Abstract
Metal-organic frameworks (MOFs) with high porosity have gained significant interest in organic reactions. In this study, copper-doped ZIF-8 (Cu-ZIF-8) was synthesized at room temperature, followed by oven drying at 60 °C, and subsequently employed as a highly efficient heterogeneous catalyst for both Ullmann cross-coupling and Biginelli reactions. The catalyst was characterized using various techniques, including FT-IR, XRD, BET, BJH, TGA, EDX, ICP, and SEM. These analyses confirmed that the ZIF-8 preserved its structural integrity after copper incorporation. Cu-ZIF-8 demonstrated remarkable catalytic activity in the Ullmann reaction, achieving a 93% yield with a small catalyst loading of 50 mg. In a similar manner, utilizing ethanol as a green solvent under reflux conditions along with loading 30 mg of the catalyst produced a 90% product yield in the Biginelli reaction. The synthesized products were further verified through melting point determination and NMR spectroscopy.
Similar content being viewed by others
Introduction
Cross-coupling reactions are fundamental to the field of organic synthesis, serving as the cornerstone for advancements in materials science, agriculture, electronics, and pharmaceuticals. For nearly two decades, these reactions have been instrumental in the development of new drugs1.
While palladium catalysts have traditionally dominated this area, their high cost and potential toxicity have necessitated the exploration of alternative catalysts2. In response, researchers have turned their attention to other transition metals, such as nickel3, copper4, iron5, zinc6, and cobalt7, in search of more affordable and environmentally options. Copper, in particular, has emerged as a promising candidate due to its low cost, low poisoning risk, and versatility8. It has shown exceptional effectiveness in forming new chemical bonds, particularly between carbon and other atoms, hence expanding the potential for chemical synthesis9. Despite their substantial investigation, homogeneous copper catalysts, pose limits in terms of reusability and product contamination due to copper residues. To overcome these challenges, heterogeneous catalysis has emerged as a promising alternative, in contrast to their homogeneous counterparts, heterogeneous catalysts operate as distinct phases, and enabling simple separation through centrifugation or filtration10. This characteristic facilitates catalyst reuse, thereby reducing waste and costs11. Moreover, heterogeneous catalysts often operate under milder conditions and with eco-friendly solvents, which is consistent to the green chemistry principles12. In response to these benefits, researchers have focused on immobilizing copper catalysts onto solid supports such as metal oxides13, silica14, alumina15, metal organic frameworks16, activated carbon13, and polymers17.
Among all these, MOFs have garnered significant attention due to their highly tunable structures, large surface area, specific pore size, and functionality18. Metal-organic frameworks (MOFs) are a class of crystalline materials composed of metal ions or clusters connected by organic linkers19. These linkers, typically organic molecules containing benzene rings or other aromatic groups, bridge the metal centers to form a highly ordered porous structure20. The resulting MOF material possesses a network of interconnected channels and cavities, providing a large surface area for various applications, including catalysis18, gas storage, and separation procedures21. Zeolite imidazolate frameworks (ZIFs) have emerged as a prominent material among the several MOFs explored, regarding their exceptional properties22. Zeolite imidazolate frameworks (ZIFs) have emerged as a prominent material among the several MOFs explored regarding their exceptional properties22. ZIF-8, a new class of MOFs characterized with a coordinated transition-metal cation (M) and a bridging imidazole ligand22, offers high surface area, providing an extensive platform for catalytic interactions, and enabling efficient molecular adsorption23. The well-defined pore size of ZIF-8 not only offers selectivity, minimizing undesirable side reactions and enhancing product purity, but also, coupled with its robust chemical stability, ensures the catalyst’s durability and consistent performance24, while its facile solvothermal synthesis method makes it an accessible and economical option, thereby expanding its potential for industrial applications25. Additionally, the environmentally nature of its synthesis process aligns with sustainable chemistry principles, further reinforcing ZIF-8 as a versatile and promising catalyst for a wide range of applications, particularly given its unique properties and potential for functionalization, making it an attractive candidate for ongoing research and development in this field26. The metal ions within ZIF-8, typically zinc (Zn), play a crucial role in the catalytic activity. Bimetallic based MOF gained much more attention due to their synergistic effect to expand the efficiency of catalyst through the each of component than mono metallic catalyst27.
Ullmann coupling reactions are particularly valuable for forming C-N bonds, which are crucial in biological, pharmaceutical, and material fields28. However, traditional Ullmann reactions require harsh conditions, making them less desirable for green chemistry applications29. While copper-ligand complex30 with various ligands such as diamines31, different amino acids32, amino alcohols30, diols33, diethylamines34, diphosphines35, and phenanthrolines36 can facilitate milder Ullmann coupling reactions. A recent example is the work of Bagheri et al. (2022), who synthesized a metal-organic framework (SB-Cu1) with Cu(I) and BBP ligand as heterogeneous catalytic sites for the N-arylation reaction, demonstrating a dual efficient heterogeneous catalyst for Cu-catalyzed Ullmann-type reactions. High-quality synthesized products, maintaining the catalyst efficiency after 5 times the reaction, and performing the reaction in green conditions using ethanol as an environmentally friendly solvent have been reported as advantages of the work37. A recent study by Ghosh et al. represents these advancements. They successfully synthesized a two-dimensional, paddle-wheel metal-organic framework (MOF) named Dy(NDC)(NO3) (DMA)2n via a solvothermal method. X-ray diffraction confirmed the MOF’s structure, and the catalyst demonstrated excellent activity for the N-arylation reaction under heterogeneous conditions, particularly when applied to the reaction between substituted bromobenzene and N-containing heterocycles38. on the other side, multicomponent reactions (MCRs) have gained significant interest due to their efficiency in synthesizing heterocyclic compounds, essential building blocks in drug discovery39. However, MCRs offer significant advantages beyond pharmaceuticals. They enable the fabrication of diverse functional materials with unique properties, such as tailored polymers for electronics, coatings, and composites40. MCRs also facilitate the production of natural products with applications in medicine41, agriculture, and material science42. Furthermore, MCRs, support sustainable chemistry, which minimizes reaction steps and generates less waste than traditional methods. Additionally, they can be designed to utilize environmentally friendly solvents and catalysts, aligning with green chemistry principles43. Some well-known examples of multicomponent reactions (MCRs) include the Hantzsch reaction, Passerini reaction, Ugi reaction, and the Biginelli reaction44. The Biginelli reaction holds a special significance among multicomponent reactions due to the valuable properties of its products, namely dihydropyrimidin-2(1 H)-ones (DHPMs)45. These compounds are synthesized through a three-component reaction involving an aldehyde, a β-keto ester, and urea or thiourea in protic solvents46. DHPMs exhibit a wide range of biological activities, making them highly attractive for pharmaceutical applications47.
