Abstract
Metal-organic frameworks (MOFs) have aroused the interest of the majority of researchers due to their prominent features, especially in the field of heterogeneous catalysts. As state-of-the-art literature, this research designed and developed a new nanomaterial with a perfect crystalline nature through the combination of two MOFs. Here, the Zn-organic framework (Zn-MOF) as a 3D hybrid scaffold was synthesized, followed by functionalizing with urea ligand which contains two amine functional groups. The second step involved coating Zn-MOF-NH2 with zeolitic imidazolate framework-8 (ZIF-8) by the widespread growth of Zif-8 crystals on the surface of urea-modified Zn-MOF. Eventually, the new nanocomposite with MOF-on-MOF architecture was incorporated for the adsorption and stabilization of Cu nanoparticles through a simple liquid impregnation-reduction procedure. The chemical structure of ZIF-8 MOF was discussed and confirmed in detail by different analytical techniques. Based on the results, the Zn-MOF-NH2-ZIF-8@Cu demonstrated unparalleled catalytic performance in the synthesis of propargyl amine derivatives. The prominent characteristics such as novelty, high surface area, wonderful catalytic activity, and excellent ability to reuse, make the Zn-MOF-NH2-ZIF-8@Cu system an ideal paradigm in the developing of industrial-scale heterogeneous catalysts.
Similar content being viewed by others
Introduction
Propargylamines are considered as important scaffolds and valuable precursors in the synthesis of a wide range of pharmaceutical products, biologically active compounds, and agrochemicals1. In addition, propargylamine moieties have been used as essential skeletons in structures of antidepressant, anti-Alzheimer’s, and anti-Parkinson’s medicines2. Traditionally, the propargylamines derivatives are produced through the direct amination reaction of propargyl acetates, phosphates, and halides using strong bases such as LDA, organomagnesium compounds, or butyl lithium3. High moisture sensitivity, strictly complicated reaction conditions, and stoichiometric ratios of these reagents are disadvantages that limit them for frequent use4.
A3-coupling is one of the most useful and powerful synthetic procedures for the preparation of propargylamine, which allows the one-pot three-component coupling of a secondary amine, an aldehyde, and a terminal alkyne by using transition-metal catalysts5. This synthetic strategy proceeds via C-H activation of the terminal alkyne and has been majorly catalyzed by homogeneous systems6.
Heterogeneous catalysts with achievements such as recyclability, much less leaching of precious metal, saving energy, produce minimal toxic waste, and eco-friendliness, are effective and proven strategies to improve the performance of homogeneous catalysts7. The introduction and stabilization of metal nanoparticles (NPs) onto the surface of suitable support is one of the best and most fascinating techniques in the design and development of heterogeneous catalysts8.
In the meantime, metal-organic frameworks (MOFs) are ideal and encouraging materials with comprehensive properties and high adsorption capacity for the immobilization of active catalytic species9,10,11. One, two, and three-dimensional lattices of metal-organic frameworks are constructed by connecting the organic and inorganic units based on the principle of reticular chemistry12,13. The advantages such as expanded surface area, adjustable porosity, facility to design, trouble-free trapping of guest species, and great structural versatility enable MOF to be an attractive platform with bi-/multifunctional applications14,15,16,17. Controlled entanglement with other materials such as porous compounds, crystalline substrates, nanomaterials, and polymers, is a fascinate technique for enhancing the capabilities of MOF18. So far, several studies have been conducted to fabricate hybrid composites of MOF with other common materials, and interesting results have been obtained. For example, Soman et al. has reported UiO-DOX@CS-FA nanomaterial for targeted drug delivery of anticancer drug doxorubicin (DOX). UiO-DOX@CS-FA was synthesized by functionalization of UiO-66 (NH2) MOF with chitosan (CS) and folic acid (FA) and presented an 82% efficiency of DOX encapsulation19. Kiani et al. have prepared Fe3O4@CFC-COOH@TMU-16@Cu by combining of a 3D dimensional MOF of TMU-16 with magnetic activated carbon obtained from chicken feather (CFC) and Cu nanoparticles. Fe3O4@CFC-COOH@TMU-16@Cu nanocomposite was incorporated as a new adsorbent for the removal of methylene blue and heterogenous catalyst in the synthesis of tetrahydrobenzo[b]pyran derivatives20. In another study, Karumbaiah et al. have investigated the sensing properties of CNT-HKUST-1 composite as a sensitive humidity sensor. This composite was synthesized from carbon nanotubes (CNT) and HKUST-1 MOF by spin coating technique21. The dual MOF-on-MOF composites are new amazing materials that present enhanced performance, synergistic effect, hierarchical nanostructure, and inimitable tunability, compared with one single MOF22.
