Abstract
Posttraumatic stress disorder (PTSD) is characterized by exaggerated responses to trauma-relevant cues and impairments in relation to contextual information. However, it is not clear whether this dysfunction is related only to memory processes, or whether early perceptual and attentional processing is already dysfunctional. We examined early processing and retrieval of trauma-related cues and neutral contexts in 20 individuals diagnosed with PTSD (PTSD) and 20 trauma-exposed controls without diagnosis of PTSD (NPTSD) using simultaneous high-density electroencephalography and eye-tracking. A group of 20 non-trauma-exposed healthy controls (HC) was employed to test for responses to trauma-unrelated cues and contexts. The earliest visual event-related potential (C1) was positive for individuals diagnosed with PTSD and negative for NPTSD, suggesting enhanced early visual processing of the cue. Eye-tracking showed that PTSD but not NPTSD displayed significantly longer latencies before looking at contexts than at trauma-related cues. The PTSD group performed significantly worse than the NPTSD group in correctly retrieving rearranged cue/context associations compared to consistent associations. Memory strength for rearranged cue-context pairs was significantly predicted by the early processing measures of the context. Perception of traumatic cues in neutral contexts is biased in PTSD at early processing stages and contributes significantly to the impairment in context-relational memories. For trauma-unrelated cues and contexts no significant differences emerged between PTSD and trauma-exposed as well as non-trauma-exposed controls. Treatments for individuals diagnosed with PTSD should focus on early processing, perception and attention of cue/context traumatic associations in addition to contextual memory.
Similar content being viewed by others
Introduction
Posttraumatic stress disorder (PTSD) develops in response to a traumatic event and is characterized by a number of distressing symptoms. These include re-experiencing of the traumatic event (intrusive memories, thoughts and/or flashbacks), heightened general arousal (hyperattention and continuous monitoring for potential threats, enhanced startle reactivity), avoidance of reminders of the traumatic event and negative alterations in cognitions and mood1.
Several psychobiological mechanisms have been proposed for the development of PTSD. Among them are heightened fear learning and memory, increased salience/threat detection, diminished executive control and deficient emotion regulation with respect to trauma cues. In addition, it has been proposed that the heightened cue reactivity might be related to deficient contextual processing2,3,4. For example, while individuals diagnosed with PTSD may show preserved or even enhanced explicit memory for trauma-related information, particularly for central or emotionally salient cues5, contextual aspects of memory, including the specificity of the conditioned response, are often impaired6. By ‘explicit memory,’ we refer to the conscious recall of factual information or events, while ‘contextual aspects’ relate to details such as time, place, and the relationships between different elements of an experience. However, it is important to acknowledge that memory for trauma remains a complex and debated topic. While vivid intrusions of traumatic images and sensations are hallmark features of PTSD7, a substantial body of literature has also documented amnesia, delayed recall, and disruptions in autobiographical memory, particularly in cases of childhood or developmental trauma8,9,10,11,12,13.
Disturbances in autobiographical memory in PTSD, characterized by poor elaboration and deficient contextualization, may prevent individuals from realizing that trauma reminders in a safe context are no longer threatening. This can lead to a persistent feeling of being unsafe due to deficient safety signal learning and overgeneralization6,14. Deficient contextual processing is believed to result in dysfunctional representations of stored trauma cues3,4, potentially explaining the intrusions and flashbacks characteristic of PTSD. The frequently observed hippocampal impairments in PTSD15 might be related to poorer performance in contextually based memories such as extinction, extinction recall, and fear renewal. Steiger et al. (2015) found that, compared to healthy controls, individuals diagnosed with PTSD exhibited lower accuracy in identifying threatening and safe contexts during a context discrimination task16. Furthermore, individuals diagnosed with PTSD failed to extinguish conditioned contextual and cued fear responses and showed increased fear to both dangerous and safe contexts16. These deficient contextual fear memory mechanisms suggest a predominance of elemental (cue-based) representations of trauma at the expense of contextual representations4,17. Neurobiological studies have shown that the amygdala is involved in elemental processing, while the hippocampus and peri- and postrhinal systems mediate contextual processing18,19. Negative emotions have been found to favor amygdalar item memory and disrupt hippocampal associative memory20. This hippocampal impairment can also be related to a lack of differentiation between dangerous and safe contexts19.
While many aspects of fear memory have been extensively studied in individuals diagnosed with PTSD2,3,6, the role of sensory and early processing has only recently gained research attention.
Recent evidence has highlighted alterations in early visual and midbrain circuits associated with trauma-related processing in PTSD. Notably, the superior colliculus, a structure involved in the earliest stages of visual attention and orienting responses, has been implicated in both basic perceptual mechanisms and trauma-related hypervigilance21,22. Moreover, aberrant alpha-band activity in visual cortex and disrupted connectivity with default mode and sensorimotor networks have been reported in individuals with PTSD, further supporting the notion of early sensory hyperresponsivity23,24,25,26. These findings suggest that abnormalities in early perceptual processing may play a critical role in the maintenance and expression of PTSD symptoms. Magnetic resonance imaging (MRI) studies have linked structural integrity in early visual regions in trauma-exposed individuals to susceptibility to PTSD27 and to symptom severity in individuals diagnosed with PTSD compared to trauma-exposed individuals without diagnosis of PTSD (NPSTD)28. Modulation of the visual network by transcranial magnetic inhibitory stimulation has been shown to reduce the emotional intensity of intrusions in non-trauma-exposed individuals exposed to distressing content29.
Perceptual and attentional modulation of incoming sensory information occurs at very early stages (20–80 ms) of processing30,31. This altered perception of trauma-related information could contribute to a mnemonic bias in PTSD. In visual perception, the earliest component of the event-related potential (ERP), C1, is a deflection originating mainly in primary visual processing areas32. C1 exhibits polarity reversal when stimuli are presented in different parts of the visual field32,33, such as the lower/upper part. Perceptual34 and emotional learning35 also modulate C1.
We hypothesized that individuals diagnosed with PTSD might show a bias toward the location of trauma-related cues (mainly placed in the lower part of the pictures) at the expense of the surrounding context, resulting in early perceptual modulation of C1 indicated by polarity inversion. Eye-tracking studies have revealed faster initial fixations to traumatic words and images in individuals with PTSD compared to those without PTSD36,37. A positive correlation between PTSD symptoms and increased attention to and better recognition of fear faces in negative rather than in neutral or positive contexts38 has also been reported. Our study examined early perceptual and attentional processes to cues and contexts and their effects on memory by combining eye-tracker and EEG recordings during free viewing of traumatic cues embedded in neutral contexts. This session was followed by a memory retrieval test on the viewed material where we used two types of stimuli: those with consistent cue-context pairs and those with rearranged pairs. We expected individuals diagnosed with PTSD to show an enhanced focus on trauma-related cues, resulting in an early affective and perceptual bias compared to NPTSD, as indicated by the polarity reversal of the visual C1 component and shorter fixation latencies to the cue with longer fixation latencies to the context in eye-tracking. Additionally, we anticipated that individuals diagnosed with PTSD would demonstrate better retrieval of pictures with consistent cue-context pairs. Their heightened attentional bias toward the cue may facilitate recognition of both previously seen and newly learned cue-context pairs, as these can be processed through familiarity-based and recall-to-reject recognition rather than effortful recollection39. Familiarity-based recognition refers to recognizing something without recalling specific details, while ‘recall-to-reject’ involves remembering specific features to correctly reject incorrect options. In contrast, identifying rearranged pairs requires detecting and resolving inconsistencies between elements or associations, engaging hippocampal and prefrontal-dependent recollection processes, a domain particularly affected in PTSD40. These brain-based processes are involved in retrieving specific relational information, such as how elements were connected in the original experience. We therefore, expected that EEG amplitudes to the cue-context picture pairs and eye-tracking attentional data to contexts would predict memory for rearranged cue-context pairs. By contrast, ERP and eye-tracking data of the trauma cues would predict memory for consistent cue-context pairs. From this point forward, we refer to memory retrieval for consistent pairs as item memory and retrieval for rearranged pairs as association memory.
