Abstract
Existing evidence suggests that the components of metabolic syndrome (MS) are sensitive to PM2.5, especially in the elderly population, and related results targeting different regions and populations are inconsistent. This study aims to quantify the risk of association between PM2.5 and MS components in the elderly population, as well as the moderating effect of physical exercise (PE) for this association. Biochemical data, demographic data and health behavior data were obtained from CHARLS dataset in 2011 and 2015, and the individual information was matched to obtain the two-wave panel data. We match meteorological data by region to obtain population exposure indicators. Subsequently, the directed acyclic graphs was used to control confonding, then instrumental variable method and fixed effects model were employed to evaluate the causal relationship between PM2.5 and MS components and the moderating effect of PE. A total of 6125 individuals were included. The prevalence of MS was 34.5% and 32.9% in 2011 and 2015 respectively. The instrumental variable probit regression indicated that high concentration PM2.5 exposure (coefPM2.5 = 0.007, P < 0.001) may increase the risk of MS, and PM2.5 had a significant impact on the components of MS, with a positive impact on waist circumference (WC) (coef = 0.052, P < 0.001) and systolic blood pressure (SYS) (coef = 0.214, P < 0.001), and with a negative impact on high-density lipoprotein cholesterol (HDL-C) (coef = − 0.030, P = 0.021), Triglyceride (TG) (coef = − 0.275, P = 0.048) and diastolic blood pressure (DIA) (coef = − 0.030, P = 0.007). Specifically, for each 1 SD increase in PM2.5 exposure, HDL-C decreased by 0.57 mg/dL, TG decreased by 5.29 mg/dL, DIA decreased by 0.57 mmHg, waist circumference increased by 1.001 cm, and SYS increased by 4.11 mmHg. Additionally, low-intensity physical exercise may alleviate the effect of PM2.5 on WC and SYS, while the high intensity exercise may increase the effect of PM2.5 on WC. Exposure to PM2.5 is associated with the occurrence of MS in the elderly population, and has a significant impact on the components of MS in different directions. The moderating effect of physical activity on PM2.5 and MS varies by component. These results may provide scientific support for the prevention and treatment of MS in the elderly.
Similar content being viewed by others
Introduction
Metabolic syndrome (MS), also known as Syndrome X, is a pathology characterized by abdominal obesity, insulin resistance, hypertension and hyperlipidemia (atherosclerosis). It is a complex group of metabolic disorders status1. Although definitions vary slightly in different organization, most focus on obesity, abnormal blood pressure, abnormal blood glucose and abnormal blood lipids. Globally, the prevalence of metabolic syndrome ranges from 20 to 30%, with an increasing trend in recent years, and is a public health problem that needs to be addressed urgently. According to data published by the CDC in 2017, about one-third of American adults have metabolic syndrome2, and the standardized prevalence in China is 24.5%3. Previous studies4,5,6 have shown that metabolic syndrome has a significant impact on the onset and development of many diseases, especially diabetes and cardiovascular disease. Evidence suggests that the underlying causes of the metabolic syndrome are multifactorial, including poor dietary behavior and lifestyle2,7,8. Also, exposure to pollutants can influence the occurrence of metabolic syndrome9,10,11.
Several epidemiological studies have shown that PM2.5, PM10 and ozone are associated with cardiovascular disease12, obesity13, and metabolic syndrome14. Zheng et al. noted that10 chronic exposure to PM2.5 was associated with metabolic syndrome, dyslipidemia and impaired fasting glucose, and there was a significant interaction between PM2.5 and exercise on high TG. Lederer et al. found that12 considerable associations between air pollution, particularly PM2.5, and hypertension, acute coronary syndromes, and cardiac arrhythmias, with mechanisms contributing to these prominent adverse effects including, but not limited to, oxidative stress, inflammatory responses, endothelial dysfunction, and autonomic imbalance. In addition, the COX regression results with time-dependent covariates11 suggest that increased annual mean concentrations of PM2.5 lead to the increased risk of metabolic syndrome components such as abdominal obesity and hypertriglyceridemia, and that PM2.5 and NO2 may increase the risk of MS in people who already have MS components. Furthermore, some scholars have worked on the interaction of exercise status with PM2.5 on metabolic syndrome. In a cross-sectional study, Hou indicated15 that long-term exposure to ambient air pollutants was associated with an increased risk of metabolic syndrome, while physical activity can attenuate the relevance. The results of Zheng also suggested that10 people who exercised less were at greater risk of the effects of PM2.5 on high TG and high FPG. However, the application of these findings in elderly population remains uncertain.
