Abstract
To evaluate the role of high glucose variability (High-GV) in early pregnancy as a potential mediating factor between pre-pregnancy overweight/obesity and late-onset HDP (LoHDP), where effective preventive strategies remain limited. This multicenter retrospective study analyzed data from 802 pregnancies across 14 facilities. Pregnancies with a 75 g-OGTT performed by 20 weeks of gestation were included. Structural equation modeling (SEM) was used to evaluate direct and indirect effects of body mass index (BMI), High-GV, and covariates (e.g., age, ART, primiparity) on LoHDP. Overweight/obese women had significantly higher rates of High-GV (26.1 vs. 16.4%, p = 0.001) and LoHDP (17.6 vs. 7.9%, p < 0.001) compared to non-overweight/obese women. SEM revealed that BMI influenced LoHDP through both direct and indirect pathways. BMI had a direct effect on LoHDP (β = 0.20, p < 0.01), and an indirect effect mediated by High-GV, with BMI significantly associated with High-GV (β = 0.15, p < 0.01), and High-GV positively associated with LoHDP (β = 0.12, p < 0.01). In Non-GDM pregnancies, High-GV showed an even stronger association with LoHDP (β = 0.25, p < 0.001). This study identifies High-GV as a key mediator linking pre-pregnancy overweight/obesity to LoHDP. These findings suggest that targeting glucose variability in early pregnancy could mitigate LoHDP risk, particularly in overweight/obese women, regardless of GDM status. Future preventive strategies should integrate multifaceted approaches addressing maternal BMI and glucose regulation to improve maternal and neonatal outcomes.
Similar content being viewed by others
Introduction
Hypertensive disorders of pregnancy (HDP) include conditions in which hypertension develops after 20 weeks of gestation, with pre-pregnancy overweight and obesity (overweight/obesity) being remarkable risk factors1. Recent studies have increasingly highlighted distinct pathophysiological differences between early-onset HDP (EoHDP), occurring before 34 weeks of gestation, and late-onset HDP (LoHDP), occurring after 34 weeks2,3,4. Overweight/obesity is more strongly associated with LoHDP, the more frequent subtype, than with EoHDP2,5,6,7,8,9,10. Accordingly, addressing pre-pregnancy overweight/obesity is expected to reduce the risk of HDP, and particularly of LoHDP.
Currently, low-dose aspirin is the primary preventive strategy available during pregnancy and is mainly targeted at preventing EoHDP, the more severe subtype11,12,13. Conversely, for LoHDP, the preventive approach during pregnancy is to reduce gestational weight gain (GWG)14,15,16, although evidence supporting specific methods to achieve appropriate GWG is lacking16,17,18. LoHDP has a similar long-term impact on women’s health postpartum than EoHDP19. Accordingly, understanding the mechanism by which overweight/obesity contributes to LoHDP onset is crucial for developing comprehensive preventive measures available during pregnancy.
The present study focused on high glucose variability (High-GV), which is increasingly being recognized as a risk factor for cardiovascular diseases (CVD)20,21, mainly due to its link with endothelial dysfunction, a mechanism shared with HDP22,23,24. Our previous report concluded that High-GV in early pregnancy was independently associated with subsequent LoHDP development25. However, we had not yet clarified what contributes to the High-GV in early pregnancy.
Research on glucose metabolism has identified overweight/obesity as a significant contributor to increased High-GV26,27. Furthermore, High-GV has been established as an intermediate factor between overweight/obesity and CVD21,28,29. Therefore, this study aims to analyze whether High-GV could serve as a mediating factor in the relationship between prepregnant overweight/obesity and LoHDP in non-diabetic pregnant women.
Results
Baseline characteristics and outcomes
Table 1 presents the baseline characteristics and pregnancy outcomes of the overweight/obesity (n = 272) and non-overweight/obesity (n = 530) groups. The mean maternal age was similar between the two groups (33.3 ± 4.9 years vs. 32.6 ± 5.2 years, p = 0.073). The rate of primiparity was lower in overweight/obesity group (38.6 vs. 47.2%, p < 0.001). The rates of assisted reproductive technology (ART) use was similar between the groups, with no significant difference (11.4% vs. 14.6%, p = 0.215). However, a higher percentage of women in the overweight/obesity group were classified as High-GV and GDM (26.1 vs. 16.4%, p = 0.001 for High-GV; and 61.8 vs. 50.0%, p = 0.002 for GDM). Regarding HDP, the incidence of LoHDP was significantly higher in the overweight/obesity group compared to the Non- overweight/obesity group (17.6 vs. 7.9%, p < 0.001).
