Abstract
A subgroup analysis of a randomized study demonstrated that patients with advanced or metastatic liposarcoma treated with eribulin had longer overall survival and progression-free survival compared to those treated with dacarbazine, suggesting eribulin as a therapeutic option for advanced liposarcoma. Therefore, this study aims to evaluate the cost-effectiveness of eribulin versus dacarbazine in the treatment of advanced liposarcoma. We established a 10-year Markov model to compare the cost-effectiveness of eribulin and dacarbazine regimens. Clinical data were sourced from a subgroup analysis of a multicenter, randomized, open-label phase 3 trials. Quality-adjusted life years (QALYs), and incremental cost-effectiveness ratios (ICERs) were computed. The total cost of the dacarbazine scheme was $10,895.558, with a QALY of 0.533. In contrast, the total cost of the eribulin scheme was $16,961.891, with a QALY of 0.698. The ICER between the eribulin and dacarbazine schemes was $36,736.467, which is below the willingness-to-pay (WTP) threshold in China ($37,877.469). From the perspective of the Chinese healthcare system, eribulin is cost-effective compared to dacarbazine at the WTP threshold.
Similar content being viewed by others
Introduction
Sarcomas represent rare solid tumors, accounting for approximately 1% of all adult malignancies. Around 80% of sarcomas originate from soft tissues, while the remaining 20% arise from bone1. Soft tissue sarcomas (STS) encompass over 50 histological subtypes but constitute less than 1% of all newly diagnosed malignancies in adults, contributing to approximately 2% of cancer-related mortality2,3. Following gastrointestinal stromal tumors, leiomyosarcoma and liposarcoma are the most common subtypes of STS4,5. Patients with advanced or metastatic STS have a poor prognosis, with an estimated median survival period of 12 to 15 months6,7. The Chinese Society of Clinical Oncology (CSCO) guidelines for the diagnosis and treatment of bone and soft tissue tumors state that trabectedin, eribulin, and dacarbazine are used to treat liposarcoma. However, the economics between the cost and efficacy of these drugs are not yet known.
Eribulin has demonstrated the ability to induce vascular remodeling, inhibit cancer cell migration and invasion, and reverse the epithelial-to-mesenchymal transition in numerous cancer cell lines8,9. It has been employed in the treatment of locally advanced or metastatic breast cancer patients who have previously received at least two chemotherapy regimens, including anthracyclines and taxanes10. In a non-randomized, phase 3 study involving patients with advanced STS, eribulin exhibited both activity and manageable tolerability1. Subgroup analysis of the study further showed that it exhibited more pronounced efficacy in the treatment of liposarcoma11. In the subgroup analysis of this study, there were 143 patients with liposarcoma (LPS), with 71 in the eribulin group and 72 in the dacarbazine group. In the LPS subgroup, treatment with eribulin significantly improved overall survival (OS) compared to dacarbazine, with a median OS of 15.6 months (95% CI, 10.2 to 18.6 months) in the eribulin group, versus 8.4 months (95% CI, 5.2 to 10.1 months) in the dacarbazine group. Progression-free survival (PFS) was also significantly improved in the subgroup, with median PFS of 2.9 months in the eribulin group and 1.7 months in the dacarbazine group (HR, 0.521; 95% CI, 0.35 to 0.78; nominal P = 0.0015). These findings suggest that eribulin is an important treatment option for patients with liposarcoma. Based on the findings of this study, the Food and Drug Administration (FDA) approved eribulin for the treatment of advanced liposarcoma patients who have failed anthracycline-based therapy.
However, the pricing of this new therapy is an important factor that must be considered in cancer treatment. The escalating costs of cancer treatment highlight the necessity of conducting cost-effectiveness analyses to enable policymakers to more effectively utilize limited resources12. In this study, our aim was to investigate the costs and effects of the eribulin regimen compared to the dacarbazine regimen in treating patients with advanced liposarcoma from the perspective of the Chinese healthcare system, using a ten-year Markov model. To our knowledge, this study represents the first cost-effectiveness comparison of eribulin and dacarbazine in the treatment of advanced liposarcoma.