In this particular study, a new bilayer support based on the MOF-on-MOF strategy with two MOFs of Zn-MOF and ZIF-8 was synthesized to boost the adsorption and stabilization of Cu nanoparticles. Functionalization of Zn-MOF with an N-rich ligand of urea as the modifier agent provides an extraordinary and ideal surface for growing Zif-8 through the creation of suitable coordination sites. The hybrid composite of Zn-MOF-NH2-ZIF-8@Cu was achieved after the successful loading of Cu nanoparticles on the surface of Zn-MOF-NH2-ZIF-8 and applied as a heterogenous and crystalline nanocatalyst in the A3-coupling reaction. The reusability of Zn-MOF-NH2-ZIF-8@Cu is also evaluated and the green chemistry standards are investigated.
Experimental
Materials and measurements
All the required materials were prepared from Merck and Aldrich company with a purity of over 99% and without any purification. The ligand of 2,5-bis(4-pyridyl)−3,4-diaza-2,4-hexadiene (4-bpdh) which was synthesized according to previously reported methods23.
A complete set of analytical techniques including Fourier transform infrared (FT-IR, TENSOR BRUKER 27), X-ray diffraction patterns (XRD, BRUKER D8-Focus Bragg − Brentano), Brunauer- Emmett- Teller (BET, BELSORP mini ΙΙ), field emission scanning electron microscope (FE-SEM, TESCAN MIRA3), Elemental mapping (ESD-MAP, TESCAN MIRA3), thermogravimetric analysis (TGA, STA504 (BAHR)), and inductively coupled plasma mass spectrometry (ICP-MS, SPECTRO ARCOS GERMANY) were applied to characterization and confirm the successful synthesis of Zn-MOF-NH2-ZIF-8@Cu catalyst in all stages of synthesis. Thin layer chromatography (TLC) with commercial silica gel-coated TLC plates (60 F254) was employed to verify the progress of the reaction.
Synthesis of Zn-MOF-NH2
Zn-MOF was synthesized according to the previously described method24. 500 mg of fully activated Zn-MOF was dispersed in 50 mL of deionized H2O under ultrasonic irradiation. 500 mg of urea was dissolved in 50 mL deionized H2O and then added dropwise to this suspension and was subsequently stirred for 24 h at 50 °C. The achieved solid Zn-MOF-NH2 was collected and washed with H2O several times to remove unreacted urea. At the end, it was dried at 70 °C under vacuum for 5 h.
Synthesis of Zn-MOF-NH2-ZIF-8
To prepare the Zn-MOF-NH2-ZIF-8 composite, certain amounts of Zn-MOF functionalized with urea (40 mg) were added to 10 mL MeOH and subjected to ultrasonic irradiation to well-dispersed. After 30 min of dispersion, 297 mg of Zn (NO3)2.6H2O in 25 mL MeOH was added to this suspension and sonicated for 8 h, followed by the addition of 328 mg of 2-methylimidazole in 25 mL MeOH. Then the mixed solution was stirred at room temperature for 6 h. After washing and filtration, the obtained product was completely dried at 100 °C under a vacuum.
Synthesis of Zn-MOF-NH2-ZIF-8@Cu
For the preparation of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst, 0.1 g of Zn-MOF-NH2-ZIF-8 was added to 20 ml dry MeOH and sonicate for 15 min to well-dispersed. Then a solution of Cu(OAc)2.2H2O (0.01 g) in 10 mL methanol was added to this mixture and allowed to agitated at 25 °C overnight. A solution of sodium borohydride (20 mg) in MeOH (12 mL) was added drop and drop and allowed stirring for another 5 h to convert Cu ions to Cu nanoparticles. Finally, the prepared Zn-MOF-NH2-ZIF-8@Cu was collected by centrifuging, washed with MeOH two times (2*10 mL), and dried at 70 °C in a vacuum oven for 8 h then were subjected to further studies.
General procedure to synthesize of Propargylamine derivatives
In a round bottom flask containing a certain amount of catalyst a mixture of aldehyde (1 mmol), amine (1.2 mmol), phenylacetylene (1.5 mmol), was poured and allowed to reaction was run under solvent-free conditions at 90 °C for a certain designed time. After finishing the reaction, 5 mL of ethyl acetate was added into the reaction vessel and then the catalyst was isolated from reaction mixtures via a simple separation and washed with ethyl acetate. The resulting remainder was further purified using short column chromatography on silica gel to afford the pure product.
