Abstract
In this study, we conducted a comprehensive comparative analysis of the physicochemical properties of natural musk (NM) and artificial musk (AM), focusing on their ultrastructure, volatile constituents, alcohol-soluble compounds, amino acid composition, inorganic element content and anti-inflammatory activity. Advanced analytical techniques such as scanning electron microscopy (SEM), gas chromatography-mass spectrometry (GC-MS), liquid chromatography-mass spectrometry (LC-MS), amino acid auto analyzer (AAA), and inductively coupled plasma-mass spectrometry (ICP-MS) were utilized to evaluate the differences in their physicochemical properties. The results of the study showed that SEM could distinguish NM and AM based on their surface morphology. GC-MS and LC-MS results identified 15 volatile compounds and 25 alcohol-soluble compounds in NM, whereas AM contained 8 volatile compounds and 22 alcohol-soluble compounds, indicating a more complex chemical constituent in NM. Amino acid analysis revealed that AM contained 787.1 mg/g total amino acids, significantly higher than the 156.4 mg/g found in NM. The determination of inorganic elements highlighted the differences in elemental content and identified the characteristic elements in both musks Specifically, K, Al, Ni, As, and Hg were characteristic of NM, while Na, Fe, Al, Co, Cu, Ag, Ba, and Pb were characteristic of AM. In addition, the results of in vitro anti-inflammatory tests showed the starting concentration of NM (0.025 mg/mL) and AM (0.05 mg/mL), indicating that NM and AM cannot be used interchangeably in clinical applications. This study provides a valuable reference for differentiating between NM and AM, providing insights into their utilization and development.
Similar content being viewed by others
Introduction
Musk is the dry secretion from the scent sacs of adult males of the musk deer (Moschus berezovskii Flerov, Moschus sifanicus Przewalski, or Moschus moschiferus Linnaeus) (Fig. 1), which is known for its effects on the human body, including enhancing mental clarity, stimulating blood circulation, promoting menstruation, reducing swelling, and relieving pain1. Natural musk (NM) has a long history of medicinal use, is classified as a top quality in the Divine Husbandman’s Classic of the Materia Medica, and used to treat coma, stroke, neurasthenia, convulsions, angina pectoris, myocardial infarction, heart disease, ulcers and sores and other diseases. It has been used for 2000 years2,3,4,5. Modern research indicates that the primary component of musk, musk ketone, offers protective effects on the central nervous system and cardiovascular system. Furthermore, it demonstrates pharmacological benefits such as anti-inflammatory, anti-tumor, anti-dementia, and anti-ischemic effects6. A male musk deer produces only 15–20 g of musk per year. Due to its limited production and extensive use, musk has historically been in short supply, leading to its price in Europe at one time being five times that of gold7. In China, due to long-term musk hunting and harvesting behaviors, musk resources have been greatly damaged, and the number of musk deer did not exceed 100,000 by the end of the last century8. The number of forest musk deer populations decreased dramatically, and musk resources became increasingly scarce9. Since 1973, the use of musk has been regulated under the Convention on International Trade in Endangered Species of Wild Fauna and Flora10. In order to cope with the shortage of musk, the Bureau of Pharmaceutical Affairs of the Ministry of Health and the Chinese Medicinal Materials Company jointly organized and released a research project on artificial musk (AM), which was successfully developed and industrialized after more than 20 years of collaborative research11. Nowadays, AM is used as one of the ingredients in more than 400 different Chinese medicinal preparations, with a market coverage of 99%12, and only a few Chinese medicinal preparations are still available with NM, as shown in Table 1.
(a) young forest musk deer, adult forest musk deer; (b) Sachets, musk kernels; (c) NM, AM; (d) Chinese medicine compound preparation containing musk.
The Ministry of Health in 1994 issued the “Ministry of Health on AM trial production management issues related to the notice” which clearly pointed out that AM is a new class of drugs, national confidential varieties, and NM is the same formula to use. NM is different because it is derived from animal secretions. Apart from containing at least 2% musk ketone, NM also comprises numerous other components, some possessing distinct physiological activities. These components can interact with other drugs to create novel pharmacologically active substances, thereby achieving a comprehensive regulatory effect13. For example, the ethyl palmitate detected in NM-containing Xiaojin Pills may result from reactions between certain compounds in NM and others14. Consequently, the state mandates that products using AM should not be labeled simply as “musk”. The correct labeling should indicate “artificial musk” or “synthetic musk” to clearly differentiate between artificial and natural musk. The gradual replacement of NM with AM is becoming inevitable, although their efficacies are unlikely to be identical. Advancements in science and technology aim to maximize the similarity in efficacy between NM and AM. Comparative studies of both musks are significant from scientific, economic, social, and ecological perspectives15.
Currently, research on NM and AM primarily focuses on their volatile components, chemical structure, and pharmacological activity16,17,18,19,20. While significant progress has been made in their development and utilization, some areas remain underexplored. For instance, comparisons of amino acid composition, alcohol-soluble compounds, and elemental content between NM and AM are seldom reported. Given the differences in sources, biosynthetic processes, and chemical compositions between NM and AM, these variations are hypothesized to influence their physicochemical properties and biological activities, potentially impacting their pharmacological effects and clinical interchangeability. Therefore, this study aims to compare NM and AM in five aspects: ultrastructure, volatile components, amino acid composition, alcohol-soluble components, elemental content, and anti-inflammatory activity. The results will provide a valuable reference for the identification and clinical use of NM and AM.