We have noted that existing research inferring the effects of air pollutants on metabolic syndrome are mostly based on cross-sectional data, lacking sufficient evidence for causal inference. Therefore, exploring the moderating role of physical activity on the impact of air pollution on metabolic syndrome, we used causal effect models to address the endogeneity associated with air pollution and to assess causal relationships influenced by omitted variables. In this study, we utilize long-term longitudinal panel data from the China Health and Retirement Longitudinal Study (CHARLS), a representative national cohort study of the elderly population in China. By integrating causal modeling methods, including DAGs, fixed-effects models and instrumental variable approaches, we aim to quantify the risk of association between PM2.5 and metabolic syndrome among the elderly, as well as the moderating effect of physical exercise (PE) for this association.
Data and methods
Sources of disease data
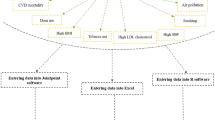
The disease data for this study were drawn from the China Health and Retirement Longitudinal Study (CHARLS) database, a project that collects representative micro-data of Chinese households and individuals aged 45 and above for interdisciplinary ageing research. The CHARLS national baseline survey was conducted in 2011, covering about 10,000 households, 17,000 people, 450 villages, and 150 counties, with follow-ups every 2–3 years16,17. As given in Fig. 1, We used the CHARLS dataset comprises blood data, demographic data and health behavior data. The 2011 data served as the baseline data, while the 2015 data constituted the follow-up data for the same cohort of subjects. Covariates such as sex, age, registration, education level, province and marriage, were derived from the baseline data. The component indicators of metabolic syndrome were obtained from both the 2011 and 2015 surveys. Subsequently, the baseline and follow-up data were integrated as two-wave panel data for further analysis. Finally, the physical exercise was categorized into subgroups for analysis based on the highest values obtained from the two surveys.
Flowchart of the sample selection process.
Sources of exposure data
The PM2.5 exposure data were derived from a dataset compiled by Liu et al.18 of annual average PM2.5 concentrations in Chinese cities. The data was obtained from global ground-observed PM2.5 concentration raster data, calibrated by geographically weighted regression at the University of Fahrenheit, St. Louis, using the Partition Statistics tool in ArcGIS. The dataset can provide data support for studies such as air pollution control in China. The meteorological data is derived from the National Centers for Environmental Information (NCEI) of the National Oceanic and Atmospheric Administration (NOAA). Based on the latitude and longitude of the weather stations and the daily average meteorological data, the daily meteorological data raster is interpolated for the whole country. The daily average meteorological data for each city is then calculated based on administrative boundary data and then calculated the annual average meteorological data.
The annual average PM2.5 concentration data in 2011 and 2015 were matched respectively according to the individual’s residence cities (in total of 117 cities of 28 provinces), which can be used to depict the long-term exposure levels of individuals included in this study19. The fusion process of multi-source data was shown in Fig. 1.
Instrumental variables approach and fixed effects models
When undertaking causal modeling of metabolic syndrome (component) and PM2.5, there may be endogeneity issues due to the problem of omitted variables and pollutant influencing residents for subsequent migrations20. This could lead to biased model estimates, whereas the instrumental variables approach can remove confounding by introducing observable mediating variables to obtain consistent estimates21. Fixed effects modeling is an analytical method applied to panel data that addresses omitted variables that do not vary over time but vary with individuals, omitted variables that do not vary with individuals but vary over time, etc. by controlling for individual, time and other fixed effects, thus reflecting the advantages of panel data. The fixed effects are introduced in the form of dummy variables that can control for factors that do not vary over time or regions, without having to measure them explicitly.
This study combined an instrumental variable approach with a fixed effects model for instrumental variables fixed effects estimation (IVFE), which was robust to endogeneity bias in the presence of omitted variables or measurement error. Since the time-spanning and geographical differences of data in this study, we developed separate time fixed effects models and regional fixed effects models (and two-dimensional fixed-effects models) to select the appropriate fixed effects model to be combined with the instrumental variables for further analysis. The application of instrumental variable need to satisfy two conditions: (1) Correlation of instrumental variables: instrumental variables need to be correlated with endogenous explanatory variables. (2) Exogeneity of instrumental variables: instrumental variables cannot be correlated with perturbation terms. In conjunction with the related studies22,23, we chosen wind speed, rainfall, sunshine, inversion temperature and PM2.5 hysteresis. as instrumental variables for PM2.5. Wind speed usually refers to the speed of air movement (m/s) at 10 m above sea level and can often be combined with surface wind patterns to influence the accumulation and dispersion of pollutants24. At the same time, pollutants emitted in the region are generally in a constant flow state and an important influence on this flow behavior is wind speed. Except in extreme cases, it is difficult to imagine that wind speed could influence the metabolic syndrome and component levels through pathways other than pollutants, also making wind speed an ideal instrumental variable to influence the metabolic syndrome through pollutant concentrations. Rainfall, sunshine, inversion temperature and lagged PM2.5 also satisfy the usage conditions of instrumental variables. Based on these variables, we established the following model of the impact of PM2.5 on MS:
\({Y}_{MS\_rt}\) represents the biochemical index of individual components of metabolic syndrome, \({\alpha }_{0}\) is a constant term, \({PM}_{2.5\_rt}\) is the exposure concentration of PM2.5, \({Social\_adapt}_{rt}\) is the included covariate, \({\alpha }_{1}\) and \({\alpha }_{2}\) are their coefficients, respectively. The term \({\tau }_{t}\) is the fixed-effect term at time t, and \({\omega }_{r}\) is the fixed effect term at the region. After incorporating instrumental variables, the model is as follows:
\(IV\) is the corresponding instrumental variable (IV), and \({\beta }_{1}\) and \({\beta }_{2}\) are the coefficients of the instrumental variable and corresponding covariate, respectively. \(\widehat{{PM}_{2.5\_rt}}\) is the estimated value of PM2.5. The interpretation of other parameters is the same as the model (1).