Reusability of the catalyst
To assess the re-buildability of Zn-MOF-NH2-ZIF-8@Cu, at the termination of the first reaction, it was separated from the mixture reaction. Then, the isolated catalyst was used in the next reaction after three cleaning steps with EtOH. The process relating to catalyst recycling was successfully carried out six times with high catalytic efficiency.
Results and discussion
Synthesis and characterization
Metal-organic frameworks as up-and-coming materials are at the forefront of scientific research and the development of nanocatalysts. The superior advantages of MOFs allow them functionalization and controllable integration of catalytically active species to obtain a high-performance nanocatalyst25. Considering the strong requirement of today’s modern industry to use heterogeneous catalysts with excellent performance and reusability, Zn-MOF-NH2-ZIF-8@Cu nanocatalyst was designed and proposed. The detailed synthetic processes of Zn-MOF-NH2-ZIF-8@Cu are shown in Fig. 1. Initially, Zn-MOF was synthesized via the solvothermal interaction of terephthalic acid, 4-bpdh, and zinc salt within DMF. Subsequently, the free -NH2 of Zn-MOF reacted with the carbonyl group of urea to increase the number of suitable sites with electron-donating properties for the organization and extensive synthesis of ZIF-8 crystalline networks. Next, the growth of ZIF-8 crystals was successfully conducted on the surface of Zn-MOF-NH2 by the strong coordination interaction between nitrogen-containing groups and zinc centers. Finally, Cu nanoparticles were successfully loaded into Zn-MOF-NH2-ZIF-8 which has an ultrahigh surface area by an impregnation-reduction method using NaBH4. The characterization and analysis were done to prove the correct synthesis.
FTIR spectrum is a powerful technique to analyze and provide detailed information about functional groups in the structure of compounds. FTIR spectra of Zn-MOF, Zn-MOF-NH2, pure ZIF-8, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu are displayed in Fig. 2. Given that the crystal network of Zn-MOF consists of the ligands of pyridine 4-pbdh and carboxylate containing the free amine group, it is expected that the characteristic peaks corresponding to amine, carboxyl, hydroxyl, imine, alkene, and ether functional groups appear in the infrared spectrum. Therefore, the strong peaks that observed at regions 1258, 1375, 1429, 1627, and 3463 cm−1 can be attributed to the vibrational frequencies of bonds -C-N, -C-O-C, -C = C, -C = N, and -NH, respectively. Additionally, the spectrum showed peaks at 1334 and 1665 cm−1, which correspond to the asymmetric and symmetric stretching vibrations of the coordination carboxyl group in Zn-MOF. The FTIR analysis provides evidence that the O = C-O coordinated to Zn in the Zn-MOF structure, indicating the successful synthesis of Zn-MOF26,27. The spectra of Zn-MOF-NH2 had typical infrared peaks appointed to Zn-MOF, suggesting that the Zn-MOF structure is preserved intact after modification with urea. In pure ZIF-8, the five absorption bands at 2923, 1574, 1257, 771, and 692 cm−1 are represented the characteristic stretching vibrations C-H, C = N, C-N, Zn-O, and Zn-N, respectively28. FTIR spectrum of Zn-MOF-NH2-ZIF-8 and Zn-MOF-NH2-ZIF-8@Cu exclusively display FTIR spectra pure ZIF-8. This document proves that the applying of the functionalized metal-organic framework as a scaffold for the synthesis of crystals of ZIF-8 and then immobilizing of copper nanoparticles did not suffer damage to the coordination of the 2-methylimidazole ligand to the zinc (II) centers and thus the integrity of ZIF − 8. The presence of copper nanoparticles in the system did not change the position of the absorption bands and only slightly reduced the intensity of the peaks.
X-ray diffraction is a fundamental instrument for distinguishing crystalline and non-crystalline samples. To evidence the crystal structure of the designed heterogeneous catalyst, XRD patterns of Zn-MOF, Zn-MOF-NH2, pure ZIF-8, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu are prepared and presented in Fig. 3. The XRD pattern of parent Zn-MOF shows all the hypothesized features that substantiate its successful synthesis and high crystallinity. As seen in Fig. 3, the XRD pattern of urea-modified Zn-MOF persevered unaltered compared to the unmodified Zn-MOF, which indicates the high stability of the structure and the preservation of crystallinity after post-synthesis modification. As Fig. 3 indicates, the XRD pattern of Zn-MOF-NH2-ZIF-8 is at reliable compliance to the XRD pattern of the pristine ZIF-8, indicating that the incorporation of Zn-MOF-NH2 did not damage the crystal growth and purity of the Zif-8. Also, by not changing the XRD pattern of the Zn-MOF-NH2-ZIF-8@Cu, it can be concluded that the crystallinity of the nanocatalyst is still maintained with the stabilization of copper nanoparticles.