Materials and methods
Materials and reagents
NM (Batches: N1-N3) were supplied by Chongqing Institute of Medicinal Plant Cultivation (Lot#:013, 037, 048), and AM (Batches: A1-A3) were from Beijing Lianxin Pharmaceutical Co., Ltd (Lot#:01984, 01989, 01982). The standard of muscone (Batch no.10730, purity > 98%) was purchased from Shanghai Standard Biotechnology Co., Ltd. (Shanghai, China). 3α-hydroxy-5β-Androstan-17-one (Batch no.13356500, purity > 98%) was purchased from Dr.Ehrenstorfer (Augsburg, Germany). Cholesterol (Batch no.013679926, purity > 98%) was purchased from Adamas (Shanghai, China).
The amino acids mixture standard solution (Type H, 2.5 µmol/mL) containing threonine (Thr), glutamic acid (Glu), aspartic acid (Asp), glycine (Gly), serine (Ser), alanine (Ala), proline (Pro), histidine (His), valine (Val), methionine (Met), leucine (Leu), isoleucine (Ile), phenylalanine (Phe), lysine (Lys), tyrosine (Tyr), arginine (Arg) and cysteine (Cys). The Ninhydrin Coloring Solution Kit was produced by FUJIFILM Wako Pure Chemical Co. (Osaka, Japan), and the MCI BUFFER™ pH-KIT buffer system was purchased from Mitsubishi Chemical Co. (Tokyo, Japan). Multi-element calibration standard 2 A (containing elements including Na, Ca, K, As, Fe, Se, Al, Rb, V, Sr, Cr, Ag, Mn, Cd, Co, Cs, Ni, Ba, Hg, Pb), ICP-MS internal std mix (containing Tb, Li, Bi, In, Ge, Sc, Rh, ), and Hg standard solution, were supplied from Agilent Technologies (California, USA). Lipopolysaccharides (LPS, from Escherichia coli serotype 055:B5) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Cell Counting Kit-8 (CCK-8) and the Griess reagent were provided by Beyotime (Shanghai, China).
Observation of the surface properties of NM and AM powder via SEM
The characteristics of NM and AM powder were examined using a scanning electron microscope (SEM, G6, Thermo Fisher Scientific). Approximately 1 cm2 of conductive adhesive was applied onto the specimen holder. NM and AM powders were uniformly spread onto the adhesive surface, ensuring the removal of any loose particles with a dust canister. To enhance conductivity, a 10 nm gold coating was applied via vacuum deposition using a gold sprayer. The surface morphology was then observed and imaged using a benchtop SEM at a working distance of 8 mm and an accelerating voltage of 10 kV, with magnifications of 1200x and 5000x, respectively.
Analysis of the volatile components in NM and AM
Preparation of test solution
The sample powders from each of the three batches of NM and three batches of AM were thoroughly mixed to create representative samples for the two groups of musk. The 0.50 g sample of musk powder was accurately weighed and placed in a stoppered test tube. Ten milliliters of anhydrous ethanol was added using a pipette, and the mixture was weighed. The sample was then ultrasonically treated for 30 min. Anhydrous ethanol was added to compensate for any weight loss. The resulting solution was filtered using a 0.22 μm microporous membrane to obtain the final test solution.
Test conditions for gas chromatography-mass spectrometry (GC–MS)
GC-MS analysis was performed using a Shimadzu TQ8040 instrument equipped with an Agilent DB-17MS capillary column (0.25 μm×0.25 mm×30 m). The column temperature was programmed from 50 °C to 280 °C in three stages, with specific holding times. A 1 µL injection volume was used at 250 °C with a split ratio of 50:1. Mass spectrometry employed an electron impact (EI) ion source, scanning an m/z range of 50–600 amu, with helium as the carrier gas at a linear flow rate of 1.0 mL/min and a 3-minute solvent delay. Components were identified via the NIST14 database and standard. Their relative contents were determined through the area normalization method.
Analysis of the alcohol-soluble composition in NM and AM
Preparation of test solution
50 µL of the test solution from “Preparation of test solution” section was taken and placed into a sample tube, then diluted to 1 mL with anhydrous ethanol.
Test conditions for alcohol-soluble composition
The alcohol-soluble components were analyzed using a Thermo Scientific Orbitrap Exploris 120 (Thermo OE 120) LC-MS under specified chromatographic and mass spectrometry conditions. A ZORBAX Eclipse Plus C18 column was used with a gradient elution of 0.1% formic acid in water (A) and acetonitrile (B) at a flow rate of 0.3 mL/min. Mass spectra were acquired in Data-Dependent Acquisition (DDA) mode, where the top four most intense precursor ions (TopN = 4) from each full MS scan (m/z 70-1050) were selected for MS/MS fragmentation. Stepped normalized collision energies (NCE 20, 40, 60) were applied to optimize fragmentation efficiency. An electrospray ionization source (ESI) was used, with a resolution of 60,000 for full MS scans and 15,000 for MS/MS scans. Key operational parameters included spray voltages of 3.5 kV (ESI+) and 2.5 kV (ESI-), and a transfer tube temperature of 325 °C.
