Abstract
This study assessed the intensity of salt stress in the two tomato varieties by measuring variables associated with the water regime, chlorophyll content, normalized difference vegetation index, gas exchange, and yield. The cultivars Amalia and Claudia, which represent tolerance and susceptibility to salinity, were evaluated. Three treatments were established in plastic pots, using a completely randomized design: T1, saline soil (ECse = 6.9 dS m−1 without QuitoMax application); T2, nonsaline soil (ECse = 0.95 dS m −1 with QuitoMax application); and T3, saline soil (ECse = 6.9 dS m−1) with QuitoMax application. The QuitoMax was applied at a rate of 300 mg L-1, during the flowering phenophase. QuitoMax caused an increase in the variables evaluated in both varieties (tolerant and susceptible) of tomato, with a lower contribution of QuitoMax to the variables related to water regime and the greatest contributions to chlorophyll content and photosynthetic activity. QuitoMax contributed positively to all variables and was superior to stress intensity for most of the variables evaluated in the tolerant variety (Amalia), except for stem thickness and the number of flowers per bunch. In the susceptible variety (Claudia), the five variables of stress intensity exceeded the contribution of QuitoMax, with the strongest effects on osmotic potential, fruit mass, and yield per plant. The present work demonstrates the feasibility of using this biostimulant to increase the tolerance of tolerant varieties and maintain tolerance in tomato varieties susceptible to salinity, reducing the intensity of saline stress and increasing plant performance under salinity conditions.
Similar content being viewed by others
Introduction
Salt is an abiotic stress condition that affects plant development and productivity1. Annually, salinization attributable to human activities leads to the abandonment of millions of hectares of traditional crops2,3. Salinity can have detrimental effects on crops through nutritional interference4. Moreover, in the medium term, it can lead to ionic toxicity by causing an accumulation of salt in vacuoles and other organelles, thereby impacting various organs, including the photosynthetic apparatus5,6. Salinity can also limit nutrient uptake capacities due to reduced leaf expansion and biological yield7. The plant- and crop-level effects mentioned above significantly diminish the profitability of saline soils and heighten vulnerability in terms of food security8.
A comprehensive solution to soil salinization is very difficult to develop at the moment due to the multifactorial effects of agricultural output9. The employment of efficient microorganisms, promoting growth, and sustaining nutrient availability has proven to be highly successful10,11. Efforts are underway to develop biostimulants that can confer tolerance to salinity12.
Some crops, such as tomato (Solanum lycopersicum L.), show moderate tolerance to salinity13. Due to the importance of this crop for human nutrition, significant efforts have been made to increase its yield and quality even under saline soils14. Tomato has been distributed globally, demonstrating its ability to adapt and produce under various environmental conditions. It is considered one of the most economically valuable vegetables in the world15. Tomato hybrids and cultivars have also helped to boost yields in salty soils16. The primary restriction of their contributions to tolerance is their cost17, particularly for small-scale agricultural projects.
Currently, research is conducted on the genetic improvement of salinity tolerance through the introgression of genes conferring tolerance18,19, particularly for specific cation uptake and evasion of Na+ 14, to obtain genetic materials with tolerance to salinity. Nevertheless, these advancements often require significant time to integrate into production systems and remain inaccessible to many producers, thereby limiting their application in agricultural practices.
Given that salinity tolerance is a trait dominated by a set of genes20, it is possible to monitor its accessible germplasm. This strategy would allow for the recommendation of varieties taking into account the saline levels that are present in diverse agricultural systems, contributing to ecosystem sustainability21. Assessing the intensity of generated stress enables decisions regarding the continuation of using a particular cultivar or altering land use practices22. In addition to salinity tolerance or susceptibility research with differential or commercial cultivars, the use of biostimulants can help reduce the intensity of saline stress in crops23. QuitoMax has been shown to improve salinity tolerance in tomato24,25,26 and lettuce27, improve transpiration in bell pepper28, reduce the incidence and severity of Alternaria solani leaf spots in tomato29, and stimulate initial tomato stand development30,31. QuitoMax is a chitosan-based liquid formulation produced from the chitin present in the lobster’s exoskeleton. It was initially designed as a developmental promoter under saline conditions32. However, a detailed study of its contribution to developmental and yield variables under saline conditions may lead to its subsequent use as a salinity attenuator in tolerant and susceptible varieties31.
The current study aimed to determine the effects of the commercially available biostimulant QuitoMax on the salinity tolerance of various tomato cultivars. The study was specifically designed to evaluate the effect of QuitoMax on several developmental traits, water status, gas exchange parameters, and yield components of tomato cultivated under saline conditions. With the current research, it will be feasible to explain how QuitoMax(R) promotes plant metabolic and physiological activities. Additionally, assess the level of stress with the administration of the biostimulant in tolerant and susceptible varieties, as well as its contribution to the improvement of agricultural productivity.
Material and method
Experimental site
The trials were established in the seedling production greenhouse of the Instituto Tecnológico del Valle del Yaqui, Sonora, Mexico, which is located in Block 611 of the Yaqui Valley, where vertisoil soils predominate33 with 49% clay. During the experimental period, there was an average temperature of 21 °C, and an average relative humidity of 37% was maintained.
Soil and plant material characteristics
The soil used for the research was characterized physiochemically at the Tepeyac Soil Laboratory in Ciudad Obregón, Sonora, Mexico. The soil properties are similar to that reported26. Based on the soil characteristics (data not shown), a saline treatment (electrical conductivity in the saturation extract of 6 dS m-1) and a control treatment (electrical conductivity in the saturation extract of ECse = 0.98 dS m-1 were used.
Two different tomato varieties were used: one tolerant (Amalia) and one susceptible (Claudia) to salinity, as previously described34. Both varieties have a specific growth habit and a biological cycle of approximately 125 days. Seedling production was carried out under controlled conditions. Plants of an average height of 14 cm were selected 28 days following emergence. The plants were transferred to plastic pots with a soil volume of 4.63 kg (width: 21 cm and height: 19.5 cm), where they were maintained until the fruit harvest stage.
Crop management
Fertilization was carried out35 for tomato crops under greenhouse conditions using TRIPLE-19 fertilizer (19% of N, P, and K, respectively) at a rate of 450 kg ha-1 (0.002 kg pot-1). The bottom application rate for all treatments was 50%. The remaining 50% of fertilizer was applied 45 days after germination, following the instructions of the producer.
Treatments and experimental design
The treatments were randomly assigned following a completely randomized experimental design, utilizing a unifactorial arrangement. The type of soil, with or without salinity, was considered as the source of variation, while the varieties were not taken into account as a factor. Each treatment was replicated three times. Three treatments were established: T1: saline soil without QuitoMax; T2: nonsaline soil + QuitoMax; and T3: saline soil + QuitoMax. These treatments were applied to both varieties. Six replications per treatment were established.
The suggested dose of QuitoMax was 300 mg L-136, and it was administered foliarly five days after transplanting. The same treatment rate was repeated after ten days. The application time was not considered a source of variation in the current trial.
