Abstract
Ubiquitin-specific peptidase 18 (USP18) is a specific interferon-stimulated gene 15 demodifying enzyme that plays an important role in apoptosis. In this study, we investigated the role of USP18 in apoptosis in hepatocellular carcinoma cells, especially its ability to regulate apoptosis through endoplasmic reticulum (ER) stress. We found that protein levels of Bcl-2-associated protein x and cytochrome c were down-regulated by USP18, which suppressed the classical mitochondrial-mediated apoptosis pathway. USP18 also inhibited apoptosis through the unfolded protein response (UPR) pathway by inhibiting the phosphorylation of protein kinase RNA-like endoplasmic reticulum kinase (PERK) and the expression of CCAAT/enhance-binding protein homologous protein, which is a downstream marker molecule of ER stress. The UPR triggered by ER stress eventually led to the cleavage of downstream effecter proteases, including caspase-3, leading to apoptosis. Furthermore, USP18 combined with a PERK agonist regulated apoptosis through the PERK–eukaryotic initiation factor–2α-activating transcription factor 4 axis of the UPR. Our results show that USP18 participates in the regulation of hepatocellular carcinoma cell apoptosis through different pathways, especially the ER stress pathway, and that it plays a complex role in cell stress responses and apoptosis regulation.
Similar content being viewed by others
Introduction
As one of the major cancer types, hepatocellular carcinoma (HCC) is an important public health problem. According to GLOBOCAN 2020, HCC deaths are estimated at 830,180, accounting for 8.30% of new cancer deaths worldwide in 20201. Traditional treatments for HCC include surgical resection, liver transplantation, chemoembolization, and radiotherapy. In recent years, targeted therapy and immunotherapy have become new directions for treatment of HCC. In addition, immune checkpoint inhibitors such as PD-1/PD-L1 inhibitors have shown some efficacy in some patients with advanced liver cancer, which provides a new option for individualized treatment for HCC2. However, the effectiveness of these treatments varies from person to person, and they have certain side effects and limitations. Therefore, further study of the molecular mechanism of HCC pathogenesis and elucidation of effective treatment methods are needed for the treatment of HCC and other types of tumors.
Ubiquitin-specific peptidases (USPs) belong to the cysteine protease family and are also important members of the deubiquitinating enzyme (DUB) family. USPs can cleave monoubiquitin molecules and/or poly-ubiquitin chains of substrate proteins, affect signal transduction, and mediate proteasome-dependent protein degradation of substrate proteins3. Previous studies confirmed that USP18 has DUB activity, as it can specifically remove ubiquitin-like protein interferon-stimulated gene 15 from the substrate proteins, thus protecting the substrate proteins from degradation4. The expression of USP18 is up-regulated in many types of tumors, which results in inhibition of cell apoptosis and promotion of tumor formation5. Data from Gene Expression Profiling Interaction Analysis (http://GEPIA.cancer-pku.cn/), which is a platform based on TCGA data, showed that USP18 expression significantly increased in esophageal, pancreatic, cervical, head and neck squamous cell, and bladder cancers, which suggests that USP18 may play roles in the occurrence and development of these types of tumors6. Additionally, knockdown of USP18 inhibited the proliferation of HCC cells and induced G1 cell cycle arrest and early apoptosis. Mechanistically, USP18 can directly bind to anti-apoptotic protein BCL2L1 and up-regulate its expression, thus exerting its anti-apoptotic role7.
Based on different starting stages, apoptosis can be divided into three main pathways: mitochondrial, endoplasmic reticulum (ER), and death receptor pathways8,9,10. The ER is the main intracellular reservoir of calcium ions, and it is one of the main sites of protein synthesis in cells11. The stability of the ER internal environment is the basic condition required for ER function. However, many factors can lead to the imbalance of ER function and the formation of ER stress, including ER calcium metabolism disorders and ischemia-reperfusion injury12. ER stress is activated in a variety of cancers and is closely related to tumor growth, metastasis, and chemotherapy resistance13.
The molecular mechanism of ER stress-induced apoptosis is an active cell death process that is regulated by multiple genes. Under persistent or severe ER stress, the adaptive response of the unfolded protein response (UPR) cannot completely restore ER homeostasis, resulting in initiation of the apoptosis pathway. ER stress can induce apoptosis through multiple pathways, including the ER-specific caspase-12, CCAAT/enhance-binding protein homologous protein (CHOP), and serine/threonine-protein kinase/endoribonuclease inositol-requiring enzyme 1 alpha (IRE1α) pathways. In addition, Bcl-2-associated protein x (Bax) and Bcl-2 homologues antagonist/killer (Bak) proteins in the B-cell lymphoma-2 (Bcl-2) family can promote mitochondrial pathway-induced apoptosis by regulating the release of ER calcium14,15,16.