To demonstrate the positive effect of using a metal-organic framework as a template for the synthesis of another metal-organic framework with improved properties, the nitrogen adsorption-desorption isotherms (Fig. 4) and the corresponding BET surface area (Table 1) of all samples were calculated. The BET surface area of Zn-MOF-NH2 is higher than unmodified urea Zn-MOF which may be due to the presence of free -NH2 groups in the surface. It seems that amine groups with nitrogen atoms have effectively increased the absorption of nitrogen gas3. According to the IUPAC classification, Zn-MOF, Zn-MOF-NH2, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2@ZIF-8@Cu exhibited type I isotherm characteristic of microporous materials24,29,30. As can be seen, the BET surface area obtained for Zn-MOF-NH2-ZIF-8 is about 2050 m2g−1, which is much higher than the surface area for pure ZIF-8 (1241 m2g−1). These results prove that applying the urea-functionalized metal-organic framework provided an ideal substrate with a large number of sites for the successful growth of high-purity ZIF-8 crystals. After stabilizing the copper nanoparticles, the obvious decrease in the BET surface area of Zn-MOF-NH2@ZIF-8@Cu is observed (1660 m2g−1). This phenomenon is probably due to the high dispersion of Cu on the surface and the blocking of the cavities of ZIF-8.
The morphological details of Zn-MOF, pure ZIF-8, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu were studied by FE-SEM and depicted in Fig. 5. According to the FE-SEM image of Zn-MOF, the untouched metal-organic framework exhibits an uneven and porous surface. FE-SEM image in Fig. 4b clearly presents the characteristics of pristine ZIF-8 particles such as rhombic dodecahedral morphology, smooth surfaces, well-defined facets, corners, and sharp edges31. The FE-SEM image of Zn-MOF-NH2-ZIF-8 demonstrates that ZIF-8 crystals are well-loaded and have covered the entire surface. The successful growth of ZIF-8 on the surface of the support may be due to the strong interactions between the ZIF-8 crystals and the organic linkers containing nitrogen atoms in Zn-MOF-NH2. As can be seen, after the deposition of palladium, the porosity is still high and metal nanoparticles like spherical shapes have decorated the entire surface with uniform and homogeneous distribution.
In spectrum EDS, the presence of the copper peak along with carbon, nitrogen, oxygen, and zinc peaks as the main peaks of the prepared support structure indicates the successful immobilization of copper nanoparticles (Fig. 6). The uniform and homogeneous distribution of Cu nanoparticles throughout the Zn-MOF-NH2-ZIF-8@Cu composite was further confirmed by the corresponding element mapping analysis (Fig. 7).
The stability and non-decomposition of the structure in harsh temperature conditions is one of the remarkable advantages of the compounds, which makes it possible to use them in the industry. The thermal stability of the Zn-MOF-NH2-ZIF-8@Cu nanocatalyst at all stages of preparation was evaluated by the thermogravimetric analysis. Figure 8 shows the TGA curves of Zn-MOF, Zn-MOF-NH2, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu materials. According to TGA results, all samples showed a slight weight loss of about 100 °C, which is related to the loss of moisture adsorbed to the surface or the release of the solvent molecules remaining in the pores. A noteworthy weight loss at around 350 °C is observed for both unmodified and urea-functionalized MOF which is associated to the collapse of the Zn-MOF network due to -NH2 elimination and decomposition of the dicarboxylate linkers. The Zn-MOF-NH2-ZIF-8 exhibits a higher thermal stability and its chart trend is more moderate than that of Zn-MOF, and Zn-MOF-NH2, which further confirms that ZIF-8 was successfully loaded onto the surface of the prepared support. The TGA diagram of Zn-MOF-NH2-ZIF-8@Cu shows that the heterogeneous catalyst containing copper nanoparticles has provided excellent thermal stability with approximately maintenance of 85% of its weight up to 600 °C.
ICP-MS analysis is a fascinating and reliable tool to confirm the presence of copper nanoparticles as catalytically active species on the surface of the introduced template. According to the outcomes, the actual amount of Cu immobilized on the support was 3.87 Wt%.