Morphophysiological variables evaluated
Plant height (PH, cm) was measured from the stem base to the apical apex. The root length (RL, cm) was measured from the root collar to the main root boundary37. Both variables were measured with a TRUPER brand tape or flexometer, with a measurement error of 0.0001 cm. Plant stem thickness (ST, cm) was measured with a digital caliper (TRUPER) starting two centimeters above the base of the stem. The leaf number (LN, unit) was determined by counting the number of leaves on each plant. All these variables were evaluated at the flowering phenophase. The sample size for these variables was five plants per replicate, totaling 15 plants per treatment. The PH/RL ratio was calculated for a total of 15 plants per treatment using the ratio between root length and plant height. For better experimental precision, data were gathered from the same plants used to analyze the pH and RL variables38.
The root volume (RV, cm3) was obtained through the destructive method described by Shabbir et al.39. Five plants were randomly sampled for each treatment and carefully washed to separate the soil adhering to the roots with low-pressure water. Subsequently, the plants were individually introduced to a 500 mL graduated cylinder with a known volume of 150 mL of distilled water, and by volume difference, the volume of roots corresponding to each replication was determined, and the average of the five plants per replication in each treatment was obtained39.
Developmental dynamics were assessed on a total of eight randomly selected plants per treatment. These plants were marked and numbered to accurately assess the relative growth rate (RGR) at seven-day intervals. This measurement was made from the time of transplanting until the flowering phenophase. The final height of the plants was divided by the number of days elapsed, and the RGR, expressed in cm day-1, was obtained40.
The normalized difference vegetation index (NDVI) was measured with a GreenSeeker portable sensor between 10:00 h and 11:00 h in the flowering phenophase. In each treatment, 20 measurements were taken at a height of 0.60 m from the seedling canopy, according to the sensor reference41. This variable was evaluated to compare, in each treatment, the NDVI value; -1 < NDVI > 1, whose interpretation is related to a quick and directed diagnosis of the nutritional conditions of the crop (especially nitrogen) and the possible incidence of stress. NDVI values close to 1 represent a better nutritional status42.
The chlorophyll concentration (CL) was measured with a SPAD 501 portable meter (Minolta Camera Co., Ltd., Osaka, Japan). Measurements were taken in the middle part of the leaf during the flowering phenophase between 10:00 and 11:00 h43. Readings were taken in the middle part of the plant, taking an average of three readings from eight plants of each treatment.
Water regime variables
To assess water potential (ΨL), 24 random samples were taken from five plants (from the two leaves most exposed to sunlight) per treatment at 5:00 (predawn) and 10:30 AM. Immediately after collection, the samples were separated into their organs (roots and leaves), and the water potential was measured with a Schollander pressure pump (PMS-100; PMS Instrument Company, Albany, OR, USA). During the measurements, the samples were quickly placed in double zip lock bags and sealed. The leaves were then placed in the chamber sample holder, and pressure was applied until sap exudate was observed in the developed cut. The criteria for classifying water stress in plants were determined based on the values of ΨL and were defined as follows: no stress when ΨL > -1.0 MPa, moderate stress between − 1.0 > ΨL > -1.4 MPa and severe stress when the water potential fell to ΨL < − 1.4 MPa44.
To determine the saturated osmotic potential (Ψs), measurements were performed under saturated weight conditions. 24 samples of roots and leaves per treatment were taken and placed in Petri dishes for rehydration with distilled water. They were then placed in double zip lock bags and kept at 8 °C for 12 h. The samples were then wrapped with aluminum foil for freezing in liquid nitrogen and stored in a freezer at -80 °C. Subsequently, the samples were thawed at room temperature and centrifuged at 3,000 rpm for 3 min to obtain cell juice from roots and leaves. Ψs was determined from 100 ml aliquots with a vapor pressure osmometer (Vapro 7120; ELITech Group, Smithfield, RI, USA). The osmotic adjustment was measured as the difference in the saturated osmotic potential of leaves between the control and saline treatments45.
Gas exchange variables
Photosynthetic activity (A) was measured in leaves 10 days after biosimulant application in the flowering phenophase and was evaluated with a portable system (LI-6400XT, LI-COR, Inc.). The IRGA (infrared spectrum gas analyzer) measures CO2 and water vapor concentrations using a spectrometer that operates in the infrared spectrum of the equipment. Measurements were taken between 10:00 am and 11:00 am on sunny days. For this measurement, the three leaves most exposed to direct solar radiation (replicates per plant) were inserted, by their central part, into a 3.0 × 2.0 cm gas exchange daylight chamber. All measurements were performed with a light intensity greater than 1500 µmol m-2 s-1 and with a CO2 concentration of 400 µmol CO2 m-2 s-1 at a constant flow rate of 500 µmol s-1. The variables measured were maximum photosynthesis (A, µmol CO2 m-2 s-1) and transpiration (E, µmol H2 O m-2 s-1). Water use efficiency (WUE) was determined by the quotient of maximum photosynthetic activity and transpiration46.
Yield component variables evaluated
At the end of the experiment, the number of bunches per plant (NV-P, unit) number of flowers per bunch (NFl-V, unit), number of fruits per vunch (NFr-V), fruit mass (FM, g), and yield (Y, kg plant-1) were evaluated for five plants per replicate within each treatment. The variables NV-P, NF-V, NFl-V, and NFr-V were evaluated by counting each at plant and bunch level, respectively. FM was assessed by separately weighing the fruits harvested from each plant and determining the average weight of the fruits per replication in each treatment. Yield was obtained by weighing all commercial and noncommercial fruits from each plant within each replicate.
Stress intensity (SI) and contribution of Quitmax® (QC) to varietal development and yield
The stress intensity was determined between saline and nonsaline treatments with the application of QuitoMax following the equation [Eq. 1] proposed by47 where some nominal modifications were made:
Vst and Vnst represent the average of the evaluated variables in the saline and non-saline treatments, respectively.
The contribution of QuitoMax (QC) was determined in the saline treatments using the following equation [Eq. 2] proposed by the authors of the present work:
V(st-Q) and V(st + Q) represent the average of the evaluated variables without and with QuitoMax application, respectively.
Statistical analysis
For each variable evaluated, the normality of the data was evaluated48, and then a simple classification analysis of variance was carried out based on a linear fixed effects model49. When detecting significant differences between the three established treatments, Tukey’s multiple comparisons test50 was used for 5% and 1% significance levels. The standard error of the mean of the treatments (SEx), coefficient of variation (CV), and unadjusted coefficient of determination (R2) were determined and presented. The correlation coefficient between Chlorophyll content and NDVI was determined. All the statistical analyses were performed in the statistical software STATISTICA, version 14.0 for Windows51.