In a previous study, we silenced the expression of USP18 in the human hepatoblastoma cell line HepG2.2.15, which is derived from HepG2 cells and stably transfected with the hepatitis B virus genome, continuously producing HBV particles, and screened the differentially expressed genes using gene chip technology. A total of 436 differentially expressed genes were screened, including CHOP, glucose-regulated protein 78 (GRP78), and homocysteine inducible ER protein with ubiquitin like domain 1 (HERPUD1). Among them, CHOP, which is a marker molecule of ER stress, was significantly up-regulated relative to the control group after interfering with USP18 expression17. GRP78 (also known as BiP) is the molecular chaperone of the UPR and another marker molecule of ER stress. HERPUD1 is a protein expressed under ER stress, and it can alleviate ER stress by promoting the degradation of misfolded proteins. We also previously reported that the expression of GRP78 and HERPUD1 were slightly increased in HepG2.2.15 cells after interfering with USP18 expression17. However, the molecular mechanism responsible for apoptosis induced by USP18 in HCC and its relationship with ER stress remained unknown.
The goal of this study was to determine if USP18 could regulate apoptosis and ER stress in HCC cells. We assessed whether USP18 suppresses the UPR in ER stress-mediated apoptosis by inhibiting the protein kinase RNA-like endoplasmic reticulum kinase (PERK)–eukaryotic initiation factor-2α (eIF2α)–activating transcription factor 4 (ATF4) signaling pathway to enhance the proliferation of HCC cells. Our results provide a theoretical basis for the clinical application of USP18 in the treatment of liver cancer.
Results
USP18 promoted HCC cell proliferation by inhibiting apoptosis
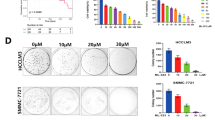
Survival analysis was carried using data from the UALCAN, KM-plotter and HPA databases. Results of survival analysis revealed that high expression of USP18 was associated with poor prognosis in HCC patients (Fig. 1a), indicating that USP18 might serve as a potential biomarker for prognosis of HCC. Subsequently, the protein expression levels of USP18 in HCC cell lines (SMMC-7721, Bel-7402, HepG2, and Huh7) and the normal liver cell line LO2 were detected by western blotting analysis (Fig. 1b). The expression of USP18 in HCC cell lines was significantly higher than that in LO2 cells, suggesting that its expression may be related to the development of HCC. Huh7 and SMMC7721 cells were selected for further study, as these two cell lines expressed high levels of USP18.
USP18 regulated the proliferation of HCC cells. (a) Correlation between expression level of USP18 and overall survival of HCC patients from UALCAN, KM-plotter and HPA databases, n = number of samples. (b) Expression levels of USP18 protein and mRNA in HCC cell lines were significantly higher than that in normal liver cells. (c, d) Huh7 and SMMC7721 cells were transfected with siRNAs or overexpression plasmids, respectively. Protein expression of USP18 was detected by western blot. (e, f) Overexpression or knockdown of USP18 regulated cell proliferation of Huh7 and SMMC7721 cells, as shown in cell viability assays using the Cell Counting Kit-8. Data are presented as the mean ± SD. Statistical analyses were performed with one-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001, ns = not significant.
To evaluate the biological functions of USP18 in the proliferation of HCC cells, the expression of USP18 in Huh7 and SMMC7721 cells was first silenced with the knockdown fragment siUSP18s (siUSP18-1, siUSP18-2, siUSP18-3) and then analyzed at protein level. The results showed that siUSP18-1 had the highest knockdown efficiency in both cell types (Fig. 1c), so it was selected for subsequent experiments. Transfection of the USP18 over-expression plasmid vector significantly increased the expression of USP18 in both cell lines relative to the control (Fig. 1d). Next, a cell viability assay was performed to test whether suppression of USP18 significantly inhibited cell proliferation in both Huh7 and SMMC7721 cells (Fig. 1e). Overexpression of USP18 significantly promoted cell proliferation in both cell types (Fig. 1f). Taken together, these results demonstrate that USP18 plays a role in promoting HCC cell proliferation in vitro.