Analysis of the amino acid composition in NM and AM
Preparation of test solution
A quantity of 0.05 g of NM powder and 0.025 g of AM powder was accurately weighed into PTFE tubes. To each tube, 10 mL of 6 mol/L hydrochloric acid solution was added, followed by a 2-minute flushing with argon gas. The digestion process was carried out under the optimized microwave conditions outlined in Table 2. After digestion, rinse the hydrolysate three times with ultrapure water and transfer it to a 50 mL volumetric flask. The solution was cooled to room temperature before being diluted to the mark with ultrapure water. Finally, the diluted solution was then filtered using a 0.22 μm membrane. A 0.5 mL aliquot of the filtered solution was precisely measured into a 1.5 mL EP tube and subjected to vacuum drying at 60℃. The resulting residue was mixed in 1.0 mL sodium citrate buffer (0.2 mol/L Na+, pH = 2.2). The test solution was thoroughly vortexed and stored at 4℃ in a refrigerator for further analysis.
Test conditions for amino acid
The amino acid analysis was performed using a Hitachi LA8080 automatic amino acid analyzer from Hitachi High-Tech Science Co. (Tokyo, Japan). The system was equipped with an analytical column (4.6 mm×60 mm, 3 μm) and an ammonia filter column (4.6 mm×40 mm). The buffer system was operated at a flow rate of 0.40 mL/min. The analytical column temperature was set at 57℃, with an injection volume of 10 µL. The resulting derivatives, produced after color development at a reaction temperature of 135℃, were simultaneously measured by UV detection at wavelengths of 570 nm and 440 nm.
Inorganic element analysis in NM and AM
Preparation of test solution
Representative samples of NM and AM were each weighed to 50 mg, and then placed into dissolution tanks containing 7 mL of concentrated nitric acid. The tanks were sealed, and the samples were dissolved using a microwave dissolution instrument according to the program outlined in Table 3. After dissolution, the tanks were placed on a graphite hot plate and heated at 180 °C to evaporate the HNO3, reducing the solution volume to approximately 1 mL. The solution was transferred to a 10 mL volumetric flask, and the solution was filtered through a 0.22 μm membrane to obtain Test Solution I, which was used for the determination of Al, V, Mn, Ni, Cr, Co, Hg, Se, Cs, Sr, Cu, Cd, Ba, Pb, As, and Ag. The sample was then analyzed using the graphite hot plate method. The solution was diluted to obtain Test Solution II, which was used for the determination of Na, Fe, Ca, and K.
Test conditions for inorganic elements
For the simultaneous detection of multiple inorganic elements, the ICP-MS technology was applied. The 7800 ICP-MS system, equipped with Mass Hunter 5.1 software (Agilent Technologies, USA), was utilized for data acquisition and analysis. The parameters measured by the optimized ICP-MS were detailed in Table 4. The analysis was conducted in full quantitative mode. In the “He” mode, different internal standards were selected for various elements: Sc for K, Mg, Na, Al, Fe, Mn, Ni, Co, V, and Cr; Ge for Sr, Ga, Se, As, and Cu; In for Ba, Cs, and Cd; and Bi for Hg, and Pb.
Comparison of in vitro Anti-Inflammatory activity of NM and AM
Cell culture and CCK-8 assay for cell viability
The macrophage RAW 264.7 cells in the logarithmic growth phase were seeded into 96-well plates at a density of 1 × 104 cells/mL and incubated for 24 h. Following incubation, cells were exposed to various concentrations of NM (0.025, 0.05, 0.1, 0.25, 0.5, and 1 mg/mL) and AM (0.025, 0.05, 0.075, 0.1, and 0.15 mg/mL) in 100 µL of the medium. The blank group received an equal volume of basal medium. Each group had three replicate wells. After being cultured at 37 °C with 5% CO₂ for another day, 100 µL of CCK-8 reagent was added to each well. The absorbance at 450 nm was measured using a microplate reader to calculate the cell survival rate.
Analysis of NO production
RAW 264.7 cells were divided into 11 groups: blank group, LPS inflammation model group, dexamethasone group, NM groups (0.025, 0.05, 0.075, 0.1 mg/mL), and AM groups (0.025, 0.05, 0.075, 0.1 mg/mL). The cells were seeded into 96-well plates at a density of 5 × 104 cells/mL. After treatment with different doses of NM and AM for 3 h, the cells were stimulated with 1 µg/mL LPS for 24 h. NO production in the cell culture supernatant was measured using the Griess assay. Each group’s supernatant was mixed with an equal volume (50 µL) of Griess reagent, and the absorbance at 540 nm was measured to calculate the nitrite concentration.
Data analysis
Statistical analysis was conducted using one-way analysis of variance (ANOVA) with SPSS 17.0 and GraphPad Prism 8. The values of the different parameters tested within each group are presented as the mean ± SD (n = 3).
Results and discussion
Microscopic surface properties of NM and AM
SEM is a crucial tool for identifying traditional Chinese medicines (TCM) and is extensively employed in the identification of plant, animal, and mineral-based TCM21. The surface morphology and structure of musk can be observed under the SEM (Fig. 2).
