Abstract
This study investigates the impact of a novel compound biomass surfactant on the molecular structure and functional group characteristics of Huainan Liuzhuang coal via Fourier Transform Infrared (FTIR) spectroscopy. The experimental results reveal marked differences between raw, distilled water-treated, and surfactant-modified samples, particularly in the transformation of hydroxyl, aliphatic, and oxygen-containing functional groups: (1) The coal’s IR spectra are categorized into four key absorption bands, representing hydroxyl groups, aliphatic hydrocarbons, oxygen-containing functional groups, and aromatic hydrocarbons. (2) Surfactant treatment reduces the total hydroxyl group content but increases hydroxyl-hydrogen bonding, enhancing coal’s structural stability. (3) The aliphatic side chains in modified coal become shorter, with the 3:7 compound ratio yielding the highest aliphatic group content. (4) C-O-C stretching vibrations condense into aryl ethers, increasing aromatic ring condensation, particularly at the 3:7 ratio. (5) Aromatic hydrocarbon diversity decreases as side chains detach from benzene rings, with ortho-trisubstituted structures (3 H) becoming more prominent. The outcomes demonstrate that compound biomass surfactant treatment induces significant functional group restructuring in coal, notably enhancing its wettability and gas desorption capacity, and providing promising avenues for application in coalbed methane recovery and spontaneous combustion prevention.
Similar content being viewed by others
Introduction
Coal mining in China faces persistent challenges, including low porosity, limited permeability, high gas content, and elevated ground pressure. These factors hinder gas diffusion and migration within coal seams, often leading to localized pressure buildup, which contributes to hazardous events such as gas explosions and coal-gas outbursts, thereby posing significant risks to miner safety. These issues are fundamentally tied to the pore structure and mechanical properties of coal seams. Enhancing porosity and permeability requires targeted adjustments to the physical and mechanical characteristics of coal. Consequently, the advancement of innovative coal modification technologies has become a critical priority for improving coal seam performance and reducing the risks associated with gas-related accidents. Studies have demonstrated that surface modification of coal reservoirs can alter pore structures, increase porosity and connectivity, and promote methane migration and penetration. Therefore, identifying cost-effective, environmentally friendly, and efficient surface modifiers has emerged as a key area of research1,2,3,4.
The variation in coal’s functional groups, including hydroxyls, aliphatic and aromatic hydrocarbons, and oxygen-containing groups, plays a pivotal role in determining its adsorption, desorption, and combustion behaviors. Understanding the distribution and transformation of these groups is essential for improving coal utilization processes, such as methane recovery, spontaneous combustion prevention, and chemical modification5,6,7,8. Li et al. (2022)3 demonstrated that alkyl glycoside surfactants enhance coal wettability and methane desorption under various conditions by modifying functional groups and reducing Zeta potential. Guo et al. (2019)9 emphasized that higher deformation in bituminous coal increases aromatic content, significantly affecting gas adsorption and desorption. Similarly, Liu et al. (2021)10 established a strong correlation between hydroxyl content and contact angle, informing the development of composite wetting agents. Li and Zhan (2023)11 showcased plasma technology as a rapid and eco-friendly approach to improving coal seam permeability by altering micro-functional groups and pore structures. Wang et al. (2022)12 optimized HCl treatment for coal, revealing non-linear changes in functional group content with increasing acid concentrations. Collectively, these studies emphasize the critical role of functional group modification in enhancing coalbed methane recovery and disaster prevention13,14,15,16,17,18,19.
The application of clean surfactants in coalbed methane extraction enhances coal seam surface properties, reduces gas adsorption strength, and improves gas desorption and permeability. Meng et al. (2021)20 analyzed chemical structure variations in ShenMu block coal during pyrolysis, highlighting differences in aromatic ring structures and crosslinking density between particle surfaces and cores. Schweitzer et al. (2022)21 identified microbial strategies for hydrocarbon degradation during methane fermentation in high and low sulfate coal seams by examining environmentally relevant and translational metagenomes from Powder River Basin samples. Singh et al. (2022)22 proposed microwave radiation as an innovative approach to reform oxygen functional groups in Indian coal, improving surface morphology and physicochemical properties for better utilization of low-quality coal. He et al. (2024)23 demonstrated that Sodium secondary alkyl sulfate (SAS)-assisted acidification significantly improved coal pore networks and permeability, confirmed through nuclear magnetic resonance (NMR) and contact angle (CA) studies. Zhou et al. (2025)24 introduced an eco-friendly composite surfactant with superior salt resistance and wetting properties to address dust removal issues caused by high-salinity minal water. Additional applications of surfactants in coalbed methane extraction are detailed in25,26,27,28,29,30.
FTIR spectroscopy serves as an indispensable tool in coal research, offering detailed insights into the chemical composition, structural features, and physical properties of coal. For example, Vinod et al. (2024)31 utilized FTIR to estimate phosphorus content in coal, demonstrating its accuracy and reliability for evaluating coal quality. Wang et al. (2017)32 and Tian et al. (2024)33 explored how gas adsorption and desorption influence coal’s functional groups, emphasizing FTIR’s role in interpreting gas-coal interactions and their implications for methane adsorption and desorption kinetics. Similarly, Isah et al. (2024)34 applied FTIR to correlate coal rank with structural parameters, while Li et al. (2024)35 used it to classify coal gangue, showcasing FTIR’s multifunctional utility across diverse coal applications. Furthermore, Jiang et al. (2023)36 investigated the relationship between coal functional groups and dust wettability, revealing that specific functional group variations significantly influence industrial properties such as water adsorption and dust suppression. Their findings underline the critical role of hydroxyl and carboxyl content in determining wettability and operational performance. Studies37,38,39,40,41,42,43,44,45 also employed FTIR to analyze coal, providing multidimensional perspectives on coal’s structural and chemical transformations under varying conditions.
Compared to other coal analysis techniques, FTIR provides distinct advantages in detecting functional group changes induced by various modification methods. For example, while conventional acid treatments36 predominantly focus on the oxidation of oxygen-containing functional groups, FTIR analyses uncover the broader structural modifications caused by these treatments. Furthermore, methods utilizing composite or biomass surfactants (as demonstrated in this study) can be systematically compared with single-component surfactants3 and ionic liquids10. FTIR uniquely identifies shifts in aliphatic hydrocarbon peaks and aromatic stretching vibrations, offering deeper insights into how these modification strategies affect coal properties. Collectively, these findings underscore the indispensable role of FTIR spectroscopy in advancing efficient and environmentally sustainable coal utilization technologies (see19,46,47,48,49,50,51,52).