Criteria for metabolic syndrome
Currently, metabolic syndrome can be diagnosed by different criteria, such as the National Cholesterol Education Program-Adult Treatment Panel III (NCEP-ATP III) criteria, the International Diabetes Federation (IDF) criteria, the World Health Organization (WHO) criteria and the Chinese Medical Association Diabetes Sub-Committee (CDS) criteria for the Chinese population. Considering the authority and representativeness, the IDF criteria were used for the selection and analysis of the components of metabolic syndrome in our study, including WC, TG, HDL-C, blood pressure and blood glucose.
The new definition of metabolic syndrome is given by the International Diabetes Federation (IDF): an individual must have the following conditions to be defined as having metabolic syndrome: central obesity (waist circumference ≥ 90 cm for Chinese men and ≥ 80 cm for Chinese women) plus any 2 of the following 4 factors, (1) Elevated triglyceride (TG) levels: > 1.7 mmol/L(150 mg/dl) or have been treated for this dyslipidemia. (2) Decreased high-density lipoprotein cholesterol (HDL-C): < 1.0 mmolL (40 mg/dl) in men and < 1.3 mmol/L (50 mg/dl) in women, or have been treated for this dyslipidemia. (3) Increased blood pressure: systolic blood pressure ≥ 130 MMHG (1 mmHg = 0.133 kPa) or diastolic blood pressure ≥ 85 mmHg, or have been diagnosed with hypertension and started treatment. (4) Elevated fasting blood glucose (FPG) ≥ 5.6mmolL (100 mg/dl), or already diagnosed with type 2 diabetes; If fasting blood glucose is higher than 5.6mmolL (100 mg/dl), an oral glucose tolerance test (OGTT) is highly recommended, but OGTT is not necessary to diagnose metabolic syndrome.
Covariates
Directed acyclic graphs (DAGs) are an increasingly popular approach for identifying confounding factors that require conditioning when estimating causal effects. First, our study used the directed acyclic graph (DAGs) to explore the influence path of covariates including sex, age, education level, marital status, registration type, province, and physical exercise to adjust for confounding factors. The confounding factors identified by DAGs will be included in the following step modelling. And then we employ the two-way fixed effects model (incorporating both time and region fixed effects) to control the time-varying and region-varying confounders. Finally, we utilize the Instrumental variable (IV) methods to control the unobserved factors such as dietary patterns, which may be induce the endogenous.
Sensitivity analysis
First, utilizing ordinary least squares (OLS) regression, a fixed-effects model for regions, a fixed-effects model for time points and the final model to compare the regression results of MS components under different modeling approaches. Second, examining whether the effects of PM2.5 exposure on metabolic syndrome were consistent across various subgroups, such as sex, region, and education level.
Type variables including sex, education, marriage, registration, MS, number of MS components, physical exercise and region. Numeric variables including age, height, weight, BMI, WC, TG, HDL-C, SYS, DIA, Glu, PM2.5, windspeed, temperature, rainfall, sunshine. All analyses were performed by STATA 15. Two-sided P values were used and P < 0.05 was considered statistically significant.
Results
Descriptive statistics
This study was based on the CHARLS database, which collected metabolic indicator data from the Chinese elderly population in 2011 and 2015, and formed a two-wave panel data of 6125 individuals. Tables 1, 2, and Figure S1 presented the descriptive statistics for the categorical variables (including metabolic syndrome) and the continuous variables (including the components of metabolic syndrome). Table S1 show annual mean of PM2.5 and meteorological data in partly cities.
According to the IDF’s metabolic syndrome diagnostic criteria, a total of 2113 (34.5%) people had metabolic syndrome in 2011 and by 2015 the number of people with the condition was 2017 (32.9%). The metabolic syndrome group score changed between the two surveys, but not significantly. The mean PM2.5 concentration increased from 51.69ug/m3 in 2011 to 55.36ug/m3 in 2015. The mean wind speed in 2011 was 4.41 m/s, slightly less than the mean wind speed in 2015 (4.80 m/s). The average age of the subjects in 2015 was 62.85 years.