Using flow cytometry analysis, we assessed whether the regulation of USP18 could affect cell apoptosis in Huh7 and SMMC7721 cells (Fig. 2a, b). Results showed that overexpression of USP18 attenuated apoptosis, whereas USP18 knockdown augmented apoptosis. Considering that USP18 has an effect on proliferation and apoptosis of HCC cell lines, we next assessed the effect of regulation of USP18 expression levels on apoptosis-related proteins in the classical mitochondrial pathway. The results of western blotting showed that the expression of anti-apoptotic protein Bcl-2 decreased and the expression of pro-apoptotic proteins Bax, Cyto-C, and cleaved-caspase-3 increased after knockdown of USP18 in Huh7 and SMMC-7721 cells (Fig. 2c). In contrast, overexpression of USP18 could up-regulated the expression of Bcl-2 and down-regulated the expression of Bax, Cyto-C, and cleaved-caspase-3 in Huh7 and SMMC-7721 cells (Fig. 2d). These results suggest that USP18 can promote the continuous proliferation of HCC cells by affecting apoptosis.
USP18 regulated apoptosis of HCC cells. (a, b) Flow cytometry analysis showed that expression change of USP18 affected apoptosis of Huh7 and SMMC7721 cells. (c, d) Western blotting results showed that regulation of USP18 expression levels affected apoptosis-related proteins in the classical mitochondrial pathway in Huh7 and SMMC7721 cells. Data are presented as the mean ± SD. Statistical analyses were performed with one-way ANOVA. *P < 0.05, ***P < 0.001.
USP18 inhibited apoptosis via ER stress
Because apoptosis can be realized not only through the mitochondrial pathway but also through ER stress, we next validated the role USP18 in regulating ER stress in Huh7 cells. The results of flow cytometry showed that knockdown of USP18 promoted apoptosis, the ER stress inhibitor 4-phenylbutyric acid (4-PBA) inhibited apoptosis, and knockdown of USP18 partially reversed the inhibitory effect of 4-PBA on apoptosis (Fig. 3a). Reciprocally, the ER stress agonist tunicamycin (TM) promoted apoptosis and overexpression of USP18 partially reversed the effect of TM on apoptosis (Fig. 3a).
USP18 inhibited apoptosis via ER stress. Huh7 cells were treated with 5 mM ER stress inhibitor 4-PBA or 3 µM ER stress agonist TM, respectively. The final concentration of DMSO were 0.05% and 0.03% in vehicle control groups, respectively. (a) Flow cytometry analysis showed that apoptosis in Huh7 cells was regulated by expression change of USP18, ER stress agonist, or ER stress inhibitor. (b) Western blotting analysis showing levels of apoptosis-related protein cleaved-caspase-3 and ER stress-related proteins CHOP and GRP78 in Huh7 cells. (c) The fluorescence intensity of CHOP was detected by immunofluorescence assay. USP18 in red, CHOP in green, DAPI nuclear staining in blue. The representative images were acquired under × 200 magnification. Data are presented as the mean ± SD. Statistical analyses were performed with one-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001.
Western blotting analysis showed that knockdown or overexpression of USP18 in Huh7 cells had no significant effect on the expression of GRP78. Knockdown of USP18 increased the expression of ER stress-related protein CHOP and apoptosis-related proteins cleaved-caspase-3. In contrast, 4-PBA had an inhibitory effect on CHOP and cleaved-caspase-3, while knockdown of USP18 partially reversed the effects of 4-PBA on ER stress and apoptosis (Fig. 3b). Overexpression of USP18 inhibited the expressions of CHOP and cleaved-caspase-3. TM promoted the expression of CHOP and cleaved-caspase-3, which was partly reversed by overexpression of USP18 (Fig. 3c).
The results of cellular immunofluorescence analysis showed that USP18 was strongly expressed in the USP18 overexpression group and the USP18 overexpression + TM group but weakly expressed in the siUSP18-1 group and the siUSP18-1 + 4-PBA group. CHOP was more strongly expressed in the siUSP18-1 group and the over-control vector + TM group. Compared with the control group, CHOP was weakly expressed in the si-control + 4-PBA group and the USP18 overexpression group (Fig. 3d). These results suggest that USP18 can regulate apoptosis and ER stress through the GRP78/CHOP signaling pathway.
USP18 May regulate apoptosis through PERK-eIF2α-ATF4 signaling
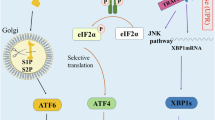
USP18 overexpression combined with TM was used to treat Huh7 cells, and the levels of proteins in the UPR signaling pathway (p-IRE1α/IRE1α, ATF6, p-PERK/PERK, and XBP-1s) were measured using western blotting. Results showed that the overexpression of USP18 promoted the expression of p-IRE1α and inhibited the level of p-PERK but had no significant effect on ATF6, XBP-1s, and IRE1α expression (Fig. 4a). These results indicate that USP18 might regulate apoptosis through the UPR signaling pathway, possibly via the PERK singling pathway. Furthermore, the combination of the PERK agonist CCT020312 with plasmid effectively increased the levels of ER stress-related proteins, including p-PERK, ATF4, p-eIF2α, and CHOP, in Huh7 cells (Fig. 4b). These results indicate that USP18 may regulate apoptosis through PERK-eIF2α-ATF4 signaling (Fig. 5).