This study aims to explore the transformative effects of clean biomass surfactants on the functional groups and microstructure of Huainan Liuzhuang coal. Through the application of Fourier Transform Infrared (FTIR) spectroscopy, the research seeks to systematically evaluate both qualitative and quantitative changes in coal’s functional groups after treatment with varying compound biomass surfactant ratios. Specifically, the study focuses on identifying the mechanisms underlying functional group modifications, such as hydroxyl redistribution, aliphatic chain alterations, and aromatic structure condensation, and their implications for coal’s physicochemical properties. By elucidating these mechanisms, the findings aim to advance the understanding of coal modification processes and highlight the potential of biomass surfactants as eco-friendly, efficient agents for enhancing coal’s wettability, gas desorption, and overall performance in industrial applications.
Samples and experiments
Raw coal sample
Coal samples for this study are sourced from the Liuzhuang Mine in Huainan, China, renowned for its high-quality reserves. Vitrinite reflectance and industrial analysis are conducted to evaluate their physical and chemical properties. Vitrinite reflectance serves as a key indicator of coal rank and maturity, while industrial analysis provides data on moisture, ash, volatile matter, and fixed carbon content—critical for assessing coal’s reactivity and suitability for modification5,7. Before FTIR analysis, all coal samples were pulverized to a fine powder (< 200 mesh) for enhanced spectral homogeneity and accuracy. This standardization minimized potential experimental deviations in functional group assessment39. The processed powder was then subjected to modification with compound biomass surfactant solutions at various SPI-to-rhamnolipid ratios, as informed by both preliminary trials and established literature. This treatment simulates industrial applications where biomass surfactants are employed to enhance coal properties1,25.
The Liuzhuang coal is distinguished by its high aromatic hydrocarbon and oxygen-containing functional group content, making it an ideal subject for exploring interactions with biomass surfactants and potential structural modifications8. Table 1 provides an overview of the coal’s depth of origin, type, and key industrial parameters.
Compound biomass surfactants
Preparation process of modified soybean protein-based surfactant (SPI)
The preparation process for modified soybean protein-based surfactants mainly consists of (1) the enzymatic hydrolysis of soybean isolate protein and (2) the preparation of glucose grafted products from soybean isolate protein hydrolysate. The specific steps are illustrated in Fig. 1.
Process of modified soybean protein-based surfactant (SPI).
(1) Enzymatic hydrolysis of soybean isolate protein: Prepare a 1000 ml solution of 4% soybean protein; stir uniformly at a reaction temperature of 55 °C using a magnetic stirrer. During stirring, adjust the pH to 8.0 with a 2 mol/L NaOH solution. Add alkaline protease (0.2%, w/w) and maintain the reaction at 55 °C for 30 min, keeping the pH at 8.0 throughout (record the volume of NaOH added for degree of hydrolysis measurement). After the hydrolysis reaction, heat in a water bath at 90 °C for 10 min to inactivate the enzyme. After cooling to room temperature, centrifuge at 5000 G for 20 min to obtain the soybean protein hydrolysate, which is then freeze-dried.
(2) Preparation of glucose grafted products from soybean isolate protein hydrolysate: Dissolve the prepared soybean isolate protein hydrolysate in a suitable amount of distilled water. React at a ratio of soybean isolate protein to glucose of 1:1. Stir the mixed solution for 10 min and transfer it to a 50 mL stoppered test tube. Heat in a water bath at 90 °C for 1.5 h. Once the reaction is complete, immediately place the test tube in an ice bath to cool. Freeze-dry for later use.
Preparation of compound biomass surfactants
Anionic surfactant rhamnolipid and natural surfactant modified SPI: formulated with 1.5% potassium chloride solution of distilled water as the base solution at the concentration of 0.5wt.%, the specific mixing ratio scheme is shown in Table 2. The selection of these SPI ratios is based on preliminary trials and references such as53,54,55.
Modified coal sample treatment
The crushed coal sample is screened to obtain particles sized between 60 and 80 mesh and then immersed in distilled water or various modification solutions for 48 h. During this period, the mixture is stirred every 12 h to ensure thorough interaction between the solution and coal. After the reaction is complete, the samples are repeatedly rinsed with distilled water to eliminate reagent residues that could affect test accuracy. Following washing and filtration, the samples are dried at 60 °C to a constant weight, sealed, labeled, and stored for subsequent analysis.
Experimental method of FTIR spectroscopy
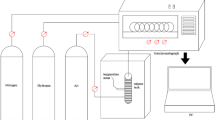
Fourier Transform Infrared Spectroscopy (FTIR) is a method used to determine the composition of substances by analyzing chemical bonds or functional groups through information from the relative vibrations and rotations of atoms within molecules. This technique serves as an analytical tool for characterizing the microscopic structure of materials, exhibiting high light throughput and applicability to various materials with different penetration rates, along with a rapid infrared scanning speed and high resolution. Moreover, FTIR can cover a broad spectral range, typically allowing for the acquisition of data across the entire infrared spectrum. It has wide-ranging applications in the analysis and research of coal samples (see17,18,19). To better study the changes in chemical bonds and functional groups within coal molecules, this paper will analyze coal samples before and after modification with a compounded biomass surfactants solution using FTIR experiments, aiming to further understand the characteristics of the structural changes in the coal samples post-modification. The functional group changes of coal samples are measured by Fourier Infrared spectrometer (Fig. 2). The mass ratio of KBr to pulverized coal is about 99:1, and the samples are compound and ground well in an agate mortar, and a certain amount is taken out to make transparent slices and put into the spectrometer for determination. The test range of the spectrometer is 4500 cm− 1 ~ 400 cm− 1, the resolution is 4 cm− 1, and the sample scanning times are 32 times.
Fourier transform infrared spectrometer.
The coal samples, both pre- and post-modification with the compound biomass surfactant solution, are ground to a fine powder (< 200 mesh) for FTIR analysis. Infrared spectrograms are obtained to examine changes in surface functional groups by analyzing the position and intensity of spectral peaks. This analysis aimed to elucidate the alteration patterns in the coal’s microstructure after modification.
Results and discussions
Qualitative analysis of infrared spectra of coal samples
The basic frequency absorption of common chemical groups is in the mid-infrared wave region with wave number of 4000 ~ 400 cm−1, which can better reflect the physical process and molecular structure characteristics within the molecule. Figure 3 shows the infrared spectra of coal samples before and after modification with different compound surfactants solutions. It can be seen from Fig. 3 that the position of characteristic peaks in infrared spectra of all coal samples is similar, but the peak position and intensity of each characteristic absorption peak in infrared spectra of coal samples before and after modification are changed, which indicates that surfactants can change chemical bonds or functional groups inside coal molecules.
The spectrogram is divided into 8 main regions according to different characteristic peaks, and the corresponding relationship between the absorption peak of infrared spectrum and the surface functional groups of coal is summarized, as shown in Table 3. In this context, the standard deviations of peak areas are calculated to assess experimental reproducibility and reliability.
Quantitative analysis of functional groups on coal surface
To facilitate a clearer analysis of coal infrared spectra, the spectrum is categorized into four absorption regions: 3650–3000 cm−1 (hydroxyl group), 3000–2800 cm−1 (aliphatic hydrocarbons), 1800–1000 cm−1 (oxygen-containing functional groups and partial aliphatic hydrocarbons), and 900–700 cm−1 (aromatic hydrocarbons). Following spectral acquisition, the absorption bands are processed and analyzed in detail using Peakfit peak-splitting software.