We confined our analyses of trauma-related cues and neutral contexts to trauma-exposed individuals with and a without diagnosis of PTSD, as non-trauma-exposed individuals did not have traumatic events to report and rated them as less relevant. However, we compared trauma-unrelated cues and contexts across all groups to determine whether there were alterations in perception and memory beyond trauma-relevant cues in the PTSD and both control (trauma-exposed and non-trauma-exposed) groups.
Results
Event-related potential (ERP) data during processing of traumatic cues and neutral contexts
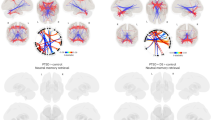
In the ERPs, we compared the polarity of the earliest visual ERP component (C1) between the PTSD (n = 16) and NPTSD (n = 16) groups, while they were viewing traumatic cues embedded in neutral contexts. Firstly, we identified a C1 deflection with an onset peaking at 40 ms, which was maximal over posterior parieto-occipital and occipital sites. The C1 peak of the PTSD group was positive, whereas that of the NPTSD group was negative. This difference in polarity was statistically significant: peak amplitude, effect group: F(1, 30) = 4.939, p = .034, partial η2 = 0.141 (Fig. 1ABCD). Neither laterality nor group by laterality were statistically significant (F(1,30 = 0.254, 0.119, p = .732, partial η2 = 0.004). We used independent component analysis to better characterize our C1 deflection. These components were grouped into several clusters on the basis of dipole location, power, average ERP in the 20–60 ms window of interest, mean event-related spectral perturbation (ERSP) and phase coherence41. Six clusters of independent components explained most of the variance in our ERP epoch following stimulus onset (see Fig. 2), with two clusters showing dipoles in the visual cortex. The other clusters were located laterally and medially in line with the limbic anterior (ACC) and posterior cingulate cortex (PCC), and subcortical (thalamus and hippocampus) regions (see Table S3 in the Supplement). The dipole locations and clusters are shown in Fig. 2. The approximated estimated Talairach coordinates, as defined by the Yale BioImage Suite software website (http://www.bioimagesuite.org), and the nearest gray matter of the mean equivalent current dipole of each cluster are presented in Fig. 2 and Table S3 in the Supplement (see also41,42.
Inverted polarity of the early visual C1 in indivisuals diagnosed with PTSD and trauma controls. In panel A, the grand average event-related potentials for the left and right parieto-occipital clusters, highlighted in gray is the time window of the C1 whose peak was significantly different between individuals diagnosed with posttraumatic stress disorder (PTSD) and the trauma-exposed control group without PTSD diagnosis (NPTSD). Panel B shows scalp maps at specified latencies in the PTSD (orange) and NPTSD (black) groups. In panel C, the mean waveform of the two clusters showing the entire epoch and the identified visual components. In panel D, for the layout of the electrode array, the electrodes used for statistical analyses are shown in blue, with midline electrodes separating the right and left clusters. P100 refers to the positive deflection peaking at ~ 100 ms, N200 refers to the negative deflection at ~ 200 ms, and P300 refers to the positive deflection at ~ 300 ms; these parameters were not analyzed in this paper.
Source localization (dipole clusters). Equivalent dipole localization plots showing the centroid of (left) and the source cluster (right) of each independent component in the Montreal Neurological Institute (MNI) template brain. In orange individuals diagnosed with posttraumatic stress disorder (PTSD) and in black the trauma-exposed control group without PTSD diagnosis (NPTSD).
Eye-tracking results for traumatic cues and neutral contexts
We compared the latency to the first fixation of the traumatic cues and the neutral contexts between the PTSD (n = 19) and NPTSD (n = 18) groups during picture viewing. For the time to first fixation, there was a significant interaction between stimulus type (cue versus context) and group, F(1,35) = 5.121, p = .030, partial η2 = 0.128 (Fig. 3A). Post hoc tests showed that the PTSD group had a significantly longer latency for looking to the context than the NPTSD group (F(1,35) = 6.220, p = .018), whereas there was no significant group difference in the latency to fixation of the cue (F(1,35) = 0.441, p = .511). The latency interval between looking at the cue and looking at the context was also significantly longer in PTSD compared with NPTSD (t(36) = 2.496, p = .017). In addition, individuals diagnosed with PTSD showed significantly longer latencies to first fixation on the images compared to individuals in the NPTSD group, regardless of whether the stimuli were cues or contexts, group: F(1,35) = 4.278, p = .046, partial η2 = 0.109. Across all participants, cues were generally fixated more quickly than contexts, stimulus type: F(1,35) = 68.415, p < .001, partial η2 = 0.662.
Differences in perceptual processing (eye-tracking data) and memory retrieval. Panel A shows the mean eye-tracking time to first fixation. Individual diagnosed with posttraumatic stress disorder (PTSD) show significant longer latency to look at the context than those in the trauma-exposed controls without diagnosis of PTSD (NPTSD). Panel B shows that individual diagnosed with PTSD perform significantly worse than those NPTSD in correctly retrieving rearranged (association) versus consistent cue-context pairs (item).
Results of the memory test related to trauma cues and neutral contexts
We compared memory strength (correct responses) and accuracy (hit rates adjusted for false alarm rates) for retrieval of consistent versus rearranged old/new traumatic cues and neutral context pairs between the PTSD (n = 19) and NPTSD (n = 19) groups during a picture retrieval task. We refer to memory retrieval for consistent pairs as item memory and retrieval for rearranged pairs as association memory (see Methods for details). For memory strength, we observed a significant memory type (item/associative) by group interaction, F(1, 36) = 4.956, p = .032, partial η2 = 0.121 (Fig. 3B). Compared to the NPTSD group, the PTSD group reported significantly reduced memory strength scores in retrieving pictures of the association category than in retrieving pictures of the item category. There was also a significant effect of memory type, with item associations generally better retrieved than context associations (F(1,36) = 7.380, p = .010, partial η2 = 0.170), whereas the factor group was not significant (F(1,36) = 0.006, p = .938). The repeated measures ANOVA with adjusted hit rates yielded similar results. We observed a significant memory by group interaction, F (1, 36) = 4.814, p = .035, partial η2 = 0.118. Compared to the NPTSD group, the PTSD group had significantly lower accuracy in retrieving pictures of the association than of the item category. There was also a significant effect of memory type, with item generally more accurately retrieved than association memory (F (1, 36) = 28.484, p < .001, partial η2 = 0.442), whereas the factor group was not significant (F (1, 36) = 0.063, p = .803).
Results of the hierarchical regression related to trauma cues and neutral contexts
We examined whether early visual processing (C1 ERP) and initial attention allocation (time to first fixation) of traumatic cues in neutral contexts could predict association and item memory strength. For association memory, we found that the best fitting model included the mean C1 ERP peaks (left and right) and the latency to first fixation of the context. These combined ERP and eye-tracking variables significantly predicted the mean memory association scores, reaching ~ 50% of the explained variance (R2 = 0.551, F(3, 27) = 9.811, p < .001; adjusted R2 = 0.495). C1 alone explained ~ 30% of the variance in the memory scores (R2 = 0.355, F(2, 27) = 6.884, p < .001; adjusted R2 = 0.304). The addition of the latency to first fixation of the context (ΔR2 of 0.196, ΔF(1, 24) = 10.456, p = .004) but not of the cue (ΔR2 of < 0.001, ΔF(1, 23) = 0.003, p = .954) to the ERP C1 data led to a statistically significant increase in the prediction of association memory (see Table 1). Item memory was not significantly predicted by any of these variables.