USP18 regulates ER stress and PERK-eIF2α-ATF4 signaling in Huh7 cells. (a) Levels of ER stress-related proteins in Huh7 cells were detected by western blotting. Cells were treated with 3 µM ER stress agonist TM. (b). Western blotting analysis of UPR-related proteins p-PERK, ATF4, p-eIF2α, and CHOP in Huh7 cells. Cells were treated with 2.5 µM PERK agonist CCT020312. The final concentration of DMSO is 0.025% in vehicle control group. Data are presented as the mean ± SD.
A proposed working model of apoptosis regulated by USP18 through the PERK-eIF2α-ATF4 signaling pathway. USP18 inhibits the expression of CHOP, a marker gene of ER stress, by regulating the PERK-eIF2α-ATF signaling pathway, which relieves the inhibition of downstream anti-apoptotic protein Bcl-2, thereby inhibiting cell apoptosis. USP18 can also directly affect apoptosis by regulating the classical mitochondrial-mediated apoptosis pathway. Solid arrows indicate promotion, and dashed arrows indicate inhibitory effects.
Discussion
Acting as one of the key molecules in the regulation of ubiquitination balance, USP18 regulates the stability and activity of proteins by removing ubiquitin chains from target proteins through deubiquitination activity or through interaction with ubiquitin ligase. These actions affect the homeostasis of cancer-associated proteins, such as survival signaling proteins and metabolic enzymes, and thereby affect the biological behavior of tumors. Depletion of USP18 not only induces the expression of interferon-stimulated genes, but also activates the expression of genes that induce cancer apoptosis and pyroptosis, indicating that USP18 can be used as an effective tool for cancer immunotherapy18. Because HCC cells generally express high mRNA and protein levels of USP187,19, regulation of USP18 expression may affect HCC cell growth and apoptosis in vitro. Recent studies showed that USP18 plays crucial roles in regulating the cell cycle, apoptosis, and the microenvironment of tumors20,21. And the data from TCGA also showed that high expression level of USP18 is associated with poor overall survival in HCC patients, indicating that USP18 might serve as a prognostic marker for overall survival in HCC. Nevertheless, whether USP18 affects the apoptosis of HCC cells and the underlying mechanism for such an effect were previously unknown.
In this study, we found that USP18 promoted the growth of HCC cell lines and suppressed apoptosis. These actions were followed by up-regulation of the anti-apoptotic protein Bcl-2 and down-regulation of the pro-apoptotic protein Bax, which in turn led to inhibition of the downstream apoptotic-inducing executive protein caspase-3. Therefore, the results demonstrated that USP18 plays an oncogenic role in HCC by regulating the classical mitochondrial signaling pathway to reduce apoptosis.
As members of the DUB subfamily of proteases, USPs acts on ubiquitinated proteins in the ubiquitin-proteasome system and can regulate cell growth, cell cycle, and signal transduction. For example, USP20 can specifically cleave K48- and K63-linked ubiquitin chains, thereby stabilizing the substrate and promoting reticulophagy, which is a selective process through which impaired ER subdomains are removed via autophagy-mediated lysosomal degradation22. Studies have shown that the ER stress-PERK axis is responsible for chemoresistance to a USP7 inhibitor that has been found to have anticancer efficacies in in vitro and in vivo models. Additionally, inhibition of PERK may be a potential strategy to improve the anticancer efficacy of USP7 inhibitors23.
Previously, USP18 was reported to regulate apoptosis by regulating the stability of 14-3-3ζ in lung cancer cells24 and to promote the proliferation and migration of ovarian cancer cells by activating protein kinase B/mammalian target of rapamycin (AKT/mTOR) signaling25. The chemotherapy drug bortezomib was reported to elicit cellular stress and extrinsic apoptotic pathway, which can be repressed in cells overexpressing USP1826. USP18 has also been reported to affect protein folding and stability by removing interferon-stimulated gene 15 modifications, thus indirectly affecting the processing capacity of ER stress27. Thus, the correlation between USP18 and ER stress is mainly reflected in its regulation of interferon signal transduction. USP18 can also reduce intracellular inflammation and stress signaling by inhibiting the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway, which predisposes cells to ER stress and potentially affects CHOP expression28.