Directed acyclic graph
By drawing the directed acyclic graph of PM2.5, MS components and demographic information, the influence path and confounding factors of PM2.5 on MS were determined. Figure 2 shows that registration, age, sex, education and marriage are confounding factors between PM2.5 and MS components that need to be controlled. Physical exercise is viewed as a mediating variable in the subgroup analyses. The MS components may be in the intermediate path from PM2.5 to MS. Therefore, in the regression we controlled for registration, age, sex, education and marriage.
The directed acyclic graph used to identify confounding factor.
Regression results
The impact of pollutants on metabolic syndrome
The six components of the metabolic syndrome diagnostic criteria were represented by WC, HDL-C, TG, GLU, SYS and DIA, respectively, and PM2.5 was regressed on the six components with covariates such as demographic characteristics. The regression results for each component of the metabolic syndrome under different models are given in Table S2, where OLS stands for the least squares estimation model, FE1 stands for the time-point fixed effects model and FE2 stands for the region(province) fixed effects model. We performed F-tests to test for the existence of fixed effects. If both time point and region-fixed effects exist, a bidirectional test will be conducted.
The estimated coefficients of the three models were inconsistent, and F-tests were conducted to select the appropriate model. For two measures, waist circumference and DIA, the F-tests indicated that the models had regional fixed effects but not time-point fixed effects; the models had both time-point and regional fixed effects for four biochemical indicators, HDL-C, TG, GLU, and SYS. Appropriate models were selected for each component and then instrumental variables were incorporated to construct the final models.
To investigate the effect of PM2.5 on metabolic syndrome, suitable instrumental variables were selected and validated for further exploration. Table S3 shows the selection of instrumental variables in different models. The above component models of metabolic syndrome were selected and incorporated into the instrumental variables, and the results of the series tests of the instrumental models are shown in Table S2. For HDL-C and GLU, the endogeneity test indicated that the original model had no endogenous variables, so we still selected the time-point and region-fixed effects model as the final model. Similarly, the DIA model had no endogenous variables. In contrast, the results of the Hausman/DWH test for WC, TG and SYS indicated (P < 0.05) that there were endogenous variables in the explanatory variables, and PM2.5_lag, wind speed and temperature inversion were included as instrumental variables for PM2.5, and subsequent tests were conducted. The weak instrumental variable test showed that the model had no weak instrumental variables (P < 0.001), meaning that the correlation between endogenous and instrumental variables was satisfied.
The final regression results are summarised in Table 3, where regional fixed effects models incorporating instrumental variables (iv = PM2.5_lag and rainfall) were selected for WC, and regional-time point fixed effects models were selected for HDL-C and GLU. Regional-time point fixed effects models incorporating instrumental variables were selected for TG (iv = PM2.5_lag and wind speed) and SYS (iv = Rainfall and sunshine). Regional fixed effects models were selected for DIA. PM2.5 had a significant effect on all components of the metabolic syndrome at the α = 0.05 level, with a negative effect on HDL-C (coef = − 0.030), TG (coef = − 0.275), and DIA (coef = − 0.030), and a positive effect on WC (coef = 0.052) and SYS (coef = 0.214). Changes in exposure concentrations of PM2.5 affect the component markers of metabolic syndrome. Specifically, for each 1 s.d.(\(\sigma\)) increase in PM2.5 exposure, HDL-C decreased by 0.57 mg/dL, TG decreased by 5.29 mg/dL, DIA decreased by 0.57 mmHg, waist circumference increased by 1.001 cm, and SYS increased by 4.11 mmHg. The impact of PM2.5 on GLU is not significant.
Effect modification of physical exercise on the metabolic syndrome of PM2.5 effects
We stratified the data by whether or not they performed light, moderate, or heavy physical activity to test whether they altered the effect of PM2.5 on the metabolic syndrome (component) in individuals. We used similar regression models to explore whether the effect of PM2.5 on the metabolic syndrome (component) varied by physical activity levels in older age groups.
For the light exercise intensity stratum, physical activity reduced the effects of PM2.5 on WC and SYS, and increased the effects on HDL-C, TG, GLU, and DIA. However, for the heavy exercise intensity stratum, physical activity exacerbated the effects of PM2.5 on WC, and alleviated the effects of PM2.5 on HDL-C, TG, GLU, SYS, and DIA. For moderate exercise intensity stratum, the effects of PM2.5 on MS components were basically consistent with total effects. Figure 3A illustrates how the three physical activities moderated the effect of PM2.5 on the metabolic syndrome component.
Effect plot of PM2.5 and MS components subgroup analysis (A) The physical exercise grouping. (B) The sex grouping. (C) The region grouping. (D) The education grouping.