Association between overweight and glucose parameters
The distributions of 75 g-OGTT parameters were compared among overweight (red), normal-weight (blue), and underweignt (yellow) groups using the density plot (Fig. 1A). Overweight/obesity individuals demonstrated higher levels of fasting plasma glucose level (FPG), 1 h post-load plasma glucose (1hPG), and 2 h post-load plasma glucose (2hPG) compared to non-overweight/obesity groups. The magnitude of the initial-increase was also more pronounced in overweight/obesity participants, indicating a steeper glucose response following the oral glucose load. However, the subsequent-decrease did not show notable differences.
Relationships between OGTT parameters, glucose variability, and late-onset hypertensive disorders of pregnancy. (A), The X-axis of the density plot represents the value of 75 g-OGTT parameters. Red, blue, and yellow parts represent the overweight, normal-weight, and underweight, respectively. (B,C) Each panel visually represents the hypothesized relationships in the SEMs. Model 1 presents the SEM results exploring the relationships between OGTT glucose parameters (FPG, 1hPG, 2hPG) and LoHDP, with BMI as an influencing factor (B). Model 2 the relationship between BMI and LoHDP from the perspective of GV (C). The standardized regression coefficients (β) indicate the strength and direction of the relationships, while the asterisks highlight the statistical significance of these relationships. Significance levels are denoted by asterisks: *p < 0.05, **p < 0.01, ***p < 0.001. BMI body mass index, FPG fasting plasma glucose, 1hPG 1-h post-load plasma glucose, 2hPG 2-h post-load plasma glucose, LoHDP late-onset hypertensive disorders of pregnancy.
Structural equation model
Model 1 explored the relationships between OGTT glucose parameters (FPG, 1hPG, 2hPG) and LoHDP, with body mass index (BMI) as an influencing factor using structural equation model (SEM) analyses (Fig. 1B and Table S1). BMI exhibited a significant direct effect on FPG (β = 0.24, p < 0.001) and 1hPG (β = 0.14, p < 0.001). Positive relationships were observed between FPG, 1hPG, and 2hPG. Interestingly, FPG did not show a significant direct effect on LoHDP, while 1hPG had a positive association with LoHDP (β = 0.18, p < 0.01), and 2hPG exhibited a negative association (β = -0.13, p = 0.003). Additionally, BMI had a significant direct effect on LoHDP (β = 0.20, p < 0.001), independent of glucose parameters. Model 2 re-evaluates the relationship between BMI and LoHDP from the perspective of GV (Fig. 1C and Table S1). The SEM results align with and support the findings from Model 1. BMI showed a significant direct effect on initial-increase (β = 0.14, p < 0.001), which in turn strongly predicted subsequent-decrease (β = 0.58, p < 0.001). Subsequent-decrease also had a significant positive association with LoHDP (β = 0.14, p < 0.001). These results indicate that BMI influences LoHDP indirectly through its effects on initial-increase and subsequent-decrease, in addition to its direct effect on LoHDP (β = 0.20, p < 0.001).
Model 3 evaluates the relationship between BMI and LoHDP by conceptualizing HighGV, defined as the condition where both the initial-increase and subsequent-decrease are maximized (Fig. 2A and Table S2). The SEM results indicate that BMI influences LoHDP through both direct and indirect pathways. BMI had a direct effect on LoHDP (β = 0.20, p < 0.001) and an indirect effect mediated by High-GV, where BMI influenced High-GV (β = 0.15, p < 0.001), and High-GV was positively associated with LoHDP (β = 0.12, p < 0.001). Model 4 expands on the findings from Model 3 by incorporating additional covariates, including age, ART, and primiparity (Fig. 2B and Table S2). The SEM results confirm that the relationships identified in Model 3 remain consistent even after adjusting for these factors. BMI continues to influence LoHDP through both direct and indirect pathways. BMI retains a direct effect on LoHDP (β = 0.21, p < 0.001) and an indirect effect mediated by High-GV, with a significant association between BMI and High-GV (β = 0.15, p < 0.001) and between High-GV and LoHDP (β = 0.12, p < 0.001).
Direct and indirect effects of BMI and glucose variability on late-onset hypertensive disorders of pregnancy. Each panel visually represents the hypothesized relationships in the SEMs. Model 3 illustrating the relationships between BMI,FPG, High-GV, and LoHDP (A). Model 4 includes Age, ART, and Primi into the Model 3 (B). The standardized regression coefficients (β) indicate the strength and direction of the relationships, while the asterisks highlight the statistical significance of these relationships. Significance levels are denoted by asterisks: *p < 0.05, **p < 0.01, ***p < 0.001. BMI body mass index, FPG fasting plasma glucose, High-GV high glycemic variability, LoHDP late-onset hypertensive disorders of pregnancy, ART assisted reproductive technology, Primi primiparity.