Methods
Clinical data
The clinical data were sourced from a subgroup analysis of liposarcoma in a multicenter, randomized, open-label phase 3 trial targeting patients with histologically confirmed locally recurrent, locally advanced, or metastatic soft tissue sarcoma11. Eribulin mesylate was administered intravenously at a dose of 1.4 mg/m2 on days 1 and 8 of a 21-day cycle. Dacarbazine was administered at doses of 850 mg/m2, 1000 mg/m2, or 1200 mg/m2, infused intravenously over 15–60 min on day 1 of a 21-day cycle. Clinical outcomes including PFS, OS, and Grade 3 adverse events (AEs) are summarized in Table 1.
Model construction
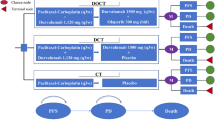
We established a Markov model based on clinical trials to assess the clinical and economic outcomes of eribulin and dacarbazine in patients with advanced liposarcoma (Fig. 1a). The cycle length was three weeks, consistent with the treatment cycles in the clinical trial, with a horizon of ten years. We assumed an age of 55 years for all enrolled patients, which corresponds to the median age of the trial. Our model comprised three health states: PFS, progressive disease (PD) and death, as illustrated in Fig. 1b. We assumed all patients started in a stable state and received treatment with either eribulin or dacarbazine until disease progression or the occurrence of unacceptable AEs. Patients in a progressed state could receive subsequent treatments until death. Patients in both progressed and stable states faced a risk of death, transitioning from one state to another or remaining in the same state within a cycle.
The model outcomes included costs, QALYs and ICERs. According to the Guidelines for Pharmacoeconomic Evaluations in China, health utilities and costs were discounted annually at a rate of 5%13. The Markov model was programmed using TreeAge Pro 2011 (TreeAge Software, Williamstown, MA) and statistical analysis was conducted using R software (version 4.3.3, https://www.r-project.org).
Markov model of advanced liposarcoma. (a) Markov model for comparing two treatment strategies for advanced liposarcoma. (b) Three transitional health states.
Model survival and transition probabilities
In each Markov cycle, the model reassigned patients to the three different health states based on transition probabilities. The transition probabilities between health states were derived from trial data. Kaplan-Meier survival curves for PFS and OS were digitized using Engauge Digitizer software to reconstruct individual patient data (IPD). The IPD was then reconstructed using R statistical software. Goodness-of-fit tests were conducted among survival distributions including Exponential, Gamma, Gompertz, Weibull, Log-logistic, and Log-normal curves. Ultimately, based on the Akaike information criterion and Schwarz Bayesian criterion, the Log-normal distribution was chosen to fit the PFS and OS survival models. Time-dependent probabilities of transitioning from PFS to PFS were computed based on the parameter model of the PFS curve, as described by the following formula.
where ‘t’ represents the current cycle number in the Markov model. Table 2 presents the estimated model parameters. Figure 2 presents the curves for PFS and OS fitting for eribulin and dacarbazine regimens and the results of the survival curve simulation. Assuming a mortality rate of 7.37‰ from PFS to death, reflecting the natural mortality rate in China in 202214, a constant transition probability from PFS to death was computed using the method of diminishing life expectancy indices15. The parameter model of the OS curve was utilized to measure the overall mortality rate for each cycle. Subsequently, transition probabilities from PD state to PD state were calculated based on the natural mortality rate and overall mortality rate.
Kaplan Meier curves and parametric model fitting. (a) PFS for the original trial and model estimated data. (b) OS for the original trial and model estimated data.
Costs
We calculated costs from the perspective of the Chinese healthcare system. All direct medical expenses considered include medication costs, AE management costs, examination fees, outpatient fees, and costs related to disease progression, as detailed in Table 3. Drug prices and laboratory testing data were sourced from the Zhejiang Provincial People’s Hospital. Medication costs were calculated by multiplying drug dosages. Hence, medication selection and dosage regimen were based on the opinions of Chinese clinical experts and followed prescription guidelines. For dosage calculations, we utilized the typical Chinese body surface area (BSA) of 1.72m216,17. Costs were converted to US dollars using the exchange rate from the Chinese National Bureau of Statistics for the year 2023 (US$ 1.00 = ¥ 7.0774), based on the historical midpoint rate14. We set the World Health Organization’s 2023 per capita gross domestic product (GDP) ($12,625.823) as the extremely cost-effective threshold and three times the per capita GDP ($37,877.469) as a cost-effective threshold, following WHO recommendations18,19.