These compounds display medicinal properties such as antibacterial, antiviral, antitumor, antioxidant, and antidiabetic activities48. For instance, Biginelli dihydropyrimidines containing an azole ring have been subjected to synthesis, computational studies, and evaluation for their α-glucosidase enzyme inhibition and anti-bacterial activities49. Additionally, the synthesis and evaluation of new thiophene-dhpms dihydropyrimidines with potential anticancer activity against breast cancer have also been explored50.
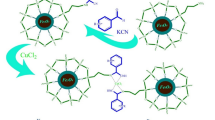
Movaheditabar et al., in 2022, successfully synthesized a new bio-based Zn-MOF through a solvothermal approach. The obtained curcumin-based MOF shows excellent catalytic performance in the preparation of several tetrahydroquinazolinone derivatives and the Biginelli reaction at room temperature in ethanol, which is a green solvent51. In this article, the bimetallic Cu-Zn @ZIF8 as a heterogeneous nanocatalyst was synthesized to perform Ullmann and Bigenili reactions, which represent a more sustainable alternative to copper-ligand complexes due to these combined benefits. High recyclability, high efficiency of the catalyst, carrying out the reaction in mild conditions, and the introduction of a heterogeneous catalyst with the ability to be reused in line with the principles of green chemistry are the advantages of this scientific work, Fig. 1.
The application of the bimetallic Cu-Zn@ZIF8 as a heterogeneous nanocatalyst for Ullmann cross coupling reaction, and Bigenili reaction.
Experimental
Materials and solvents
Solvents and all materials were purchased and used from Merck. The progress of the reactions was done by thin layer chromatography (plates prepared by silica gel F256 60 with a diameter of 0.2 mm and grain size of 25–30 micrometers) and separation for several products was done through silica gel plate chromatography.
Synthesis of ZIF-8 and Cu@ZIF-8
ZIF-8 was synthesized according to different articles52,53. In continuation, to synthesize the Cu@ZIF-8 catalyst, solutions with different concentrations of Cu(NO3)2·6H2O salt were prepared. In five 10 mL volumetric flasks, solutions with concentrations of 100, 200, 300, 400, and 500 ppm of copper salt were prepared and sonicated for 2 min. Then, 0.2 g of ZIF-8 dispersed in methanol was added to each of the solutions. These mixtures were stirred for 20 min using a magnetic stirrer and then centrifuged. The resulting precipitates were washed twice with 10 mL of methanol and dried in an oven at 60 °C for 14 h. Finally, a blue precipitate was produced (Fig. 2). To evaluate the amount of copper adsorbed by ZIF-8, ICP analysis was used, and the results showed that the concentration of 300 ppm, with 0.13% copper adsorption by ZIF-8, had the best performance. To use the Cu@ZIF-8 nanocatalyst in the Ullmann N-arylation reaction, Cu(II) was reduced to Cu(I) using NaBH4 (as a reducing agent), and the efficiency of the nanocatalyst in this reaction was investigated.
Synthesis of Cu@ZIF-8.
General method of derivatization of Ullmann reaction
For the derivatization of Ullmann’s N-arylation reaction, the reactions of various aryl halide derivatives (1.0 mmol), aniline (1.5 mmol), K2CO3 (2.00 mmol), Cu@ZIF-8 catalyst (0.05 g) by adding DMSO (2 ml) solvent were prepared. The progress of the reactions was investigated through TLC using different ratios of n-hexane and ethyl acetate, 6:4. The catalyst was separated from the reaction mixture using a centrifuge. Using ethyl acetate and water solvents, the reaction products were extracted, then the solvent was evaporated by rotary evaporation, and then the products were identified by measuring the melting point, 1H NMR, and 13C NMR spectra.
General method of derivation of Biginelli reaction
For the derivatization of this reaction in the presence of urea (1.5 mmol) and using various derivatives of aldehyde compounds (1 mmol) and 1,3 dicarbonyl compounds (1 mmol), (0.03 g) of Cu@ZIF-8 catalyst was prepared with 5 ml of ethanol solvent. It was done according to the optimal conditions explained in the previous section. The progress of the reactions was investigated through TLC using different ratios of n-hexane and ethyl acetate, 4:6. The catalyst was separated from the reaction mixture by filtration. Then ice pieces were added to the desired composition, and the precipitate obtained was crystallized using ethanol solvent. And then the products were identified by measuring the melting point, 1H NMR, and 13C NMR spectra.