Table S3 presents the fit indices for each model. The fit indices indicate that every model provided a good fit to the data (χ2/df ≤ 4.50, RMSEA ≤ 0.07, NFI ≥ 0.95, IFI ≥ 0.97, TLI ≥ 0.93, CFI ≥ 0.97, GFI = 0.99, AGFI ≥ 0.96, and SRMR ≤ 0.03), further supporting the robustness of the proposed SEM framework. These results underscore the importance of considering multiple components when analyzing factors influencing HDP, with High-GV emerging as a significant mediator between BMI and LoHDP.
Subgroup-analysis
Table 2 summarizes the results of the subgroup analysis stratified by GDM status. In the Non-GDM subgroup, the frequency of High-GV was significantly higher in the overweight/obesity group compared to the non-overweight/obesity group. Specifically, 25 out of 104 participants in the overweight/obesity group (24.0%) had High-GV, while in the non-overweight/obesity group, the frequency was 25 out of 265 (9.4%). Additionally, among non-overweight/obesity individuals, 20.0% of participants with High-GV developed LoHDP, compared to 6.3% of those without High-GV (p = 0.013). Similarly, in the overweight/obesity group, 44.0% of participants with High-GV developed LoHDP, compared to 8.9% of those without High-GV (p < 0.001). In the GDM subgroup, differences were observed based on overweight/obesity status. Among non-overweight/obesity individuals, participants with High-GV had a higher frequency of LoHDP (16.1% [10/62]) compared to those without High-GV (5.9% [12/203]; p = 0.011). In contrast, among overweight/obesity individuals with GDM, no significant difference in LoHDP frequency was observed between those with High-GV (15.2% [7/46]) and those without High-GV (21.7% [10/46]; p = 0.583).
Figures S1 present the SEM analyses focusing on Non-GDM individuals, evaluating the relationships among BMI, HighGV, and LoHDP as in the analyses conducted for Fig. 2. These results indicate that the findings observed in Fig. 2 remain consistent in the Non-GDM subgroup. BMI retains a significant direct effect on HighGV (β = 0.20, p < 0.01), and HighGV continues to exhibit a strong positive association with LoHDP (β = 0.25, p < 0.001), demonstrating an even stronger trend compared to the broader cohort analyzed in Fig. 2.
Discussion
This study contributes to the existing body of knowledge regarding the relationship between maternal characteristics and HDP. This is the first study to evaluate High-GV in early pregnancy as a mediating factor between pre-pregnancy overweight/obesity and development of LoHDP. By identifying High-GV as a critical intermediary, this study provides novel insights into mechanisms linking maternal pre-pregnancy BMI with HDP, and particularly LoHDP. Our findings underscore the importance of timely glucose regulation in pregnant women with higher BMI to mitigate the risk of LoHDP.
This study highlighted the complex interplay between the maternal BMI, High-GV, LoHDP, Age, ART, and primiparity. The inclusion of each additional variable provides a more comprehensive understanding of these relationships, with BMI consistently showing a strong influence on High-GV and LoHDP across all models. These results are consistent with those of previous research8,9,10. In non-pregnant individuals, high GV has been shown to contribute to the development of CVD30. Given that both CDV and HDP share vascular endothelial dysfunction as a central pathological feature31, it is plausible that high GV during pregnancy is also associated with the onset of HDP. Additionally, we demonstrated an association between High-GV and LoHDP even in non-GDM individuals. Nevertheless, GDM is reported to be associated with an increased risk of HDP32,33, and this may be explained by the higher prevalence of GDM observed in women with High-GV in this study. However, an interesting observation in our study was the lack of association between High-GV and LoHDP among overweight/obese women with GDM. This may be explained by distinct vascular and metabolic adaptations in this subgroup compared to non-overweight/obese women with GDM34. The coexistence of GDM with overweight/obesity leads to pre-existing vascular remodeling, potentially diminishing endothelial sensitivity to GV34. Consequently, the incidence of LoHDP remains consistent regardless of GV levels, suggesting that chronic endothelial dysfunction in overweight/obese women with GDM may blunt the vascular impact of GV.