Furthermore, the costs of managing AE (≥ 3 grades) were calculated by multiplying the reported incidence rates in clinical trials by the unit costs derived from published literature. Table 1 summarizes the incidence rates of each AE (≥ 3 grades), sourced from clinical trials. Management strategies for grade 3 AEs were based on expert opinions and clinical practice. The costs of treating AEs were calculated based on patient records from local hospitals.
Utilities
The mean health utility values obtained from published literature for stable and progressed states are 0.68 and 0.42, respectively, assuming similar utility values for both groups post-progression20,21. Our analysis also accounts for the negative utility associated with treatment-related serious adverse events (SAEs), sourced from published literature22.
Sensitivity analysis
To assess the robustness of the model and address the uncertainty in estimated parameters, we conducted univariate sensitivity analysis and probabilistic sensitivity analysis (PSA). For the univariate sensitivity analysis, with the exception of the discount rate ranging from 0 to 8%, one variable was varied by ± 20% while holding other variables constant. This was done to ascertain the impact of each independent variable on the outcomes within a specified range. Tornado diagrams were used to display the results of the univariate sensitivity analysis.
For PSA, we performed 1,000 Monte Carlo simulations, sampling different values from statistical distributions (Beta distribution for utilities; Gamma distribution for costs)23. The results were utilized to generate cost-effectiveness acceptability curves, indicating the probability of cost-effectiveness within the WTP threshold range. Additionally, further exploration was conducted at WTP levels corresponding to one or two times the per capita GDP. Table 3 illustrates the range of values and distribution types utilized in sensitivity analysis.
Results
Base case analysis
Table 4 presents the baseline scenario analysis results over a 10-year time horizon. The total cost for the dacarbazine regimen amounts to $10,895.558, yielding 0.533 QALYs, while the total cost for the eribulin regimen is $16,961.891, generating 0.698 QALYs. Relative to the dacarbazine regimen, eribulin incurs incremental costs and effectiveness of $6,066.333 and 0.165 QALYs, respectively. Consequently, the ICER of eribulin versus dacarbazine stands at $36,736.467 per QALY, which is below the WTP threshold. Thus, within the WTP threshold, eribulin demonstrate cost-effectiveness.
Sensitivity analysis
The results of the ICER sensitivity analysis comparing the eribulin group to the dacarbazine group are illustrated in Fig. 3. It is evident from this analysis that the cost of eribulin medication has the most significant impact on the model outcomes. Variations within the sensitivity analysis range have minimal influence on the ICER results, indicating the robustness of the model.
The cost-effectiveness scatterplot of the study group is depicted in Fig. 4a, where each point represents a simulated ICER value. The scatterplot is predominantly located in the first quadrant, indicating higher costs but favorable efficacy in the eribulin group compared to the dacarbazine group. Moreover, most of the scattered points are within the WTP threshold line. Consequently, eribulin treatment for patients with advanced liposarcoma has economic viability.
The WTP value directly impacts the determination of whether a treatment regimen is cost-effective. As observed from the cost-effectiveness acceptability curve (Fig. 4b), with an increase in the WTP value, the likelihood of eribulin being cost-effective also increases. When the threshold exceeds $36,736.5, the probability that eribulin is economically justified surpasses 50%. And when we reduced the cost price of eribulin and reintroduced the model, we found that the probability of eribulin being cost-effective in the WTP range was 100% when the cost of eribulin drugs was reduced by about 30%.
The tornado plot shows the ICER of eribulin versus dacarbazine for different input parameters.
Probabilistic sensitivity analysis results. (a) Scatter plot of ICER between the eribulin and dacarbazine groups. (b) Cost-effectiveness acceptability curves of eribulin versus dacarbazine at different WTP thresholds.
Discussion
Soft tissue sarcoma is a heterogeneous malignant tumor, and its treatment is essentially palliative, aiming to delay tumor growth, preserve organ function, and reduce morbidity when progression and severe symptoms occur24. For patients with unresectable and non-curable STS, treatment typically involves cytotoxic chemotherapy, most commonly anthracycline-based (primarily doxorubicin) regimens and other chemotherapy drugs, such as dacarbazine, trabectedin, and eribulin25,26. Liposarcomas, which represent 20% of all adult sarcomas, are the most common histological type of malignant soft tissue tumors27. Currently, the main treatment options for liposarcoma include surgery and radiotherapy (RT). Recently, several chemotherapy agents targeting various LPS subtypes have been developed based on molecular genetic alterations, and these are at different stages of clinical trials. For chemotherapy agents that are already in use, such as dacarbazine, trabectedin, and eribulin, the cost of these medications is also a significant factor to consider. Therefore, it is essential to conduct pharmacoeconomic analyses of these chemotherapy treatments to balance economic burdens with health outcomes.