Catalytic activity studies
Considering the importance of propargyl amines as biologically active skeletons32, pharmacologically relevant molecules33, and valuable precursors in synthesizing multifunctional amino derivatives34, developing new catalytic systems with desirable performance is currently a highly essential goal. Hence the catalytic activity of Zn-MOF-NH2-ZIF-8@Cu as a heterogeneous catalyst was evaluated for one-pot synthesis of propargyl amines via A3-coupling reaction. To explore the feasibility of the suggested method and find the best reaction conditions, the reaction of benzaldehyde, morpholine, and phenylacetylene was regarded as a simple model reaction.
Solvents usually notably influence the percentage of product production in heterogeneous catalytic reactions. The effect of the reaction solvent on the A3-coupling reaction was investigated by applying different solvents such as CHCl3, CH3CN, EtOH, and DMF using 10 mg catalyst, conducted for 15 min under reflux conditions. As can be seen, EtOH exhibited the highest efficiency between the tested solvents (Table 2). In order to steer the protocol towards green conditions, and reduce waste materials that may cause harm to researchers, laboratory personnel, and the environment, the reaction was evaluated under solvent-free conditions. It was observed that the desired product was achieved in 75% yield when the reaction was carried out without using solvent. By analyzing the results, the condition without solvent was chosen as the most suitable condition to continue the optimization experiments (Table 2).
Operating temperature as one of the affecting factors on the A3-coupling reaction was studied. The experimental outcomes demonstrated that with increasing temperature from 60 to 90 °C, the efficiency increased up to 97%, though at 25 °C, the influence of temperature on efficiency was insignificant. Thus, it can be concluded the 90 °C temperature favors the A3-coupling reaction using Zn-MOF-NH2-ZIF-8@Cu (Table 2).
The catalyst dosage is a very substantial parameter since it directly affects the number of catalytical active centers that can be available to organic reagents. No product was identified in the absence of the Zn-MOF-NH2-ZIF-8@Cu or Zn-MOF-NH2-ZIF-8 without Cu nanoparticles active centers. As reported in Table 2, the conversion of benzaldehyde was increased from 30 to 95% by increasing the amount of Zn-MOF-NH2-ZIF-8@Cu catalyst from 5 to 20 mg. By further increasing the amount of catalyst dose no positive impact was observed in the efficiency of the reaction, which may be due to the saturation of metal active sites. Hence, 20 mg of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst was selected as the optimum condition reaction for further investigations in the synthesis of other derivatives (Table 2).
To assess the efficacy, applicability, and generality of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst, various aromatic and aliphatic aldehydes with different groups (attractors and donors) were reacted with morpholine and phenylacetylene. With electron-withdrawing–containing aldehydes desired products were obtained in excellent yields of 90–98% after 10–35 min. The aldehyde with electron donor functionality demonstrates good efficiency of the corresponding product (85%) in reaction times between 45 and 50 min (Table 3). These results prove that Zn-MOF-NH2-ZIF-8@Cu is an excellent heterogeneous catalyst, that can be applied to synthesize propargyl amine derivatives without any co-catalyst.
Reusability of Zn-MOF-NH2-ZIF-8@Cu
In the design and development of heterogeneous catalysts, the reusability and durability of the catalysts is a substantial issue not only from the economically but also for environmental reasons. In this regard, the reusability of Zn-MOF-NH2-ZIF-8@Cu was examined. Six sequential experiments were designed, each of which used the recycled catalyst from the previous step. The recyclability tests showed that the efficiency decreased from 95 to 82% after six sequential runs, which can be concluded that the stability and performance of the Zn-MOF-NH2-ZIF-8@Cu was well maintained (Fig. 9). Comparison of the FTIR spectra, XRD patterns of the reused nanocatalyst with the fresh one unambiguously confirmed that the structure of the catalyst was preserved. Also, the FESEM image of the Zn-MOF-NH2-ZIF-8@Cu nanocatalyst showed the porosity of the structure and the good distribution of copper nanoparticles on the surface after 6 reuses (Fig. 10).
As mentioned, heterogeneous catalysts are extremely important in the industry due to their easy separation from the reaction mixture, reusability, and also the effective reduction of costs for the implementation of the process. To check the heterogeneity of the catalyst, the amount of Cu nanoparticles in the unused and re-used catalyst was determined by ICP-MS analysis. The amount of loaded Cu in Zn-MOF-NH2-ZIF-8@Cu was 3.53 Wt% in the recuperated catalyst, very near to the Cu content in the fresh catalyst (3.87 Wt%), which indicates a really partial copper leaching and high stability of the catalyst during the reaction process.