Spectral data for Ullmann and Biginelli products
Diphenylamine (Table 1Entry 1and 9).
Entry 1, Weight: 0.157.17 g and Entry 9, Weight: 0.1183 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 6.82) t, J = 7.2 Hz ,2 H ، CHAr ), 7.08(d, J = 7.6 Hz,4 H. CHAr), 7.23(t, J = 7.2 HZ, 4 H, CHAr ), 8.15 (1H, S, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = (117.1, 1200, 129.6, 143.8, CHAr).
4-Nitrodiphenylamine (Table 1Entry 2 and 3).
Entry 2, Weight: 0.2033 g and Entry 3, Weight: 0.14552 g.
1H NMR (CDCl3, 400MHZ): δ (ppm) = 7.07) d, J = 9.2 Hz ,2 H, CHAr ) 7.11 (t, J = 7.2 Hz,1H CHAr) 7.25 (d, J = 8.4 HZ, 2 H, CHAr ), 7.38 (t, J = 8.4 Hz, 2 H. CHAr), 8.09 (d, J = 9.2 HZ, 2 H, CHAr) 8.42 (S ,1H, NH). 13C NMR (CDCl3, 62 MHz): δ ppm 112.9, 121.1, 123.9, 125.5, 128.9, 138.8, 149.5.
2-Nitrodiphenylamine (Table 1Entry 4 and 11).
Entry 4, Weight: 0.13696 g and Entry11, Weight: 0.0898 g.
1HNMR (CDCl3, 250 MHz): δ ppm 6.79–6.90 (d, 1H, NH₂, J = 5–6 Hz), 7.50–7.73 (m, 3 H, Ar-H, J = 7–10 Hz), 7.73–7.96 (m, 4 H, Ar-H, J = 7–10 Hz), 9.42–9.92 (d, 1H, NO₂, J = 2–3 Hz). 13C NMR (CDCl3, 62 MHz): δ ppm 115.5, 116.7, 123.6, 124.9, 125.9, 129.0, 132.5, 134.9, 137.9, 142.3.
N-phenylnaphthalen-1-amine (Table 1Entry 5 and 13).
Entry 5, Weight: 0.16206 g and Entry13, Weight: 0.12264 g.
1HNMR (CDCl3, 300 MHz): δ ppm 6.02 (s, 1H, N–H), 6.93–6.98 (m, 1H, Ar-H, J = 7–8 Hz ), 7.02–7.05 (m, 2 H, Ar-H, J = 7–8 Hz, ortho/para), 7.27–7.33 (m, 2 H, Ar-H, J = 7 Hz), 7.41–7.44 (m, 2 H, Ar-H, J = 7 Hz), 7.50–7.54 (m, 2 H, Ar-H, J = 7–8 Hz ), 7.59–7.63 (m, 1H, Ar-H, J = 7 Hz), 7.89–7.92 (m, 1H, Ar-H, J = 7 Hz), 8.04–8.08 (m, 1H, Ar-H, J = 7 Hz). 13C NMR (CDCl3, 75 MHz): δ ppm 115.9, 117.4, 120.5, 121.8, 123.0, 125.7, 126.0, 126.1, 127.8, 128.6, 129.4, 134.8, 138.7, 144.7.
4-Methoxydiphenylamine (Table 1Entry 6).
Entry 6, Weight: 0.14328 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 3.71) S, 3H, OCH3 ) 6.70 (t, J = 7/2 Hz,1H CHAr) 6.86 (d, 3JHH =8.8HZ, 2 H, CHAr ), 6.90 (d, 3JHH = 7.6 Hz, 2 H, CHAr), 7.03 (d, J = 8.8 HZ, 2 H, CHAr) 7/15 (t, J = 7.6 HZ, 2 H, CHAr) 7.84 (,1H, S, NH).
Triphenylamine (Table 1Entry 7 and 14).
Entry 7, Weight: 0.1617 g and Entry 14, Weight: 0.11025 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 6.99) t, 3JHH = 7.2 Hz ,2 H ، CHAr) 7.02 (t, J = 7.6 Hz,1H. CHAr), 7.28(t, J = 7.2 HZ, 2 H, CHAr ). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = (123.3, 124.1 ,129.9 ,147.7 CHAr).
4-Hydroxydiphenylamine (Table 1Entry 8).
Entry 8, Weight: 0.09065.
1H NMR (CDCl3, 250 MHz): δ ppm 4.112–4.138 (s, 2 H, CH₂), 5.301–5.578 (m, 2 H, NH/OH) ,7.255–7.988 (m, 9 H, Ar-H, J = 7–10 Hz, ortho/meta). 13C NMR (CDCl3, 62 MHz): δ ppm 114.9, 115.3, 115.8, 116.3, 118.8, 119, 119.4, 119.9, 121.6, 128.5, 135.0.
5-Ethoxycarbonyl-4-(4-phenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 1).
Entry 1, Weight: 0.234 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 1.16) 3H, 3JHH = 7.7 Hz, t) ,2.33 (3H, s) ,4.3 (2 H, 3JHH = 7.3 Hz, q), 5.4 (1H, 3JHH = 2 Hz, d), 5.72 (1H, s, NH), 7.26–7.32 (5 H, m, CHAr), 8.52 (1H, s, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.1 ,18.6, 55.5, 60.1, 101.3, 126.4, 127.2, 128.8, 143.4, 146.1, 153.1, 165.3.
5-Ethoxycarbonyl-4-(3-bromophenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 2).