Results
Morphophysiological variables in response to salt stress and the application of QuitoMax
Significant differences were found between the treatments evaluated for both varieties and for all the variables evaluated (Table 1). The PH value of the tolerant variety Amalia (F = 8.25; p = 0.00226) was greatest in the T2 treatment (nonsaline soil + QuitoMax), with a 30.1 cm plant height, which was significantly different from that of the T1 treatment (saline soil - QuitoMax). Similarly, the cultivar Claudia (F = 162.95; p = 0.00003) exhibited the same trend, with a greater effect in the T2 treatment (32.76 cm). The behavior of the salinity-susceptible variety Claudia (Table 1) was similar to that of Amalia (tolerant), but the values were greater than those of the latter. The treatments with the biostimulant stood out compared to the control treatments (T1). This result indicates the feasibility of using susceptible varieties to validate the protective and development-promoting effects of this biostimulant against soil salinity. This result demonstrated the remedial power of QuitoMax when applied to tomatoes, regardless of the genetics of the cultivar used.
The other four morphophysiological variables evaluated are shown in Table 1, and a similar behavior to that obtained for PH was obtained. Accordingly, the RL (F = 93.60; p = 0.00006 for Amalia and Claudia varieties F = 103.24; p = 0.00006), SS (Amalia with F = 57.19; p = 0.00011 and for Claudia with F = 69.98; p = 0.00001), LN (F = 120.30; p = 0.00002, in Amalia and F = 160.25; p = 0.00005 in Claudia), and RV (F = 24.19; p = 0.00004 in Amalia and F = 80.14; p = 0.00012 in Claudia)] had similar results. Overall, the T2 treatment was superior to that of the T1 treatment and did not significantly differ from the T3 treatment in terms of RL and RV variables for the tolerant variety Amalia or in PH for the susceptible cultivar Claudia (Table 1). The highest morphophysiological values were obtained when the bio-stimulant was applied under non-saline conditions (T2), and the lowest values (T1) were obtained when QuitoMax was not applied under saline conditions (Table 1).
There were significant differences between the two varieties (F = 17.5; p = 0.03752 and F = 18.25; p = 0.03321 for Amalia and Claudia, respectively). There was greater variability in the susceptible variety (Claudia) than in the tolerant variety (Amalia) (Table 2). The linear fixed effects mathematical model for the analysis of variance explained 94% of the total variability in the effect of the treatments established in the susceptible variety (Claudia), while in the tolerant variety (Amalia), it explained only 54% (R2). An analysis of the PH/RL ratio (Table 2) revealed a value of 0.81 in T1 (saline soil without QuitoMax) for both varieties, which demonstrated the effects of salinity on the root system of the plants regardless of their relative tolerance to salinity. When the bio-stimulant was applied, an increase in both parameters was observed, with the greatest increase occurring at T3 (0.89) in the Amalia variety. However, in the Claudia variety, there was no promotion effect since the indicator was reduced (Table 2). No results were found from other research using this bio-stimulant in tomato cultivation under salinity conditions where this variable is evaluated.
NDVI and chlorophyll content
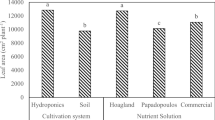
When the NDVI was evaluated in tomato for both varieties, significant differences were found between the treatments evaluated (Fig. 1). The highest values were obtained in T2 for both varieties, and the lowest were in T1. In the varieties Amalia and Claudia, the application of the bio-stimulant resulted in increases of 0.14 and 0.12 units, respectively, in the Normalized Difference Vegetation Index (NDVI) when comparing the plants subjected to salinity alone (T1 and T3) (Fig. 1). Higher NDVI values (-1 < NDVI > 1) represent better plant nutritional status. The results obtained for this indicator revealed the positive influence of the application of the bio-stimulant QuitoMax on tomato under salinity-stressed conditions.
Effect of QuitoMax application on the normalized difference vegetation index (NDVI) of tomato plants of the Amalia and Claudia varieties under semi controlled conditions subjected to three treatments: T1, saline soil without QuitoMax; T2, non-saline soil + QuitoMax; and T3, saline soil + QuitoMax. The rectangular bars represent the standard deviation of the mean of the treatments. R2: unadjusted coefficient of determination; ES: standard error of the mean of treatments; CV: coefficient of variation.
Chlorophyll levels measured in the leaves of Amalia significantly differed between treatments (F = 194.15; p = 0.00001). The highest values were obtained in T2, and the lowest was obtained in T1; however, when the bio-stimulant was applied to the saline medium, the values did not exceed those in the QuitoMax treatment under non-saline conditions, and the values decreased by only 4.33 units and exceeded those in T1 by more than 20 units (Table 3). In the Claudia variety, significant differences were found between T1 and the other treatments (F = 31.59; p = 0.00006). The highest values were obtained in T2, and the lowest was obtained in T1; however, when the bio-stimulant was applied to the saline medium (T2), the values did not exceed those in the QuitoMax treatment under non-saline conditions (T3), and no differences were found between them (Table 4).
Water regime under salt stress and QuitoMax application
The differences in the water potentials of the varieties in the different treatments were highly significant [F = 108.5; p = 0.00023, F = 112.46; p = 0.000321 for Amalia and Claudia, respectively (Fig. 2a and b)], and a similar response was found for the osmotic potential (F = 207.32; p = 0.00461, F = 201.31; p = 0.00117 for Amalia and Claudia, respectively). However, in Claudia, treatments T1 and T3 did not result in differences in water potential (Fig. 2b). This result indicates that in the susceptible variety, the applied dose of QuitoMax was not sufficient to achieve a reduction in water potential. The highest values were found in T2 for both varieties, and the lowest were found in T3 for both the varieties and indicators evaluated. This response demonstrated the ability of QuitoMax to promote plant water balance under nonsaline conditions. The differences in water and osmotic potentials between the saline and nonsaline treatments indicate the capacity of plants of both varieties to adjust their metabolism and maintain water intake under these stress conditions (Fig. 2a and b). However, it is important to note that when QuitoMax was applied, the best results were obtained in this medium.
Water potential (WP) and osmotic potential (OP) in tomato plants of the Amalia (a) and Claudia (b) varieties under semi controlled conditions subjected to three treatments: T1, saline soil without QuitoMax; T2, nonsaline soil + QuitoMax; and T3, saline soil + QuitoMax. The rectangular bars represent the standard deviation of the mean of the treatments.
Under saline conditions, when QuitoMax was applied, the OP values decreased in both varieties of plants concerning WP but increased in Amalia (Fig. 2a). This result is important because it indicates that under salinity stress conditions, the application of the bio-stimulant could have influenced the metabolic readjustment to reduce their water potential and be able to absorb water under saline conditions. Under saline conditions, the plants of the two varieties underwent significant osmotic adjustment due to the increase in the concentration of osmotically active solutes concerning that in T2 (Fig. 3), with the highest value being found at T3 in the Amalia variety (2.71 MPa).