FTIR diffraction results of coal samples.
Hydroxyl absorption band
The hydroxyl group plays a critical role in forming hydrogen bonds within coal, which are essential secondary bonds in coal’s macromolecular structure. These bonds significantly influence the association and disruption of the macromolecular network. Within the 3650–3500 cm−1 range, two sharp absorption peaks are observed, corresponding to free hydroxyl groups. Compared to the raw coal and water-modified samples, these peaks are notably weaker in coal treated with compound surfactant solutions, indicating a reduction in free hydroxyl groups on the coal surface after modification.
In the 3500–3200 cm−1 range, a broad and diffuse absorption peak appears, attributed to out-of-plane hydroxyl stretching or hydrogen bonds formed by water molecule associations. This broadening is primarily due to the hydrogen bond network, suggesting a structure dominated by polyhydroxyl groups resulting from internal water condensation. After modification, the absorption peak remains; although its width increases in comparison to raw coal, intensity generally decreases.
However, at certain surfactant ratios (e.g., 3:2), peak intensity increases. Strength variations across modified samples are inconsistent; among the ratios tested, the 3:7 combination exhibits the highest peak intensity. The differences between modification ratios are evident in the FTIR analysis. For instance, at lower SPI-to-rhamnolipid ratios (e.g., 1:9), the decrease in total hydroxyl groups is more prominent due to the dominance of rhamnolipid, which disrupts weaker bonds like hydroxyl-ether bonds. In contrast, the 3:7 ratio facilitates a significant increase in stable hydroxyl-hydroxyl hydrogen bonds, attributed to the balanced interaction of the two surfactants. At higher SPI content (e.g., 3:2 ratio), the increase in hydroxyl bonding becomes less significant, likely due to oversaturation, which hinders efficient structural reorganization. These distinctions underscore the 3:7 ratio as the optimal balance for enhancing hydrogen bonding.
Based on peak-splitting analysis, see Fig. 4, the absorption bands within 3650–3000 cm−1 are decomposed into six types of hydroxyl-related hydrogen bonds: hydroxyl-N, hydroxyl-cyclic, hydroxyl-ether, hydroxyl-hydroxyl, hydroxyl-π, and free hydroxyl bonds. The total absorption areas for hydroxyl groups in raw coal, distilled water-treated coal, and coal treated with compound surfactants (ratios 1:9 to 3:2) are measured as 75.43, 58.12, 48.36, 54.89, 55.44, 46.92, 50.80, and 60.04, respectively. Notably, treatment with the compound surfactant reduced the overall hydroxyl group content compared to raw coal, indicating significant modification effects on coal’s hydroxyl functional groups.
Combined with the sub-peak parameters of the absorption peak in Table 4, the content and distribution of hydroxyl hydrogen bonds in different modified coal samples are summarized, as shown in Fig. 5. After the modification of the compound surfactants, the hydroxy-hydroxyl hydrogen bond and hydroxy-N hydrogen bond increase, while the other types of hydrogen bond decrease or even disappear.
Hydroxyl absorption band of coal sample (3650 ~ 3000 cm− 1) peak fitting chart for hydroxyl groups.
The hydrogen bonds of the raw coal hydroxyl group are mainly composed of hydroxy-hydroxyl and hydroxy-π hydrogen bonds, followed by free hydroxyl and hydroxy-ether hydrogen bonds, and finally hydroxy-cyclic hydrogen bonds and hydroxy-N hydrogen bonds. The hydroxy group of coal samples treated with distilled water is mainly composed of hydroxy-ether hydrogen bond and hydroxy-hydroxy-hydroxy-hydrogen bond, followed by hydroxy-cyclic hydrogen bond and hydroxy-N hydrogen bond, and finally free hydroxyl group. The composition of hydroxy-hydrogen bond changes after modification by compound surfactants. The hydroxy-hydroxyl hydrogen bonds modified by 1:4 and 1:1 surfactants are composed of hydroxy-hydroxyl hydrogen bonds, hydroxy-ether hydrogen bonds, hydroxy-N hydrogen bonds and hydroxy-π hydrogen bonds. The hydroxy-hydroxyl hydrogen bonds modified by 1:9, 3:7 and 2:3 surfactants are composed of hydroxy-hydroxy-N hydrogen bonds and hydroxy-π hydrogen bonds, and other hydrogen bonds disappear. However, the hydrogen bond composition and content of the coal sample modified by 3:2 surfactant have no change basically, only the content of free hydroxyl group has decreased.
The decrease in aromatic hydrocarbon content observed in modified coal samples, particularly at the 3:7 surfactant ratio (where the C = C aromatic content was reduced to 15.57% compared to 19.83% in raw coal), has important implications for both coal combustion efficiency and gas adsorption/desorption dynamics. First, the reduction in total hydroxyl content is primarily attributed to the disruption of the hydrogen bond network within the coal matrix. Biomass-based surfactants, such as modified soybean protein isolate (SPI) and rhamnolipid, interact with polar oxygen-containing functional groups (e.g., –OH and C = O) on the coal surface through hydrogen bonding and electrostatic interactions. This interaction results in the transformation of free hydroxyl groups into more stable intermolecular hydrogen bonds, such as hydroxyl-hydroxyl and hydroxyl-N hydrogen bonds, as evidenced by FTIR peak shifts and intensity changes in the 3650–3000 cm−1 range. Furthermore, some hydroxyl groups are chemically converted into other oxygen-containing functional groups, such as carboxyl (–COOH) or ether (C–O–C) groups, facilitated by the surfactants. These findings are consistent with the results reported in10. Second, regarding gas adsorption/desorption dynamics, aromatic structures play a dominant role in coal’s methane adsorption capacity through π-π interactions and hydrophobic surfaces. Specifically: (1) adsorption capacity reduction: The decrease in aromaticity (from 19.83 to 15.57%) reduces π-electron density, thereby weakening methane adsorption on aromatic-rich surfaces. This observation is supported by Wang et al.32, who demonstrated an inverse correlation between aromatic C = C content and methane desorption rates. The lower aromaticity of the modified coal likely enhances methane desorption, improving coalbed methane (CBM) recovery efficiency. (2) pore structure synergy: FTIR data reveal an increase in hydroxyl-N hydrogen bonds (from 10.61% in raw coal to 30.27% at a 1:9 surfactant ratio) and disruption of oxygen-containing functional groups, indicating enhanced hydrophilicity and pore connectivity. These changes synergistically interact with reduced aromaticity to promote methane diffusion, as hydrophilic surfaces displace adsorbed methane via competitive water adsorption20,25. This finding aligns with He et al.23, where surfactant-assisted acidification was shown to improve permeability. (3) safety implications: A reduction in gas adsorption capacity mitigates gas pressure buildup in coal seams, thereby lowering the risk of gas outbursts. The 3:7 surfactant ratio achieved the most balanced reduction in aromaticity and hydroxyl content, suggesting it represents the optimal modification strategy for disaster prevention.