Entry 2, Weight:0.30171 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 1.22) 3H ,3JHH =7.2 Hz, t) ,2.37 (3H, s) ,4.05–4.14 (2 H, m) ,5.2 (1H, 3JHH = 2.6, d), 5.4 (1H, s, NH) ,7.22 (1H, 3JHH =7.7, t, CHAr), 7.26–7.29 (1H, m, CHAr), 7.28 (1H, s, NH), 7.7 (1H, m, CHAr), 7.43 (1H, 3JHH =7.7 Hz, 4JHH = 2 Hz, CHAr. 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.3 ,19.2, 55.3, 60.1, 99.3, 122.6, 125.2, 129.9, 130.3, 131.2, 142.3, 145.6, 152.3, 165.2.
5-Ethoxycarbonyl-4-(4-chlorophenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 3).
Entry 3, Weight: 0.27048 g.
1H NMR: (CDCl3, 400 MHZ): δ (ppm) = 1.17) 3H, 3JHH = 6.6 Hz, t) ,2.36 (3H, s) ,4.11 (2 H, 3JHH = 7.3 Hz, q), 5.37 (1H ,3JHH = 2.4 ,s), 5.93 (1H, s, NH), 7.26–7.28 (4 H, m, CHAr), 8.17 )1H, s, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.3 ,18.6, 55.2, 60.3, 101.2, 128.1, 128.7, 133.6, 142.2, 146.3, 153.1, 165.4.
5-Ethoxycarbonyl-4-(3-chlorophenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 4).
Entry 4, Weight: 0.2793 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 1.11) 3H ,3JHH = 6.7 Hz, t) ,2.24 (3H, s) ,3.94–4.03 (2 H, m), 5.13 (1H, 3JHH = 3.7, d), 7.18–7.22 (2 H, m, CHAr), 7.32–7.34 (1H, m, CHAr), 7.3 (1H, 3JHH = 7.5 Hz, t, CHAr), 7.9 (1H, s, NH), 9.27 (1H, s, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.3 ,18.4, 54.03, 59.5, 99.1, 125.4, 126.9, 127.9,, 130.8, 133.4, 147.7, 149.5, 152.4, 165.8.
5-Ethoxycarbonyl-4-(2,4-diphenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 5).
Entry 5, Weight: 0.26286 g.
1H NMR: (CDCl3, 400 MHZ): δ (ppm) = 1.02) 3H ,3JHH = 6.7 Hz, t) ,2.20 (3H, s), 2.27 (3H, s), 2.36 (3H, s), 3.82–3.92 (2 H, m), 5.33 (1H, 3JHH = 2.7, d), 6.92 (1H, s, CHAr), 6.92 (1H, 3JHH = 8 Hz, d, CHAr), 7.03 (1H, 3JHH = 7.5 Hz, d, CHAr), 7.55 (1H, s, NH), 9.10 (1H, s, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.3 ,18.2, 19.1, 21.2, 50.06, 59.3, 99.5, 126.7, 127.3, 131.2, 134.7, 136.6, 140.8, 148.4, 152.2, 165.8.
5-Ethoxycarbonyl-4-(4-nitrophenyl)-6-methyl-3,4-dihydropyrimidine-(1 H)2-one (Table 2Entry 6).
Entry 6, Weight: 0.29798 g.
1H NMR: (CDCl3, 400MHZ): δ (ppm) = 1.11) 3H, 3JHH = 6.8 Hz, t) ,2.36 (3H, s) ,4.15 (2 H, 3JHH = 6.7 Hz, q), 5.3 (1H ,3JHH = 2 Hz, d), 5.80 (1H, s, NH), 7.52 (2 H ,3JHH = 7.3 Hz, d, CHAr), 8.1 (2 H, 3JHH = 7.1 Hz, d, CHAr), 9.17 (1H, s, NH). 13C NMR: (100 MHZ, CDCl3): δ) ppm) = 14.3 ,18.2, 55.3, 60.2, 100.3, 124.2, 127.4, 135.3, 144.2, 147.3, 150.1, 165.2.
Results and discussion
XRD
Powder X-ray diffraction (XRD) patterns of ZIF-8 and Cu@ZIF-8 nanocrystals are shown in Fig. 3. Both materials exhibit characteristic diffraction peaks at 2θ values of 7.36° (011), 10.41° (002), 12.76° (112), 14.72° (022), 16.48° (013), and 18.04° (222), consistent with the body-centered cubic (BCC) topology of ZIF-854. The sharp, well-defined peaks, particularly the prominent (011) peak at 7.36° (d = 12.01 Å), indicate high crystallinity. The loading of copper did not significantly alter the characteristic peaks of Cu@ZIF-8 compared to ZIF-8, suggesting that the ZIF-8 crystalline structure remained intact. A slight decrease in the relative peak intensities was observed due to a slight increase in grain size. No characteristic peaks of metallic copper were detected, indicating that the copper nanoparticles were highly dispersed and had small particle sizes. Figure 3 confirms that the incorporation of Cu2+ into the ZIF-8 matrix did not affect the crystal structure or lattice parameters of ZIF-8.
X-ray diffraction patterns for two compounds Cu@ZIF-8 and ZIF-8.