Effect modification of other factors on the metabolic syndrome of PM2.5 effects
The impacts of PM2.5 on different components vary between males and females, and most of its effects on males are higher than those on females (See Fig. 3B). As shown in Fig. 3C, D, in different regions, there were also differed in the impact of PM2.5 on the components of metabolic syndrome, with an overall trend of higher effects in the northwest than in the north, and in the north than in the south. Compared to other regions, the population in the northwest region was more affected by PM2.5 on metabolic syndrome, mainly manifested in SYS and DIA. PM2.5 had a more pronounced impact on WC, DIA and SYS, among the low-education level population. The sensitive analysis results were mostly consistent with the main results of our final model, which validated the robustness of our results (See Fig. 3 and Table S3).
Results of sensitivity analysis
As given in Table S2, comparing the regression results for MS components under different models and comparing them with the final model results shows the direction and significance of the key variable coefficients were consistent. Second, we have verified that the effects of PM2.5 on metabolic syndrome are basically the same across subgroups in Fig. 3 (e.g., sex region and education level,).
Discussion
In this study, we constructed a causal model of PM2.5 and metabolic syndrome (components) using the CHARLS database, a representative Chinese longitudinal cohort data, and a fixed-effects model combined with an instrumental variables approach, meanwhile we also developed separate models for different components according to the conditions of model applicability. We developed regional fixed-effects models in the measurement components of waist circumference and DIA. Temporal and regional fixed-effects models with instrumental variables (use wind speed and PM2.5 lag. as an instrumental variable for PM2.5) were built in the components of biochemical indicators such as TG and SYS. We found a causal relationship between PM2.5 and components of the metabolic syndrome and also identified the moderating role of physical activity in this relationship.
The results indicated that, after adjusting for confounding factors, including gender, age, education level and lifestyle habits, PM2.5 concentration significantly influences most components of the metabolic syndrome. The results shown that higher PM2.5 concentration is associated with higher WC (coef = 0.052, P < 0.001) and SYS (coef = 0.214, P < 0.001). Conversely, the impact on HDL-C (coef = − 0.030, P < 0.05), TG (coef = − 0.275, P < 0.05), and DIA (coef = − 0.030, P < 0.05) was negative. The observed PM2.5 induced the increase of WC is consistent with emerging evidence of particulate matter’s adipogenic effects25,26. Inhalation of PM2.5 particulate matter may induce systemic inflammation, activating NLRP3 inflammasomes in adipose tissue macrophages and promoting adipocyte hypertrophy through PPAR-γ mediated lipid accumulation27,28. Multiple studies have found that PM2.5 exposure significantly raises systolic BP, with a higher effect size in our study than the previous in NEJM29,30,31. One of the possible reasons is that the period from 2011 to 2015 is a critical time window for pollution control in China, reflecting the significant impact of severe pollution on health. It should be noted that the effect size was low in this study compared to a German cohort study. The differences in PM2.5 exposure levels in different countries, the PM2.5 concentration in Europe is generally lower than that in China, and the marginal effect of long-term exposure per unit PM2.5 at high concentration may be decreasing32. A group study by Sun33 based on Wuhan data found that short-term exposure to PM2.5 can lead to a decrease in TG and TC, and an increase in HDL-C. That may be related to the lower concentrations of PM2.5 and the differences in the study populations.
The biological mechanisms underlying PM2.5 and the metabolic syndrome are not known but may be explained by the relationship between its components and pollutants. The six components of the metabolic syndrome (WC, TG, HDL-C, GLU, SYS and DIA) can be grouped into four areas: obesity, lipids, blood glucose and blood pressure. The effects of PM2.5 on obesity in organisms can be mediated through increased risk of inflammation, altered energy metabolism, and altered dietary behavior. It has been shown that exposure to PM2.5 is associated with systemic pro-inflammatory responses in humans and animals27,34, including inflammation, endothelial dysfunction and upregulation of endothelial damage mediators, with significant increases in inflammatory mediators such as tumor necrosis factor (TNF) alpha, prostaglandin E2 and C-reactive protein, and vasoconstrictors. This may also lead to an increase in human blood pressure. Meanwhile, one of the main causes of obesity is due to abnormal energy metabolism35, organisms exposed to PM2.5 may affect their own and their offspring’s energy balance by inducing BAT (brown adipose tissue) bleaching and regulating food intake, causing fat accumulation. PM2.5 inhalation comes into contact with lung epithelial cells and lung macrophages and releases large amounts of cytokines and ROS, which can induce cellular lipid peroxidation and thus lead to abnormalities in blood lipids36, which is the main reason for its impact on the development of cardiovascular disease. It has also been suggested that it can affect blood lipid levels through the Notch signaling pathway37. The mechanism by which PM2.5 affects blood glucose in humans can be explained by its effect on insulin resistance, and the reduced insulin sensitivity caused by pollutant exposure may be one of the ways in which it affects blood glucose38. Organic matter (OM) in the composition is a major component of the association28. Also, PM2.5 may mediate insulin resistance or diabetes by affecting the gut microbiota and altering sphingolipid metabolism39.