Osmotic adjustment saturated osmosis in tomato plants of the Amalia and Claudia varieties under semicontrolled conditions subjected to three treatments: T1, saline soil without QuitoMax; T2, nonsaline soil + QuitoMax; and T3, saline soil + QuitoMax. The rectangular bars represent the standard deviation of the mean of the treatments.
Gas exchange variables in response to salt stress and the application of QuitoMax
When comparing the A (max) values obtained, significant differences were found between the treatments evaluated for both varieties (Fig. 4). The highest values were recorded in the non-saline medium enhanced with the biostimulant (T2), while the lowest was noted in the saline medium devoid of biostimulant (T1), underscoring the detrimental impacts of salinity on the photosynthetic machinery of tomato plants. When comparing the values obtained in treatments one and three, increases of 17.69 and 11.78 µmol CO m s22−1 were observed when QuitoMax was applied to the Amalia and Claudia varieties, respectively.
Gas exchange [photosynthesis (A) and transpiration (E)] in tomato plants of the Amalia (a) and Claudia (b) varieties in saline media and the control under semicontrolled conditions subjected to three treatments: T1, saline soil without QuitoMax; T2, nonsaline soil + QuitoMax; and T3, saline soil + QuitoMax. The rectangular bars represent the standard deviation of the mean of the treatments. R2: unadjusted coefficient of determination; ES: standard error of the mean of treatments; CV: coefficient of variation.
The increases observed in this investigation when QuitoMax was administered to the saline media are noteworthy since photosynthetic activity is the key physiological variable that leads to yield. The increases in photosynthetic activity observed when the biostimulant was applied in this study demonstrated, for the first time, the effectiveness of QuitoMax in increasing photosynthesis levels in tomato under saline conditions. When the effects of QuitoMax on tomato plants under salt stress were studied, significant differences were found between treatments in both varieties (Fig. 4). Transpiration was greater in the biostimulant treatments; the highest transpiration values were obtained in T2, and the lowest transpiration values were obtained in T1 for both varieties.
When QuitoMax was applied under saline conditions, greater transpiration was obtained, which shows that under saline stress, the plants can maintain higher levels of physiological activity than those where the bio-stimulant was not applied. No results were found on the effects of QuitoMax under saline conditions for this indicator in tomatoes or in other species, thus demonstrating the importance of the results obtained in the present study.
The WUE in the Amalia showed the highest values at T3 when QuitoMax was applied under saline conditions, and the lowest values under stress conditions without the application of the QuitoMax (Table 3). The application of the bio-stimulant did not result in a significant difference in this indicator among the Claudia variety, indicating its lack of response. Moreover, in the saline medium treatments, the values of the indicator were either similar to or greater than those observed in the treatment without salinity.
Yield components variability in response to salt stress and the application of QuitoMax
Significant differences were found for the variables NRP (F = 66.12; p = 0.00431), MF (F = 100.16; p = 0.00221), and Y (F = 79.54; p = 0.04121). However, the number of flowers and fruits per bunch did not significantly differ between treatments for either variety (Table 5). Among the three variables that showed significant differences, the effect of the treatments explained more than 95% of the total variability (R2) (Table 5).
The best results for both varieties were obtained in treatment T2 (non-saline soil + QuitoMax), demonstrating the ability of the biostimulant to promote development and yield (Table 5). With the application of the biostimulant under saline conditions before transplanting, a positive effect on fruiting efficiency (12%) and yield per plant was obtained for both varieties (Table 5). The results indicate that QuitoMax stimulates plant development and yield indicators.
Evaluation of QuitoMax contribution and the stress intensity in the variables evaluated
The contribution of QuitoMax to the varieties studied was positive. The highest percentages were found for the number of leaves, chlorophyll content, NDVI, maximum photosynthesis, and transpiration in both varieties. This result shows that the biostimulant used had a greater effect on the leaf area than on the root system. For example, QuitoMax had a similar contribution to photosynthetic activity, which was 70% for both varieties (Fig. 5).
QuitoMax contribution (QC) and stress intensity (SI) in a Amalia (tolerant) and b Claudia (susceptible) to salinity based on 6 dS m-1 in the saturated extract of the soil. PH plant height, RL root length; SS: stem thickness; LN: number of leaves; RV: root volume; RGR: relative growth rate; NDVI: normalized difference vegetation index; CHL: Chlorophyll; WP: water potential; OP: osmotic potential; A: photosynthesis; E: transpiration; NV-P: number of vunch per plant; NFl-V: number of flowers per vunch; NFr-V: number of fruits per plant; FM: fruit mass; Y: yield.
The stress intensity of the evaluated variables remained between 1% and 20%. The variables that presented the highest stress intensities were stem thickness (SS), osmotic potential (OP), and transpiration (E) in both varieties (Fig. 5). Therefore, even with the application of the bio-stimulant, the salinity condition likely affects the water regime of the plant. In the tolerant variety Amalia (Fig. 5a), the stress intensity was greater than that in the susceptible variety (Claudia) (Fig. 5b), although this indicator was surpassed by the contribution of QuitoMax, with greater values in the Claudia variety. In this susceptible variety, stress intensity exceeded the contribution of QuitoMax to three morphological variables (RV, WP, and OP), which shows that it significantly affects the water regime of the plants. SI also outperformed QC in terms of fruit mass yield (FM) and yield per plant (Y). This result indicates its susceptibility to salt stress.
Discussion
The QuitoMax has a pH-promoting effect on tomato under saline conditions. Other researchers studied the impact of QuitoMax on tomato crops and found similar results, albeit without the presence of salt stress29,52,53,54,55. A study conducted by55 indicated the efficiency of QuitoMax, even under non-saline conditions, which is consistent with that obtained in the T2 treatment for both differential varieties (Table 1)30. reported a 7% increase in the root length with QuitoMax compared to the control under non-saline conditions. The dose applied by these authors is considered high in relation to that applied here (300 mg L-1) and to that reported by other studies56,57. Additionally58, obtained similar results for root length, (5% increase) with respect to plants not treated with QuitoMax under non-saline conditions59. reported an RL-promoting effect by applying 90 mg L-1 of chitosan, obtaining an increase of 15% in root development similar to the results achieved here24. applied chitosan (a precursor of QuitoMax) and reported a positive and significant effect (40%) on root length in tomato under saline and non-saline conditions. The response of both varieties to QuitoMax application indicates that QuitoMax stimulates RL under saline conditions regardless of the variety. This result is of utmost importance since plants need a root system architecture that responds to the water and nutrient demand of aerial organs. Rapid root elongation is an important indicator of stress tolerance in tomato cultivation60.
The results obtained for SS in this study were consistent with the findings reported by55. In their study, they investigated the impact of chitosan application at a concentration of 150 mg L-1 on tomato plants and found a positive correlation between the application of chitosan and salinity tolerance. QuitoMax has not been previously studied under saline conditions in this crop; nonetheless, this bio-stimulant is acknowledged as a promoter of stem development under non-saline conditions30,54,61. In a study conducted by21, where the effects of exogenous application of chitosan at a concentration of 100 mg L-1 were investigated on lettuce plants (Lactuca sativa L.) exposed to salinity levels of 100 mM NaCl. The study reported a stimulating effect on growth rates in response to the chitosan treatment.