Results with non-traumatic cues and non-trauma-exposed individuals
We compared C1 polarity, latency to the first fixation of the non-traumatic (negative, positive and neutral) cues and the neutral contexts, as well as item/association memory related scores between the (resp. PTSD (n = 16; n = 19; n = 19) and NPTSD (n = 16; n = 18; n = 19) and HC (n = 16; n = 16; n = 19) groups (see Supplement for details). There was no significant effect that survived follow-up tests neither in ERP, eye-tracking or memory (see the Supplement for details).
Discussion
We investigated early perceptual and attentional processing and retrieval of competing realistic information (trauma cues embedded in neutral contexts) in individuals diagnosed with PTSD compared to trauma-exposed NPTSD controls. Our data on the earliest visual ERP (C1), the time to first fixation and the retrieval of cue/context pairs indicate early altered processing of traumatic materials as well as a deficit in the retrieval of rearranged pairs (associations) versus consistent pairs (items). Traumatic cues are attended with shorter latencies than neutral contexts in both groups, but only in the PTSD group this is followed by a significantly longer latency to look at the contexts, compared to the NPTSD group. Reduced memory strength in individuals diagnosed with PTSD for associations was predicted by poorer contextual processing.
During early processing of the pictures, the PTSD, compared with the NPTSD group, showed a very early perceptual difference, as indicated by the polarity (positive in PTSD versus negative in NPTSD) of the first identified visual ERP deflection (C1). C1 has been described with latencies ranging between 20 and 80 ms (see43 for a review) for complex stimuli. Changes in the polarity of the C1 deflection have been related to differences in upper/lower visual field sensory processing due to its retinotopic properties44,45. In the case of complex stimuli such as pictures, changes in polarity have been related to the processing of different but concurrent information in the visual field competing for neural representation46. Our results suggest that individuals diagnosed with PTSD prioritize trauma cues in the lower visual field while neglecting contextual details. In contrast, NPTSD individuals focus more on the context. The amplitude of the C1 deflection has also been associated with affective top-down modulation in aversive learning35 and was found to be modulated by anxiety states and emotional processing47. Like in this study, Yoneda et al. (1995), using EEG and magnetoencephalography (MEG), observed an early visual response at ~ 40 ms after the stimulus, likely generated in the striate cortex48. Another MEG study also showed that stimulus-type (faces-objects-abstract patterns) and task-dependent (comparison of pairs of faces instead of objects or individual faces) modulations in the early latency window (30–60 ms) and amplitude were present49. Interestingly, and in line with our results, only the task that required an association and use of faces versus neutral objects led to faster and greater neural responses.
Our deflection starts at 20 ms and peaks at approximately 40 ms. Braeutigam et al. (2001) described the finding of an earlier onset for C1 as consistent with suggestions of anatomical pathways between thalamic nuclei and subcortical as well as cortical locations that may be activated simultaneously with or even before the striate cortex49. In our source location results, we found that cortical sensory and limbic regions (occipital and cingulate regions) as well as subcortical structures (thalamus and hippocampus) explained significant variance in the ERP signal. The primary visual cortex, specifically the calcarine fissure, has been for long described to be the main generator of the visual C150 and to be impermeable to attentional changes. In contrast, several studies using emotional material35,45,47 and a recent meta-analysis on the C1 showed an effect of attention of moderate size51, suggesting that input from several regions could contribute to the computation of prior activations in the primary visual cortex. The type of used stimuli should be considered, as many of the initial studies on the C1 generators implied non-emotionally relevant stimuli. In this study, we found that the anterior and posterior cingulate cortices (ACC, PCC), together with prefrontal regions, were identified as sources contributing to the ERP signal. While we did not perform formal network analyses, these regions could be part of a frontoparietal attentional network involved in amplifying relevant and suppressing irrelevant input, thus increasing afferent information-related sensory representations (Bayer et al., 2017). Notably, the PCC is typically associated with the default mode network52, and it may also be flexibly recruited during attentional processing, depending on task demands. Animal studies described not only feedback but also feedforward modulation to the primary visual cortex from other cortical regions53 enhancing visual representation and contributing to context-dependent modulation of visual processing54. Anatomical studies in primates have shown that higher-order occipital areas, temporal regions such as the inferotemporal cortex (IT), and subcortical structures including the pulvinar and superior colliculus contribute in parallel to the hierarchical organization of the visual system55. Hippocampus and thalamus generators of our ERP signal could then send information for the contextualization and the salience portion of the visual stimuli. In line with Qin et al., stimulus-dependent (e.g. color, location, intensity) and observer-dependent information (e.g. contextualization biases) are integrated dynamically, thereby supporting an efficient selection of the most relevant information51.
The eye-tracking results of the viewing task are in line with the neural ERP results. Individuals diagnosed with PTSD had significant longer latencies to look at the neutral backgrounds versus the trauma cues compared to the NPTSD group. The NPTSD group looked at the cue as fast as the PTSD group. This suggests that both groups do not differ much in the processing of trauma cues, but that the difference resides in contextual processing. These results add to previous studies that used eye-tracking to examine differences in the processing of trauma or other emotional stimuli in individuals with PTSD, but that did not consider the context. For example, individuals diagnosed with PTSD showed significantly more first fixations of traumatic words than did those with NPTSD or HCs36. Thomas et al. (2013) showed that individuals diagnosed with PTSD compared to HC have significantly more initial fixations of trauma-related images, with the trauma-exposed NPTSD group in between37. Consistent with our primary findings, no significant interactions emerged when analyzing non-traumatic cues embedded in neutral contexts. Although a significant two-way interaction between item-association and group was observed, post hoc comparisons were not significant, limiting interpretability. This suggests that alterations in cue-context memory in PTSD may be particularly pronounced when processing trauma-related cues, rather than extending to non-traumatic emotional stimuli. Further research is needed to clarify whether these patterns generalize beyond trauma-related material. Partially in contrast to this, a recent study recording saccade latency, reported enhanced overt attentional allocation towards emotional material (positive, negative and violent) in PTSD than in HC56. In their study, participants were intentionally instructed to orient their gaze in a given direction, thus it can be that such paradigm may have been more suitable in highlighting differences in explicit orientation of attentional processes.
Many studies have shown that PTSD is associated with heightened vigilance and increased attention to threat-related information, which is collectively referred to as a threat-related attentional bias36,37,57,58. Our results add to this research by showing that individuals diagnosed with PTSD, compared to NPTSD, fail to adequately focus on the context of a traumatic event without significant differences in fixations latencies. A recent review found little support for enhanced threat detection, hypervigilance and attentional avoidance and supported instead the concept of sustained attention to threat (i.e. attention maintenance) in PTSD59. Difficulty in disengaging attention from a stimulus has been reported also in other studies56. Our results suggest that difficulties in the processing of the context in which a stimulus is embedded may be partially responsible for this bias.
The results of the memory tests are in line with the assumption that association memory is impaired in individuals diagnosed with PTSD4,11. Individual diagnosed with PTSD performed significantly worse in flexibly retrieving cue/context associations than did those in the NPTSD group. This finding is supported by the notion that deficient contextual memory enhances traumatic cue retrieval20. This impairment in associative retrieval is consistent with findings in both acute and chronic post-trauma conditions. For example, a study demonstrated that individuals with acute stress disorder (ASD) exhibited marked deficits in associative memory compared to item memory for neutral stimuli, suggesting that impaired binding of event components might be an early cognitive consequence of trauma exposure60. This deficit, particularly in visual associative memory, was found to persist even after ASD symptoms resolved, highlighting a potentially enduring cognitive vulnerability60. Similarly, research on acute stress in non-clinical samples has shown that stress impacts associative recognition more than item recognition, particularly for pictorial stimuli61. These findings support the idea that associative memory, rather than simple item recognition, is more susceptible to stress-related disruptions, which could explain the pronounced contextual memory deficits observed in PTSD.