SEM Results of NM and AM. (a) NM; (b) AM.
SEM observations of NM revealed a sparse and scattered sample overall. At 1200x magnification, the left side of the NM exhibited a lamellar texture, showing an aggregation of small particles of varying sizes with numerous depressions or cavities. At 5000x magnification, the sample displayed small clusters of overlapping, intermingled coral-like structures with extremely uneven surfaces, featuring pores and depressions. In contrast, SEM observations of the AM revealed a relatively flat sample overall, with fewer gaps and a relatively tight arrangement. At 1200x magnification, the AM appeared as agglomerates of similar size and shape. At 5000x magnification, the sample appeared as flakes that slightly overlap and fuse in a rock-like structure with an uneven surface, displaying a compact and homogeneous texture with relatively few interstitial spaces. Significant differences existed in the surface microstructures of NM and AM, which could be taken as one of the differences between the physicochemical properties of NM and AM.
Differential analysis of volatile components of NM and AM
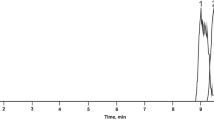
The total ion chromatograms of NM and AM were depicted in Fig. 3. Volatile components were identified using the NIST14 database, and the relative percentage content of each component was determined using peak area normalization. Detailed results were presented in Table 5.
GC-MS TIC of NM (a) and AM (b).
The components in the NM and AM samples were identified and analyzed using the NIST 14 database and standards. Specifically, 15 volatile components were identified in NM and 8 in AM, with musk ketone and cholesterol present in both types. According to the area normalization method, the percentage of muscone in AM is significantly higher than that in NM, nearly three times. In NM, the highest relative contents were observed for cholesterol and musk ketone, averaging 35.53% and 26.24%, respectively. AM exhibited 8 detected compounds, including 6 not found in NM. It is worth noting that ethylparaben was not detected in NM, while its relative peak area was 0.82% in AM. Ethylparaben may be introduced as a preservative in the production process of AM to ensure the stability and safety of the product. The highest relative contents in AM were musk ketone and dehydroepiandrosterone, averaging 67.45% and 16.08%, respectively.
The comparative analysis indicated that NM contains cyclotridecanone, (Z)-7-hexadecenal, musk ketone, 5-cyclohexadecen-1-one, 5α-androstane-3α,17β-diol, cis-9-octadecadienal, dehydroepiandrosterone sulfate, 3α-hydroxy-5β-androstan-17-one, benzoic acid, and cholesterol. These findings align with previous reports in the references22,23,24,25. In contrast, most of these components are lacking in AM, which contains only glycerol, ethyl nipagin, musk ketone, linoleic acid, and dehydroepiandrosterone26. The type of volatile components can be one of the distinguishing substances between NM and AM. In comparison with related literature, this experiment also detected other components in NM, such as 3,3-dimethylpentane, 2-cyclobutyl-2-propane, p-cresol, acetic acid, and benzene acetic acid, which were not reported in previous literature. These discrepancies may arise due to various factors including the expansion of musk production in forest environments, different species, origins, breeding conditions, years of growth, and harvest times, all of which can influence the composition of musk.
Differential analysis of alcohol-soluble components in NM and AM
The extracts of NM and AM were analyzed under the conditions specified in “section Test conditions for alcohol-soluble composition”. The TIC for both NM and AM, obtained in positive and negative ion modes, were presented in Fig. 4.
TICs of NM in the positive (a) and negative (b) mode; TICs of AM in the positive (c) and negative (d) mode.
A total of 26 components were identified in both NM and AM using a self-constructed database in combination with MS Dial ver. 5.1 software for analysis and processing. Among the identified compounds, 25 were found in NM, categorized into 9 classes: 3 alkaloids, 9 organic acids, 3 nucleosides, 2 saccharides, 2 esters, 1 macrocyclic ketone, 1 amino acid, 1 pyrrolidine, and 3 others (organic nitrogenous compounds and ketones). In AM, 22 compounds were identified, also classified into 9 categories: 3 alkaloids, 6 organic acids, 3 nucleosides, 2 saccharides, 2 esters, 1 macrocyclic ketone, 1 amino acid, 1 pyrrolidine, and 3 others. Detailed information were provided in Table 6.
From Fig. 5, it could be visualized that nine classes of compounds account for different proportions in NM and AM. In NM, the compounds were present in the following descending order: alkaloids > macrocyclic ketones > organic acids > other classes > nucleosides > saccharides > esters > pyrrolidines > amino acids. In AM, the order was organic acids > alkaloids > macrocyclic ketones > other classes > pyrrolidines > saccharides > esters > amino acids > nucleosides. The contents of alkaloids, macrocyclic ketones, and organic acids were higher in both NM and AM, accounting for 71.08% in NM and 75.65% in AM. A review of previous literature established that NM contains 15 amino acids, creatine, lactic acid, allantoin, inosine, palmitoleic acid, muscone, stearic acid, and linoleic acid27,28,29. Additional constituents were identified and reported for the first time in this study.
Proportion of NM and AM components. (a) NM; (b) AM.