Previous studies found that physical activity can improve insulin resistance and lipid disorders, thereby reducing the risk of metabolic syndrome40,41. However, physical activity, especially outdoor exercise, may also increase the risk of exposure to air pollutants. Another study using the CHARLS database showed that at high pollution concentrations, high-intensity exercise increases the threat of PM2.5 exposure to people42. A study on hypertension also found that the protective effect of exercise would be cancelled out when PM2.5 concentration was too high43. Our results suggest that moderate-intensity physical activity is more susceptible to the effect of PM2.5 on SYS than low-intensity and high-intensity physical activity. However, for WC component, participants who engaged in vigorous physical activity were more likely to be affected by PM2.5 exposure. The reason may be that is a multi-pathway interaction between each component of MS, and the intensity of physical activity and the different exercise environment (indoor and outdoor), resulting in different and complex regulatory effects of exercise on each component and PM2.5. Besides, the characteristics of exercise patterns in the elderly population may be different from those in the general adult population. One study showed that 68% of exercise places for the elderly are located in green areas such as parks, and the interception effect of vegetation on PM2.5 (19–37% reduction in concentration) may form a protective barrier23,44,45. The high-intensity exercise group may mostly concentrate around traffic roads, and the increase of PM2.5 exposure may counteract the benefits of exercise, resulting in the differences of the effect in each component.
Our study used a combination of fixed effects models and instrumental variables to address endogeneity issues arising from the modelling process, and directed acyclic graphs to identify and control for potential confounders, allowing for a more accurate and reliable causal relationship between PM2.5 and MS. Based on the metabolic decline and unique physical exercise patterns in middle-aged and older adults, our study is based on the period of higher pollutant severity in China to explore the cumulative effects of long-term exposure to PM2.5, and in conjunction with emerging research advances in recent years. We also analyzed the causal effects of PM2.5 on the components of MS. In fact, the development of MS is a cumulative effect of environmental exposure, and the pathological mechanisms, such as lipid metabolism disorders and insulin resistance, often require several years or even longer to become apparent. As a critical transition stage for China’s pollution control, Period 2011 to 2015 can provide an important time window to reflect the impact of serious pollution on health, and provide a baseline reference for subsequent policy effectiveness assessment. In addition, the key diagnostic indicators of MS (e.g. waist circumference, blood pressure, blood glucose, blood lipids, etc.) have remained stable over time in terms of medical definitions and clinical thresholds. The data in 2011 can be viewed as a baseline reference and data in 2015 can used to measure the health outcome of long-term effects of PM2.5 exposure. Thus, using longitudinal data from 2011 to 2015, it is possible to capture the long-term cumulative effects of PM2.5 exposure on MS, especially in the middle-aged and elderly population, where metabolic function decline is more significant, and the findings are more valuable for clinical reference.
Current studies provide important evidence for the causal relationship between PM2.5 and MS, however, this study has some limitations. Firstly, our data were obtained from the 2011 and 2015 data from CHARLS, and for the older age groups after a long follow-up period, the large number of dropouts resulted in a small sample size after matching, which had some impact on the analysis results. Thus, extending the follow-up period to include a larger and more representative population, combined with integrating additional cohort data (e.g., the China Longitudinal Healthy Longevity Survey, CLHLS46), would facilitate multicenter studies and enable a more robust assessment of the cumulative effects of long-term PM2.5 exposure on MS. Secondly, due to the use of municipal exposure level as a proxy variable for individual exposure, it is difficult to reflect the real exposure of individuals, and the estimated effect may be biased. Based on the satellite remote sensing47, land use regression models or personal monitoring devices48 to improve the estimation accuracy of individual PM2.5 exposure data is necessary to improve the spatial resolution of PM2.5 exposure. Thirdly, combining more factors such as diet intake, energy expenditure, socioeconomic status (SES), green space and considering the moderating effects of these factors on the association between PM2.5 and metabolic disease is of significance49. These issues should be considered in future.
Conclusion
Exposure to PM2.5 is associated with the occurrence of metabolic syndrome in the elderly population and has a significant impact on several types of components. Such as the effect of PM2.5 on waist circumference and SYS was positive, while the effect on HDL-C, TG and DIA was negative. Exposure to high concentrations of PM2.5 increases the occurrence of metabolic syndrome components, and exercise can regulate this effect. The moderating effect of physical activity on PM2.5 and metabolic syndrome varies with different component. In terms of different regions, the impact effect of PM2.5 on MS shows a trend of higher in the Northwest region than in the north region and higher in the South region.
Data availability
Data is publicly available for download from https://charls.charlsdata.com/pages/data/111/zh-cn.html.