FT-IR
In Fig. 4, the FT-IR spectra for ZIF-8 and Cu@ZIF-8 are shown. A sharp peak at 423 cm− 1 is related to the Zn-N stretching, which depicts that the zinc atom in the ZIF-8 structure is linked to the nitrogen atoms of 2-MeIm. the peaks at 690 cm− 1 and 760 cm− 1 were associated with aromatic sp2 C-H bending. Several peaks between 900 and 1310 cm− 1 are related to the bending vibration of rings and trembling C-N in the imidazole ring. Carbon-carbon double bond stretch (C = C stretch) is assigned a peak at 1445 cm− 1, while C = N stretch has a sharp peak at 1584 cm− 1, which confirmed the well-synthesized ZIF-8 structure. The peaks appearing in the 3100 and 3000 − 2850 cm− 1 range can be attributed to SP2 and sp3 C-H stretching vibrational modes of the imidazole ring, and of the methyl group present in the linker, respectively. The peaks appeared in 550 cm− 1, is demonstrating that Cu is properly doped to the nitrogen atom in the imidazole ring and this modification doesn’t affect the ZIF structure55.
FT-IR spectrum of ZIF-8 and Cu@ZIF-8.
BET
Nitrogen adsorption isotherms Fig. 5 were employed to investigate the surface area and porosity properties of Cu-doped ZIF-8 materials. Type-I isotherms, indicative of a microporous nature, were observed for both samples, with BET surface areas of 1630 m2/g and 1275.93 m2/g for pristine ZIF-8 and Cu/ZIF-8, respectively. Due to the occupation of ZIF-8 pores by copper nanoparticles, the surface area of Cu@ZIF-8 has decreased significantly at low relative pressures (P/P0 < 0.08), and the adsorbed volume exhibited a notable increase. At P/P0 approaching 1, the observed N2 adsorption-desorption hysteresis loop suggests the presence of interparticle mesopores, further corroborating the dual micro- and mesoporous nature of Cu-doped ZIF-8 crystals. Pore size distribution analysis revealed that the pores of ZIF-8 and Cu/ZIF-8 were within the micro- and mesoporous regions. The pore size distribution centered around 1–2 nm aligns perfectly with the structural model of ZIF-8. The measured surface areas and micropore volumes of the Cu-doped ZIF-8 materials were found to be comparable to those of pure ZIF-8 (SBET = 1630 m2/g and V micro = 0.9934 cm³/g). Notably, the pore volumes of nanocrystals prepared with Cu doping (1.1943 cm³/g) were significantly higher than those measured for pristine ZIF-8 (0.48), suggesting the following order: Cu/ZIF-8 > ZIF-8 (Table 3)56.
Analysis of surface isotherms of nitrogen adsorption-desorption. (A) BET and (B) BJH ZIF-8 and Cu@ZIF-8.
TGA
Thermal degradation studies of solid materials are essential due to their widespread applications that rely on thermal stability. The TGA analysis of ZIF-8 revealed a negligible weight loss of approximately 6.87% within the temperature range of 200–440 °C, indicating its remarkable thermal resilience up to this temperature. However, a pronounced weight loss of over 61% was observed as the temperature ascended from 440 to 570 °C, signifying the onset of ZIF-8 thermal decomposition within this temperature interval. Post-decomposition, approximately 23.22% of the initial mass remained, corresponding to the formation of zinc oxide. In contrast, the Cu@ZIF-8 sample exhibited a minor weight loss of 3.92% between 200 and 310 °C, likely attributed to the removal of moisture and volatile residues. Subsequently, the organic linker and zinc metal underwent decomposition, resulting in a sharp decrease in weight up to 570 °C. Within this temperature range, Cu@ZIF-8 experienced a significant weight loss of 61.44%. Following decomposition, approximately 17.67% of the original mass persisted (Fig. 6)57.
Thermal mass spectrometric analysis (TGA) of ZIF-8 and Cu@ZIF-8.
EDX-MAP
As shown in the EDX analysis of ZIF-8 (A) and Cu@ZIF-8 (B) the presence of C, O, N, Zn, and Cu elements is confirmed (Fig. 7). Using MAP, the dispersion of metals used is shown in Fig. 8.
Energy dispersive spectroscopy data (EDX) of (A) ZIF-8 and (B) Cu@ZIF-8.
Elemental mapping of A: ZIF-8 and B: Cu@ZIF-8, revealing the elemental distributions of C, N, O, Zn, and Cu elements.
SEM
The shape and surface texture of ZIF-8 and Cu-ZIF-8 were examined using scanning electron microscopy (SEM). As shown in (Fig. 9A–C), ZIF-8 crystals displayed a characteristic rhombic dodecahedron shape with a smooth surface and an average diameter of approximately 1.07 micrometers. Following the introduction of copper ions, the resulting Cu-ZIF-8 maintained the original rhombic dodecahedron structure and particle size. However, a noticeable difference was observed in the surface texture, which became rougher (Fig. 9D–F).
SEM images of ZIF-8 (A–C) and Cu@ZIF-8 (D–F).
The copper content in Cu@ZIF-8 was quantified using Inductively Coupled Plasma (ICP) analysis to investigate the effect of copper loading on catalytic activity. Cu@ZIF-8 was synthesized with copper precursor concentrations ranging from 100 to 500 ppm. To evaluate the catalytic efficiency of each sample, all were tested in both the Ullmann and Biginelli reactions. As shown in Table S1, the sample prepared with a 300 ppm copper precursor, corresponding to a copper loading of 0.13 wt%, exhibited the highest catalytic performance, achieving a 93% yield in the Ullmann N-arylation reaction (Table 4, Entry 3) and a 90% yield in the Biginelli reaction (Table 5, Entry 3). Lower copper loadings (100–200 ppm) resulted in insufficient active sites, while higher loadings (400–500 ppm) likely led to copper aggregation, both of which contributed to reduced catalytic efficiency. The superior performance at 0.13 wt% copper loading is further supported by high turnover numbers (TON: 143.1 for Ullmann, 230.8 for Biginelli) and turnover frequencies (TOF: 6.5 h−1 for Ullmann, 153.8 h−1 for Biginelli), which are attributed to the synergistic interaction between Cu and Zn and the high porosity of the ZIF-8 framework.