Differential analysis of the amino acid components of NM and AM
Method validation
The 16 amino acid standards were analyzed under the conditions detailed in “section Test conditions for amino acid” and a standard curve was constructed with mass concentration (µg/mL) versus peak area. When the signal-to-noise ratio (S/N) of each amino acid content was 3, as the limit of detection (LOD), and the signal-to-noise ratio (S/N) was 10 as the limit of quantitative (LOQ). Corresponding mass concentrations, calculated from each amino acid’s molar mass, are presented in Table 7. Correlation coefficients exceeded 0.9991 for all amino acids, demonstrating excellent linearity. The LODs and LOQs ranged from 0.0116 to 0.0322 µg/mL and 0.0312–0.1013 µg/mL, respectively, indicating high sensitivity. Precision tests (n = 5) showed RSD values of 0.20-1.83%, confirming the instrument’s accuracy for sample analysis. Accurately weigh 0.025 g of NM into a dissolution tube. Add a precise volume of mixed amino acid standard solution to achieve a final concentration of 40 nmol/mL for each amino acid. Prepare six replicates following this procedure. Similarly, weigh 0.0125 g of AM into a dissolution tube and add the mixed amino acid standard solution to attain a concentration of 100 nmol/mL for each amino acid, also in six replicates. Each test solution was digested according to the “section Preparation of test solution” and measured according to the “section Test conditions for amino acid” chromatographic conditions. The average recovery for the 16 amino acids in NM ranged from 93.82 to 112.3%, with RSDs between 0.69% and 3.88%. For AM, the average recovery were between 94.66% and 115.5%, with RSDs from 0.47 to 4.01%.
Amino acid composition and content
The NM and AM samples were analyzed under the chromatographic conditions described in “section Test conditions for amino acid”. The chromatogram was shown in Fig. 6. A total of 15 amino acids were detected in NM, whereas 16 amino acids were detected in AM, which contains an additional amino acid, cystine (Cys), compared to NM. The average total amino acid (TAA) content was 156.4 mg/g in NM and 787.1 mg/g in AM. NM and AM contain a diverse array of amino acids, including six essential amino acids (EAA): Phe, Lys, Ile, Leu, Thr, and Val. Additionally, they include non-essential amino acids (NEAA) such as Asp, His, Ser, Gly, Glu, Tyr, Arg, Ala, and Pro. As shown in Fig. 7, in NM, the content of the six EAAs ranked in descending order was Leu > Lys > Phe > Thr > Val > Ile, comprising 42.95–43.46% of the TAA content. In AM, the order of the six EAAs was Leu > Val > Lys > Phe > Thr > Ile, accounting for 43.27–46.08% of the TAA content. NM exhibited the highest content of Glu and Leu, averaging 24.04 mg/g and 23.28 mg/g, respectively, which constitute 15.37% and 14.89% of the TAA content. On the other hand, AM showed the highest content of Leu and Asp, averaging 123.9 mg/g and 92.93 mg/g, respectively, accounting for 15.74% and 11.81%.
Chromatogram of amino acids in musk. (a) Mixed standard solution of 17 amino acids; (b) NM; (c) AM.
Comparison of the differences in amino acid composition and content between NM and AM. (a) Comparison between EAAs; (b) Comparison between NEAAs; (c) Comparison of EAA, NEAA, and TAA content; (d) Amino acid composition and content of NM and AM. * P < 0.05, ** P < 0.01.
The EAA content in NM and AM was 71.70 mg/g and 403.3 mg/g, respectively, while the NEAA content was 84.65 mg/g and 383.8 mg/g, respectively. Both EAA and NEAA levels were higher in AM compared to NM. Analyzing the amino acid composition revealed that acidic amino acids such as Asp and Glu accounted for 21.71% of the total in AM and 26.42% in NM.
Yu et al.30 isolated and purified a glycoprotein, Mu-1 (pI 3.0, molecular weight 6000, 15 amino acids), from musk powder, which aligns with the findings of this experiment. The experimental results clearly showed that the amino acid content in AM was significantly higher than in NM. This difference is likely related to the origin of the peptide components. Since AM is a nationally secret recipe, it is hypothesized that a different protein source or biosynthetic method may have been used, leading to the production of more or different types of amino acids. In contrast, the amino acid composition of NM is derived from the natural secretions of the male musk gland and is determined by the metabolic processes of the musk deer31,32.
Differential analysis of inorganic elements in NM and AM
Method validation
Mixed standard solutions of 20 elements were analyzed using the chromatographic conditions in “section Test conditions for inorganic elements”. Regression curves plotted with element mass concentrations (µg/kg) versus internal standard ratios showed excellent linearity, with correlation coefficients above 0.9990. LOD and LOQ were determined through 11 replicate blank measurements, calculated as LOD = 3s/k and LOQ = 10s/k (where s: response standard deviation, k: calibration slope), with results presented in Table 8. Precision tests with a 200 µg/L standard solution (n = 6) resulted in RSDs of 2.8-9.5%, while stability tests over 12 h yielded RSDs of 0.83–6.89% (Table 9).
For reproducibility, 0.05 g NM samples (n = 6) showed RSDs of 1.50–8.62%, and spiked recovery tests at low, medium, and high concentrations (5, 50, and 5000 ng/mL) achieved recoveries of 90.68-98.16%, with RSDs between 1.25 and 7.29% (Table 9). These results demonstrate the method’s precision, stability, and reproducibility.