LoHDP is also associated with lipid metabolism35,36. Although our study did not include lipid metabolism parameters, BMI and LoHDP had significant direct effects independent of High-GV, suggesting that factors related to lipid metabolism may be involved. Furthermore, the association between primiparity or ART and LoHDP was consistent with that of previous studies1,37,38.
Althogh women diagnosed with GDM before 20 weeks of pregnancy have poorer perinatal outcomes39, the evidence on how to evaluate and manage GDM when diagnosed at this stage is still limited, which is why OGTT is not widely recommended in early pregnancy40. However, from the perspective of LoHDP prevention, our study suggests that assessing and addressing GV evaluation could be a beneficial strategy. Previously, the mediating factors linking overweight/obesity and HDP remained unclear, limiting preventive strategies for HDP in overweight/obese pregnant women to weight management before or during pregnancy41,42,43,44. However, our study identified GV as a potential mediator, suggesting that a multifaceted, seamless preventive approach targeting glycemic control from the preconception period through pregnancy may now be explored.
This study has several strengths. First, the evaluation of a large and diverse dataset from multiple centers enhances the generalizability of the findings. The inclusion of data from both tertiary centers and private maternity facilities ensures a comprehensive representation of the population, accounting for various socioeconomic and healthcare backgrounds. Moreover, the use of SEM allowed for a nuanced exploration of the complex interrelationships among maternal characteristics and pregnancy outcomes45. Lastly, the rigorous exclusion criteria ensured that the study focused specifically on pregnant women without diabetes mellitus (DM), eliminating potential confounding effects of pre-existing DM and chronic hypertension. This targeted approach strengthens the validity of the conclusions drawn regarding the impact of BMI and High-GV on HDP.
Limitations
Although this study offers valuable insights, several limitations should be considered when interpreting the results. First, the retrospective design may have introduced selection bias and analytical methods were applied, the bias inferent to retrospective studies cannot be completely ruled out. Second, relying on self-reported maternal height and weight to calculate pre-pregnancy BMI could introduce measurement errors. However, participants were weighed in the first trimester; therefore, discrepancy between self-reported and actual values was likely to be minimal. Third, the study population included only women with random blood glucose (RBG) ≥ 100 mg/dL in Japan, which may limit the generalizability of the findings to other populations or ethnic groups. The definition and clinical significance of High-GV may vary across populations due to racial and ethnic differences in glucose metabolism and pregnancy outcomes. Despite these limitations, the study makes an important contribution to improve our understanding of the relationship between maternal BMI, GV, and HDP, and highlights areas for further research and potential intervention.
Conclusion
This study advances our understanding of the pathophysiological mechanisms linking maternal overweight/obesity to HDP, particularly LoHDP, through the intermediary role of High-GV. These findings provide a basis for more targeted and efficient preventive strategies, ultimately contributing to better maternal and neonatal health outcomes through the development of seamless and multifaceted preventive measures from pre- to post-conception. However, as the study included only women with RBG ≥ 100 mg/dL, these conclusions should be interpreted cautiously and validated in broader populations.
Methods
Study design and participants
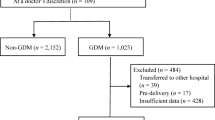
This multicenter retrospective study used electronic data from two tertiary centers (Nagoya University Hospital and TOYOTA Memorial Hospital, Aichi, Japan) and 12 private maternity facilities (Kishokai Medical Corporation, Aichi, Japan). The dataset included women aged ≥ 15 years who gave birth between 2009 and 2019, with available data from before 20 weeks of gestation. The study specifically included pregnancies where an 75 g-OGTT was performed by 20 weeks. The exclusion criteria were women with multiple pregnancies, stillbirth before 20 weeks, chronic hypertension, pre-existing DM, overt-DM, and missing data on blood pressure before 20 weeks and pre-pregnant BMI. Despite chronic hypertension being a subtype of HDP, this group was excluded to specifically assess the association between GV and the development of HDP (Fig. 3A).
Characteristics of study participants. (A) Flowchart of study enrolment. Among 30,393 individuals, 802 were eligible after excluding 29,591 individuals. (B) Schematic representation of OGTT results and definition of initial-increase and subsequent-decrease. Initial-increase was defined as the increase in in blood glucose levels from FPG to 1hPG. Subsequent-decrease was defined as the decrease in blood glucose levels from 1 to 2hPG. (C) Three-dimensional bar chart representing the frequency of overweight and non-overweight (n = 802). The frequencies of overweight (red-cuboid) and non-overweight (blue-cuboid) are represented on the Z axis, with the classified initial-increase and subsequent-decrease forming the axes of X and Y, respectively. High-GV was defined as individuals with a significant initial-increase of ≥ 74 mg/dL with a notable subsequent-decrease of ≥ 28 mg/dL. BMI body mass index, DM diabetes mellitus, RBG random blood glucose level, wk weeks of gestation, OGTT oral glucose tolerance test, FPG fasting plasma glucose, 1hPG 1-h post-load plasma glucose, 2hPG 2-h post-load plasma glucose, High-GV high glycemic variability.