Plausible mechanism
According to the reported catalyzed A3-coupling reaction, a plausible mechanism for the synthesis of propargylamines derivatives using the Zn-MOF-NH2-ZIF-8@Cu nanocatalyst was suggested and is depicted in Fig. 11. The reaction begins with the interaction of the phenylacetylene with the catalyst through the coordination of the terminal C–H bond to the copper active sites on the surface of the Zn-MOF-NH2-ZIF-8@Cu followed by the formation of deprotonated copper-acetylide as an intermediate. At the same time, a condensation reaction between the aldehyde and secondary amine occurs to create an iminium ion. Then, with the nucleophilic addition of the iminium intermediate to copper-acetylide, the corresponding propargylamine is produced. The catalytic cycle continues with the regenerated catalyst until all raw materials are consumed.
Comparison
To reveal the worthiness and generality of the Zn-MOF-NH2-ZIF-8@Cu a comparative study between the performance of the mentioned system with other MOF-based heterogeneous counterparts previously reported for the synthesis of propargylamine derivatives was performed (Table 4). It can be concluded from the results that the Zn-MOF-NH2-ZIF-8@Cu system has superiority in terms of short reaction time, high yields of products, and higher TON/TOF values to some of the previously reported heterogeneous catalysts.
Conclusions
A novel nanomaterial Zn-MOF-NH2-ZIF-8@Cu with MOF-on-MOF architecture was successfully synthesized and incorporated as a heterogeneous nanocatalyst in the synthesis of propargylamine derivatives. The structure of the nanocatalyst from the first step to the end of the process was investigated, analyzed, and verified. When the A3 coupling reaction was carried out using Zn-MOF-NH2-ZIF-8@Cu with a catalyst dosage of 0.72 mol%, the yield of 95% could be achieved after 15 min of reaction under solvent-free conditions. This high-yield synthesis technique provides encouraging approaches to developing nanocatalysts with advanced functionalities in the industrial scale. The low activation energy, splendid target product selectivity, and solvent-free conditions of the established reaction system encouraged the development of green catalytic processes for organic reactions.
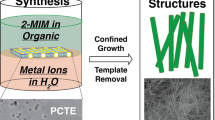
Schematic representation of the production of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst.
FT-IR spectra of Zn-MOF, Zn-MOF-NH2, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu.
XRD patterns of Zn-MOF, Zn-MOF-NH2, pure ZIF-8, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu.
Nitrogen adsorption-desorption isotherms of (a) Zn-MOF, (b) Zn-MOF-NH2, (c) pure ZIF-8, (d) Zn-MOF-NH2-ZIF-8, (f) Zn-MOF-NH2-ZIF-8@Cu.
FE-SEM images (a) Zn-MOF, (b) pure ZIF-8, (c) Zn-MOF-NH2-ZIF-8, and (d) Zn-MOF-NH2-ZIF-8@Cu.
The EDS analysis of the Zn-MOF-NH2-ZIF-8@Cu nanocatalyst.
The elemental mapping of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst.
The TGA curves of Zn-MOF, Zn-MOF-NH2, Zn-MOF-NH2-ZIF-8, and Zn-MOF-NH2-ZIF-8@Cu.
Recycling experiments of Zn-MOF-NH2-ZIF-8@Cu nanocatalyst in A3 coupling reaction.
(a) FT-IR image, (b) XRD pattern, and (c) FE-SEM image for the fresh and reused catalyst after catalysis.
The plausible mechanism of the A3 coupling reaction catalyzed by Zn-MOF-NH2-ZIF-8@Cu nanocatalyst.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Zarei, M., Saidi, K. & Sheibani, H. Preparation and investigation of catalytic activities of Cu-Ni nanoparticles supported on the biochar derived from pomegranate shells in the A3-coupling reactions, Biomass Convers. Biorefinery 1–13. (2022).
Kardan, M. et al. DABCO-based ionic liquid-modified magnetic nanoparticles supported gold as an efficient catalyst for A3 coupling reaction in water. J. Iran. Chem. Soc. 19, 3417–3430 (2022).
Kiani, A., Alinezhad, H. & Ghasemi, S. TMU-16-NH2 MOF modified with organic ligands for stabilization of copper nanoparticles: an efficient and crystalline heterogeneous catalyst for the synthesis of propargyl amines derivatives under green reaction condition. Appl. Organomet. Chem. e6676 (2022).