In a subgroup analysis study by George D Demetri et al., eribulin was shown to produce a longer OS and PFS than dacarbazine for liposarcoma11. We estimated that eribulin could result in higher QALYs, but its cost is higher. Hence, a comprehensive cost-effectiveness comparison between dacarbazine and eribulin is warranted. Utilizing clinical trial data, previously reported utility data, published cost estimates, and local costs, we compared the cost-effectiveness of eribulin and dacarbazine in treating advanced liposarcoma patients. The clinical trial reported 95% confidence intervals for both PFS and OS. Quality of life scores from the clinical trial showed no difference between the two groups; thus, the utility values for both groups are considered equivalent.
In this study, we conducted a model-based cost-effectiveness analysis of eribulin and dacarbazine as third-line treatments for advanced liposarcoma in China. Based on our base-case analysis results, patients receiving eribulin treatment would gain an additional 0.165 QALYs at an additional cost of $6,066.333, resulting in an ICER of $36,736.467 per QALY gained. The Chinese Pharmacoeconomic Evaluation Guidelines suggest that if the ICER is below the per capita GDP, the additional cost of the new therapy is considered worthwhile; if the ICER falls between the per capita GDP and three times the per capita GDP, the additional cost of the new therapy is considered acceptable; if the ICER exceeds three times the per capita GDP, the additional cost of the new therapy is not justified13. The three times the per capita GDP threshold is the most commonly used cost-effectiveness threshold when evaluating new therapies in China28. In our analysis, the ICER for eribulin compared to dacarbazine was $36,736.467 per QALY gained, lower than three times the per capita GDP. Therefore, from the perspective of the Chinese Pharmacoeconomic Evaluation Guidelines, eribulin is cost-effective for the treatment of advanced liposarcoma.
Economic evaluations of eribulin in the treatment of malignant tumors have been limited, with only a few studies reporting cost-effectiveness analyses of eribulin in treating metastatic breast cancer (MBC). Gabriel Tremblay et al. evaluated the cost-effectiveness of eribulin for MBC from the perspective of Spanish society29. The ICER for eribulin compared to capecitabine and vinorelbine was €35,149 per LY and €37,152 per QALY. In Spain, an ICER between €30,000 and €45,000 is reportedly reasonable. For eribulin, both ICERs fall within this range, suggesting that eribulin is a cost-effective treatment in both settings. This finding is consistent with our study results, suggesting that eribulin can improve patients’ health outcomes while allowing for more efficient utilization of financial resources. However, at present, the price of eribulin remains relatively high in China. If the cost of eribulin were to decrease by approximately 30%, it would become 100% cost-effective for the treatment of advanced liposarcoma in China.
However, our study has some limitations. Firstly, clinical data are derived from foreign trials, with the majority of enrolled patients being Caucasian, and a small proportion of Asians. This may slightly impact our results. This limitation is currently unavoidable, but the latest clinical data of advanced liposarcoma patients in the Chinese population may enhance accuracy in the future. Secondly, the clinical outcomes and costs in our study are obtained from the Chinese population, which may differ from other countries. Additionally, in actual clinical practice, many adjuvant therapies, such as traditional Chinese medicine and immunotherapy, can influence the total cost of cancer treatment. In recent years, the monopoly pattern of imported drugs in the Chinese pharmaceutical market has changed. Our study results provide valuable information for rational drug use, selecting specific drugs for patients, and informing decisions by government health policy makers.
Conclusion
We constructed a Markov model to explore the cost-effectiveness of eribulin and dacarbazine in the treatment of advanced liposarcoma patients from the perspective of Chinese society. Since the ICER is within three times the per capita GDP in China, eribulin treatment for advanced liposarcoma is considered cost-effective in the country. Furthermore, as the cost of eribulin continues to decrease, it would become highly cost-effective for the treatment of advanced liposarcoma in China.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.All data supporting the findings of this study are available within the paper. The mean health utility values obtained from published literature for stable and progressed states are 0.68 and 0.42, respectively, assuming similar utility values for both groups post-progression20-21. Example from: https://doi.org/10.3747/co.21.1899, https://doi.org/10.1155/2018/6703963. Our analysis also accounts for the negative utility associated with treatment-related serious adverse events (SAEs), sourced from published literature22. Example from: https://doi.org/10.1093/annonc/mdq339.