In our previous study, knockdown of USP18 in HepG2.2.15 cells significantly altered gene expression associated with PERK-mediated UPR17. Although we lacked direct evidence of the interaction between USP18 and ER stress, we speculated that USP18 might affect ER stress indirectly through its roles in regulating protein stability, type I interferons, and apoptosis. In the present study, we demonstrated that combining an ER-stress inhibitor or agonist with vectors that regulate USP18 expression synergistically affected HCC cell apoptosis in vitro. USP18 attenuated the expression of CHOP, suggesting that USP18 has an effect on UPR signaling pathway. Further assessment showed that USP18 promoted the expression of ATF6 and p-IRE1α and inhibited the level of p-PERK but had no significant effect on XBP-1s. However, activation of PERK effectively increased the levels of ER stress-related proteins, including p-PERK, ATF4, p-eIF2α, and CHOP, which could be reversed by overexpression of USP18. These results indicate that USP18 may regulate apoptosis through PERK-eIF2α-ATF4 signaling. However, there are still some defects in our study. For example, the proteins that interact with USP18 and the way in which USP18 regulates the level of p-PERK should be explored in depth.
Conclusions
In conclusion, the results of this study provide evidence that USP18 promotes growth and suppresses apoptosis of HCC cells, which might help to expand the research directions and clinical applications of USP18. Our mechanistic studies showed that USP18 not only regulates apoptosis via the classical mitochondrial pathway, but also via the ER stress-mediated UPR, especially the PERK-CHOP axis. Because USP18 regulates apoptosis through these pathways to maintain the balance of the intracellular environment, it is may be a new target for regulating the progression of HCC.
Materials and methods
Cell culture and transfection
The human HCC cell lines (SMMC-7721, Bel-7402, HepG2, and Huh7) and normal liver cell line LO2 used in this study were purchased from Pricella (Wuhan, China) and preserved in the Key Laboratory of Chinese Medicine for Prevention and Treatment of Autoimmune Diseases, Chongqing Hospital of Traditional Chinese Medicine, China. HCC cell lines SMMC-7721, Bel-7402 and normal liver cell line LO2 were cultured in 1640 medium (Gibco, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco) and 1% penicillin/streptomycin at 37˚C in a 5% CO2 incubator until cells reached 90% confluence. HCC cell lines HepG2 and Huh7 were cultured in Dulbecco’s modified Eagle’s medium (Gibco) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin at 37 °C in a 5% CO2 incubator until cells reached 80% confluence.
To evaluate the biological functions of USP18 in the proliferation of HCC cells, Huh7 and SMMC-7721 cells were subsequently seeded in 6-well plates (2 × 105 per well for Huh7 and 5 × 105 per well for SMMC-7721) and incubated for 24 h prior to transduction. Diluted small interfering RNAs (si-USP18-1 (Target sequence CCAGGGAGTTATCAAGCAA), si-USP18-2 (Target sequence CATCCGGAATGCTGTGGAT), si-USP18-3 (Target sequence GAAGCGAGAGTCTTGTGAT), and nonspecific control siRNA (si-Control, Genemine Bio-Tech Co., Ltd., Chongqing, China)), or plasmids (over-Control and over-USP18, Genemine Bio-Tech Co., Ltd., Chongqing, China)), and appropriate amount of lipofectamine 2000 (Thermo FisherScientific, Waltham, MA, USA) were added with a dilution ratio of 1:2. Cells were incubated for 6 h, after which the medium containing siRNAs or plasmids was removed and replaced with fresh complete medium. After 24–72 h of transfection, cells were harvested for further analysis. For the signal pathway regulation experiment, Huh7 cells were treated with the ER stress inhibitor 4-PBA (MedChemExpress, Monmouth Junction, NJ, USA), the ER stress agonist TM (Macklin Biotech Co., Ltd., Shanghai, China), the PERK agonist CCT020312 (MedChemExpress) or the vehicle control dimethyl sulfoxide (DMSO) for 24 h, respectively.
Cell viability
To assess cell viability, Huh7 and SMMC-7721 cells were seeded on 96-well plates, respectively. The original medium was discarded at 1, 2, 3, and 4 days after inoculation, and 100 µL of fresh medium were added to each well. Next, 10 µL of Cell Counting Kit-8 solution (Dojindo, Kumamoto, Japan) was added to each well, and the mixture was incubated in the dark at 37 °C for 2 h. The absorbance value at 450 nm was measured by enzyme-linked immunosorbent assay (Bio-tek ELX800, Winooski, VT, USA), and the growth curves of HCC cells were plotted.