Elements content in NM and AM
The test solution was prepared according to “2.6.1”, and the determination was conducted under the conditions specified in “Test conditions for inorganic elements” section. The results could be found in Table 10. Due to instrument and reagent limitations, Na and Fe were not detected in NM. The trace elements with higher concentrations were Al and Rb, averaging 18.06 µg/g and 3.939 µg/g, respectively. AM contained macronutrient elements such as Na, K, and Fe. The highest content observed was Na, averaging 14.34 mg/g. Among the trace elements, Al, Rb, and Cu were found in higher concentrations, averaging 37.70 µg/g, 11.38 µg/g, and 8.732 µg/g, respectively. The differences in elemental contents in different batches of NM and AM were small, and the RSD was lower than 15% and 20%, respectively. Both NM and AM contained 14 trace elements such as Al, Mn, Ni, Cr, As, Se, Rb, Cu, Ag, Cs, Ba, Hg, Pb, Cd, etc., and the AM contained extra Co and V elements, while the NM contained extra Sr element. Among the trace elements, the content of Cr, Ni, Mn, As, Rb, Ag, Cu, Cd, Hg, Ba, and Cs in AM was higher than that in NM. Chen et al.33 identified the primary inorganic elements in NM—including Na, Si, Mg, P, Al, S, Ag, Cl, K, Ga, and Fe—using electron probe microanalysis. Other studies on element determination in musk have primarily focused on proprietary Chinese medicines containing musk.
Multivariate analysis: PCA
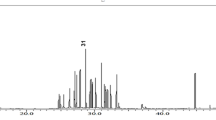
PCA is a statistical method used to reduce the number of variables in a dataset and retain as much information as possible from the original data34,35. PCA of 16 inorganic elements in NM and 19 inorganic elements in AM was conducted using IBM SPSS Statistics 26 software. The results are presented in Fig. 8; Table 11.
PCA scree plot of NM (a) and AM (b).
In Fig. 8a, the first two principal components for NM exhibited significant variance, with initial eigenvalues exceeding 1 and a cumulative contribution reaching 99.91%. This indicated that these two components account for 99% of the variable information of the inorganic elements in NM. Based on this, the first two principal components were selected to generate the component matrix (Table 12). In this matrix, the elements K, Al, Ni, As, and Hg had high loadings, suggesting that these elements were characteristic inorganic components of NM. Similarly, for AM, the component matrix was rotated, and two principal component factors were obtained by combining the mutation point in Fig. 8b, as shown in Table 12. The elements Na, Fe, Al, Co, Cu, Ag, Ba, and Pb exhibited high loadings, indicating that these elements were characteristic inorganic components of AM.
Comparison of in vitro anti-inflammatory activity of NM and AM
Cytotoxicity of NM and AM on RAW 264.7 macrophages
The survival rates of RAW 264.7 cells following exposure to different dosages of NM (0.025, 0.05, 0.1, 0.25, 0.5 mg·mL− 1) (Fig. 9a) and AM (0.025, 0.05, 0.075, 0.1 mg·mL− 1) (Fig. 9b) were consistently above 95% after 24 h, as determined by a CCK-8 experiment. None of the treatment groups showed a significant decrease in cell viability compared to the control (p > 0.05), indicating no cytotoxic effects at concentrations up to 0.1 mg·mL− 1. Therefore, the possibility of musk-induced cytotoxicity as a cause of observed bioactivities was ruled out. Concentrations equal to or below 0.1 mg·mL− 1 were selected for further experiments.
Effects of NM (a) and AM (b) on RAW 264.7 macrophage viability and on LPS-induced NO production in RAW 264.7 cells (c). #### P < 0.0001 vs. control group; * P < 0.05, ** P < 0.01, *** P < 0.001, and **** P < 0.0001 vs. LPS group.
Effects of NM and AM on the production of NO in LPS-induced RAW 264.7
To assess the anti-inflammatory effects of NM and AM, nitric oxide (NO) production was quantified using Griess reagents. RAW 264.7 cells induced with lipopolysaccharide (LPS) exhibited significantly higher NO production compared to the control group, which was notably suppressed by the addition of NM or AM at certain concentrations (p < 0.05). The inhibitory effects of both types of musk on NO generation increased with higher concentrations. NM significantly reduced NO production at a concentration of 0.075 g·L− 1 (p < 0.01) and demonstrated a strong concentration dependence, while AM significantly reduced NO production at a concentration of 0.1 g·L− 1 (P < 0.01). These results were presented in Fig. 9c.
Further analysis revealed that NM exhibited a more potent anti-inflammatory effect compared to AM at all tested concentrations, with a notably stronger inhibition of NO production at lower doses, highlighting its superior efficacy. Compared to dexamethasone (Dex), NM exhibited comparable anti-inflammatory activity at higher concentrations, while AM, though effective, required higher doses to achieve similar results. This further supported the idea that the superior anti-inflammatory efficacy of NM may be attributed to its richer and more diverse chemical composition. Therefore, although both NM and AM have shown potential as anti-inflammatory agents, NM was proved to be more effective, particularly at lower doses, making it a more potent option.