References
Huang, P. L. A comprehensive definition for metabolic syndrome. Dis. Model Mech. 2(5–6), 231–237 (2009).
Saklayen, M. G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 20(2), 12 (2018).
Li, R. et al. Prevalence of metabolic syndrome in Mainland China: A meta-analysis of published studies. BMC Public Health 16, 296 (2016).
Gurusamy, J. et al. Exercise, diet and educational interventions for metabolic syndrome in persons with schizophrenia: A systematic review. Asian J. Psychiatr 36, 73–85 (2018).
Bassi, N. et al. Lifestyle modification for metabolic syndrome: A systematic review. Am. J. Med. 127(12), 1241–1242 (2014).
Lemieux, I. & Despres, J. P. Metabolic syndrome: Past, present and future. Nutrients 12(11), 3501 (2020).
Li, Y. et al. Metabolic syndrome prevalence and its risk factors among adults in China: A nationally representative cross-sectional study. PLoS ONE 13(6), e199293 (2018).
Martinez-Ferran, M. et al. Metabolic impacts of confinement during the COVID-19 pandemic due to modified diet and physical activity habits. Nutrients 12(6), 1549 (2020).
Park, S. K. et al. Particulate air pollution, metabolic syndrome, and heart rate variability: The multi-ethnic study of atherosclerosis (MESA). Environ. Health Perspect. 118(10), 1406–1411 (2010).
Zheng, X. Y. et al. Effects of long-term PM(2.5) exposure on metabolic syndrome among adults and elderly in Guangdong, China. Environ. Health 21(1), 84 (2022).
Chen, Y. C. et al. Long-term exposure to air pollution and the occurrence of metabolic syndrome and its components in Taiwan. Environ. Health Perspect. 131(1), 17001 (2023).
Lederer, A. M. et al. Cardiovascular effects of air pollution: current evidence from animal and human studies. Am. J. Physiol. Heart Circ. Physiol. 320(4), H1417–H1439 (2021).
Wei, Y. et al. Chronic exposure to air pollution particles increases the risk of obesity and metabolic syndrome: findings from a natural experiment in Beijing. FASEB J. 30(6), 2115–2122 (2016).
Poursafa, P. et al. DNA methylation: a potential mediator between air pollution and metabolic syndrome. Clin. Epigenet. 14(1), 82 (2022).
Hou, J. et al. Long-term exposure to ambient air pollution attenuated the association of physical activity with metabolic syndrome in rural Chinese adults: A cross-sectional study. Environ. Int. 136, 105459 (2020).
Zhao, Y., Strauss, J., Yang, G., et al. China Health and Retirement Longitudinal Study, 2011–2012 National Baseline Users’ Guide. (National School of Development, Peking University, 2013).
Zeng, P. et al. Association of metabolic syndrome severity with frailty progression among Chinese middle and old-aged adults: a longitudinal study. Cardiovasc Diabetol 23(1), 302 (2024).
Liu, H. PM2.5 Annual average concentration dataset for 342 Cities in China (2000–2021) (Science Data Bank, 2023).
Liu, C. et al. Effect of long-term exposure to PM2.5 on the risk of type 2 diabetes and arthritis in type 2 diabetes patients: Evidence from a national cohort in China. Environ Int 171, 107741 (2023).
Shen, F. et al. Air pollution and migration decision of migrants in low-carbon society. Int. J. Environ. Res. Public Health 20(1), 870 (2023).
Lousdal, M. L. An introduction to instrumental variable assumptions, validation and estimation. Emerg Themes Epidemiol. 15, 1 (2018).
Schwartz, J., Bind, M. A. & Koutrakis, P. Estimating causal effects of local air pollution on daily deaths: Effect of low levels. Environ. Health Perspect 125(1), 23–29 (2017).
Ju, K. et al. Causal effects of air pollution on mental health among Adults–An exploration of susceptible populations and the role of physical activity based on a longitudinal nationwide cohort in China. Environ. Res. 217, 114761 (2023).
Xie, J. et al. Quantitative evaluation of impacts of the steadiness and duration of urban surface wind patterns on air quality. Sci. Total Environ. 850, 157957 (2022).
Mazidi, M. & Speakman, J. R. Ambient particulate air pollution (PM2.5) is associated with the ratio of type 2 diabetes to obesity. Sci. Rep. 7(1), 9144 (2017).
Tamayo-Ortiz, M. et al. Exposure to PM(2.5) and obesity prevalence in the greater Mexico City area. Int. J. Environ. Res. Public Health 18(5), 2301 (2021).
Calderon-Garciduenas, L. et al. Systemic inflammation, endothelial dysfunction, and activation in clinically healthy children exposed to air pollutants. Inhal Toxicol. 20(5), 499–506 (2008).
Shen, Y. et al. Long-term exposure to fine particulate matter and fasting blood glucose and diabetes in 20 million chinese women of reproductive age. Diabetes Care 47(8), 1400–1407 (2024).