To better capture these memory processes, we computed separate scores for item and association memory. Item memory was derived from responses to consistent cue-context pairs (identical and new/new), where participants were not explicitly required to evaluate the cue, context, or their relationship but rather judged whether the image as a whole was entirely new or previously seen. In contrast, association memory was extracted from rearranged cue-context pairs (new/old, old/new, and old/old), where participants had to determine whether recombination of cues and contexts had been encountered before.
This distinction aligns with theoretical models suggesting that consistent cue-context pairs can be supported by familiarity-based recognition, while rearranged cue-context pairs require more effortful relational retrieval, engaging hippocampal and prefrontal-dependent processes that are particularly impaired in PTSD39,40. Thus, recognizing a familiar picture as a whole may rely on a general sense of familiarity, while recognizing that specific elements have been rearranged demands more complex memory processes that involve linking parts together, a function often disrupted in PTSD. The present results suggest that the PTSD-related memory impairment is specific to associative retrieval rather than general deficits in recognizing previously encountered stimuli or correctly rejecting novel ones, a process that may involve recall-to-reject mechanisms (Yonelinas, 2002). Given the central role of context in flexibly representing and retrieving information, as well as in resolving ambiguity, particularly through interactions within a hippocampal-prefrontal-thalamic network, deficits in contextual processing may lead to a heightened focus on cues62. This, in turn, could contribute to various aspects of posttraumatic psychopathology, including exaggerated emotional responses, impaired emotion regulation, and reduced flexibility in behavioral choices2.
Finally, our hierarchical regression results showed which perceptual variables are predictive of the memory impairment in PTSD. Only measures associated with contextual processing, both in ERPs and eye-tracking, significantly predicted variation in memory strength in the association category.
It is not clear which mechanisms could underlie this contextual processing deficit. Levy-Gigi et al.63 reported that individuals repeatedly exposed to traumatic experiences cannot properly integrate contextual information even when they did not (or not yet) develop PTSD63. In line with this study, several lasting sequelae to the brain have been reported as caused by traumatic stress, including increased cortisol and norepinephrine responses to subsequent stressors64 and have been associated with gray matter reduction of relevant structures, like the hippocampus65. Developing more comprehensive models is important for understanding PTSD. These models could link dysfunction in neuroendocrine, genetic, immunological, and psychophysiological markers in relevant brain circuits to the mechanisms of symptom development and disease maintenance2. Longitudinal studies are needed to validate these models.
This study has several limitations. C1 studies usually require specific paradigms with extensive usage of simple stimulus repetition in different parts of the visual field, which we could not implement with the use of complex stimuli followed by a memory test. To overcome this limitation, we combined shorter EEG recordings with simultaneous eye-tracking and the use of a limited number (30 pictures) of traumatic cues in neutral contexts. When referring to group differences in trauma cue processing throughout the manuscript, we refer specifically to early visual attention and perception of static images. Importantly, this interpretation is limited to the specific experimental context and methodology employed, including the analysis of the early visual ERP component (C1) and eye-tracking measures. Our design does not allow for conclusions about deeper neural structures or the processing of dynamic or ecologically richer stimuli. Before conducting the study, a power analysis was performed to estimate the necessary sample size. This analysis was based on detecting an effect size of Cohen’s f = 0.40, with a power of 0.80 and a significance level of α = 0.05. The calculation suggested that a sample of n = 18 participants per group would be sufficient for identifying effects of this magnitude. We collected data of 20 participants to be on the safe side but could not use all because of technical reasons. After running the study, the observed effect sizes for the interaction effects were somewhat lower than anticipated. Specifically, the memory × group interaction yielded a partial η2 of 0.118 (Cohen’s f ≈ 0.36), while similar interactions in eye-tracking and EEG data showed partial η2 values of 0.128 and 0.141, respectively. While these are still meaningful effects, they fall below the originally anticipated effect size of partial η2 ≈ 0.14–0.16 (Cohen’s f ≈ 0.40) that would ensure optimal statistical power. This difference suggests that the study may have profited from a increased sample sizes to better capture smaller but meaningful interaction effects and to further enhance the generalizability of the results. However, it is important to note that, despite these considerations, the study still identified significant effects across multiple measures, reinforcing the robustness of the findings. We limited our main investigation to traumatic cues and neutral contexts. We separately examined the processing and retrieval on non-traumatic cues and neutral contexts in comparison with HC. For this more complex design our sample might have been underpowered and therefore the effects found were not significant. Further studies with a central interest in non-traumatic stimuli should investigate this more extensively. The presence of trauma heterogeneity provides ecological validity to the study, but also needs to be considered as a possible source of noise in the data. Further studies focusing specifically on one trauma type might be helpful.
In conclusion, individuals diagnosed with PTSD exhibited altered early visual and attentional processing, showing a bias toward cues over contexts and significantly slower orientation toward the neutral background surrounding the trauma reminder. In contrast, individuals who did not develop PTSD processed the context more extensively and displayed a balanced attentional distribution between cues and contexts. This perceptual pattern may serve as a valuable indicator not only for assessing avoidance and generalization phenomena but also for understanding threat detection mechanisms and attentional allocation in response to both traumatic and non-traumatic materials. Memory retrieval data demonstrated that explicit associative links between cues and contexts were not properly retrieved in individuals diagnosed with PTSD, and this impairment was closely related to deficits in perceptual processing of contextual information. Our findings contribute to the growing body of literature on PTSD mechanisms by highlighting that not only trauma- and context-related learning and memory but also the perceptual processing of contextual information is altered. These findings are consistent with recent neuroimaging and electrophysiological studies showing altered sensory processing in PTSD, including disrupted early visual and midbrain activity during trauma cue exposure21,22. Evidence of visual cortical hyperactivity and aberrant oscillatory dynamics further supports the idea that trauma-related sensory input is processed differently from very early stages of23,24,25,26. This insight has important implications for both assessment and treatment approaches, emphasizing the need to enhance perceptual processing of contextual information in competition with dominant trauma-related cues. For instance, Narrative Exposure Therapy (NET)66 works in this direction, by helping individuals process the particular elements of their trauma network, including sensory, affective and cognitive memories, thereby enhancing their ability to contextualize these cues and reduce their emotional impact. Another form of therapy worth considering in this context is Deep Brain Reorienting (DBR67, which specifically targets early orienting responses, including activity at the level of the superior colliculus. DBR may complement exposure-based methods by addressing basic sensory mechanisms implicated in trauma-related perceptual processing.
Methods
Participants
We compared the responses of 20 trauma-exposed persons with PTSD and 20 trauma-exposed persons without PTSD (NPTSD) as a trauma control group to individualized traumatic cues and neutral contexts. Twenty HCs (the non-trauma control group) were added for the comparison of non-traumatic standardized positive, negative and neutral cues and neutral contexts. Individuals with PTSD were recruited via the outpatient clinic of the Central Institute of Mental Health, Mannheim, and were self-referred based on press coverage and information on the website of the institute. The Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I)68 was used to assess mental disorders, and the Structured Clinical Interview for DSM-IV Axis II Disorders (SCID-II) German version was used to assess personality disorders69. The Clinician-Administered PTSD Scale (CAPS for DSM-IV)70 was used to assess the current diagnosis of PTSD together with the Posttraumatic Diagnostic Scale (PDS)71. Depressive symptoms were assessed using the German version of the Center for Epidemiological Studies Depression Scale72, and the level of trait anxiety was assessed with the German version of the State-Trait Anxiety Inventory73. Participants in the trauma control group were only included if they had a history of a criterion A trauma for at least 3 months before participation in the study. Seven people with PTSD also met the criteria for current major depressive episodes (MDEs), panic disorder (PD), social phobia (SP) or specific phobia. Three trauma-exposed controls also met the criteria for PD, one for SP and one for current MDE. Exclusion criteria were comorbid borderline personality disorder, history of schizophrenia-spectrum psychosis, bipolar type I affective disorder, current substance abuse, neurological disorders, traumatic head injuries, mental impairments and lack of German language skills. Exclusion criteria for healthy controls (HC) comprised also any personality disorder or Axis I disorder assessed by the German Version of the Structured Clinical Interview for DSM-IV Axis I and II Disorders (SKID-II and SKID-I);68,69, or any history or current psychiatric treatment, and history of trauma.