This study was approved by the ethics board of Nagoya University Hospital (approval number: 2015–0415) following the guidelines of the Declaration of Helsinki and was conducted according to the STROBE guidelines. The requirement for informed consent was waived because of the retrospective study design.
Definitions
HDP was defined as the development of new hypertension (systolic blood pressure of ≥ 140 mmHg or diastolic blood pressure of ≥ 90 mmHg) after 20 weeks of gestation1,46. HDP was classified as LoHDP if it occurred at ≥ 34 weeks, and EoHDP if it occurred at < 34 weeks47,48. GV was evaluated using initial-increase and subsequent-decrease (Fig. 3B). The initial-increase was defined as the increase in blood glucose levels from FPG to the 1hPG, and the “subsequent-decrease” was defined as the decrease from 1hPG to 2hPG25,49. High-GV was defined as initial-increas of ≥ 74 mg/dL (4.1 mmol/L) followed by a notable subsequent-decrease of ≥ 28 mg/dL (1.6 mmol/L), as shown in (Fig. 3C)25,49. These cutoff values were determined based on the tertiles of each respective change.
Self-reported maternal height and body weight were used to calculate pre-pregnant BMI (kg/m2). In this study, we defined individuals with a BMI of < 18.5 kg/m2 as underweight, and of ≥ 23.0 kg/m2 as overweight/obesity, in accordance with the World Health Organization criteria for the Asian population50. ART included in vitro fertilization or intracytoplasmic sperm injection.
Diagnosis of overt-DM/GDM
Pregnant women were screened for RBG levels at approxymately 12–14 weeks of gestation, according to the guidelines; the cut-off value was set at RBG of ≥ 100 mg/dL (5.6 mmol/L)17,51. For screening-positive patients, the FPG level was measured usually within 1–2 weeks, and patients with FPG of ≥ 126 mg/dL (7.0 mmol/L) were diagnosed as overt-DM (excluded from this analysis). For non-overt-DM individuals, the 75 g-OGTT was conducted with cutoff values of ≥ 92 mg/dL (5.1 mmol/L) for FPG, ≥ 180 mg/dL (10 mmol/L) for 1hPG, and ≥ 153 mg/dL (8.5 mmol/L) for 2hPG17. GDM was diagnosed if at least one of the three aforementioned glycemic levels was above the threshold value17. In Japan, oral diabetes medications are contraindicated for pregnant women, so treatment was by diet and insulin injections.
Outcome
The primary outcome is to determine whether GV in early pregnancy serves as a mediating factor in the relationship between pre-pregnancy BMI and the development of LoHDP in non-diabetic pregnant women.
Statistical analysis
Baseline characteristics of overweight/obese and non-overweight/obese pregnant women were compared using the independent samples t-test and Chi-square test. Based on the clinical relevance, an initial SEM was constructed to synthesize path-reflective relationships between variables and to explore the direct and indirect effects of overweight status and GV on LoHDP. The analysis included the calculation of standardized beta coefficients (β) to quantify the strength and direction of these relationships, thereby providing a comprehensive understanding of both direct and mediated pathways within the model. The following ideal-fit-indices were used to evaluate the model: χ2/df < 3 [acceptable < 5], a root mean square error of approximation (RMSEA) < 0.05 [acceptable ≤ 0.08], normed fit index (NFI) ≥ 0.95 [acceptable ≥ 0.90], incremental fit index (IFI) ≥ 0.95 [acceptable ≥ 0.90], Tucker–Lewis index (TLI) ≥ 0.95 [acceptable ≥ 0.90], comparative fit index (CFI) ≥ 0.95 [acceptable ≥ 0.90], goodness of fit index (GFI) ≥ 0.95 [acceptable ≥ 0.90], adjusted goodness-of-fit index (AGFI) ≥ 0.90 [acceptable ≥ 0.85], and standardized root mean square residual (SRMR) < 0.05 [acceptable ≤ 0.08]52,53,54,55,56.
Data availability
The raw data underlying the conclusions of this article can be accessed upon reasonable request, subject to the approval of Nagoya University, TOYOTA Memorial Hospital, and Kishokai Medical Corporation. Contact information: Sho Tano, tano.sho.v4@f.mail.nagoya-u.ac.jp.