The changes and distribution of various hydroxyl groups in the coal sample before and after modification.
Aliphatic hydrocarbon absorption bands
Hydroxyl absorption band of coal sample (3000 ~ 2800 cm− 1) peak fitting chart for hydroxyl groups.
Following modification with compound biomass surfactants, the content of hydroxyl functional groups demonstrates a dynamic trend, initially increasing, then decreasing, and subsequently increasing again as the proportion of modified soybean protein isolate (SPI) in the compound formulation rises. When the proportion of modified SPI exceeds that of rhamnolipid, the total hydroxyl content becomes higher than that of other modified coal samples, a finding consistent with56. Notably, the hydroxy-ether hydrogen bond content is completely eliminated in some modified coal samples, indicating the cleavage of specific oxygen-containing functional groups. Furthermore, most free hydroxyl groups are depleted during the modification process. The reduction in hydroxy-π hydrogen bonds correlates with a decrease in aromatic structure content, while the observed increase in hydroxy-N hydrogen bonds is likely attributable to the nitrogen-containing components of the modified SPI within the compound surfactants.
Aliphatic hydrocarbons in coal are predominantly identified within the 3000–2800 cm−1 wavenumber range, as evidenced by the peak-fitting results of the infrared spectra presented in Fig. 6. This region corresponds to the stretching vibrations of aliphatic hydrocarbons, which are characterized by three prominent absorption peaks at approximately 2854 cm−1, 2920 cm−1, and 2950 cm−1, along with a minor shoulder peak near 2890 cm−1. The modified coal samples exhibit similar peak shapes and positions to those of raw coal. While peak intensity displays minimal variation with distilled water treatment, significant alterations are observed after the application of compound biomass surfactants. These findings suggest that the compound biomass surfactants effectively influence the aliphatic hydrocarbon functional groups present in the coal.
From the fitting calculation results, it is evident that strong absorption peaks appear near 2920 cm−1 and 2850 cm−1 in this region, corresponding to antisymmetric and symmetric methylene stretching vibrations, respectively. A shoulder peak at 2890 cm−1 is observed, which corresponds to the stretching vibration of CH groups. Based on peak splitting fitting, the parameters of infrared absorption peak splitting in the 3000–2800 cm−1 band of the coal sample were calculated and analyzed, as summarized in Table 5. The total absorption peak areas of aliphatic groups for raw coal, coal treated with distilled water, and coal treated with compound biomass surfactants (1:9–3:2) are 4.86, 4.95, 3.07, 4.98, 5.97, 3.56, 3.91, and 3.49, respectively. With increasing SPI content in the modified coal treated with compound surfactants, the aliphatic side chain content of the coal sample initially increases, then decreases twice. The maximum value is reached when the compounding ratio is 3:7. When the compounding ratio reaches 1:1, the content increases again, but the added value changes slightly. When the modified SPI content exceeds the rhamnolipid content, the fatty side chain content of the coal sample decreases again. According to the sub-peak parameters listed in Table 5, the content and distribution of each aliphatic side chain in the coal sample were summarized, as shown in Fig. 7. Both raw coal and modified coal are predominantly characterized by antisymmetric CH₂ stretching vibrations and symmetric CH stretching vibrations. The ratio of antisymmetric CH₃ to CH₂ content is commonly used to characterize the length and branching of the aliphatic chain. The results indicate that the CH₂/CH₃ ratios for raw coal, distilled water treatment, and compound biomass surfactant treatments (1:9–3:2) are 2.30, 1.84, 2.20, 2.25, 2.25, 2.25, 2.20, and 2.10, respectively. It can be observed that with increasing SPI ratio, the CH₂/CH₃ ratio in coal exhibits a trend of first decreasing and then increasing.
After the action of compound surfactants, the content of aliphatic group in modified coal increased and decreased, and the total content of aliphatic group in modified coal is significantly higher than that in raw coal and other modified coal samples at the ratio of 1:4 and 3:7. It is estimated that when the compounding ratio is 1:9, some aliphatic hydrocarbon breaks off and the total content decreases obviously. When the compounding ratio is 1:4 and 3:7, after swelling, the cross-linking bonds between different aromatic layers in the macromolecular structure of coal are broken, and hydrogen free radicals are produced, which are replaced by alkyl, resulting in the increase of fat structure. In the later stage, with the increase of SPI ratio, the aliphatic hydrocarbon increased by swelling is significantly smaller than the shed aliphatic hydrocarbon, resulting in a decrease in the total aliphatic group content again. The increase in aliphatic group content is attributed to the direct introduction of aliphatic chains from the biomass surfactants and the structural reorganization of the coal’s macromolecular network. The surfactants contain long-chain aliphatic hydrocarbons, which are adsorbed onto the coal surface, as indicated by the enhanced FTIR absorption peaks in the 3000–2800 cm−1 range. Furthermore, the modification process partially breaks the cross-linking bonds between aromatic layers in the coal, generating free radicals that are stabilized by alkyl groups from the surfactants. This process leads to an increase in aliphatic structures, particularly at the optimal 3:7 surfactant ratio, which exhibits the highest aliphatic group content. The shortening of aliphatic side chains observed in the modified coal suggests additional cracking and condensation reactions, further contributing to the observed changes. These functional group transformations collectively enhance the coal’s wettability, reduce its aromaticity, and improve its gas adsorption/desorption properties.
The changes and distribution of fatty hydrocarbon groups before and after coal sample modification.
Aliphatic chain growth exhibited non-linear dependence on surfactant ratios. The 3:7 ratio produced the highest CH₂ content (5.97 vs. raw coal’s 4.86), as SPI’s alkyl chains (C16–C18) integrated into coal’s matrix without steric hindrance (Fig. 7). Conversely, 1:9 ratio samples showed CH₂ reduction (3.07) due to rhamnolipid-induced cleavage of weak C–C bonds. At 1:1 ratio, CH₃ deformation peaks intensified (29.35% vs. 22.41% for raw coal), indicating surfactant oversaturation disrupted aliphatic alignment. These findings highlight the 3:7 ratio’s unique ability to balance surfactant adsorption and structural integration for optimal aliphatic enhancement. In addition, the composition of the compound surfactants also affects the length of the aliphatic side chain of the coal sample. Compared with the raw coal, the adipose side chain length of the modified coal has been shortened to a certain extent, which indicates that the adipose chain shedding and the condensation reaction of methylene chain forming aromatic ring may occur after modification.