The PTSD, NPTSD and HC groups did not significantly differ in age, sex, handedness or education. Individuals diagnose with PTSD scored significantly higher than the NPTSD individuals and HCs in terms of depressive symptoms and anxiety levels.
None of the participants was medicated and all were examined by trained clinical psychologists (see Table 2 for the clinical and demographic characteristics of the sample).
Depending on the trauma experienced, the participants in the PTSD or NPTSD groups belonged to seven trauma types: car accident (PTSD = 4; NPTSD = 6), fire (PTSD = 3; NPTSD = 1), hospital (PTSD = 1; NPTSD = 2), war (PTSD = 2; NPTSD = 2), rape (PTSD = 7; NPTSD = 4), suicide (PTSD = 1; NPTSD = 3), and aggression (PTSD = 2; NPTSD = 2). There were no significant differences in the proportions of patients and trauma controls in the different trauma types as assessed by Fisher’s exact test, p = .787. All participants reported to have slept seven or more hours on the day of the assessment. All participants had normal or corrected to normal vision. After informing the participants, by explaining the procedures of the study (EEG, eye-tracking) and the possible emotional content of the stimuli, we obtained written consent of the participants. The study was approved by the Ethics Committee of the Medical Faculty Mannheim of the Heidelberg University and adhered to the Declaration of Helsinki.
Procedure
The participants underwent a two-day experimental paradigm (see Fig. 4 for a schematic representation of the experimental paradigm and of the used stimuli). On day one, simultaneous high-density 128-channel EEG and eye-tracker recordings were performed during free viewing of visual stimuli (acquisition details in the Supplement). Thirty traumatic cues per trauma type (car accident, aggression, rape, hospital, fire, suicide and war) were embedded in neutral contexts (see details regarding the images in the Supplement; Tables S1, S2 and S3). Each participant saw only the traumatic images related to their specific trauma.
Schematic representation of the experimental paradigm and of the used stimuli. Panel A shows the experimental paradigm. On day 1 (left), high-density electroencephalography (EEG) and eye gaze recording occurred during free viewing of visual stimuli. Each trial began with a fixation cross with variable intertrial interval (ITI) duration between 2 and 4 s, followed by the picture of interest lasting 6 s; on day 2 (right) a memory retrieval test took place on the pictures seen on the previous day. Each trial began with a picture lasting 3 s, followed by questions concerning the cues, the contexts and their associations respect to day one. Panel B, below, shows an example of stimuli preparation, with the cues extracted from their original pictures and inserted in neutral contexts. The pictures shown are for illustration purposes, they were not used in the study. The pictures were taken from us and processed in a final image using the software Adobe Photoshop CS5 Extended Version 12.0 https://www.adobe.com/de/products/photoshop.html.
For the comparison with the HCs, we added nontraumatic stimuli, which consisted of 30 neutral, negative and positive cues embedded in neutral contexts for 120 images (for details, see the Supplement). Each image was presented for six seconds. The intertrial (ITI) interval varied from two to four seconds.
On day two, the subjects participated in a memory test (retrieval) of the pictures seen on the previous day. The pictures were created by manipulating the presence of old/new cues and contexts and their associations to assess the differential impact of the cue/context manipulation on memory retention. The test included five picture categories composed of six slides each (see below a more detailed description) (identical – new/new – new/old – old/new – old/old). The identical category included pictures which were exactly the same as those presented on the day before; the new/new category referred to pictures where both cue and context were new, not at all seen on the previous day; the new/old and old/new categories included an old cue inserted in a new background and a new cue inserted in a previously seen background, respectively; and in the old/old category, both the cue and context were already seen on the previous day but rearranged in a different pair (i.e., the cue was in a different context). The memory test was administered using Presentation® software (Version 18.0, Neurobehavioral Systems, Inc., Berkeley, CA, USA, www.neurobs.com). Every image was shown for six seconds, followed by five forced choice questions: “Please choose which one of the following statements best describes this picture”. The answers included “the same pair”, “a new pair”, “only the background is old”, “only the object is old” or “a rearranged pair”. After the memory test, we collected self-reported data on the traumatic pictures, valence, arousal, relevance to self and item/background balance (asking to indicate their relative presence in the picture to control for identification biases) (see Supplement for details and Table S1).
Data analysis
EEG
The EEG data were analyzed off-line with EEGLAB74 running in MATLAB 8.2 (MathWorks, Natick, MA, USA) (see Supplement). To specifically assess early visual activity, peak-to-baseline mean values were extracted from parieto-occipital and occipital posterior channels and grouped into two hemispheric clusters (E63, E64, E65, E66, E68, E69, E70, E73, and E74; for the left hemisphere; and E82, O2, E84, E89, E90, E94, E95, and E99 for the right hemisphere), which surround and approximate Oz and were averaged according to the latency of interest (20–60 ms)49,75,76. Because of technical reasons and artefacts (less than < 75% good epochs) we had to exclude EEG data of four subjects per group (PTSD = 16/NPTSD = 16/HC = 16).
Eye-tracking
Fixations were automatically determined by the eye-tracking software that uses a dispersion-based algorithm, with a threshold of 1º radius. The eye movement parameter total time to first fixation (TTtFF), the time from stimulus onset until the first fixation, was sampled with the pupil center corneal reflection method and extracted after manually tracing the main object called area of interest (AOI) aka CUE, in each image. Everything outside the AOI would be then considered to be the context. This procedure allowed us to obtain separate values for cues and contexts. Eye-tracking data of three (PTSD = 19/NPTSD = 18) and of four participants in the HC group (HC = 16) could not be used because of data acquisition issues. For details on used hardware and software please refer to the “Eye-tracking data acquisition” paragraph in the Method section of the Supplement.
Memory retrieval
Correct responses for each of the five memory categories (identical, new/new, new/old, old/new, and old/old) were summed and averaged to obtain a single score per category and subject. These categories were analyzed separately for traumatic cues in neutral contexts and non-traumatic cues in neutral contexts. Given the limited sample size, we sought to avoid analyzing each category in isolation and instead applied principal component analysis (PCA) as a data reduction method. The PCA identified two distinct components that corresponded to the different memory processes required by the task. The first component, item memory (consistent cue-context pairs), loaded on the identical and new/new categories, representing memory for pictures where the cue-context association remained unchanged. Recognition of these pairs was likely supported by familiarity-based retrieval, allowing participants to judge whether an image had been encountered before as a whole rather than explicitly evaluating the relationship between its components. The second component, association memory (rearranged cue-context pairs), loaded on the new/old, old/new, and old/old categories, which required participants to detect and resolve recombined cue-context associations. Accurate responses in these conditions relied on effortful associative retrieval, as participants had to determine whether the cue and context had been previously presented together or in a different configuration39,77.