To optimize the Ullmann reaction conditions, the reaction between iodobenzene and aniline was chosen as a model. Several parameters were investigated to determine their effect on the reaction, including solvent, temperature, base, and catalyst amount. A range of protic and aprotic solvents (DMSO, DMF, EtOH, THF, toluene, CH3CN, EtOAc, and H₂O) were evaluated to assess their impact on the solubility of reactants, catalyst stability, and reaction kinetics (Table 4, Entries 3, 9–15). DMSO was selected as the optimal solvent, achieving a 93% yield (Table 4, Entry 3), due to its high polarity, which stabilizes polar transition states and intermediates, enhancing the nucleophilicity of aniline for C-N bond formation. DMSO’s excellent solvating properties also improved the solubility of reactants and the base (K2CO3), facilitating interactions with the Cu@ZIF-8 catalyst.
In the Ullmann reaction, a base is crucial for deprotonating the amine, thereby enhancing its nucleophilicity for coupling with an aryl halide. Five bases K2CO3, NaOH, Et3N, NaOAc, and K3PO4—were tested in DMSO with 50 mg of catalyst at 120 °C. K2CO3 proved most effective, achieving 93% conversion, followed by NaOH (75%), NaOAc (70%), Et3N (63%), and K3PO4 (60%). The absence of a base yielded no conversion, underscoring its necessity. K2CO3’s superior performance likely stems from its moderate alkalinity, which optimally deprotonates aniline to facilitate coupling without promoting side reactions or catalyst deactivation. Stronger bases like NaOH may trigger side reactions, while less basic or organic bases like Et₃N provide insufficient deprotonation. Thus, K2CO3 was selected as the optimal base for its balanced alkalinity and compatibility.
The reaction’s rate and efficiency in Ullmann coupling are also directly influenced by catalyst quantity. Catalyst amounts of 0, 40, 50, 60, and 70 mg were tested in DMSO with K2CO3 at 120 °C (Table 4, Entries 1–5). The 50 mg amount was optimal, yielding 93% (Entry 3), as it provided an ideal density of Cu(I) active sites (0.13 wt% copper) for efficient catalysis. No reaction occurred without a catalyst (0%, Entry 1), confirming the necessity of Cu@ZIF-8. A lower amount (40 mg) resulted in a reduced yield of 78% (Entry 2) due to fewer active sites, while higher amounts (60 mg, 70% yield, Entry 4; 70 mg, 60% yield, Entry 5) led to lower yields, likely due to catalyst aggregation or side reactions caused by excess copper. The 50 mg amount was chosen to maximize catalytic activity while minimizing adverse effects.
Three temperatures (110 °C, 120 °C, and 140 °C) were tested in DMSO with 50 mg of Cu@ZIF-8 and K₂CO₃ (Table 4, Entries 3, 21, 22). The temperature of 120 °C was optimal, yielding 93% (Entry 3), as it provided sufficient thermal energy to overcome the activation barrier for the C-N coupling reaction without causing thermal decomposition of reactants, intermediates, or the catalyst. At 110 °C, the yield dropped to 65% (Entry 21) due to insufficient energy for efficient coupling, while at 140 °C, the yield decreased to 50% (Entry 22), likely due to side reactions or catalyst deactivation caused by excessive heat. The choice of 120 °C balances reaction kinetics with catalyst stability.
In entry 6, ZIF-8 was employed without Cu(NO3)2·6 H₂O as a catalyst, resulting in a trace yield. This negligible conversion indicates that ZIF-8, in the absence of copper, is ineffective at catalyzing the Ullmann reaction, underscoring copper’s role as an essential catalytic component. In contrast, entry 7 utilized Cu(NO3)2·6H2O as the catalyst, achieving a yield of 25%, a significant improvement over the trace yield in entry 6. This result confirms that copper is a critical catalytically active species in the Ullmann reaction, capable of driving the reaction to some extent even without ZIF-8.
A control experiment was performed under optimized conditions, in which copper nitrate and ZIF-8 were added separately to the reaction flask. As shown in Entry 8, this approach resulted in a significantly lower yield compared to the 93% yield obtained using the pre-synthesized Cu@ZIF-8 catalyst under identical conditions (Entry 3). The diminished catalytic activity suggests that the physical mixture does not enable effective copper–ZIF-8 interaction, likely due to the absence of stable copper incorporation into the ZIF-8 framework. This incorporation is achieved only through the controlled doping method described in Experimental Section (Table 4).
To investigate the scope of the Ullmann N-arylation reaction, various aniline and aryl halide derivatives were reacted under the optimized conditions described previously. The resulting products were characterized using melting point determination and NMR spectroscopy (Table 1). The reaction efficiency was influenced by the electronic nature of the substituents on both the aryl halide and aniline derivatives. Aryl halides with electron-withdrawing groups (entries 2 and 10) generally exhibited higher yields, while those with electron-donating groups (entries 6 and 8) were less reactive. Conversely, aniline derivatives with electron-donating groups (entries 5 and 13) tended to provide higher yields.