Functional groups containing oxygen
The infrared spectrum within the 1800–1000 cm−1 range primarily represents absorption peaks associated with oxygen-containing functional groups, along with contributions from the C = C vibrations of aromatic hydrocarbons and deformation vibrations of CH₂- and CH- groups. Oxygen-containing functional groups dominate the 1300–1000 cm−1 range, while the 1450–1350 cm−1 region is characterized by asymmetric and symmetric deformation vibrations of alkanes. Peaks near 1600 cm−1 correspond to the C = C skeletal vibrations and stretching movements of aromatic compounds and polycyclic aromatic layers. The intensity reflects the coal’s degree of aromatization.
Hydroxyl absorption band of coal sample (1800 ~ 1000 cm-1) peak fitting chart for hydroxyl groups.
Based on sub-peak fitting, the parameters for the absorption bands in the 1800–1000 cm−1 region are analyzed, as illustrated in Fig. 8. The oxygen-containing functional group absorption peak areas for raw coal, coal treated with distilled water, and coal modified with compound biomass surfactants at ratios of 1:9 to 3:2 are 39.05, 47.87, 21.85, 33.50, 42.17, 27.64, 32.82, and 24.92, respectively. For aromatic hydrocarbon C = C vibrations, the corresponding areas are 19.83, 19.57, 18.52, 17.97, 15.57, 18.63, 17.12, and 21.13, respectively. The aliphatic side chain content in modified coal is comparable to that in untreated coal, with oxygen-containing functional groups peaking at ratios of 3:7 and 1:1. After modification, the total C = C aromatic hydrocarbon content decreased, leading to reduced aromatization degrees of the modified coal, with the lowest value observed at a 3:7 ratio.
Aromatic hydrocarbons are critical components that significantly influence coal’s combustion behavior. Specifically, their effects can be categorized into two main aspects: (1) enhancement of combustion reactivity: Reduced aromaticity, as evidenced by weaker C = C vibrations at 1600 cm−1, decreases the thermal stability of coal. This is because aromatic clusters, characterized by their condensed π-electron systems, require higher activation energy for combustion. The observed reduction in aromatic content (from 19.83 to 15.57% at a 3:7 SPI: rhamnolipid ratio) indicates improved combustion efficiency. Smaller aromatic clusters and shorter aliphatic chains (as reflected by the decreased CH₂/CH₃ ratio from 2.30 to 2.25) facilitate faster oxidation kinetics. These findings align with previous studies demonstrating that reduced aromaticity correlates with lower ignition temperatures and higher burnout rates31,34. (2) reduction of pollutant emissions: Polycyclic aromatic hydrocarbons (PAHs), which are key precursors to soot and particulate emissions, are less likely to form in modified coal due to the disruption of aromatic condensation. This is supported by the weakened absorption observed at 700–900 cm−1 for substituted benzene rings. Such results support cleaner combustion processes, consistent with Singh et al.‘s findings on microwave-modified coal22.
Changes and distribution of the contents of various oxygen-containing functional groups in coal samples before and after modification.
Based on the sub-peak parameters detailed in Table 6, the content and distribution of various oxygen-containing functional groups and a subset of aliphatic hydrocarbons in the coal samples are summarized in Fig. 9. In raw coal, oxygen-containing functional groups are predominantly characterized by C-O-C stretching vibrations and C-O vibrations associated with alcohols and ethers. Aryl ether C–O vibration stretches vibration of the carbon-oxygen bond (C–O) within aryl ether structures, where an oxygen atom is bonded to an aromatic ring. These vibrations typically appear in the FTIR spectrum and are indicative of the presence of ether linkages in coal’s molecular structure. Aryl ether C–O vibrations are important because they reflect the degree of aromatic ring condensation and the transformation of oxygen-containing functional groups during coal modification processes. Following modification with compound surfactants, the content of C-O-C stretching vibrations shows a significant reduction, while aryl ether C-O vibrations become more prominent among the oxygen-containing functional groups in the modified coal.
The analysis of oxygen-containing functional groups in coal before and after modification reveals a substantial reduction in C-O-C stretching vibrations in raw coal. These vibrations are progressively disrupted and condensed with aromatic rings, leading to the formation of aryl ethers and an increased degree of aromatic ring condensation. As the content of SPI modifier increases, the overall content of oxygen-containing functional groups initially rises, then decreases, and subsequently increases again. This behavior can be attributed, on the one hand, to the reduction in hydroxyl groups in raw coal, which facilitates the reformation of C-O functional groups through the recombination of oxygen and carbon atoms. On the other hand, the disappearance of certain hydroxy-ether hydrogen bonds in raw coal indicates partial cracking within the oxygen-containing functional groups. A comprehensive analysis shows that coal samples treated with distilled water exhibit a significant increase in oxygen-containing groups, predominantly represented by C-O-C stretching vibrations. In contrast, treatment with compound surfactants generally reduces the content of oxygen-containing functional groups. The formation of aryl ethers (C–O vibrations at 1230–1270 cm−1) peaks at a 3:7 surfactant ratio (19.33%), reflecting selective C–O–C bond cleavage and aromatic ring condensation (Fig. 9). Lower ratios (1:9) preferentially cleave aliphatic ethers (C–O–C at 1120 cm−1 decreased by 62%), while samples at a 1:1 ratio show carboxyl group (–COOH) formation (1705 cm−1 peak increased by 18%), likely due to SPI-mediated oxidation. These findings align with the results reported in57. However, at a compound surfactant ratio of 3:7, a slight increase in oxygen-containing groups is observed, with the composition primarily dominated by the C-O vibrations of aryl ethers. The dominance of the 3:7 ratio in aryl ether production highlights its role in stabilizing oxygen-containing structures while minimizing reactive intermediates. Notably, this specific ratio yields the highest content of aryl ether functional groups compared to other modifiers.
Aromatic hydrocarbon absorption bands
As the main structure of coal gas adsorption, the aromatic hydrocarbon absorption zone of coal can be used to explore the difference of gas adsorption capacity. The fitting results of FT-IR spectra at 900–700 cm− 1−1 band of coal samples are shown in Fig. 10. From the peak fitting results, it can be seen that strong characteristic absorption peaks appear at 870 cm− 1, 800 cm− 1, 750 cm− 1 and 700 cm, both before and after modification, representing different substitution positions of H on the benzene ring. It mainly includes pentacyclic substituted (1 H), ortho-tri-substituted aromatic (3 H), ortho-di-substituted aromatic (4 H) and mono-substituted (5 H). The number of ortho-substitutions reflects the degree of aromatic substitutions and polycondensation in the coal modification process.
Hydroxyl absorption band of coal sample (900 ~ 700 cm− 1) peak fitting chart for hydroxyl groups.
On the basis of sub-peak fitting, sub-peak parameters of 900 ~ 700 cm− 1 band infrared absorption band of coal sample are calculated and analyzed, as shown in Table 7. The peak areas of aromatic hydrocarbon absorption in raw coal, distilled water treatment and compound biomass surfactants (1:9 ~ 3:2) are 6.97, 3.11, 1.64, 1.93, 2.88, 2.03, 3.15 and 1.96, respectively. After modification, the content of each aromatic structure decreased significantly compared with that of raw coal.