Conclusion
The aim of this experiment was to compare the physicochemical properties and anti-inflammatory activity of NM and AM. Significant differences in their surface structures were observed through comparative analysis. Additionally, volatile components, alcohol-soluble compounds, amino acid profiles, inorganic elements, and anti-inflammatory activities were investigated using various analytical techniques, including GC-MS, LC-MS, AAA, ICP-MS, and Griess assays. This study provided practical insights for distinguishing between NM and AM.
The results revealed that NM and AM differ significantly in ultrastructure, with NM exhibiting a more complex and diverse composition of volatile and alcohol-soluble compounds, while AM showed higher levels of essential amino acids and inorganic elements. The differences may be attributed to variations in their biosynthetic pathways, aging processes, and environmental influences during formation. NM undergoes long-term biochemical transformation within musk glands, leading to the accumulation of a wide range of secondary metabolites, which contribute to its pharmacological potency. In contrast, AM is synthesized or semi-synthesized through controlled formulations, often lacking the intricate molecular interactions and gradual maturation seen in NM. In vitro anti-inflammatory tests demonstrated that NM was more effective than AM, indicating that substituting NM with an equal amount of AM may not achieve the same therapeutic effects in clinical applications. Further pharmacokinetic and in vivo studies are necessary to determine precise initial doses and provide better clinical guidance.
These findings underscore the importance of further research into the quantitative and qualitative relationships to better guide clinical use. Moreover, it is recommended that the quality control of musk products be enhanced by including a variety of indicator components (e.g., steroids, proteins) in addition to muscone, to ensure a more comprehensive assessment of musk quality.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
National Pharmacopoeia Commission & Pharmacopoeia, C. China Medical Science and Technology, 402–403 (2020).
Guo, J. Research advances in artificial musk. Acta Acad. Med. Sin. 36, 577–580 (2014).
Ayuob, N. N., Ali, S. S., Suliaman, M., Wahab, M. G. & Ahmed, S. M. The antidepressant effect of musk in an animal model of depression: A histopathological study. Cell Tissue Res. 371, 377–378. https://doi.org/10.1007/s00441-016-2468-9 (2018).
Du, Y. Q. et al. Hypoxia-Inducible factor 1 alpha (HIF-1α)/vascular endothelial growth factor (VEGF) pathway participates in angiogenesis of myocardial infarction in muscone-treated mice: Preliminary study. Med. Sci. Monit. 24, 8870–8877. https://doi.org/10.12659/msm.912051 (2018).
Liu, K. et al. Zoology, chemical composition, pharmacology, quality control and future perspective of musk (Moschus): A review. Chin. Med. 16, 46. https://doi.org/10.1186/s13020-021-00457-8 (2021).
Wang, J. et al. Pharmacological effects and mechanisms of muscone. J. Ethnopharmacol. 262, 113120. https://doi.org/10.1016/j.jep.2020.113120 (2020).
Volker, H. On the scent: Conserving musk deer. The uses of musk and Europe’s role in its trade (1999).
Li, X. Y. & Jiang, X. L. Implication of musk deer (Moschus spp.) depletion from Hunter reports and Dung transect data in Northwest Yunnan, China. J. Nat. Conserv. 22, 474–478. https://doi.org/10.1016/j.jnc.2014.05.004 (2014).
Yang, Q. S., Meng, X. X., Xia, L. & Feng, Z. J. Conservation status and causes of decline of musk deer (Moschus spp.) in China, biological conservation. 109 333–342 (2003). https://doi.org/10.1016/s0006-3207(02)00159-3
Do, J. C., Kitatsuji, E. & Yoshi, E. Study on the components of musk. I. Ether soluble components. Chem. Pharm. Bull. 23(3), 629–635 (1975).
Development Industrialization of artificial musk, the 65th-anniversary development achievement of Chinese traditional medicine. Chin. Mod. Tradit. Med. 23, 395 (2021).
Tang, X. W. & Zhang, Q. Artificial musk: Let the natural beauty of human fragrance. Sci. Technol. Dly. 11, 004 (2016).
Liu, C. S. Because of natural, so precious–musk and Pientzehuang. Med. World. 04, 71 (2008).
Yuan, L. Y. et al. GC-MS study on characteristics of Xiaojin pills containing natural musk and artificial musk Chinese. J. Exp. Tradit. Med. Formulae. 23, 43–47. https://doi.org/10.13422/j.cnki.syfjx.2017020043 (2017).
Zhu, X. J., Li, H. T., Yu, B. & Wu, Q. B. Progress in the study of the effects of natural and artificial musk on the cardiovascular system. J. Nanjing Univ. Tradit. Chin. Med. 25, 316–317 (2009).
Jiang, Q. Y., Luo, Y., Tan, T., Yang, M. & Liao, Z. G. Comparison of volatile chemical compositions of Moschus from different species and producing areas by GC-MS and chemometric analysis. Chin. J. Exp. Tradit. Med. Formulae. 24, 49–55 (2018).
Liang, Y. & Wang, X. G. Preliminary analysis of natural musk and artificial musk by GC-MS. Tradit. Chin. Drug Res. Clin. Pharmacol. 16, 204–205 (2005).