Although the mechanisms obtained from the calculations for forming the C-N bond from the reaction between aryl halides and amines are available in the sources, the mechanism information obtained through experience is still scarce. However, in several experimental studies, the structure and behavior of intermediates in the catalytic cycle have been investigated. The mechanism for the Ullmann reaction, as depicted in Fig. 10, involves a Cu(I)-mediated catalytic cycle. Initially, the base (K2CO3) deprotonates aniline, enhancing its nucleophilicity. The deprotonated amine coordinates with Cu(I) sites on Cu@ZIF-8 (0.13 wt% Cu) to form a copper-amine complex (Complex A). This complex undergoes oxidative addition with the aryl halide (e.g., iodobenzene), forming a Cu(III) intermediate (Complex B) with the aryl and amine ligands. Reductive elimination then occurs, forging the C-N bond to yield the N-arylated product (e.g., diphenylamine) and regenerating the Cu(I) species. The high polarity of DMSO and the microporous structure of Cu@ZIF-8 (BET surface area: 1275.93 m2/g, Table 3) facilitate the stabilization of polar intermediates and efficient substrate-catalyst interactions, contributing to the high yield. This cycle, highlights the role of Cu(I) as the active species, with ZIF-8 enhancing accessibility and recyclability.
Proposed mechanism for the N-arylation Ullmann reaction.
The results of recycling and reuse of Cu@ZIF-8 nanocatalyst to perform N-arylation Ullmann reaction
In order to check the recyclability of the catalyst, to a 10 ml flask containing DMSO solvent, iodobenzene (1 mmol, 0.112 ml), aniline (1.5 mmol, 0.136 ml), base K2CO3 (2 mmol, 0.276 g) was added along with 0.05 g of Cu@ZIF-8 catalyst. The resulting mixture was stirred at 120 °C. The reaction was followed by TLC. It was investigated using different ratios of n-hexane and ethyl acetate solvents. After the end of the reaction, the catalyst was separated from the reaction mixture by centrifugation, then it was washed with DMSO (1 × 5 ml) and ethanol (3 × 5 ml) and dried in an oven at 65 °C and The reaction was repeated four times under the same conditions. The conversion percentages, which directly reflect the reaction yields for each run, were as follows: Run 1: 93%, Run 2: 92%, Run 3: 90%, Run 4: 86%, and Run 5: 84%. Overall, this trend indicates a slight and gradual decline in yield with each reuse, thereby demonstrating that the Cu@ZIF-8 catalyst retains good catalytic activity and stability over consecutive cycles. The observed decrease is likely due to minor catalyst loss during the recovery and washing steps. Notably, no significant change in catalytic efficiency was observed, as illustrated in Fig. 11.
To further confirm the heterogeneous nature of Cu@ZIF-8, a hot filtration test was performed for the Ullmann reaction, (Figure. S41), After 6 h at 80 °C, the catalyst was removed by hot filtration, and the filtrate was allowed to react for an additional 22 h. No further conversion was observed indicating that catalytic activity is confined to the solid Cu@ZIF-8 framework, with no contribution from leached copper species. This result, combined with low leaching (ICP, < 0.01 wt%), underscores the catalyst’s stability and heterogeneity.
Reusability of the Cu@ZIF-8 in the optimum Ullmann reaction condition.
To optimize the conditions of the Biginelli reaction, the reaction between benzaldehyde, urea, and ethyl acetoacetate was chosen as the model reaction;, such as the type of solvent, temperature, reaction time, and the amount of catalyst, were optimized. The solvent contributes a relevant role to the Biginelli reaction; the solvent significantly influences reactant solubility, intermediate stability, and catalyst activity. Seven solvents (EtOH, MeOH, H2O, EtOAc, CH3CN, toluene, CHCl3) were tested with 30 mg of Cu@ZIF-8 for 90 min under reflux (or 80 °C for non-refluxing solvents like H2O and toluene). Ethanol yielded the highest conversion (90%, Table 5, Entry 3), outperforming methanol (59%, Entry 14), acetonitrile (52%, Entry 11), ethyl acetate (40%, Entry 10), chloroform (25%, Entry 13), toluene (10%, Entry 12), and water (0%, Entry 9). Ethanol’s efficacy arises from its moderate polarity, which effectively solubilizes both polar (urea) and less polar (benzaldehyde, ethyl acetoacetate) reactants, creating a homogeneous reaction environment. Its protic nature likely stabilizes transition states via hydrogen bonding during condensation steps. Nonpolar (toluene) and chlorinated (chloroform) solvents resulted in low yields due to poor solubility of polar intermediates, while water’s inability to dissolve organic reactants led to no product formation. Ethanol was thus selected as the optimal solvent for its balance of solubility and catalytic efficiency.
The catalyst is essential for the reaction process by facilitating the formation of key intermediates such as the iminium ion and enolate. Different amounts of catalyst (0, 20, 30, 40, and 50 mg) were tested in ethanol under reflux conditions. Without a catalyst, the reaction occurred 0% (entry 1). Thus, the presence of the catalyst is essential. The catalyst with the highest yield (90%) was 30 mg (entry 3). With a lower amount (20 mg), the yield was 80% (entry 2). This may have occurred because there were not enough catalytic sites to drive the reaction effectively. Larger amounts (40 and 50 mg) also reduced yield (83% and 78%, entries 4 and 5), probably arising from aggregation of the catalyst, side reaction, or saturation of catalytic activity. The 30 mg amount is therefore the optimal amount to strike a balance of catalytic effectiveness while avoiding the reduced yield caused by excess catalyst. Temperature plays an effective role in the reaction rate and the relative stability of the reactants and intermediates in the Biginelli reactions.