Hasan, K., Joseph, R. G., Patole, S. P. & Al-Qawasmeh, R. A. Development of magnetic Fe3O4-chitosan immobilized Cu (II) schiff base catalyst: an efficient and reusable catalyst for microwave assisted one-pot synthesis of propargylamines via A3 coupling. Catal. Commun. 174, 106588 (2023).
Jesin, I. & Nandi, G. C. Recent advances in the A3 coupling reactions and their applications. Eur. J. Org. Chem. 2019, 2704–2720 (2019).
Mirabedini, M., Motamedi, E. & Kassaee, M. Z. Magnetic CuO nanoparticles supported on graphene oxide as an efficient catalyst for A3-coupling synthesis of propargylamines. Chin. Chem. Lett. 26, 1085–1090 (2015).
Dhakshinamoorthy, A. & Garcia, H. Cascade reactions catalyzed by metal organic frameworks. ChemSusChem 7, 2392–2410 (2014).
Luz, I., i Xamena, F. X. L. & Corma, A. Bridging homogeneous and heterogeneous catalysis with MOFs:Click reactions with Cu-MOF catalysts. J. Catal. 276, 134–140 (2010).
Samanta, P. & Canivet, J. MOF-Supported heterogeneous catalysts for hydroformylation reactions: A minireview. ChemCatChem 16, e202301435 (2024).
Afaq, S. et al. Amide functionalized mesoporous MOF LOCOM-1 as a stable highly active basic catalyst for Knoevenagel condensation reaction. ACS Omega. 8, 6638–6649 (2023).
Ahmad, N., Younus, H. A., Chughtai, A. H. & Verpoort, F. Metal–organic molecular cages: applications of biochemical implications. Chem. Soc. Rev. 44, 9–25 (2015).
Zhang, Y. et al. Current research status of MOF materials for catalysis applications. Mol. Catal. 555, 113851 (2024).
Ahmad, N. et al. Synthesis of 2D MOF having potential for efficient dye adsorption and catalytic applications. Catal. Sci. Technol. 8, 4010–4017 (2018).
Chughtai, A. H., Ahmad, N., Younus, H. A., Laypkov, A. & Verpoort, F. Metal–organic frameworks: versatile heterogeneous catalysts for efficient catalytic organic transformations. Chem. Soc. Rev. 44, 6804–6849 (2015).
Akram, M. U. et al. An easy approach towards once a day sustained release dosage form using microporous Cu-MOFs as drug delivery vehicles. New. J. Chem. 48, 11542–11554 (2024).
Wang, Y., Chen, L., Hou, C. C., Wei, Y. S. & Xu, Q. Multiple catalytic sites in MOF-based hybrid catalysts for organic reactions. Org. Biomol. Chem. 18, 8508–8525 (2020).
Cirujano, F. G., i Xamena, F. X. L. & Corma, A. MOFs as multifunctional catalysts: One-pot synthesis of menthol from citronellal over a bifunctional MIL-101 catalyst. Dalt Trans. 41, 4249–4254 (2012).
García-García, P., Müller, M. & Corma, A. MOF catalysis in relation to their homogeneous counterparts and conventional solid catalysts. Chem. Sci. 5, 2979–3007 (2014).
Soman, S., Kulkarni, S., Vineeth, P., George, S. D. & Mutalik, S. Harnessing nanotechnology for breast cancer management: UiO-66 (NH2) metal-organic frameworks functionalized with Chitosan and folic acid for the efficient delivery of doxorubicin. Nano-Structures Nano-Objects. 40, 101360 (2024).
Kiani, A., Alinezhad, H. & Sadatmadani, R. Copper nanoparticles supported On magnetic functionalized chicken feather-derived activated carbon/metal–organic frameworks nanocomposite: as an efficient adsorbent for the removal Of methylene blue and a unique heterogeneous catalyst for the synthesis O. Appl. Organomet. Chem. 37, e7224 (2023).
Chappanda, K. N. et al. The quest for highly sensitive QCM humidity sensors: the coating of CNT/MOF composite sensing films as case study. Sens. Actuators B Chem. 257, 609–619 (2018).
Chai, L., Pan, J., Hu, Y., Qian, J. & Hong, M. Rational design and growth of MOF-on-MOF heterostructures. Small 17, 2100607 (2021).
Masoomi, M. Y., Stylianou, K. C., Morsali, A., Retailleau, P. & Maspoch, D. Selective CO2 capture in metal–organic frameworks with azine-functionalized pores generated by mechanosynthesis. Cryst. Growth Des. 14, 2092–2096 (2014).