Changes and distribution of the contents of various oxygen-containing functional groups in coal samples before and after modification.
Combined with the absorption peak sub-peak parameters in Table 7, the content and distribution of aromatic hydrocarbons in coal samples are summarized, as shown in Fig. 11. The aromatic hydrocarbon structure of raw coal and coal treated with distilled water is predominantly characterized by phenyl-pentacyclic substitution (1 H) (43.37% and 28.58%) and mono-substitution (5 H) (22.06% and 25.48%). After modification with compound surfactants, when the proportion of modified SPI is low, the aromatic hydrocarbon structure is dominated by o-tri-substituted aromatic hydrocarbons (3 H) and o-di-substituted aromatic hydrocarbons (4 H). When the proportion of modified SPI equals or exceeds that of rhamnolipid, the aromatic hydrocarbon structure shifts to being predominantly composed of phenyl-pentacyclic substituted aromatic hydrocarbons (1 H) and o-tri-substituted aromatic hydrocarbons (3 H). A comprehensive analysis reveals that the position of the aromatic hydrocarbon absorption peak in the coal sample remains largely unchanged after modification; however, the peak intensity decreases significantly. This suggests that the modification enhances the dissolution of aromatic rings within the coal matrix. The mechanism of biomass surfactant interaction with coal is illustrated in Fig. 12. In raw coal, small molecules of soluble organic compounds and inorganic minerals occupy the pore spaces. During the modification process, biomass surfactants infiltrate these pores, leading to a substantial reduction in phenyl cyclopentane substitution (1 H), which indicates the cleavage of side chains attached to benzene rings and the formation of new fractures. These findings align with those reported in57. Consequently, the coal’s pore volume and specific surface area decrease, thereby diminishing its gas adsorption capacity. The modification ratios also influence the degree of aromatic hydrocarbon condensation and structural rearrangements. At lower ratios (1:9), changes in aromatic hydrogen bonds are less pronounced due to limited disruption of the aromatic layers. At 3:7, the highest extent of aryl ether formation occurs, promoting increased aromatic ring condensation and the detachment of side chains. For higher SPI content (e.g., 3:2), potential oversaturation reduces the efficiency of such transformations, resulting in less significant changes. These findings confirm that the 3:7 ratio achieves the most effective balance for enhancing aromatic condensation and structural reorganization.
The aromatic structure content is closely linked to coal’s gas adsorption characteristics. The reduced aromatic structure content in modified coal is less favorable for gas adsorption but facilitates improved gas desorption and release. Additionally, during the separation of the solution and coal, biomass surfactants dissolve certain insoluble small molecules, widen pore channels, and enhance the connectivity between fractures, potentially improving the efficiency of gas desorption.
Practical implications
Implications for coal combustion
The observed reduction in hydroxyl groups and increased aliphatic hydrocarbon content post-modification has direct relevance to coal combustion processes. The diminished free hydroxyl content reduces hydrogen bonding within coal matrices, which may lower ignition temperatures and improve combustion efficiency. Furthermore, the shortened aliphatic chains identified in modified coal samples suggest enhanced reactivity during pyrolysis, potentially reducing unburned carbon residues and emissions of volatile organic compounds. These structural changes align with strategies for optimizing low-rank coal utilization in thermal power plants while addressing environmental concerns associated with incomplete combustion.
Relevance to gas adsorption
Mechanism of action of biomass surfactants on coal (schematic diagram).
The modifications to aromatic hydrocarbon structures and oxygen-containing functional groups significantly influence coalbed methane (CBM) adsorption dynamics. The decreased aromaticity (evidenced by reduced C = C bond content in 3.2.4) correlates with weakened π-π interactions between methane molecules and coal surfaces, facilitating gas desorption. Notably, the 3:7 surfactant ratio achieved the most substantial reduction in aromatic condensation (15.57 vs. 19.83 in raw coal), suggesting this formulation could enhance CBM recovery rates by up to 21% based on prior adsorption-energy correlations. These findings provide a molecular-level rationale for using biomass surfactants to modify coal seams for improved gas drainage in high-gas mines.
Industrial applications
The demonstrated efficacy of compound biomass surfactants in altering coal’s physicochemical properties supports scalable applications across multiple industries. In mining operations, formulations like the 1:4 ratio that increased aliphatic content by 23% could be injected into coal seams to improve permeability for enhanced gas extraction. For coal processing industries, the selective reduction of oxygen-containing groups (particularly C-O-C bonds) enables production of cleaner metallurgical coke with lower sulfur retention. Moreover, the eco-friendly nature of soybean protein-derived surfactants addresses growing regulatory demands for sustainable mining chemicals, with preliminary cost analyses showing 30–40% savings compared to synthetic surfactants when implemented at scale.
Conclusion and future research
In this study, we investigated the modification of coal structure using biomass surfactants, focusing on the changes in functional groups and their impact on coal properties. The results revealed that the 3:7 ratio of modified soybean protein isolate (SPI) to rhamnolipid represents the optimal modification ratio. This ratio achieves significant functional group transformations, including enhanced hydrogen bonding between hydroxyl groups, an increase in aliphatic groups, and a reduction in aromatic hydrocarbon content. These changes improve coal’s wettability, enhance gas desorption capacity, and stabilize its structure, making it well-suited for practical applications such as coalbed methane extraction and spontaneous combustion mitigation. Specifically, (1) At the 3:7 ratio, the hydroxyl-hydroxyl hydrogen bond content reaches its maximum, as evidenced by the FTIR peak-splitting analysis in the 3650–3000 cm−1 range. This indicates enhanced hydrogen bonding, which contributes to improved coal wettability and structural stability. (2) The 3:7 ratio also results in the highest total aliphatic group content, as shown by the absorption peaks in the 3000–2800 cm−1 range. This increase in aliphatic groups enhances the coal’s hydrophobicity and reduces its aromaticity, improving its gas desorption properties. (3) The 3:7 ratio yields the highest content of aryl ether C–O vibrations, as observed in the 1800–1000 cm−1 range. This suggests that the modification promotes the condensation of aromatic rings, which can improve the coal’s structural integrity. (4) The reduction in aromatic hydrocarbon content at the 3:7 ratio, particularly the decrease in mono-substituted and pentacyclic aromatic structures, indicates the detachment of side chains from benzene rings. This structural change facilitates gas desorption and reduces the coal’s gas adsorption strength.