Baumgartner, J. et al. Highway proximity and black carbon from cookstoves as a risk factor for higher blood pressure in rural China. Proc. Natl. Acad. Sci. USA 111(36), 13229–13234 (2014).
Fuks, K. et al. Long-term urban particulate air pollution, traffic noise, and arterial blood pressure. Environ. Health Perspect. 119(12), 1706–1711 (2011).
Rajagopalan, S. & Landrigan, P. J. Pollution and the heart. N. Engl. J. Med. 385(20), 1881–1892 (2021).
Burnett, R. et al. Global estimates of mortality associated with long-term exposure to outdoor fine particulate matter. Proc. Natl. Acad. Sci. USA 115(38), 9592–9597 (2018).
Sun, J. et al. Association of short-term exposure to PM(2.5) with blood lipids and the modification effects of insulin resistance: A panel study in Wuhan. Toxics 10(11), 663 (2022).
Xu, X. et al. Effect of early particulate air pollution exposure on obesity in mice: Role of p47phox. Arterioscler Thromb. Vasc. Biol. 30(12), 2518–2527 (2010).
Chen, M. et al. Prenatal and postnatal mothering by diesel exhaust PM(2.5)-exposed dams differentially program mouse energy metabolism. Part Fibre Toxicol. 14(1), 3 (2017).
Guarnieri, M. & Balmes, J. R. Outdoor air pollution and asthma. Lancet 383(9928), 1581–1592 (2014).
Zhang, T. et al. Exposure to PM(2.5) affects blood lipid levels in asthmatic rats through notch signaling pathway. Lipids Health Dis. 18(1), 160 (2019).
Chen, Z. et al. Ambient air pollutants have adverse effects on insulin and glucose homeostasis in Mexican Americans. Diabetes Care 39(4), 547–554 (2016).
Zhao, L. et al. PM2.5 and serum metabolome and insulin resistance, potential mediation by the gut microbiome: A population-based panel study of older adults in China. Environ. Health Perspect. 130(2), 27007 (2022).
Myers, J., Kokkinos, P. & Nyelin, E. Physical activity, cardiorespiratory fitness, and the metabolic syndrome. Nutrients 11(7), 1652 (2019).
Gallardo-Alfaro, L. et al. Leisure-time physical activity, sedentary behaviour and diet quality are associated with metabolic syndrome severity: The PREDIMED-Plus Study. Nutrients 12(4), 1013 (2020).
Zhao, Y. et al. Long-term exposure to fine particulate matter modifies the association between physical activity and the risk of hypertension. Environ. Sci. Pollut. Res. Int. 30(15), 43690–43701 (2023).
Liu, Q. et al. Long-term exposure to fine particulate matter modifies the association between physical activity and hypertension incidence. J. Sport Health Sci. 11(6), 708–715 (2022).
Zhang, L. et al. Effect of vegetation seasonal cycle alterations to aerosol dry deposition on PM2.5 concentrations in China. Sci. Total Environ. 828, 154211 (2022).
Yao, J. et al. Dry deposition effect of urban green spaces on ambient particulate matter pollution in China. Sci. Total Environ. 900, 165830 (2023).
Center for Healthy Aging and Development Studies. The Chinese Longitudinal Healthy Longevity Survey (CLHLS)-Longitudinal Data (1998–2014) . Peking University Open Research Data Platform, 2016.
Hsu, C.-Y. et al. Exposure estimates of PM2.5 using the land-use regression with machine learning and microenvironmental exposure models for elders: Validation and comparison. Atmos. Environ. 318, 120209 (2024).
Yang, W. & Zhao, B. A real-time personal PM2.5 exposure monitoring system and its application for college students. Build. Simul. 17, 1531–1539 (2024).
Liu, L. et al. Air pollution, residential greenness, and metabolic dysfunction biomarkers: Analyses in the Chinese Longitudinal Healthy Longevity Survey. BMC Public Health 22(1), 885 (2022).
Funding
The study was funded by the Ningxia Medical University Major Science and Tchnology and Achievements Project (XJKF230204), Natural Science Foundation of Ningxia (2023AAC02033) and National Natural Science Foundation of China (12061058).
Author information
Authors and Affiliations
Contributions
Xianglong Liu and Fan Ding wrote the original draft. and Jie Wuand Jiayi Tian obtained the data acquisition and Shi Zhao reviewed and edited the main manuscript text and Yu Zhao reviewed, supervised and edited the main manuscript text. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors read and agree to submit this manuscript to your journal.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, X., Ding, F., Tian, J. et al. Causal association between PM2.5 and metabolic syndrome in the Chinese elderly population-insights from a cohort study of CHARLS. Sci Rep 15, 15028 (2025). https://doi.org/10.1038/s41598-025-00160-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00160-w