An analysis of the PH/RL ratio (Table 2) revealed a value of 0.81 in T1 (saline soil without QuitoMax) for both varieties, which demonstrated the effects of salinity on the root system of the plants regardless of their relative tolerance to salinity62. Other researchers recognize that the root-stem ratio in plants is crucial for their growth and development, as it allows plants to adjust to the amount of nutrients taken at the root level and the products of photosynthesis63.
In the Normalized Difference Vegetation Index (NDVI) when comparing the plants subjected to salinity alone (T1 and T3) (Fig. 1). Higher NDVI values (-1 < NDVI > 1) represent better plant nutritional status42,64,65. The results obtained for this indicator revealed the positive influence of the application of the bio-stimulant QuitoMax on tomato under salinity-stressed conditions. This promoted adequate nutrition and development of tomato plants regardless of the susceptibility of the variety to salinity stress, thus promoting a better response to stress in tomato plants. A study by66 demonstrated that the NDVI is related to biomass, leaf area, plant cover, and nitrogen and chlorophyll contents in plants, which reveals the importance of the results found when evaluating this indicator when QuitoMax is applied under salt stress conditions.
Regardless of susceptibility to salt stress, QuitoMax seems to be an alternative for alleviating salt stress in tomato. Some studies agree with those obtained in the present work when QuitoMax was applied under non-saline conditions, where increases of 12% in chlorophyll content were found in tomato crops obtained with the application of 1 g L-1 15 days after sowing56,57. This increase is related to increased growth as a result of increased photosynthetic input, as has been reported in a previous work with chitosan67.
When evaluating the effects of chitosan application on the adverse effects of salinity on tomato plants, positive effects were detected by quantifying the leaf chlorophyll content24,68, and similar effects were detected under water-stressed conditions69, resulting in better performance of plants under salt stressed conditions. The increase in chlorophyll content was positively correlated with the NDVI found in the saline treatment of both varieties (r = 0.98**) when QuitoMax was applied. The application of QuitoMax increased the levels of this indicator, highlighting the significance of using this biostimulant in tomato growth under saline circumstances.
Ruiz-Sánchez et al.44demonstrated that treatments T1 and T3 resulted in severe stress conditions as their water potential reduced to ΨL < − 1.4 MPa, even in soil with 88% moisture content. This result indicates that the plants are in a physiological drought state70.
The differences between varieties could be related to the intervarietal response to salinity during tomato cultivation and the degree of intrinsic tolerance of the varieties. The results in the saline treatments (T1 and T3) of this study, in which a decrease in water potential is experienced, coincide with those obtained by71 when studying the response to salinity in this crop. It is known that a reduction in osmotic potential can also originate from a net increase in the number of existing solutes; this capacity to accumulate solutes that occurs in response to a water deficit caused by salt stress determines the total or partial maintenance of cell pressure, which is called osmotic adjustment capacity and was described by72. When the tissue water potential decreases due to salt stress, a reduction in the osmotic potential minimizes the negative effects of this change on the pressure potential due to the maintenance of a water potential gradient between the soil and roots that allows water uptake from the soil73, and this is what probably occurred in this study. The response to salt stress by decreasing the water potential due to osmotic adjustment capacity is an additive and easily inherited trait in salt- and water-stress-tolerant species74. The results obtained are relevant since OA is considered a key mechanism by which plants adapt to reduced water availability. The greater the OA capacity is, the greater the capacity of cells to maintain turgor, which represents another component of water potential and is essential for maintaining growth, among other processes.
The results obtained indicate the positive effect of the use of the bio-stimulant on tomato crops under salt stressed conditions. It has also been shown that when A is low in reproductive phenophases, it leads to significant yield decreases75. When salt accumulates in chloroplasts, it reduces the chlorophyll content, affects the photosynthetic transport system, and inhibits photosystem II activity76. The increase observed in leaf development may have favored a greater fixation of CO2, which, together with the stimulation observed in the chlorophyll content, led to an increase in photosynthesis, as well as greater production of photoassimilates, which had a positive impact on the increase in growth and development of the plants treated with the bio-stimulant77.
The E levels recorded in the saline treatment group when QuitoMax was applied suggest that QuitoMax regulates transpiration. In this sense, stomatal closure has been demonstrated in plants sprayed with chitosan, which suggested that the growth-stimulating effect after stomatal closure could be related to an anti-transpiring effect on the plant28,78.
These results for WUE may be related to the difference in intervarietal response to salinity stress in tomatoes78 and to stomatal closure mechanisms that the plant activates to avoid water loss79. The WUE, as a function of the photosynthesis-transpiration ratio, is a relevant indicator in plants subjected to abiotic stress; under these conditions, it is necessary to maintain adequate levels of water inside the cells, but stomatal closure affects photosynthesis12,80. These effects have been recognized by other authors when chitosan was applied under saline conditions81 and coincide with those found in this work when QuitoMax was applied (Table 3) and considering the variable WUE.
The contribution of the bio-stimulant to the chlorophyll content was greater in the susceptible variety (Claudia). This result suggests that it contributes to the normal functioning of chlorophyllase activity even under saline conditions. It has been reported that salinity inhibits the activity of this enzyme12, which affects photosynthesis in the medium term. The results presented here demonstrate the effectiveness of bio-stimulants based on QuitoMax in protecting the photosynthetic apparatus even under saline conditions. A study by82 showed that a reduction in the adverse effect of salt stress in tomatoes occurs with the application of spermine. Regardless of the type of variety (tolerant or susceptible), the superior effect of QuitoMax applied at a dose of 300 mg L-1 concerning stress intensity on most variables confirms the effectiveness of QuitoMax in reducing the negative effects of salinity on tomato plants.
As a future perspective, the use of this biostimulant could be an economically and ecologically viable alternative to increase tomato production in ecosystems affected by salt stress as an enhancer of the metabolic capacities of plants to adapt and produce a yield equal to or greater than its productive genetic potential. The development of molecular studies of gene overexpression that confer salinity tolerance and the monitoring of the carbon of this biostimulant with the use of isotopic techniques will help validate its contribution to increasing tomato tolerance to soil salinity.
Conclusion
All the variables evaluated were influenced by salt stress, but the application of QuitoMax positively modified the response, demonstrating its contribution to the reduction in salt stress intensity in tomato plants. QuitoMax contributes to reducing stress in tomato plants while promoting a decrease in stress when considering morphoagronomic variables and variables related to the water regime and gas exchange in tomato plants. The use of QuitoMax at a dose of 300 mg ha-1 minimizes the effects of salt stress on tomato plants.
Data availability
The datasets used and/or analyses during the current study available from the corresponding author on reasonable request.
References
Hameed, A. et al. Effects of salinity stress on chloroplast structure and function. Cells 10, 2023. https://doi.org/10.3390/cells10082023 (2021).