This factor structure provided a clear, data-driven distinction between item-based and association-based retrieval and was consistent with theoretical models of memory function in PTSD. Based on this, we then computed an average of the category-specific adjusted variables to obtain a mean value representing memory retrieval for the item (consistent pair) and one for the association (rearranged pair) (see also Table 3 below). In addition to analyzing correct response rates, we calculated an adjusted measure of memory accuracy by incorporating false alarm rates, similar to prior work on associative recognition. Hit rates were defined as the proportion of correct responses per memory category, while false alarms were calculated as instances where participants misclassified a given condition as another. For each category, we subtracted the category-specific false alarm rate from the hit rate, resulting in an adjusted accuracy score that accounts for response bias. These adjusted scores were then subjected to the same principal component analysis (PCA) used for correct response scores, confirming that the two-factor structure distinguishing item memory (consistent pairs: identical, new/new) from association memory (rearranged pairs: new/old, old/new, old/old) remained stable. Based on this, we then computed an average of the category-specific adjusted variables to obtain a mean value representing memory retrieval for item memory (consistent pairs) and one for association memory (rearranged pairs).
Because of technical reasons we had to exclude memory data of two participants (PTSD = 19/NPTSD = 19).
Statistical analysis
Statistical analyses were performed in IBM SPSS Statistics for Windows version 20.0.
Data not normally distributed (as assessed through Shapiro-Wilks test) were logarithmically transformed (base 10) to achieve normality. Outliers which had studentized residual values ± 2.8 SD (N = 3; one in the memory category association, two in the TTtFF of cue and context), were replaced by the mean. The statistical significance level was set to p < .05.
For the EEG data, a mixed ANOVA was used with hemisphere (left, right) as the within-subject factor and group (PTSD, NPTSD) and for the nontraumatic picture group (PTSD, NPTSD, HC) as the between factor to test for differences in the C1 mean amplitudes. For the eye-tracking data, a mixed ANOVA was applied to the total time to first fixation data (TTtFF; see the Methods section of the Supplement), with cue/context (i.e. inside and outside the AOIs) values as the within-subject factor and either trauma-exposed groups (PTSD, NPTSD) or all groups (PTSD, NPTSD, HC) as the between-subject factor.
A mixed ANOVA was applied to the memory data with item and association values as the within-subject factor and trauma-exposed groups (PTSD, NPTSD) for the traumatic stimuli and all groups (PTSD, NPTSD, HC) for the non-traumatic stimuli as the between-subject factor.
Multiple hierarchical regressions
To assess whether early processing of traumatic cues and neutral contexts has an effect on the strength of memory retrieval, we employed two multiple hierarchical regressions. In one regression model, we used the correct memory scores for the category association as the dependent variable, and in the second model, the correct memory scores for item were predicted. We did not repeat this analysis for the non-traumatic stimuli since no significant differences in memory accuracy between the groups were found.
Control analyses
Finally, to control for the effect of major comorbidities, all analyses were subsequently also performed using anxiety (STAI scores) and depression (ADS scores) as covariates in a general linear and regression models. As the results did not significantly differ, they are not reported here (see the Results section in the Supplement).
Data availability
The datasets analyzed during the current study are not publicly available due to ethical concerns but are available from the corresponding author on reasonable request.
References
American Psychiatric Association. In Diagnostic and Statistical Manual of Mental Disorders 5th edn (American Psychiatric, 2013).
Liberzon, I. & Abelson, J. L. Context processing and the neurobiology of post-traumatic stress disorder. Neuron 92, 14–30. https://doi.org/10.1016/j.neuron.2016.09.039 (2016).
Maddox, S. A., Hartmann, J., Ross, R. A. & Ressler, K. J. Deconstructing the gestalt: Mechanisms of fear, threat, and trauma memory encoding. Neuron 102, 60–74. https://doi.org/10.1016/j.neuron.2019.03.017 (2019).
Acheson, D. T., Gresack, J. E. & Risbrough, V. B. Hippocampal dysfunction effects on context memory: Possible etiology for posttraumatic stress disorder. Neuropharmacology 62, 674–685. https://doi.org/10.1016/j.neuropharm.2011.04.029 (2012).
Wessa, M., Jatzko, A. & Flor, H. Retrieval and emotional processing of traumatic memories in posttraumatic stress disorder: Peripheral and central correlates. Neuropsychologia 44, 1683–1696. https://doi.org/10.1016/j.neuropsychologia.2006.03.024 (2006).
Jovanovic, T., Kazama, A., Bachevalier, J. & Davis, M. Impaired safety signal learning May be a biomarker of PTSD. Neuropharmacology 62, 695–704. https://doi.org/10.1016/j.neuropharm.2011.02.023 (2012).
Van der Kolk, B. A. The assessment and treatment of complex PTSD. Treat. Trauma. Survivors PTSD. 127, 156 (2002).
Van der Kolk, B. A. Trauma and memory. J. Neuropsychiatry Clin. Neurosci. 52, S52–S64 (1998).
Harvey, M. R. & Herman, J. L. Amnesia, partial amnesia, and delayed recall among adult survivors of childhood trauma. Conscious. Cogn. 3, 295–306 (1994).
Chu, J. A., Frey, L. M., Ganzel, B. L. & Matthews, J. A. Memories of childhood abuse: Dissociation, amnesia, and corroboration. Am. J. Psychiatry. 156, 749–755. https://doi.org/10.1176/ajp.156.5.749 (1999).
Brewin, C. R., Gregory, J. D., Lipton, M. & Burgess, N. Intrusive images in psychological disorders: Characteristics, neural mechanisms, and treatment implications. Psychol. Rev. 117, 210–232. https://doi.org/10.1037/a0018113 (2010).
Wolf, M. R. & Nochajski, T. H. Black holes’ in memory: Childhood autobiographical memory loss in adult survivors of child sexual abuse. Eur. J. Trauma. Dissociation. 6, 100234 (2022).
Kearney, B. E. & Lanius, R. A. Why reliving is not remembering and the unique Neurobiological representation of traumatic memory. Nat. Mental Health. 2, 1142–1151 (2024).
Ehlers, A. & Clark, D. M. A cognitive model of posttraumatic stress disorder. Behav. Res. Ther. 38, 319–345 (2000).
Joshi, S. A., Duval, E. R., Kubat, B. & Liberzon, I. A review of hippocampal activation in post-traumatic stress disorder. Psychophysiology e13357 https://doi.org/10.1111/psyp.13357 (2019).
Steiger, F., Nees, F., Wicking, M., Lang, S. & Flor, H. Behavioral and central correlates of contextual fear learning and contextual modulation of cued fear in posttraumatic stress disorder. Int. J. Psychophysiol. 98, 584–593. https://doi.org/10.1016/j.ijpsycho.2015.06.009 (2015).
Flor, H. & Wessa, M. Memory and posttraumatic stress disorder: A matter of context? Z. Für Psychologie/J. Psychol. 218, 61–63. https://doi.org/10.1027/0044-3409/a000012 (2010).
Cacciaglia, R., Pohlack, S. T., Flor, H. & Nees, F. Dissociable roles for hippocampal and amygdalar volume in human fear conditioning. Brain Struct. Function. 220, 2575–2586. https://doi.org/10.1007/s00429-014-0807-8 (2015).
Rudy, J. W. Context representations, context functions, and the parahippocampal-hippocampal system. Learn. Mem. 16, 573–585. https://doi.org/10.1101/lm.1494409 (2009).
Bisby, J. A., Horner, A. J., Horlyck, L. D. & Burgess, N. Opposing effects of negative emotion on amygdalar and hippocampal memory for items and associations. Soc. Cognit. Affect. Neurosci. 11, 981–990. https://doi.org/10.1093/scan/nsw028 (2016).
Basso, M. A., Bickford, M. E. & Cang, J. Unraveling circuits of visual perception and cognition through the superior colliculus. Neuron 109, 918–937. https://doi.org/10.1016/j.neuron.2021.01.013 (2021).