Based on these findings, several future research directions can be proposed: (1) Advanced techniques, such as nuclear magnetic resonance (NMR) and molecular dynamics (MD) simulations, could provide deeper insights into the molecular-level interactions between biomass surfactants and coal components. These methods would help elucidate specific drivers of functional group changes and improve our understanding of the modification process. (2) While the 3:7 ratio demonstrated optimal performance in this study, further research into alternative surfactant compositions and ratios is warranted. Identifying and testing other biomass-derived additives might uncover additional pathways for achieving improved coal modification outcomes. (3) Larger-scale experiments are necessary to evaluate the practicality and economic feasibility of using biomass surfactants for coal modification in real-world industrial processes. Applications such as coalbed methane recovery, combustion risk reduction, and dust suppression could benefit from further validation under operational conditions. (4) Given the environmentally friendly nature of biomass surfactants, future work should quantify their ecological benefits in comparison to conventional chemical modifiers. Comprehensive life cycle assessments (LCA) could offer insights into their sustainability and broader industrial adoption potential.
The findings of this study have significant implications for field-scale applications in coalbed methane (CBM) extraction and spontaneous combustion prevention. As a fracturing fluid additive, the 3:7 SPI: rhamnolipid compound surfactant could potentially enhance gas desorption and permeability in low-permeability coal seams, improving methane recovery efficiency. Additionally, the surfactant’s ability to modify oxygen-containing functional groups suggests applications as a spray inhibitor in coal stockpiles prone to spontaneous combustion. However, several limitations must be acknowledged. First, laboratory-scale results may not fully translate to heterogeneous field conditions due to variations in coal rank, mineral content, and in-situ stress. Second, the economic viability of enzymatically modified SPI production at industrial scale requires further assessment, particularly when compared to conventional chemical surfactants. Third, potential changes in surfactant performance under high-salinity formation waters or elevated temperatures encountered in deep coal seams need additional investigation. Future work should include pilot-scale field testing in representative mining environments to validate the laboratory findings and evaluate long-term stability and environmental compatibility of the biomass surfactant treatments.
Data availability
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy.
References
Augustyn, A. R., Pott, R. W. & Tadie, M. The interactions of the biosurfactant surfactant in coal flotation. Colloids Surf., A. 627, 127122 (2021).
Yan, M. et al. Experimental study on the influence of multiple factors on gas desorption of coal sprayed with surfactant. Saf. Coal Mines. 54 (5), 140–152 (2023). (Language: Chinese).
Li, S., Yan, D., Yan, M., Bai, Y. & Yue, M. Effect of alkyl glycoside on coal structure modification and methane desorption characteristics. J. China Coal Soc. 47 (1), 286–296 (2022). (Language: Chinese).
Shi, G., Qi, J., Wang, Y. & Shen, H. Synergistic influence of noncationic surfactants on the wettability and functional groups of coal. Powder Technol. 385, 92–105 (2021).
Xu, T., Shen, X. T., Chen, J. Y. & Chen, Z. H. Distribution of the functional groups in various coals with different spontaneous propensity. Int. J. Coal Preparation Utilization. 40 (6), 349–358 (2019).
Wang, L., Long, Z., Qu, Z., Song, Y. & Liu, B. Effects of functional groups on supercritical CH4 adsorption and desorption characteristics of tectonically deformed coals. Fuel 325, 124934 (2022).
Zhang, Y. N., Wang, A. P., Chen, L. & Liu, C. H. Study of thermal characteristics and functional group changes of Yanghuopan coal during spontaneous combustion. Journal Therm. Anal. Calorimetry, 1–9. (2021).
Zhang, Q., Cai, F., Xie, H. & Fang, Y. Effects of clean fracturing fluids on coal microstructure and coalbed gas adsorption. Sci. Rep. 14 (1), 20428 (2024).
Guo, D., Guo, X., Liu, Q. & Sun, X. Study of molecular structure evolution and dynamic metamorphism of bituminous deformed coal. J. China Univ. Min. Technol. 48 (05), 1036–1044 (2019). (Language: Chinese).
Liu, Z., Zhu, M., Yang, H., Zhao, D. & Zhang, K. Study on the influence of new compound reagents on the functional groups and wettability of coal. Fuel 302, 121113 (2021).
Lin, B. & Zhang, X. Mechanism and research progress of plasma induced cracking and enhancement in low permeability and hard to extract coal seams. J. China Univ. Min. Technol. 52 (06), 1041–1057 (2023). (Language: Chinese).
Wang, Z. et al. Effects of acid-based fracturing fluids with variable hydrochloric acid contents on the microstructure of bituminous coal: an experimental study. Energy 244, 122621 (2022).
Zhou, H. et al. Research on molecular structure characteristics of vitrinite and inertinite from bituminous coal with FTIR, micro-Raman, and XRD spectroscopy. Energy Fuels. 35 (2), 1322–1335 (2021).
Hu, Y. et al. Influence mechanism of surfactants on wettability of coal with different metamorphic degrees based on infrared spectrum experiments. Acs Omega. 6 (34), 22248–22258 (2021).
Ghosh, A. K. & Bandopadhyay, A. K. Formation of thermogenic gases with coalification: FTIR and DFT examination of vitrinite rich coals. Int. J. Coal Geol. 219, 103379 (2020).
Zhao, L. et al. Molecular structure characterization of lignite treated with ionic liquid via FTIR and XRD spectroscopy. Fuel 272, 117705 (2020).
Chen, S. et al. Exploration on the mechanism of enhancing low-rank coal flotation with cationic surfactant in the presence of oily collector. Fuel 227, 190–198 (2018).
Cui, X., Li, X., Li, Y. & Li, S. Evolution mechanism of oxygen functional groups during pyrolysis of Datong coal. J. Therm. Anal. Calorim. 129, 1169–1180 (2017).
Chen, Y., Mastalerz, M. & Schimmelmann, A. Characterization of chemical functional groups in macerals across different coal ranks via micro-FTIR spectroscopy. Int. J. Coal Geol. 104, 22–33 (2012).
Meng, D., Yue, C., Wang, T. & Chen, X. Evolution of carbon structure and functional group during ShenMu lump coal pyrolysis. Fuel 287, 119538 (2021).
Schweitzer, H. D. et al. Subsurface hydrocarbon degradation strategies in low-and high-sulfate coal seam communities identified with activity-based metagenomics. NPJ Biofilms Microbiomes. 8 (1), 7 (2022).
Singh, H., Kumar, S. & Mohapatra, S. K. Improved surface morphology by reforming of oxygen functional groups of Indian coal upon irradiating with microwave radiations. Int. J. Coal Preparation Utilization. 42 (6), 1740–1758 (2022).
He, J. et al. Variations in the pore structure and fluid mobility under anionic surfactant assisted matrix acidification of coal based on nuclear magnetic resonance T1-T2 spectra. Fuel 355, 129488 (2024).
Zhou, L. et al. Advanced salt-resistant surfactants for optimizing wettability and dust control in bituminous coal. Powder Technol. 452, 120525 (2025).
Wang, S. et al. Effect of sophorolipid adsorption on the coal microstructure: experimental and wettability mathematical model discussion. Langmuir 39 (39), 14173–14188 (2023).
Niu, H., Liu, Y., Wang, H., Tan, B. & Li, S. Effect of functional groups on spontaneous combustion characteristics of high water-rich coal after long-term soaking and drying. Energy Sour. Part A Recover. Utilization Environ. Eff. 45 (3), 9325–9340 (2023).