Harper, R. J., Dell, B., Ruprecht, J. K., Sochacki, S. J. & Smettem, K. R. J. Salinity and the reclamation of salinized lands. In Soils and landscape restoration. 193–208Academic Press,. (2021). https://doi.org/10.1016/B978-0-12-813193-0.00007-2
Stavi, I., Thevs, N. & Priori, S. Soil salinity and sodicity in drylands: a review of causes, effects, monitoring, and restoration measures. Front. Environ. Sci. 9, 712831. https://doi.org/10.3389/fenvs.2021.712831 (2021).
Alexopoulos, A. A. et al. Hedypnois cretica Land Urospermum picroides L. plant growth, nutrient status and Qucharacteristicsistics salinitylstress stress. Horticulturae 9, 65. https://doi.org/10.3390/horticulturae9010065 (2023).
Kumar, A., Behera, I., Langthasa, M. & Prakash, N. S. Effect of plant growth-promoting rhizobacteria on alleviating salinity stress in plants: a review. J. Plant. Nutr. 46, 2525–2550. https://doi.org/10.1080/01904167.2022.2155548 (2023).
Seymen, M. et al. Effects of different levels of water salinity on plant growth, biochemical content, and photosynthetic activity in cabbage seedling under water-deficit conditions. Gesunde Pflanzen. 75, 871–884. https://doi.org/10.1007/s10343-022-00788-y (2023).
Ntanasi, T. Assessment of Growth, Yield, and nutrient uptake of Mediterranean Tomato landraces in response to salinity stress. Plants 12, 3551. https://doi.org/10.3390/plants12203551 (2023).
Kumar, P. & Sharma, P. K. Soil salinity and food security in India. Front. Sustain. Food Syst. 4, 533781. https://doi.org/10.3389/fsufs.2020.533781 (2020).
Sahab, S. et al. Potential risk assessment of soil salinity to agroecosystem sustainability: current status and management strategies. Sci. Total Environ. 764, 144164. https://doi.org/10.1016/j.scitotenv.2020.144164 (2021).
Hoque, M. N. et al. Plant growth-promoting rhizobacteria-mediated adaptive responses of plants under salinity stress. J. Plant. Growth Regul. 42, 1307–1326. https://doi.org/10.1007/s00344-022-10633-1 (2023).
Kumawat, K. C. et al. Plant growth-promoting rhizobacteria: salt stress alleviators to improve crop productivity for sustainable agriculture development. Front. Plant. Sci. 13, 1101862. https://doi.org/10.3389/fpls.2022.1101862 (2023).
Khan, T. A., Saleem, M. & Fariduddin, Q. Melatonin influences stomatal behavior, root morphology, cell viability, photosynthetic responses, fruit yield, and fruit quality of tomato plants exposed to salt stress. J. Plant. Growth Regul. 42, 2408–2432. https://doi.org/10.1007/s00344-022-10713-2 (2023).
Alam, M. S., Tester, M., Fiene, G. & Mousa, M. A. A. A. Early growth stage characterization and the biochemical responses for salinity stress in tomato. Plants 10, 712. https://doi.org/10.3390/plants10040712 (2021).
Roșca, M., Mihalache, G. & Stoleru, V. Tomato responses to salinity stress: from morphological traits to genetic changes. Front. Plant. Sci. 14, 1118383. https://doi.org/10.3389/fpls.2023.1118383 (2023).
Sonawane, P. D. A BAHD-type acyltransferase concludes the biosynthetic pathway of nonbitter glycoalkaloids in ripe tomato fruit. Nat. Commun. 14, 4540. https://doi.org/10.1038/s41467-023-40092-5 (2023).
Das, S. & Jahan, M. Production and profitability intervention of summer hybrid tomato: a farm level review in Bangladesh. IJEBA 14, 21–30. https://doi.org/10.3844/ajebasp.2022.21.30 (2022).
Armenta-Bojórquez, A. D. et al. Pacific white shrimp and tomato production using water effluents and salinity-tolerant grafted plants in an integrated aquaponic production system. J. Clean. Prod. 278, 124064. https://doi.org/10.1016/j.jclepro.2020.124064 (2021).
Ali, A. A. M. et al. Analysis of salinity tolerance in tomato introgression lines based on morpho-physiological and molecular traits. Plants 10, 2594. https://doi.org/10.3390/plants10122594 (2021).
Pessoa, H. P. et al. Uncovering tomato candidate genes associated with drought tolerance using Solanum pennellii introgression lines. PLos One. 18, e0287178. https://doi.org/10.1371/journal.pone.0287178 (2023).
Shams, M. & Khadivi, A. Mechanisms of salinity tolerance and their possible application in the breeding of vegetables. BMC Plant. Biol. 23, 139. https://doi.org/10.1186/s12870-023-04152-8 (2023).
Singh, A. Soil salinity: a global threat to sustainable development. Soil. Use Manag. 38, 39–67. https://doi.org/10.1111/sum.12772 (2022).
Melino, V. & Tester, M. Salt-tolerant crops: time to deliver. Annu. Rev. Plant. Biol. 74, 671–696. https://doi.org/10.1146/annurev-arplant-061422-104322 (2023).
Rakkammal, K., Maharajan, T., Ceasar, S. A. & Ramesh, M. Biostimulants and their role in improving plant growth under drought and salinity. Cereal Res. Commun. 51, 61–74. https://doi.org/10.1007/s42976-022-00299-6 (2023).
Attia, M. S. et al. Impact of foliar application of chitosan dissolved in different organic acids on isozymes, protein patterns and physio-biochemical characteristics of tomato grown under salinity stress. Plants 10, 388. https://doi.org/10.3390/plants10020388 (2021).
Avila-Amador, C. A., Argentel-Martínez, L., Peñuelas-Rubio, O., Aguilera, J. G. & Reyna, I. F. Resposta Da Cultura do tomateiro (Solanum lycopersicum L.) à aplicação de QuitoMax em condições de salinidade. Res. Soc. Dev. 11, e10111233870. https://doi.org/10.33448/rsd-v11i12.33870 (2022).
Argentel-Martínez, L. et al. Contribution of QuitoMax to the hormonal and enzymatic metabolism in tomato under saline stress. Ciênc Agrotec. 48, e014524. https://doi.org/10.1590/1413-7054202448014523 (2024).
Zhang, G. et al. Exogenous application of Chitosan alleviates salinity stress in lettuce (Lactuca sativa L). Horticulturae 7, 342. https://doi.org/10.3390/horticulturae7100342 (2021).
Bittelli, M., Flury, M., Campbell, G. S. & Nichols, E. J. Reduction of transpiration through foliar application of Chitosan. Agr for. Meteorol. 107, 167–175. https://doi.org/10.1016/S0168-1923(00)00242-2 (2001).