Terpou, B. A. et al. The threatful self: Midbrain functional connectivity to cortical midline and parietal regions during subliminal Trauma-Related processing in PTSD. Chronic Stress (Thousand Oaks). 3, 2470547019871369. https://doi.org/10.1177/2470547019871369 (2019).
Clancy, K., Ding, M., Bernat, E., Schmidt, N. B. & Li, W. Restless ‘rest’: Intrinsic sensory hyperactivity and disinhibition in post-traumatic stress disorder. Brain J. Neurol. 140, 2041–2050. https://doi.org/10.1093/brain/awx116 (2017).
Clancy, K. et al. (ed, J.) Posttraumatic stress disorder is associated with alpha dysrhythmia across the visual cortex and the default mode network. eNeuro 7 https://doi.org/10.1523/ENEURO.0053-20.2020 (2020).
Clancy, K. J. et al. Multimodal associations between posterior hippocampus glutamate metabolism, visual cortex connectivity, and intrusive trauma reexperiencing symptoms. MedRxiv https://doi.org/10.1101/2025.01.27.25320595 (2025).
Kearney, B. E. et al. How the body remembers: Examining the default mode and sensorimotor networks during moral injury autobiographical memory retrieval in PTSD. Neuroimage Clin. 38, 103426. https://doi.org/10.1016/j.nicl.2023.103426 (2023).
Harnett, N. G. et al. Structural covariance of the ventral visual stream predicts posttraumatic intrusion and nightmare symptoms: A multivariate data fusion analysis. Transl Psychiatry. 12, 321. https://doi.org/10.1038/s41398-022-02085-8 (2022).
Harnett, N. G. et al. Acute posttraumatic symptoms are associated with multimodal neuroimaging structural covariance patterns: A possible role for the neural substrates of visual processing in posttraumatic stress disorder. Biol. Psychiatry Cogn. Neurosci. Neuroimaging. 7, 129–138. https://doi.org/10.1016/j.bpsc.2020.07.019 (2022).
Herz, N. et al. Neuromodulation of visual cortex reduces the intensity of intrusive memories. Cereb. Cortex. 32, 408–417. https://doi.org/10.1093/cercor/bhab217 (2022).
Kelly, S. P., Gomez-Ramirez, M. & Foxe, J. J. Spatial attention modulates initial afferent activity in human primary visual cortex. Cereb. Cortex. 18, 2629–2636. https://doi.org/10.1093/cercor/bhn022 (2008).
Rauss, K. S., Pourtois, G., Vuilleumier, P. & Schwartz, S. Attentional load modifies early activity in human primary visual cortex. Hum. Brain Mapp. 30, 1723–1733. https://doi.org/10.1002/hbm.20636 (2009).
Clark, V. P., Fan, S. & Hillyard, S. A. Identification of early visual evoked potential generators by retinotopic and topographic analyses. Hum. Brain. Mapp. 2, 170–187. https://doi.org/10.1002/hbm.460020306 (1994).
Jeffreys, D. A. & Axford, J. G. Source locations of pattern-specific components of human visual evoked potentials. I. Component of striate cortical origin. Exp. Brain Res. 16, 1–21 (1972).
Armstrong, T. & Olatunji, B. O. Eye tracking of attention in the affective disorders: A meta-analytic review and synthesis. Clin. Psychol. Rev. 32, 704–723. https://doi.org/10.1016/j.cpr.2012.09.004 (2012).
Stolarova, M., Keil, A. & Moratti, S. Modulation of the C1 visual event-related component by conditioned stimuli: Evidence for sensory plasticity in early affective perception. Cereb. Cortex. 16, 876–887. https://doi.org/10.1093/cercor/bhj031 (2006).
Felmingham, K. L., Rennie, C., Manor, B. & Bryant, R. A. Eye tracking and physiological reactivity to threatening stimuli in posttraumatic stress disorder. J. Anxiety Disord. 25, 668–673. https://doi.org/10.1016/j.janxdis.2011.02.010 (2011).
Thomas, C. L., Goegan, L. D., Newman, K. R., Arndt, J. E. & Sears, C. R. Attention to threat images in individuals with clinical and subthreshold symptoms of post-traumatic stress disorder. J. Anxiety Disord. 27, 447–455. https://doi.org/10.1016/j.janxdis.2013.05.005 (2013).
Milanak, M. E., Judah, M. R., Berenbaum, H., Kramer, A. F. & Neider, M. PTSD symptoms and overt attention to contextualized emotional faces: Evidence from eye tracking. Psychiatry Res. 269, 408–413. https://doi.org/10.1016/j.psychres.2018.08.102 (2018).
Yonelinas, A. P. The nature of recollection and familiarity: A review of 30 years of research. J. Mem. Lang. 46, 441–517. https://doi.org/10.1006/jmla.2002.2864 (2002).
Brewin, C. R. The nature and significance of memory disturbance in posttraumatic stress disorder. Annu. Rev. Clin. Psychol. 7, 203–227. https://doi.org/10.1146/annurev-clinpsy-032210-104544 (2011).
Milne, E., Scope, A., Pascalis, O., Buckley, D. & Makeig, S. Independent component analysis reveals atypical electroencephalographic activity during visual perception in individuals with autism. Biol. Psychiatry. 65, 22–30. https://doi.org/10.1016/j.biopsych.2008.07.017 (2009).
Rissling, A. J. et al. Cortical substrates and functional correlates of auditory deviance processing deficits in schizophrenia. Neuroimage Clin. 6, 424–437. https://doi.org/10.1016/j.nicl.2014.09.006 (2014).
Woodman, G. F. A brief introduction to the use of event-related potentials in studies of perception and attention. Atten. Percept. Psychophys. 72, 2031–2046. https://doi.org/10.3758/APP.72.8.2031 (2010).
Di Russo, F. et al. Spatiotemporal brain mapping of Spatial attention effects on pattern-reversal erps. Hum. Brain Mapp. 33, 1334–1351. https://doi.org/10.1002/hbm.21285 (2012).
Bayer, M. et al. Independent effects of motivation and Spatial attention in the human visual cortex. Soc. Cognit. Affect. Neurosci. 12, 146–156. https://doi.org/10.1093/scan/nsw162 (2017).
West, G. L., Anderson, A. A., Ferber, S. & Pratt, J. Electrophysiological evidence for biased competition in V1 for fear expressions. J. Cogn. Neurosci. 23, 3410–3418. https://doi.org/10.1162/jocn.2011.21605 (2011).
Rossi, V. & Pourtois, G. State-dependent attention modulation of human primary visual cortex: a high density ERP study. NeuroImage 60, 2365–2378. https://doi.org/10.1016/j.neuroimage.2012.02.007 (2012).
Yoneda, K., Sekimoto, S., Yumoto, M. & Sugishita, M. The early component of the visual evoked magnetic field. Neuroreport 6, 797–800 (1995).
Braeutigam, S., Bailey, A. J. & Swithenby, S. J. Task-dependent early latency (30–60 ms) visual processing of human faces and other objects. Neuroreport 12, 1531–1536. https://doi.org/10.1097/00001756-200105250-00046 (2001).
Di Russo, F., Martinez, A., Sereno, M. I., Pitzalis, S. & Hillyard, S. A. Cortical sources of the early components of the visual evoked potential. Hum. Brain Mapp. 15, 95–111. https://doi.org/10.1002/hbm.10010 (2002).
Qin, N., Wiens, S., Rauss, K. & Pourtois, G. Effects of selective attention on the C1 ERP component: A systematic review and meta-analysis. Psychophysiology 59, e14123. https://doi.org/10.1111/psyp.14123 (2022).