He, L. L. Study of the influence of artificial musk and natural moschus for the central nervous. China Med. Herald. 7, 22–23 (2010).
Quan, H. X. et al. Protection of H2O2-induced H9c2 cardiomyocyte damage by musk and its substitute artificial musk. J. Chin. Med. Mater. 41, 961–965. https://doi.org/10.13863/j.issn1001-4454.2018.04.040 (2018).
Li, D. Y. et al. The musk chemical composition and microbiota of Chinese forest musk deer males. Sci. Rep. 6, 18975. https://doi.org/10.1038/srep18975 (2016).
Lu, G. Y. et al. Applications of scanning electron microscopy in identification of traditional Chinese medicine. J. Chin. Electron. Microscopy Soc. 40, 483–487 (2021).
Jie, H. et al. Variation of chemical constituents in musk harvested at different maturity stages. . Tradit. Patent Med. 43, 144–148 (2021).
Lv, S. Q. et al. Chemical compositions and Pharmacological activities of natural musk (Moschus) and artificial musk: A review. J. Ethnopharmacol. 284, 114799 (2022).
Dong, W., Zhao, W. & Liu, C. Research progress of musk. Spec. Wild Econ. Anim. Plant. Res. 2, 48–58 (2001).
Zhou, Z., Shen, W., Yu, L., Xu, C. & Wu, Q. A Chinese patent medicine, Shexiang Baoxin pill, for Non-ST-elevation acute coronary syndromes: A systematic review. J. Ethnopharmacol. 194, 1130–1139 (2016).
Song, Z., Zhu, X., Song, W., Yu, D. & Liu, X. Modern Research on Chinese Herbal Medicine, Musk. Beijing, 204–226 (Beijing Medical University Press & China Union Medical College, 1995).
Ding, M. et al. From non-targeted to target GC-MS metabolomics strategy for identification of TCM preparations containing natural and artificial musk. Chin. Med. 17, 41. https://doi.org/10.1186/s13020-022-00594-8 (2022).
Zhang, T. X. et al. Study of compositions of musks in different types secreted by forest musk deer (Moschus berezovskii). PLOS ONE. 16, e0245677. https://doi.org/10.1371/journal.pone.0245677 (2021).
Liu, J. H., Cheng, Y. Y., Hsieh, C. H. & Tsai, T. H. Identification of a multicomponent traditional herbal medicine by HPLC–MS and Electron and light microscopy. Molecules 81, 1245–1251. https://doi.org/10.3390/molecules22122242 (2018).
Yu, D., Liu, X. & Gao, S. Studies on the anti-inflammatory principle of musk. Acta Pharm. Sin.. 15 (5), 306–307 (1980). (in Chinese).
Yang, M. N., Li, R., Li, L., Zhao, J. N. & Tan, X. R. Research progress on chemical constituents and Anti-Tumor effects of musk, Pharmacology and clinics of Chinese materia medica. 1–18 (2024).
Liu, X. M., Li, H. Q., Xiao, X. & Yu, D. Q. Studies on natural musk anti-inflammatory proteins I. Isolation and purification of Mu-a-1 and characterization of some of its properties. Curr. Zool. 03, 302–308 (1992).
Chen, J. W., Li, X. & Li, F. Rapid determination of inorganic elements in musk by electron microprobe. China J. Chin. Materia Med. 08, 487–488 (1991).
Liu, S., Liang, Y. Z. & Liu, H. T. Chemometrics applied to quality control and metabolomics for traditional Chinese medicines. J. Chromatogr. B. 15, 82–91. https://doi.org/10.1016/j.jchromb.2016.02.011 (2016).
Han, J. et al. Qualitative and quantitative evaluation of Flos puerariae by using chemical fingerprint in combination with chemometrics method. J. Pharm. Anal. 12, 489–499. https://doi.org/10.1016/j.jpha.2021.09.003 (2022).
Funding
This research was supported by grants from the Research Program of Chongqing Municipal Education Commission (KJZD-K202302804), the Natural Science Foundation of Chongqing Medical and Pharmaceutical College(ygzzd2024104), and the Chongqing Talent project (cstc2021ycjh-bgzxm0201).
Author information
Authors and Affiliations
Contributions
Shuya, Li: Investigation, Writing-original draft, Methodology. Bumarya Rahimjan: Investigation, Writing-original draft, Visualization. Hang Jie: Investigation, Resources, Data curation, Funding acquisition. Yiwu, Wang: Software, Investigation. Yuan, Shen: Validation, Data curation. Jialing, Yu: Investigation, Visualization. Lin, Yang: Conceptualization, Writing-review & editing, Funding acquisition. Dan, He: Supervision, Conceptualization, Writing-review & editing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All muskrat scenting experiments were conducted in accordance with the guidelines of the Chongqing Institute of Medicinal Plant Cultivation and Welfare Committee (Approval No. 20220608).
Consent for publication
All authors have approved the manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, S., Rahimjan, B., Jie, H. et al. Comparative physicochemical properties and anti-inflammatory activity of natural and artificial musk for quality evaluation. Sci Rep 15, 20450 (2025). https://doi.org/10.1038/s41598-025-00968-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00968-6