography. Sci Rep 15, 1497 (2025). https://doi.org/10.1038/s41598-024-82122-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82122-2