Chanaluisa-Saltos, J. S., Sánchez, A. R. Á., Reyes-Pérez, J. J. & Lizarde, N. A. Respuesta agronómica Y fitosanitaria de plantas de tomate (Solanum lycopersicum L.) a la aplicación de quitosano en condiciones controladas. Rev. Científ Agroecosist. 10, 139–145 (2022).
Reyes-Pérez, J. et al. Chitosan application increases tomato (Solanum lycopersicum L.) crop emergence, growth and yield under greenhouse conditions. Biotechnology 22, 156–163. https://doi.org/10.18633/biotecnia.v22i3.1338 (2020).
Gustavo-González, L. et al. Effect of seed treatment with QuitoMax on yield and quality of tomato seedlings ESEN and L-43 varieties. Terra Latinoam. 39, 1–6 (2021).
Falcón-Rodríguez, A. B., Menéndez, D. C., Fundora, D. G. P. & García, M. C. N. New natural products for agriculture: oligosaccharins. Cultivos Tropicales. 36, 111–129 (2015).
Verhulst, N. et al. Soil quality as affected by tillage-residue management in a wheat–maize irrigated bed planting system. Plant. Soil. 340, 453–466. https://doi.org/10.1007/s11104-010-0618-5 (2011).
Álvarez, M., Rodríguez, J., Moya, C., Plana, D. & Claudia Mercy and Mayle, three new tomato varieties for fresh consumption. Cultivos Tropicales. 29, 43–44 (2008).
Agrinova Foligreen 19-19-19. (2022). https://agri-nova.com/product/fertilizantes-foliares/foligreen-19-19-19/
Terry-Alfonso, E., Falcón-Rodríguez, A., Ruiz-Padrón, J. & Carrillo-Sosa, Y. Morales-Morales. H. Agronomic response of tomato crop to QuitoMax bioproduct. Cultivos Tropicales. 38, 147–154 (2017).
Li, J. et al. Comprehensive comparison of different saline water irrigation strategies for tomato production: soil properties, plant growth, fruit yield and fruit quality. Agric. Water Manag. 213, 521–533. https://doi.org/10.1016/j.agwat.2018.11.003 (2019).
Toribio, A. J. et al. Application of sonicated extracts of cyanobacteria and microalgae for the mitigation of bacterial canker in tomato seedlings. J. Appl. Phycol. 33, 3817–3829. https://doi.org/10.1007/s10811-021-02599-6 (2021).
Shabbir, A. et al. Effects of drip irrigation emitter density with various irrigation levels on physiological parameters, root, yield, and quality of cherry tomato. Agronomy 10, 1685. https://doi.org/10.3390/agronomy10111685 (2020).
Saha, H. et al. Effects of light quality on colonization of tomato roots by AMF and implications for growth and defense. Plants 11, 861. https://doi.org/10.3390/plants11070861 (2022).
Govaerts, B. & Verhulst, N. Mexico,. The normalized difference vegetation index (NDVI) Greenseeker (TM) handheld sensor: toward the integrated evaluation of crop management. In Concepts and Case Studies. (ed. CIMMYT13). (2010).
Inman, D., Khosla, R. & Mayfied, T. On-the-go active remote sensing for efficient crop nitrogen management. Sens. Rev. 25, 209–214. https://doi.org/10.1108/02602280510606499 (2005).
Ali, K. A., Noraldeen, S. S. & Yaseen, A. A. An evaluation study for chlorophyll estimation techniques. Sarhad J. Agric. 37, 1458–1465. https://doi.org/10.17582/journal.sja/2021/37.4.1458.1465 (2021).
Ruiz-Sánchez, M. et al. Respuesta de la planta de arroz (Oryza sativa L.) a la suspensión de la lámina de agua en tres momentos de su desarrollo. Parte I. Cultivos Tropicales. 38, 61–69 (2017).
Turner, N. C. Turgor maintenance by osmotic adjustment, an adaptive mechanism for coping with plant water deficits. Plant. Cell. Environ. 40, 1–3. https://doi.org/10.1111/pce.12839 (2017).
Wagle, P. Evaluation of water use efficiency algorithms for flux variance similarity-based evapotranspiration partitioning in C3 and C4 grain crops. Water Resour. Res. 57, e2020WR028866. (2021). https://doi.org/10.1029/2020WR028866
Fernandez, G. C.J. Effective selection criteria for assessing plant stress tolerance. (ed. Kuo, C.). 257–270. (Shanhua, Taiwan AVRDC,1992).
Kolmogorov, A. Basic Concepts of Probability Theory. 62 (Julius Springer, 1933).
Fisher, R. A. New, York, Statistical methods for research workers. (ed. Springer). (1935). https://doi.org/10.1007/978-1-4612-4380-9_6
Tukey, J. W. A survey of sampling from contaminated distributions. In Olkin, I. (Ed.), Contributions to probability and statistics: Essays in honor of harold hotelling. 448–485 pp. Stanford, CA: Stanford University Press, (1960).
StatSoft Satistica 13.3. StatSoft Incorporation Version 13.3. (2014). https://statistica.software.informer.com/13.3/
Gómez-Salazar, A., López-Salvador, G., Jerez-Mompie, E. & González-Cañizares, P. Guerrero-Domínguez, L. Influence of two biostimulants on growth and development of tomato (Solanum lycopersicum L.) plants. Cultivos Tropicales. 43, 07 (2023).
Castro-Lizazo, I. et al. Potencialidades De Dos Bioestimulantesen La germinación Y El Crecimiento De las plántulas de tomate. Cienc. Tecnol Agropecuaria. 23, e2343. https://doi.org/10.21930/rcta.vol23_num1_art (2022).
Terry-Alfonso, E., Ruiz-Padrón, J., Falcón-Rodríguez, A. & Socarrás-Armenteros, Y. Oligosacarinas stimulate the growth and yield on tomato (Solanum lycopersicum L.) under protected conditions. Cultivos Tropicales. 40, e04 (2019).
Ullah, N. et al. Mitigation the adverse effect of salinity stress on the performance of the tomato crop by exogenous application of Chitosan. Bull. Nat. Res. Cent. 44, 1–11. https://doi.org/10.1186/s42269-020-00435-4 (2020).
Rodríguez, A. B. F. et al. Oligosaccharins as biostimulants for Cuban agriculture. Acad. Cienc. Cuba. 11, 1–11 (2020).
Falcón-Rodríguez, A. B. et al. Oligosacarinas como bioestimulantes para la agricultura cubana. Annals ACC. 11, e852 (2021). http://scielo.sld.cu/scielo.php?script=sci_arttextpid=S2304-01062021000100007lng=estlng=es
González-Peña, D., Costales, D. & Falcón, A. B. Influencia De Un polímero De Quitosana en El crecimiento y la actividad de enzimas defensivas en tomate (Solanum lycopersicum L). Cultivos Tropicales. 35, 35–42 (2014).