References
Schoffski, P. et al. Eribulin versus dacarbazine in previously treated patients with advanced liposarcoma or leiomyosarcoma: a randomised, open-label, multicentre, phase 3 trial. Lancet 387, 1629–1637. https://doi.org/10.1016/S0140-6736(15)01283-0 (2016).
Amdahl, J. et al. Cost-effectiveness of pazopanib in advanced soft tissue sarcoma in the United Kingdom. Sarcoma 2014(481071). https://doi.org/10.1155/2014/481071 (2014).
Jones, N. B., Iwenofu, H., Scharschmidt, T. & Kraybill, W. Prognostic factors and staging for soft tissue sarcomas: an update. Surg. Oncol. Clin. N Am. 21, 187–200. https://doi.org/10.1016/j.soc.2011.12.003 (2012).
Van Glabbeke, M. et al. Prognostic factors for the outcome of chemotherapy in advanced soft tissue sarcoma: an analysis of 2,185 patients treated with anthracycline-containing first-line regimens–a European Organization for Research and Treatment of Cancer Soft Tissue and bone Sarcoma Group Study. J. Clin. Oncol. 17, 150–157. https://doi.org/10.1200/JCO.1999.17.1.150 (1999).
Ducimetiere, F. et al. Incidence of sarcoma histotypes and molecular subtypes in a prospective epidemiological study with central pathology review and molecular testing. PLoS One 6, e20294. https://doi.org/10.1371/journal.pone.0020294 (2011).
Santoro, A. et al. Doxorubicin versus CYVADIC versus doxorubicin plus ifosfamide in first-line treatment of advanced soft tissue sarcomas: a randomized study of the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. J. Clin. Oncol. 13, 1537–1545. https://doi.org/10.1200/JCO.1995.13.7.1537 (1995).
Patel, S. R. et al. Results of two consecutive trials of dose-intensive chemotherapy with doxorubicin and ifosfamide in patients with sarcomas. Am. J. Clin. Oncol. 21, 317–321. https://doi.org/10.1097/00000421-199806000-00025 (1998).
Funahashi, Y. et al. Eribulin mesylate reduces tumor microenvironment abnormality by vascular remodeling in preclinical human breast cancer models. Cancer Sci. 105, 1334–1342. https://doi.org/10.1111/cas.12488 (2014).
Yoshida, T. et al. Eribulin mesilate suppresses experimental metastasis of breast cancer cells by reversing phenotype from epithelial-mesenchymal transition (EMT) to mesenchymal-epithelial transition (MET) states. Br. J. Cancer 110, 1497–1505. https://doi.org/10.1038/bjc.2014.80 (2014).
Perry, C. M. Eribulin. Drugs 71, 1321–1331, https://doi.org/10.2165/11207520-000000000-00000 (2011).
Demetri, G. D. et al. Activity of Eribulin in patients with Advanced Liposarcoma demonstrated in a Subgroup Analysis from a Randomized Phase III Study of Eribulin Versus Dacarbazine. J. Clin. Oncol. 35, 3433–3439. https://doi.org/10.1200/JCO.2016.71.6605 (2017).
Lin, J., Fang, Q. & Zheng, X. Cost-effectiveness analysis of anlotinib versus sunitinib as first-line treatment for metastatic renal cell carcinoma in China. PLoS One 18, e0281402. https://doi.org/10.1371/journal.pone.0281402 (2023).
Liu, G. E., Wu, H. S., Wu, J. H., Yang, J. & Li, L. HC. China guidelines for pharmacoeconomic evaluations (Chinese-English version). (2020).
National Bureau of Statistics of China: National Data.. http://data.stats.gov.cn/index.htm. (2023).
Le, Q. A. Structural uncertainty of Markov models for advanced breast Cancer: a Simulation Study of Lapatinib. Med. Decis. Mak. 36, 629–640. https://doi.org/10.1177/0272989X15622643 (2016).
Chen, X. et al. Cost-effectiveness analysis of gemcitabine plus cisplatin versus fluorouracil plus cisplatin for first-line treatment of recurrent or metastatic nasopharyngeal carcinoma. Oral Oncol. 94, 80–85. https://doi.org/10.1016/j.oraloncology.2019.04.022 (2019).