UALCAN, HPA and Kaplan-Meier plotter databases
The University of ALabama at Birmingham Cancer data analysis Portal (UALCAN, https://ualcan.path.uab.edu/) is a comprehensive, user-friendly, and interactive web resource for analyzing cancer omics data29. Kaplan-Meier survival analysis was carried out to explore the association between USP18 expression and HCC patient outcomes using UALCAN. The Human Protein Atlas (HPA) (https://www.proteinatlas.org)30 generated by The Cancer Genome Atlas (TCGA) was used to determine the correlation between USP18 expression and overall survival of HCC patients. Kaplan-Meier Plotter (KM-plotter) is the most sophisticated online survival analysis tool that performs all calculations on gene expression data, relapse free and overall survival information using data downloaded from GEO, EGA and TCGA31. All data were anonymized, and their use complies with the access policies and ethical guidelines of the databases.
Quantitative real-time PCR
Total RNA from the five cell lines was isolated using TRIzol reagent (Invitrogen, Waltham, MA, USA) and further purified using an RNeasy Mini kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s protocol. The concentration and quality of RNA were verified by spectrophotometry (NanoDrop 2000; Thermo Fisher Scientific). cDNA was synthesized using a PrimeScript™ RT Reagent kit (Takara Biotechnology, China) following the manufacturer’s protocol. Quantitative PCR was performed using a LightCycler system (RocheDiagnostics, Basel, Switzerland) in a volume of 10 µL with the SYBR Premix Ex Taq™ II kit (Takara Biotechnology). The genespecific primers were purchased from Sangon Biotech Co., Ltd. (Shanghai, China), and the sequences of PCR primers were as follows: USP18 forward primer 5′-ATGCTCACCCTCCCACTTTC-3′ and reverse primer 5′-GTCTTCTTCCCACAGTTCTCACAG-3′ and β-actin forward primer 5′-ACCCCGTGCTGCTGACCGAG-3′ and reverse primer 5′-TCCCGGCCAGCCAGGTCCA-3’. The thermocycling conditions were as follows: 95 °C for 10 min followed by 40 cycles at 95 °C for 15 s and 57 °C for 1 min. Gene expression was analyzed using the comparative 2–ΔΔCq method and normalized to the internal reference gene β-actin. All experiments were performed in triplicate.
Flow cytometry analysis
Huh7 and SMMC-7721 cells (1–5 × 105 cells) were collected after washing three times with phosphate buffered saline (PBS). Cells were centrifuged at 300 g for 5 min, with an adjusted cell concentration of 1 × 106 cells/mL, and then they were fixed with 70% ethanol by volume fraction. Next, 100 µL of RNase A were added, and samples were place in a 37 °C water bath for 30 min and subsequently stained. The fluorescence intensity was recorded using flow cytometry detection (Agilent Technology, CA, USA).
Western blot analysis
The total cell protein was collected and extracted by adding RIPA lysis buffer [50 mM Tris (pH 7.4), 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS] and protease and phosphatase inhibitor cocktail. The protein concentration in Huh7 and SMMC-7721 cells was determined using the bicinchoninic acid method, and samples were stored at − 80 °C. Protein was added to sample buffer and boiled at 95 °C for 10 min. The protein was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. Next, the cells were blocked with 5% bovine serum albumin for 2 h at room temperature and then incubated overnight with rabbit polyclonal antibodies against USP18 (ABclonal Biotechnology, MA, USA), ATF6 (ABclonal Biotechnology), X-box binding protein 1 (XBP-1s) (ABclonal Biotechnology), Bcl-2 (Affinity Biosciences, OH, USA), Bax (Affinity Biosciences), cleaved-caspase3 (Affinity Biosciences), cytochrome c (Cyto-C, Affinity Biosciences), phospho-IRE1α (p-IRE1α, Affinity Biosciences), IRE1α (Affinity Biosciences), phospho-PERK (p-PERK, Affinity Biosciences), PERK (Affinity Biosciences), phospho-eIF2α (Affinity Biosciences), and eIF2α (Affinity Biosciences), and with mouse monoclonal antibodies against caspase 3 (Proteintech Group, Hubei, China), GRP78 (Proteintech Group), CHOP (Santa Cruz Biotechnology, Shanghai, China), and β-actin (Santa Cruz Biotechnology) at 4 °C. Subsequently, the 1:1000 dilution of secondary antibody (zsbio Biotechnology, Beijing, China) was added, and the samples were incubated at room temperature for 2 h. After washing the membrane, the protein bands were detected using the enhanced chemiluminescence method, and the gray values of the bands were recorded using FUSION Gel Imager (Vilber, Collégien, France). The quantification of protein bands related to β-actin was performed using the FUSION FX imaging system (Vilber).