Hussain, I. et al. Foliar application of Chitosan modulates the morphological and biochemical characteristics of tomato. Asian J. Agric. Biol. 7, 365–372 (2019). https://www.asianjab.com/wp-content/uploads/2019/10/5.-AJAB-2018-04-132_Okay.pdf
Álvarez, M. A., Fita, A., Ruiz, C. & Bolarín, C. Morphology and biomass variations in root system of young tomato plants (Solanum sp.) plants. Cultivos Tropicales. 37, 96–101. https://doi.org/10.13140/RG.2.1.4827.2888 (2016).
Sánchez, A. R. Á. et al. Respuesta agronómica De Plantas De Tomate Solanum lycopersicum La La aplicación De Bradyrhizobium japonicum y quitosano. Ciencia Latina Revista Científica Multidisciplinar. 5, 11361–11374. https://doi.org/10.37811/cl_rcm.v5i6.1171 (2021).
Tanveer, M. & Ahmed, H. A. I. ROS Signalling in modulating salinity stress tolerance in plants. (eds Hasanuzzaman, M. & Tanveer, M.) Salt and Drought Stress Tolerance in Plants. Signaling and Communication in Plants. (Springer Cham, https://doi.org/10.1007/978-3-030-40277-8_11 (2020).
Villalobos, E. Fisiología de la producción de los cultivos tropicales. Editorial Universidad de Costa Rica. San Pedro, San José Costa Rica. 227. (2001).
Bian, L., Wang, J., Liu, J. & Han, B. Spatiotemporal changes of soil salinization in the yellow river delta of China from 2015 to 2019. Sustainability 13, 822. https://doi.org/10.3390/su13020822 (2021).
Stefen, D. L. V., Souza, C. A., Coelho, C. M. M., Gutkoski, L. C. & Sangoi, E. L. A adubação nitrogenada durante o espigamento melhora a qualidade industrial do trigo (Triticum aestivum Cv. Mirante) cultivado com regulador de crescimento etil-trinexapac. Revista Fac. Agron. 114, 161–169 (2016). http://revista-vieja.agro.unlp.edu.ar/index.php/revagro/article/view/28/273
Zhang, T. T. Using hyperspectral vegetation indices as a proxy to monitor soil salinity. Ecol. Indic. 11, 1552–1562. https://doi.org/10.1016/j.ecolind.2011.03.025 (2011).
Dzung, N. A. Enhancing crop production with chitosan and its derivatives. In: Chitin, Chitosan, Oligosaccharides and Their Derivatives: Biological Activities and Applications (ed Se-Kwon, K.) 619–631. (Taylor & Francis, (2011).
Özkurt, N. & Bektaş, Y. Alleviation of salt stress with Chitosan Foliar Application and its effects on Growth and Development in Tomato (Solanum lycopersicum L). TÜTAD 9, 342–351. https://doi.org/10.19159/tutad.1168393 (2022).
Hassnain, M. et al. Efficacy of chitosan on performance of tomato (Lycopersicon esculentum L.) plant under water stress condition. Pakistan J. Agri Res. 33, 27–41. https://doi.org/10.17582/journal.pjar/2020/33.1.27.41 (2020).
Argentel-Martínez, L., Garatuza-Payán, J., Yépez-González, E. A. & de los Santos, S. Evaluating salinity tolerance through physiological, biochemical and agronomical indicators in Mexican wheat varieties, cultivated under field conditions in Cuba. Cultivos Tropicales. 37, 91–101. https://doi.org/10.13140/RG.2.1.3719.6407 (2016).
Morales, D., Rodríguez, P., Sánchez-Blanco, M. J. & Torresillas, A. Response to salinity of three tomato (Lycopersicon esculentum Mill) varieties. Cultivos Tropicales. 23, 71–76 (2002).
Leidis, E. Control of abscisic synthesis, protein synthesys under osmothic regulation. J. Exp. Bot. 51, 1563–1574 (2007).
Munns, R. & Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant. Biol. 59, 651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911 (2008).
Munns, R. et al. Energy costs of salt tolerance in crop plants. New. Phytol. 225, 1072–1090. https://doi.org/10.1111/nph.15864 (2019).
Prins, A. et al. Rubisco catalytic properties of wild and domesticated relatives provide scope for improving wheat photosynthesis. J. Exp. Bot. 67, 1827–1838. https://doi.org/10.1093/jxb/erv574 (2016).
Khalid, M. F. et al. Alleviation of drought and salt stress in vegetables: crop responses and mitigation strategies. Plant. Growth Regul. 99, 177–194. https://doi.org/10.1007/s10725-022-00905-x (2022).
Shahrajabian, M. H., Chaski, C., Polyzos, N., Tzortzakis, N. & Petropoulos, S. A. Sustainable agriculture systems in vegetable production using chitin and chitosan as plant biostimulants. Biomolecules 11, 819. https://doi.org/10.3390/biom11060819 (2021).
Avila-Amador, C. et al. Effect of salt stress on water regime, concentration of osmotically active compounds and photosynthetic pigments in tomato cultivars. RIA J. Agric. Res. 49, 32–40 (2023). https://ria.inta.gob.ar/wp-content/uploads/2023/06/ria-49-n-1-april-2023.pdf
Xue, F. et al. Stomatal conductance of tomato leaves is regulated by both abscisic acid and leaf water potential under combined water and salt stress. Physiol. Plant. 172, 2070–2078. https://doi.org/10.1111/ppl.13441 (2021).
Messina, C. D. et al. Limited-transpiration trait may increase maize drought tolerance in the US Corn Belt. Agron. J. 107, 1978–1986. https://doi.org/10.2134/agronj15.0016 (2015).
Hidangmayum, A., Dwivedi, P., Katiyar, D. & Hemantaranjan, A. Application of chitosan on plant responses with special reference to abiotic stress. Physiol. Mol. Biol. Plants. 25, 313–326. https://doi.org/10.1007/s12298-018-0633-1 (2019).
Pascual, L. S. et al. Exogenous spermine alleviates the negative effects of combined salinity and paraquat in tomato plants by decreasing stress-induced oxidative damage. Front. Plant. Sci. 14, 1193207. https://doi.org/10.3389/fpls.2023.1193207 (2023).
Acknowledgements
The authors wish to thank Dr. Harun Bektaş for his supports provided in English revision of the manuscript.
Author information
Authors and Affiliations
Contributions
L.A.M., O.P.R. and C.A.A. participated in experimental studies and manuscript preparation. F.S., J.G.A., J.H.S., A.M.Z., R.F.R., P.E.T. and U.A. contributed to the coordination and helped to draft the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Argentel-Martínez, L., Peñuelas-Rubio, O., Amador, C.Á. et al. Mitigating salinity stress on tomato growth, water regime, gas exchange, and yield with the application of QuitoMax. Sci Rep 14, 31755 (2024). https://doi.org/10.1038/s41598-024-82211-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82211-2
Keywords
This article is cited by
-
Effects of saline water irrigation on soil nutrients, growth performance, and yield of tomato (Solanum lycopersicum)
Journal of the Saudi Society of Agricultural Sciences (2025)