References
Wu, P., Green, M. & Myers, J. E. Hypertensive disorders of pregnancy. BMJ 381, e071653 (2023).
Bicocca, M. J., Mendez-Figueroa, H., Chauhan, S. P. & Sibai, B. M. Maternal obesity and the risk of early-onset and late-onset hypertensive disorders of pregnancy. Obstet. Gynecol. 136, 118–127 (2020).
Vatten, L. J. & Skjærven, R. Is pre-eclampsia more than one disease?. BJOG Int. J. Obstet. Gynaecol. 111, 298–302 (2004).
Egbor, M., Ansari, T., Morris, N., Green, C. & Sibbons, P. Maternal medicine: Morphometric placental villous and vascular abnormalities in early-and late-onset pre-eclampsia with and without fetal growth restriction. BJOG Int. J. Obstet. Gynaecol. 113, 580–589 (2006).
Wang, W. et al. Genetically predicted obesity causally increased the risk of hypertension disorders in pregnancy. Front. Cardiovasc. Med. 9, 888982 (2022).
Ornaghi, S. et al. Influencing factors for late-onset preeclampsia. J. Matern. Fetal Neonatal Med. 26, 1299–1302 (2013).
Gasse, C., Boutin, A., Demers, S., Chaillet, N. & Bujold, E. Body mass index and the risk of hypertensive disorders of pregnancy: the great obstetrical syndromes (GOS) study. J. Matern. Fetal Neonatal Med. 32, 1063–1068 (2019).
Ferrazzi, E., Stampalija, T. & Aupont, J. E. The evidence for late-onset pre-eclampsia as a maternogenic disease of pregnancy. Fetal Maternal Med. Rev. 24, 18–31 (2013).
Sarno, L. et al. The impact of obesity on haemodynamic profiles of pregnant women beyond 34 weeks’ gestation. Pregnancy Hyperten. 22, 191–195 (2020).
Valensise, H., Vasapollo, B., Gagliardi, G. & Novelli, G. P. Early and late preeclampsia: two different maternal hemodynamic states in the latent phase of the disease. Hypertension 52, 873–880 (2008).
Roberge, S. et al. The role of aspirin dose on the prevention of preeclampsia and fetal growth restriction: systematic review and meta-analysis. Am. J. Obstet. Gynecol. 216 (110–120), e116. https://doi.org/10.1016/j.ajog.2016.09.076 (2017).
Costantine, M. M. & Cleary, K. Pravastatin for the prevention of preeclampsia in high-risk pregnant women. Obstet. Gynecol. 121, 349–353. https://doi.org/10.1097/AOG.0b013e31827d8ad5 (2013).
Groom, K. M. et al. STRIDER NZAus: a multicentre randomised controlled trial of sildenafil therapy in early-onset fetal growth restriction. BJOG 126, 997–1006. https://doi.org/10.1111/1471-0528.15658 (2019).
Mustafa, H. J. et al. Gestational weight gain below instead of within the guidelines per class of maternal obesity: a systematic review and meta-analysis of obstetrical and neonatal outcomes. Am. J. Obstet. Gynecol. MFM 4, 100682 (2022).
Truong, Y. N., Yee, L. M., Caughey, A. B. & Cheng, Y. W. Weight gain in pregnancy: does the institute of medicine have it right?. Am. J. Obstet. Gynecol. 212, 362.e1-362.e8 (2015).
National Research Council Committee to Reexamine. Weight Gain During Pregnancy: Reexamining the Guidelines (National Academies Press, 2009).
Itakura, A. et al. Guidelines for obstetrical practice in Japan: Japan society of obstetrics and gynecology and Japan association of obstetricians and gynecologists 2020 edition. J. Obstet. Gynaecol. Res. 49, 5–53. https://doi.org/10.1111/jog.15438 (2023).
Wang, S., Rexrode, K. M., Florio, A. A., Rich-Edwards, J. W. & Chavarro, J. E. Maternal mortality in the United States: Trends and opportunities for prevention. Annu. Rev. Med. 74, 199–216. https://doi.org/10.1146/annurev-med-042921-123851 (2023).
Grandi, S. M., Reynier, P., Platt, R. W., Basso, O. & Filion, K. B. The timing of onset of hypertensive disorders in pregnancy and the risk of incident hypertension and cardiovascular disease. Int. J. Cardiol. 270, 273–275. https://doi.org/10.1016/j.ijcard.2018.06.059 (2018).