Abbasi, A. R. & Hatami, S. Comparison of structure of nano zinc Metal–Organic frameworks upon uptake and release of phenazopyridine hydrochloride. J. Inorg. Organomet. Polym. Mater. 27, 1941–1949 (2017).
Mu, X., Zhong, Y., Jiang, T. & Cheang, U. K. Effect of solvation on the synthesis of MOF-based microrobots and their targeted-therapy applications. Mater. Adv. 2, 3871–3880 (2021).
Xu, W. Q. et al. A copper based metal-organic framework: synthesis, modification and VOCs adsorption. Inorg. Chem. Commun. 92, 1–4 (2018).
Arindita, N. P. Y. & Nandiyanto, A. B. D. The Structural Characterization of Cobalt-Based Metal-Organic Framework (Co-MOF) Using Fourier Transform Infrared (FTIR), Compliment. Copy 131. (2023).
Wang, J. et al. Exploration of the adsorption performance and mechanism of zeolitic imidazolate framework-8@ graphene oxide for Pb (II) and 1-naphthylamine from aqueous solution. J. Colloid Interface Sci. 542, 410–420 (2019).
Pan, Y., Liu, Y., Zeng, G., Zhao, L. & Lai, Z. Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system. Chem. Commun. 47, 2071–2073 (2011).
Safarifard, V. & Morsali, A. Influence of an amine group on the highly efficient reversible adsorption of iodine in two novel isoreticular interpenetrated pillared-layer microporous metal–organic frameworks. CrystEngComm 16, 8660–8663 (2014).
Kiani, A., Alinezhad, H. & Ghasemi, S. Embedded palladium nanoparticles on metal-organic framework/covalently sulfonated magnetic SBA-15 mesoporous silica composite: as a highly proficient nanocatalyst for Suzuki–Miyaura coupling reaction in amino acid-based natural deep eutectic solvent, Appl. Organomet. Chem. (n.d.) e6912.
Cammarata, J. R. et al. Single and double A3-coupling (aldehyde-amine-alkyne) reaction catalyzed by an air stable copper (I)-phosphole complex. Tetrahedron Lett. 58, 4078–4081 (2017).
Eagalapati, N. P., Rajack, A. & Murthy, Y. L. N. Nano-size ZnS: A novel, efficient and recyclable catalyst for A3-coupling reaction of propargylamines. J. Mol. Catal. Chem. 381, 126–131 (2014).
Ghosh, S., Biswas, K., Bhattacharya, S., Ghosh, P. & Basu, B. Effect of the ortho-hydroxy group of salicylaldehyde in the A3 coupling reaction: A metal-catalyst-free synthesis of Propargylamine. Beilstein J. Org. Chem. 13, 552–557 (2017).
Sarkar, F. K. et al. A sustainable avenue for the synthesis of propargylamines and benzofurans using a Cu-functionalized MIL-101 (cr) as a reusable heterogeneous catalyst. Sci. Rep. 13, 12908 (2023).
Huang, Y., Fan, X., Zhou, Y., Liu, S. & Li, Y. In situ growth of Cu (BDC) on microscale Cu-based carboxymethylcellulose fibers: A new strategy for constructing efficient catalysts for A3-coupling reactions. Appl. Organomet. Chem. 37, e7041 (2023).
Kiani, A., Alinezhad, H. & Ghasemi, S. Preparation immobilized Cu nanoparticles on modified Metal-Organic framework via linker design as an effective and highly efficient nanocatalyst for the synthesis of propargyl amines derivatives Preparation immobilized Cu nanoparticles on modified Metal-, (2022). https://doi.org/10.1080/10406638.2022.2056623
Cheraghian, M., Alinezhad, H. & Ghasemi, S. Post-synthetic modification of UIO-66-NH2 as a highly efficient and recyclable nanocatalyst in the three-component coupling (A3) reaction for the synthesis of Propargylamine derivatives. J. Organomet. Chem. 1002, 122903 (2023).
Acknowledgements
This research is supported by the research grant of the University of Mazandaran (number 33/76372).
Author information
Authors and Affiliations
Contributions
HA supported the expenses including the preparation of materials and analysis in the article, HA, and AK conceived the presented idea and contributed to the data curation, methodology, and validation. AK carried out the experiment. All authors reviewed the results and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kiani, A., Alinezhad, H. Synthesis and characterization of novel nanomaterial Zn-MOF-NH2-ZIF-8@Cu based on MOF-on-MOF architecture and Cu nanoparticle as a stable nanocatalyst in solvent-free A3 coupling reaction. Sci Rep 15, 17987 (2025). https://doi.org/10.1038/s41598-025-02953-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-02953-5