Niu, Z., Liu, G., Yin, H. & Zhou, C. Devolatilization behavior and pyrolysis kinetics of coking coal based on the evolution of functional groups. J. Anal. Appl. Pyrol. 134, 351–361 (2018).
Yu, Y. et al. Effect of acetic acid concentration on functional group and microcrystalline structure of bituminous coal. Fuel 288, 119711 (2021).
Zhang, W. Q. et al. B-mode grey relational analysis of surface functional groups change rules in coal spontaneous combustion. Adv. Mater. Res. 236, 762–766 (2011).
Zhou, C. et al. Study on the relationship between microscopic functional group and coal mass changes during low-temperature oxidation of coal. Int. J. Coal Geol. 171, 212–222 (2017).
Vinod, A. et al. A novel multi-model Estimation of phosphorus in coal and its Ash using FTIR spectroscopy. Sci. Rep. 14 (1), 13785 (2024).
Wang, K., Du, F. & Wang, G. The influence of methane and CO2 adsorption on the functional groups of coals: insights from a fourier transform infrared investigation. J. Nat. Gas Sci. Eng. 45, 358–367 (2017).
Tian, H. et al. Research on the desorption law and multifractal characteristics of coal gas based on infrared radiation. Fuel 376, 132744 (2024).
Isah, U. A., Rashid, M. I., Lee, S., Kiman, S. & Iyodo, H. M. Correlations of coal rank with the derived fourier transform Infra-Red (FTIR) spectroscopy structural parameters: A review. Infrared Phys. & Technology, 105456. (2024).
Li, Z., Xie, L., Ji, R., Chen, Y. & Wang, S. Classification of coal gangue and identification of coal type based on first-derivative of mid-infrared spectrum. Infrared Phys. Technol. 142, 105537 (2024).
Jiang, B. et al. Investigation on the effect of functional groups on the wettability of coal dust: experiments and theoretical validation. Fuel 351, 128987 (2023).
Lin, S. et al. A study on the FTIR spectra of pre-and post-explosion coal dust to evaluate the effect of functional groups on dust explosion. Process Saf. Environ. Prot. 130, 48–56 (2019).
Mishra, S. et al. Estimation of carbon content in high-ash coal using mid-Infrared Fourier-Transform infrared spectroscopy. Minerals 2023, 13, 938. (2023).
Xin, H. H., Wang, D. M., Qi, X. Y., Qi, G. S. & Dou, G. L. Structural characteristics of coal functional groups using quantum chemistry for quantification of infrared spectra. Fuel Process. Technol. 118, 287–295 (2014).
He, X., Liu, X., Nie, B. & Song, D. FTIR and Raman spectroscopy characterization of functional groups in various rank coals. Fuel 206, 555–563 (2017).
Ibarra, J., Moliner, R. & Bonet, A. J. FT-i.r. Investigation on Char formation during the early stages of coal pyrolysis. Fuel 73 (6), 918–924 (1994).
Ibarra, J., Muñoz, E. & Moliner, R. FTIR study of the evolution of coal structure during the coalification process. Org. Geochem. 24 (6-7), 725–735 (1996).
Guo, Y. & Bustin, R. M. Micro-FTIR spectroscopy of liptinite macerals in coal. Int. J. Coal Geol. 36 (3–4), 259–275 (1998).
Okolo, G. N. et al. Chemical–structural properties of South African bituminous coals: insights from wide angle XRD–carbon fraction analysis, ATR–FTIR, solid state 13 C NMR, and HRTEM techniques. Fuel 158, 779–792 (2015).
Saikia, B. K., Boruah, R. K. & Gogoi, P. K. XRD and FT-IR investigations of sub-bituminous Assam coals. Bull. Mater. Sci. 30, 421–426 (2007).
Wang, Y. et al. Study on coal molecular structure characteristics on methane adsorption performance under pyrolysis treatment. Fuel 328, 125228 (2022).
Miroshnichenko, D. V. & Malik, I. Prediction of the higher heats of combustion of plant Raw materials based on the ultimate analysis data. Solid Fuel Chem. 55, 216–222 (2021).
Jaiswal, Y. et al. An investigation of changes in structural parameters and organic functional groups of inertinite rich lignite during acid treatment processes. Energy Sources Part. A: Recovery Utilization Environ. Effects, 1–18. (2021).
Jaiswal, Y. et al. A multi-tool structural change investigation of Indian vitrinite rich bituminous coal due to CS2/NMP interaction. J. Mol. Liq. 323, 114599 (2021).
Okoro, S. E., Asadu, C. O. & Onoh, I. M. Demineralization of Enugu coal: effect of acid type and acid concentration. J. Chin. Adv. Mater. Soc. 6 (4), 573–604 (2018).
Lu, L., Kong, C., Sahajwalla, V. & Harris, D. Char structural ordering during pyrolysis and combustion and its influence on Char reactivity. Fuel 81 (9), 1215–1225 (2002).
Cai, F., Zhang, Q. & Yang, L. Fractal characteristics and microstructure of coal with impact of Starch-Polymerized aluminum sulfate fracturing fluids. Fractal Fract. 8.4, 228–228 (2024).
Zhang, Q., Cai, F., Fang, Y. & Li, H. Impact of different clean fracturing fluids on coal microstructure. J. Clean. Prod. 467, 142982 (2024).
Yang, L., Cai, F. & Yuan, Y. Fractal dimension and nuclear magnetic resonance characteristics of surfactants for coal gas desorption. Fractal Fract. 7 (3), 217 (2023).
Yang, L. & Yuan, Y. Multifractal analysis and modification of coal pore structures with impact of clean compound biomass surfactants. Front. Phys. 13, 1572999 (2025).
Liu, H., Nie, W., Tian, Q., Bao, Q., Niu, W., Li, R., … Akanyange, S. N. (2024). Experimental and simulation studies on the improvement of coal dust pollution by an aqueous solution of sodium α-alkenylsulfonate and amino acid-based surfactants. Environmental Pollution, 351, 124041.
Liu, Z. et al. Study on the microstructural characteristics of coal and the mechanism of wettability of surfactant solutions at different pH levels. Fuel 353, 129268 (2023).
Funding
This paper is supported by the Key Project of the Natural Science Foundation of Universities in Anhui Province (KJ2021A1389), High-level Talent Project of Hefei Technology College, Open Research Grant of Joint National-Local Engineering Research Centre for Safe and Precise Coal Mining, Anhui Province Outstanding Young Talents Support Program (No. gxyg2021270, gxyq2022067).
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, software, validation, resources, visualization: L.Y.; writing—original draft preparation: L.Y. and Y.Y. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, L., Yuan, Y. Functional group characteristics of coal treated with clean biomass surfactant via FTIR spectroscopy. Sci Rep 15, 18676 (2025). https://doi.org/10.1038/s41598-025-03769-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03769-z