Fransson, P. & Marrelec, G. The precuneus/posterior cingulate cortex plays a pivotal role in the default mode network: Evidence from a partial correlation network analysis. NeuroImage 42, 1178–1184. https://doi.org/10.1016/j.neuroimage.2008.05.059 (2008).
Fisek, M. et al. Cortico-cortical feedback engages active dendrites in visual cortex. Nature 617, 769–776. https://doi.org/10.1038/s41586-023-06007-6 (2023).
Marques, T., Nguyen, J., Fioreze, G. & Petreanu, L. The functional organization of cortical feedback inputs to primary visual cortex. Nat. Neurosci. 21, 757–764. https://doi.org/10.1038/s41593-018-0135-z (2018).
Maunsell, J. H. & van Essen, D. C. The connections of the middle Temporal visual area (MT) and their relationship to a cortical hierarchy in the macaque monkey. J. Neurosci. Off. J. Soc. Neurosci. 3, 2563–2586. https://doi.org/10.1523/JNEUROSCI.03-12-02563.1983 (1983).
Blekic, W. et al. Eye-tracking exploration of inhibitory control in post-traumatic stress disorder: An emotional antisaccade paradigm. Eur. J. Psychotraumatol. 12, 1909281. https://doi.org/10.1080/20008198.2021.1909281 (2021).
Bar-Haim, Y., Lamy, D., Pergamin, L., Bakermans-Kranenburg, M. J. & van IJzendoorn, M. H. Threat-related attentional bias in anxious and nonanxious individuals: A meta-analytic study. Psychol. Bull. 133, 1–24. https://doi.org/10.1037/0033-2909.133.1.1 (2007).
Cisler, J. M. & Koster, E. H. Mechanisms of attentional biases towards threat in anxiety disorders: An integrative review. Clin. Psychol. Rev. 30, 203–216. https://doi.org/10.1016/j.cpr.2009.11.003 (2010).
Lazarov, A. et al. Attention to threat in posttraumatic stress disorder as indexed by eye-tracking indices: A systematic review. Psychol. Med. 49, 705–726. https://doi.org/10.1017/S0033291718002313 (2019).
Guez, J. et al. Associative memory impairment in acute stress disorder: Characteristics and time course. Psychiatry Res. 209, 479–484. https://doi.org/10.1016/j.psychres.2012.12.013 (2013).
Guez, J., Saar-Ashkenazy, R., Keha, E. & Tiferet-Dweck, C. The effect of Trier social stress test (TSST) on item and associative recognition of words and pictures in healthy participants. Front. Psychol. 7, 507. https://doi.org/10.3389/fpsyg.2016.00507 (2016).
Yadav, N., Toader, A. & Rajasethupathy, P. Beyond hippocampus: Thalamic and prefrontal contributions to an evolving memory. Neuron 112, 1045–1059. https://doi.org/10.1016/j.neuron.2023.12.021 (2024).
Levy-Gigi, E., Sudai, E. & Bar, M. Context as a barrier: Impaired contextual processing increases the tendency to develop PTSD symptoms across repeated exposure to trauma. J. Anxiety Disord. 100, 102765. https://doi.org/10.1016/j.janxdis.2023.102765 (2023).
Bremner, J. D. Traumatic stress: Effects on the brain. Dialogues Clin. Neurosci. 8, 445–461. https://doi.org/10.31887/DCNS.2006.8.4/jbremner (2006).
Gianaros, P. J. et al. Prospective reports of chronic life stress predict decreased grey matter volume in the hippocampus. NeuroImage 35, 795–803. https://doi.org/10.1016/j.neuroimage.2006.10.045 (2007).
Schauer, M., Neuner, F. & Elbert, T. Narrative Exposure Therapy: A short-term Treatment for Traumatic Stress Disorders (Hogrefe Publishing GmbH, 2011).
Corrigan, F. M., Young, H. & Christie-Sands, J. Deep Brain Reorienting: Understanding the Neuroscience of Trauma, Attachment Wounding, and DBR Psychotherapy (Taylor & Francis, 2024).
Wittchen, H. U., Gruschwitz, W. U. & Zaudig, S. M. SKID. Strukturiertes Klinisches Interview Für DSM-IV. Achse I: Psychische Störungen [Structured Clinical Interview for DSM-IV. Axis I: Mental Disorders] (Hogrefe, 1997).
Fydrich, T. et al. SKID II. Strukturiertes Klinisches Interview für DSM-IV, Achse II: Persönlichkeitsstörungen. Interviewheft. Eine deutschspeachige, erw. Bearb. d. amerikanischen Originalversion d. SKID-II von: M.B. First, M. Gibbon, J.B.W. Williams, L. Benjamin, (Version 3/96). (Hogrefe, Göttingen,). (1997).
Schnyder, U. & Moergeli, H. German version of Clinician-Administered PTSD scale. J. Trauma. Stress. 15, 487–492. https://doi.org/10.1023/A:1020922023090 (2002).
Foa, E. B. et al. Psychometric properties of the posttraumatic diagnostic scale for DSM-5 (PDS-5). Psychol. Assess. 28, 1166–1171. https://doi.org/10.1037/pas0000258 (2016).
Meyer, T. D. & Hautzinger, M. Allgemeine Depressions-Skala (ADS). Normierung an minderjährigen und erweiterung Zur erfassung Manischer symptome (ADMS). [Center for Epidemiological Studies—Depression Scale (CES-D). Norms for adolescents and extension for the assessment of manic symptoms]. Diagnostica 47, 208–215. https://doi.org/10.1026/0012-1924.47.4.208 (2001).
Laux, L., Glanzmann, P., Schaffner, P. & Spielberger, C. D. Das state-trait-angst Inventar [German state-trait Anxiety Inventory] (Hogrefe, 1981).
Delorme, A. & Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods. 134, 9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009 (2004).
Weymar, M., Keil, A. & Hamm, A. O. Timing the fearful brain: Unspecific hypervigilance and Spatial attention in early visual perception. Soc. Cognit. Affect. Neurosci. 9, 723–729. https://doi.org/10.1093/scan/nst044 (2014).
Foxe, J. J. & Simpson, G. V. Flow of activation from V1 to frontal cortex in humans. A framework for defining early visual processing. Exp. Brain Res. 142, 139–150. https://doi.org/10.1007/s00221-001-0906-7 (2002).
Ozubko, J. D., Moscovitch, M. & Winocur, G. The influence of recollection and familiarity in the formation and updating of associative representations. Learn. Mem. 24, 298–309. https://doi.org/10.1101/lm.045005.117 (2017).
Acknowledgements
This work was funded by a grant from the Deutsche Forschungsgemeinschaft (SFB636/C1 to HF) and a grant from the Deutsche Forschungsgemeinschaft (SFB1158/B03 to HF and FN). We thank Sophie Dautricourt and Andrea Vitale for their help in the piloting phases of the study. We thank Dr. Sebastian Pohlack and Christina Sundermann for help in data acquisition and quality checks. We thank Laura Zidda for graphically redesigning the pictures in the trauma categories and Dr. Daniel Wagner for providing the pictures for illustration purpose.
Author information
Authors and Affiliations
Contributions
H.F. organized the research project and conceived the experiment, H.F., F.N. and F.Z. designed the experiment, F.Z. conducted the experiment, analyzed the data and wrote the main manuscript text, M.R. carried out the implementation of the design, F.S.W. and T.W. coordinated and conducted recruitment and clinical assessment of the participants. J.A. provided feedback, supported analyses. All authors gave input and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zidda, F., Steiger-White, F., Winkelmann, T. et al. Early processing of traumatic material and contextual information in posttraumatic stress disorder and its relation to memory impairments. Sci Rep 15, 16362 (2025). https://doi.org/10.1038/s41598-025-00322-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00322-w