Wang, H. et al. Cost-utility of afatinib and gefitinib as first-line treatment for EGFR-mutated advanced non-small-cell lung cancer. Future Oncol. 15, 181–191. https://doi.org/10.2217/fon-2018-0692 (2019).
Eichler, H. G., Kong, S. X., Gerth, W. C., Mavros, P. & Jonsson, B. Use of cost-effectiveness analysis in health-care resource allocation decision-making: how are cost-effectiveness thresholds expected to emerge? Value Health 7, 518–528. https://doi.org/10.1111/j.1524-4733.2004.75003.x (2004).
Murray, C. J., Acharya, E. D. & Baltussen, A. Development of WHO guidelines on generalized cost-effectiveness analysis. Health Econ. 9(3), 235–251. (2000).
Delea, T. E. et al. Cost-effectiveness of pazopanib in advanced soft-tissue sarcoma in Canada. Curr. Oncol. 21, e748–759. https://doi.org/10.3747/co.21.1899 (2014).
Zuluaga-Sanchez, S. et al. Cost-Effectiveness of Olaratumab in Combination with Doxorubicin for Patients with Soft Tissue Sarcoma in the United States. Sarcoma 6703963. https://doi.org/10.1155/2018/6703963 (2018).
Soini, E. J. O., San Andres, G., Joensuu, T. & B. & Trabectedin in the treatment of metastatic soft tissue sarcoma: cost-effectiveness, cost-utility and value of information. Ann. Oncol. 22, 215–223. https://doi.org/10.1093/annonc/mdq339 (2011).
Ding, H. et al. Cost-effectiveness analysis of fulvestrant versus anastrozole as first-line treatment for hormone receptor-positive advanced breast cancer. Eur. J. Cancer Care (Engl) 26. https://doi.org/10.1111/ecc.12733 (2017).
Demetri, G. D. et al. Efficacy and safety of Trabectedin or Dacarbazine for Metastatic Liposarcoma or Leiomyosarcoma after failure of conventional chemotherapy: results of a phase III randomized Multicenter Clinical Trial. J. Clin. Oncol. 34, 786–793. https://doi.org/10.1200/JCO.2015.62.4734 (2016).
Garcia-Del-Muro, X. et al. Randomized phase II study comparing gemcitabine plus dacarbazine versus dacarbazine alone in patients with previously treated soft tissue sarcoma: a Spanish Group for Research on Sarcomas study. J. Clin. Oncol. 29, 2528–2533. https://doi.org/10.1200/JCO.2010.33.6107 (2011).
Zucali, P. A. et al. The old drug dacarbazine as a second/third line chemotherapy in advanced soft tissue sarcomas. Invest. New. Drugs 26, 175–181. https://doi.org/10.1007/s10637-007-9086-z (2008).
Zhao, X. et al. Prognostic factors predicting the postoperative survival period following treatment for primary retroperitoneal liposarcoma. Chin. Med. J. (Engl.) 128, 85–90. https://doi.org/10.4103/0366-6999.147822 (2015).
Liao, M. et al. Cost-effectiveness analysis of utidelone plus capecitabine for metastatic breast cancer in China. J. Med. Econ. 22, 584–592. https://doi.org/10.1080/13696998.2019.1588125 (2019).
Tremblay, G., Majethia, U., Kontoudis, I. & De Rosendo, J. Cost effectiveness analysis of Eribulin Mesylate as a treatment for metastatic breast Cancer in Spain: management in the later line of Therapy. J. Health Econ. Outcomes Res. 3, 180–193. https://doi.org/10.36469/9834 (2016).
Funding
This work was supported by the Zhejiang Provincial Health Commission Medical and Health Science and Technology Project (2022KY585), General scientific research project of Zhejiang Provincial Department of Education (Y202352079).
Author information
Authors and Affiliations
Contributions
MMZ, JLH, XCZ, PH, and XLY were responsible for study design, model building and statistical analysis. MMZ prepared the manuscript. JLH, XCZ, and PH searched literatures and collected data. XLY acquired funding. All authors critically reviewed the model structure, verifed results and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, M., Huang, J., Zheng, X. et al. Cost-effectiveness analysis of eribulin versus dacarbazine in patients with advanced liposarcoma. Sci Rep 15, 2084 (2025). https://doi.org/10.1038/s41598-024-84247-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-84247-w