Jenkins, D. J. A. et al. Glycemic index, glycemic load, and cardiovascular disease and mortality. N. Engl. J. Med. 384, 1312–1322. https://doi.org/10.1056/NEJMoa2007123 (2021).
Kim, M. K. et al. Associations of variability in blood pressure, glucose and cholesterol concentrations, and body mass index with mortality and cardiovascular outcomes in the general population. Circulation 138, 2627–2637 (2018).
Roberts, J. M. & Hubel, C. A. The two stage model of preeclampsia: variations on the theme. Placenta 30, 32–37 (2009).
Monnier, L. et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 295, 1681–1687 (2006).
Di Flaviani, A. et al. Impact of glycemic and blood pressure variability on surrogate measures of cardiovascular outcomes in type 2 diabetic patients. Diabetes Care 34, 1605–1609 (2011).
Tano, S. et al. Evaluating glucose variability through OGTT in early pregnancy and its association with hypertensive disorders of pregnancy in non-diabetic pregnancies: a large-scale multi-center retrospective study. Diabetol. Metab. Syndr. 15, 123. https://doi.org/10.1186/s13098-023-01103-z (2023).
Norton, L., Shannon, C., Gastaldelli, A. & DeFronzo, R. A. Insulin: The master regulator of glucose metabolism. Metabolism 129, 155142 (2022).
Klimontov, V. V. & Semenova, J. F. Glucose variability in subjects with normal glucose tolerance: relations with body composition, insulin secretion and sensitivity. Diabetes Metab. Syndr. 16, 102387 (2022).
Buscemi, S. et al. Effects of hypocaloric diets with different glycemic indexes on endothelial function and glycemic variability in overweight and in obese adult patients at increased cardiovascular risk. Clin. Nutr. 32, 346–352 (2013).
Buscemi, S. et al. Glycaemic variability using continuous glucose monitoring and endothelial function in the metabolic syndrome and in type 2 diabetes. Diabet. Med. 27, 872–878 (2010).
Zhou, Z., Sun, B., Huang, S., Zhu, C. & Bian, M. Glycemic variability: adverse clinical outcomes and how to improve it?. Cardiovasc. Diabetol. 19, 1–14 (2020).
Powe, C. E., Levine, R. J. & Karumanchi, S. A. Preeclampsia, a disease of the maternal endothelium: the role of antiangiogenic factors and implications for later cardiovascular disease. Circulation 123, 2856–2869. https://doi.org/10.1161/CIRCULATIONAHA.109.853127 (2011).
Coustan, D. R., Lowe, L. P., Metzger, B. E. & Dyer, A. R. The hyperglycemia and adverse pregnancy outcome (HAPO) study: paving the way for new diagnostic criteria for gestational diabetes mellitus. Am. J. Obstet. Gynecol. 202, 654.e1-654.e6 (2010).
Sweeting, A., Wong, J., Murphy, H. R. & Ross, G. P. A clinical update on gestational diabetes mellitus. Endocr. Rev. 43, 763–793 (2022).
Cornejo, M. et al. Gestational diabesity and foetoplacental vascular dysfunction. Acta Physiol. (Oxf.) 232, e13671. https://doi.org/10.1111/apha.13671 (2021).
Yao, M. et al. Identification of biomarkers for preeclampsia based on metabolomics. Clin. Epidemiol. 14, 337–360 (2022).
Gallos, I. et al. Pre-eclampsia is associated with, and preceded by, hypertriglyceridaemia: a meta-analysis. BJOG Int. J. Obstet. Gynaecol. 120, 1321–1332 (2013).
You, S.-H. et al. Population-based trends and risk factors of early- and late-onset preeclampsia in Taiwan 2001–2014. BMC Pregnancy Childbirth 18, 199. https://doi.org/10.1186/s12884-018-1845-7 (2018).
Hutcheon, J. A., Lisonkova, S. & Joseph, K. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 25, 391–403 (2011).
Liu, B. et al. Early diagnosed gestational diabetes mellitus is associated with adverse pregnancy outcomes: a prospective cohort study. J. Clin. Endocrinol. Metab. 105, e4264–e4274 (2020).
Davidson, K. W. et al. Screening for gestational diabetes: US preventive services task force recommendation statement. JAMA 326, 531–538 (2021).
Tano, S. et al. Visualizing risk modification of hypertensive disorders of pregnancy: development and validation of prediction model for personalized interpregnancy weight management. Hypertens. Res. 48, 884–893. https://doi.org/10.1038/s41440-024-02024-8 (2025).