Immunofluorescence assay
Huh7 cells transfected with siRNA (si-Control and si-USP18-1) or plasmids (over-Control and over-USP18) were fixed with immunol staining fix solution (Beyotime, Biotechnology, Jiangsu, China) for 30 min, washed three times with cold PBS, and then permeabilized in 0.5% Triton X-100 for 20 min. After blocking in 5% goat serum (zsbio Biotechnology) for 1 h, cells were incubated at 4 °C overnight with 1:100 dilution of rabbit polyclonal antibodies against USP18 and with 1:100 dilution of mouse monoclonal antibodies against CHOP diluted in blocking solution. Cells were washed three times with cold PBS, and then goat anti-rabbit or goat anti-mouse antibodies (zsbio Biotechnology) were added for 1 h incubation at room temperature. Nuclei were stained with 4, 6-diamidino-2-phenylindole for 5 min. Cells were washed three times and then examined to determine the distribution of USP18 and CHOP using a fluorescence microscope (Mshot Photoelectric Technology, Guangdong, China). The fluorescence expression intensity of CHOP was quantified using Fiji (version 2.16.0), which was processed in 20 independent fields.
Statistical analysis
The measurement data were expressed as mean ± standard deviation (SD). Statistical analysis was carried out using GraphPad Prism (version 9.4.1), SPSS (version 23.0), and R software (version 4.2.1). Comparisons among groups were conducted using one-way analysis of variance (ANOVA), and comparisons between two groups were performed using two tailed Student’s t test. Statistical analysis was performed on three biological replicates (n = 3), each consisting of three technical replicates per condition (all data points considered), unless otherwise specified. P < 0.05 was considered to be statistically significant.
Data availability
All data generated or analysed during this study are included in this published article.
References
Sung, H. et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 71(3), 209–249 (2021).
Li, Z. et al. Tumor-associated macrophages in anti-PD-1/PD-L1 immunotherapy for hepatocellular carcinoma: recent research progress. Front. Pharmacol. 15, 1382256. https://doi.org/10.3389/fphar.2024.1382256 (2024).
Huang, X. & Dixit, V. M. Drugging the undruggables: Exploring the ubiquitin system for drug development. Cell Res. 26(4), 484–498 (2016).
Liu, X. et al. The ubiquitin-specific peptidase USP18 promotes lipolysis, fatty acid oxidation, and lung cancer growth. Mol. cancer Research: MCR. 19(4), 667–677 (2021).
Mustachio et al. Deubiquitinase USP18 loss mislocalizes and destabilizes KRAS in lung cancer. Mol. cancer Research: MCR. 15(7), 905–914 (2017).
Tang, Z. et al. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 45(W1), W98–W102 (2017).
Cai, J. et al. Downregulation of USP18 inhibits growth and induces apoptosis in hepatitis B virus-related hepatocellular carcinoma cells by suppressing BCL2L1. Exp. Cell Res. 358(2), 315–322 (2017).
Jiang, Z. et al. 13-Methylpalmatine improves myocardial infarction injury by inhibiting CHOP-mediated cross-talk between Endoplasmic reticulum and mitochondria. Biomed. Pharmacotherapy = Biomedecine Pharmacotherapie. 179, 117342. https://doi.org/10.1016/j.biopha.2024.117342 (2024).
Gadet, R. et al. The Endoplasmic reticulum pool of Bcl-xL prevents cell death through IP3R-dependent calcium release. Cell. Death Discovery. 10(1), 346 (2024).
Chen, C. H. et al. Naringin induces ROS-stimulated G1 cell-cycle arrest and apoptosis in nasopharyngeal carcinoma cells. Environ. Toxicol. 39(11), 5059–5073 (2024).
Shi, H. & Zhao, Y. Astaxanthin inhibits apoptosis in a cell model of tauopathy by attenuating endoplasmic reticulum stress and unfolded protein response. Eur. J. Pharmacol. 983, 176962. https://doi.org/10.1016/j.ejphar.2024.176962 (2024).
Shi, Y., Jiang, B. & Zhao, J. Induction mechanisms of autophagy and Endoplasmic reticulum stress in intestinal ischemia-reperfusion injury, inflammatory bowel disease, and colorectal cancer. Biomed. Pharmacotherapy = Biomedecine Pharmacotherapie 170, 115984. https://doi.org/10.1016/j.biopha.2023.115984 (2024).
Kim, T. & Croce, C. M. MicroRNA and ER stress in cancer. Sem. Cancer Biol. 75, 3–14 (2021).
Abo-Zaid, O. A., Moawed, F. S., Taha, E. F., Ahmed, E. S. A. & Kawara, R. S. Melissa officinalis extract suppresses Endoplasmic reticulum stress-induced apoptosis in the brain of hypothyroidism-induced rats exposed to γ-radiation. Cell. Stress Chaperones. 28(6), 709–720 (2023).
Bagchi, A. K., Malik, A., Akolkar, G., Jassal, D. S. & Singal, P. K. Endoplasmic reticulum stress promotes iNOS/NO and influences inflammation in the development of doxorubicin-induced cardiomyopathy. Antioxid. (Basel Switzerland). 10(12), 1897 (2021).