Tano, S. et al. Annual body mass index gain and risk of hypertensive disorders of pregnancy in a subsequent pregnancy. Sci. Rep. 11, 22519. https://doi.org/10.1038/s41598-021-01976-y (2021).
Morisaki, N. et al. Pre-pregnancy BMI-specific optimal gestational weight gain for women in Japan. J. Epidemiol. 27, 492–498 (2017).
Santos, S. et al. Impact of maternal body mass index and gestational weight gain on pregnancy complications: an individual participant data meta-analysis of European, North American and Australian cohorts. BJOG Int. J. Obstet. Gynaecol. 126, 984–995 (2019).
Ives, C. W., Sinkey, R., Rajapreyar, I., Tita, A. T. N. & Oparil, S. Preeclampsia—Pathophysiology and clinical presentations. J. Am. Coll. Cardiol. 76, 1690–1702. https://doi.org/10.1016/j.jacc.2020.08.014 (2020).
Hypertension in Pregnancy. Report of the American college of obstetricians and gynecologists’ task force on hypertension in pregnancy. Obstet. Gynecol. 122, 1122–1131. https://doi.org/10.1097/01.Aog.0000437382.03963.88 (2013).
Watanabe, K. et al. Outline of the new definition and classification of “hypertensive disorders of pregnancy (HDP)”; a revised JSSHP statement of 2005. Hypertens. Res. Pregnancy 6, 33–37 (2018).
Tranquilli, A. L., Brown, M. A., Zeeman, G. G., Dekker, G. & Sibai, B. M. The definition of severe and early-onset preeclampsia. Statements from the International society for the study of hypertension in pregnancy (ISSHP). Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health 3, 44–47 (2013).
Kasuga, Y. et al. Perinatal outcomes of diet therapy in gestational diabetes mellitus diagnosed before 24 gestational weeks. Nutrients 16, 1553 (2024).
Consultation, W. H. O. E. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363, 157–163. https://doi.org/10.1016/S0140-6736(03)15268-3 (2004).
Kyozuka, H. et al. Adverse obstetric outcomes in early-diagnosed gestational diabetes mellitus: The Japan environment and children’s study. J. Diabetes Investig. 12, 2071–2079 (2021).
Xia, Y. & Yang, Y. RMSEA, CFI, and TLI in structural equation modeling with ordered categorical data: The story they tell depends on the estimation methods. Behav. Res. Methods 51, 409–428 (2019).
Kline, R. B. Principles and Practice of Structural Equation Modeling (Guilford Publications, 2023).
Bentler, P. M. EQS Structural Equations Program Manual (Multivariate software Encino, 1995).
Browne, M. W. & Cudeck, R. Alternative ways of assessing model fit. Sociol. Methods Res. 21, 230–258 (1992).
Bentler, P. M. Comparative fit indexes in structural models. Psychol. Bull. 107, 238 (1990).
Acknowledgements
We thank for Rho Emoto at Department of Biostatistics, Nagoya University Graduate School of Medicine for constuructive comments. During the preparation of this work the authors used DeepL and ChatGPT4 for translation and academic editing. After using these tools, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication. We also thank Editage (www.editage.jp) for English language editing.
Funding
ST was supported by the Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research (23K19800 and 25K21070) in the decision to submit the article for publication. The funder had no role in study including study design, data collection, analysis, interpretation or writing. The corresponding authors had full access to all the data in this study and had final responsibility for the decision to publish.
Author information
Authors and Affiliations
Contributions
S.T., T.K., and T.I. conceived the study. S.T. and F.K. performed the statistical analyses. All authors collected and interpreted the clinical data. S.T. and T.K. drafted the manuscript. All authors contributed to the interpretation of the results and the approval of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Ethical approval was granted prior to study start by the ethics board of Nagoya University Hospital (Approval Number: 2015–0415) on 4th March 2016, with amendments on 24th June 2021 (22550) and 5th September 2022 (26473).
Consent for publication
The requirement for informed consent was waived because of the retrospective study design.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tano, S., Kotani, T., Inamura, T. et al. Glucose variability as a key mediator in the relationship between pre-pregnancy overweight/obesity and late-onset hypertensive disorders of pregnancy. Sci Rep 15, 18123 (2025). https://doi.org/10.1038/s41598-025-02965-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-02965-1
Keywords
This article is cited by
-
Synergistic use of 1,5-AG and HbA1c for early prediction of gestational diabetes: capturing BMI-dependent glycemic phenotypes
Archives of Gynecology and Obstetrics (2026)