Du, M. et al. C/EBPα-p30 confers AML cell susceptibility to the terminal unfolded protein response and resistance to venetoclax by activating DDIT3 transcription. J. Experimental Clin. cancer Research: CR 43(1), 79 (2024).
Li, L., Lei, Q. S., Kong, L. N., Zhang, S. J. & Qin, B. Gene expression profile after knockdown of USP18 in Hepg2.2.15 cells. J. Med. Virol. 89(11), 1920–1930 (2017).
Arimoto, K. I. et al. Expansion of interferon inducible gene pool via USP18 Inhibition promotes cancer cell pyroptosis. Nat. Commun. 14(1), 251 (2023).
Tong, H. V. et al. Upregulation of enzymes involved in isgylation and ubiquitination in patients with hepatocellular carcinoma. Int. J. Med. Sci. 17(3), 347–353 (2020).
Diao, W. et al. USP18 promotes cell proliferation and suppressed apoptosis in cervical cancer cells via activating AKT signaling pathway. BMC cancer. 20(1), 741 (2020).
Jifu, C. et al. USP18 is associated with PD-L1 antitumor immunity and improved prognosis in colorectal cancer. Biomolecules 14(9), 1191 (2024).
Zhang, M. et al. USP20 deubiquitinates and stabilizes the reticulophagy receptor RETREG1/FAM134B to drive reticulophagy. Autophagy 20(8), 1780–1797 (2024).
Lim, C. H. et al. ER stress-activated HSF1 governs cancer cell resistance to USP7 inhibitor-based chemotherapy through the PERK pathway. Int. J. Mol. Sci. 25(5), 2768 (2024).
Chen, Z. et al. Loss of ubiquitin-specific peptidase 18 destabilizes 14-3-3ζ protein and represses lung cancer metastasis. Cancer Biol. Ther. 23(1), 265–280 (2022).
Liu, M. et al. USP18 contributes to the proliferation and migration of ovarian cancer cells by regulating the AKT/mTOR signaling pathway. Acta Biochim. Pol. 69(2), 417–422 (2022).
Potu, H., Sgorbissa, A. & Brancolini, C. Identification of USP18 as an important regulator of the susceptibility to IFN-alpha and drug-induced apoptosis. Cancer Res. 70(2), 655–665 (2010).
Basters, A., Knobeloch, K. P. & Fritz, G. How USP18 deals with ISG15-modified proteins: structural basis for the specificity of the protease. FEBS J. 285(6), 1024–1029 (2018).
Jiang, W., Wu, D. & Tang, B. H. Usp18 attenuates the anti-hepatitis B virus effect of Ifn by down regulating jak-stat pathway. Future Virol. 17(11) (2022).
Chandrashekar, D. S. et al. UALCAN: an update to the integrated cancer data analysis platform. Neoplasia (New York N Y). 25, 18–27 (2022).
Karlsson, M. et al. A single-cell type transcriptomics map of human tissues. Sci. Adv. 7(31), eabh2169. https://doi.org/10.1126/sciadv.abh2169 (2021).
Győrffy, B. Integrated analysis of public datasets for the discovery and validation of survival-associated genes in solid tumors. Innov. (Cambridge (Mass)). 5(3), 100625 (2024).
Funding
This study was supported by the Chongqing Medical Scientific Research Project (Joint project of Chongqing Health Commission and Science and Technology Bureau) (grant number 2022QNXM060); the Natural Science Foundation of Chongqing, China (grant number cstc2021jcyj-msxmX0390) and the National Traditional Chinese Medicine Advantage Specialty (Chongqing Municipal Finance Bureau, Chongqing Municipal Civil Affairs Bureau [2024] No.21).
Author information
Authors and Affiliations
Contributions
Lin Li, Qingsong Lei and Yi Huang wrote the main manuscript text. Pujuan Yang and Huabao Liu edited the manuscript text. Lin Li, Qingsong Lei, Yi Huang, Pujuan Yang, Nana Liu, Chunyan Rao prepared Figs. 1, 2, 3 and 4, and Lingna Kong prepared figure 5. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, L., Liu, N., Yang, P. et al. USP18 attenuates endoplasmic reticulum stress via the PERK-eIF2α-ATF4 axis to reduce apoptosis in hepatocellular carcinoma cells. Sci Rep 15, 15659 (2025). https://doi.org/10.1038/s41598-025-00540-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00540-2
Keywords
This article is cited by
-
Endoplasmic reticulum stress-mediated programmed cell death in the tumor microenvironment
Cell Death Discovery (2025)