Temperature affects reaction kinetics and the stability of reactants and intermediates. Five temperatures (room temperature, 60 °C, 70 °C, 80 °C, and reflux ~ 78 °C) were evaluated with 30 mg of Cu@ZIF-8 in ethanol (Table 5, Entries 3, 9, 12 15–17). Reflux conditions yielded 90% (Entry 3), providing optimal thermal energy for condensation, nucleophilic addition, and cyclization steps. Room temperature (45%, Entry 15) was insufficient to overcome activation barriers, while 60 °C (76%, Entry 16) and 70 °C (84%, Entry 17) improved yields but were less effective than reflux. Reflux was chosen as it maximizes yield under mild, green conditions without risking side reactions from excessive heat.
In entry 6, ZIF-8 was tested on its own and produced near negligible yields of product, indicating that ZIF-8 did not provide any catalytic activity toward the Biginelli reaction on its own. Consequently, copper acts as an active catalytic component. In entry 7, copper nitrate was used as the catalyst, with a higher efficiency (27%) than ZIF-8 (entry 6). It is noteworthy that copper plays a significant role in the catalytic reaction. This catalytic effect was observed even in the absence of the ZIF-8 framework.
Under optimized reaction conditions, a control experiment was carried out by adding copper nitrate and ZIF-8 separately to the reaction flask. This approach, as reported in Entry 8, resulted in a significantly reduced yield compared to the 90% obtained with the pre-synthesized Cu@ZIF-8 catalyst (Entry 3). The reduced efficiency suggests that the physical mixture does not allow for sufficient copper–ZIF-8 synergy, likely due to the absence of uniform copper dispersion or integration within the framework. In contrast, the doping procedure described in Experimental section ensures stable incorporation of copper into the ZIF-8 lattice, which appears essential for achieving optimal catalytic activity (Table 5).
The reaction of various derivatives of benzaldehyde, urea, and ethyl acetoacetate was carried out according to the optimal conditions that were reported for the Biginelli reaction in the previous section, and the yield of the reactions is in Table 2 to identify the products obtained by methods such as measuring the melting point of 1H NMR and 13C NMR used. In the Biginelli reaction, when aldehyde derivatives with electron-killing substitution (entries 2-3-4-6-8-9-11-12) show high efficiency and derivatives with electron-donating substitution (entry 5 and 10) have lower efficiency.
The Biginelli reaction mechanism, shown in Fig. 12, proceeds via a three-step process. In Step 1, Cu(II) ions in Cu@ZIF-8 coordinate with the carbonyl oxygen of benzaldehyde, increasing its electrophilicity. This enables nucleophilic attack by urea’s nitrogen, forming an iminium intermediate through condensation. In Step 2, ethyl acetoacetate tautomerizes to its enol form, catalyzed by Cu@ZIF-8, and performs a Mannich-type nucleophilic addition to the iminium intermediate, yielding a complex acyclic intermediate. In Step 3, the urea’s amide group attacks the ketonic carbonyl of this intermediate, triggering intramolecular cyclization and dehydration to form the dihydropyrimidinone (DHPM). Ethanol’s protic nature stabilizes polar transition states via hydrogen bonding, while Cu@ZIF-8’s porous structure enhances substrate access to catalytic sites. The high yields across aldehyde derivatives (Tables 2 and 71–96%) validate the efficiency of this mechanism.
A proposed mechanism for the Biginelli reaction.
The results of recycling and reuse of Cu@ZIF-8 nanocatalyst to perform the Biginelli reaction
To assess the catalyst’s recyclability, a mixture of benzaldehyde, ethyl acetoacetate, and urea was reacted in the presence of 0.03 g of Cu@ZIF-8 catalyst in ethanol at reflux temperature. The reaction progress was monitored using TLC. After the reaction, the catalyst was recovered by centrifugation, washed with ethanol, and dried. This process was repeated four times under the same conditions. Catalytic performance across five runs yielded 90%, 90%, 89%, 85%, and 82% for Runs 1 to 5, respectively. This slight downward trend suggests minor catalyst deactivation or leaching during each recovery cycle. However, the data confirm that the catalyst retains its activity well, with a relatively small decrease (~ 8%) after four successive uses. These findings highlight the good recyclability and operational stability of the catalyst system (Fig. 13).
To investigate whether leached copper ions contribute to the catalytic activity, we performed a hot filtration test under the optimized reaction conditions for the Biginelli reaction (benzaldehyde, ethyl acetoacetate, urea, 30 mg Cu-ZIF-8, ethanol, reflux, 90 min) as depicted in Figure S41. After 30 min of reaction (approximately 30–40% conversion), the reaction mixture was filtered while hot to remove the solid Cu-ZIF-8 catalyst. The filtrate was then allowed to continue reacting under the same conditions (ethanol, reflux) for an additional 60 min. Analysis of the reaction mixture showed no significant increase in product yield beyond the initial conversion achieved before filtration (yield remained at ~ 35–40%). In contrast, the reaction with Cu-ZIF-8 present for the full 90 min achieved a 90% yield (Table 5, Entry 3). To confirm minimal copper leaching, the hot filtration filtrate was analyzed by ICP-OES, revealing copper levels below the detection limit (< 0.1 ppm). This is negligible compared to the 0.13 wt% copper loading in Cu@ZIF-8, indicating that copper remains stably embedded in the ZIF-8 framework and confirming the heterogeneous nature of the catalysis.
This result indicates that the catalytic activity ceases upon removal of the solid catalyst, strongly suggesting that leached copper ions, if present, do not contribute significantly to the catalysis.
Reusability of the Cu@ZIF-8 in the optimum Biginelli reaction condition.
Table 6 provides a comparison of the catalytic performance for Cu@ZIF-8 with other reported heterogeneous systems in the Ullman cross-coupling reactions. We can conclude that the present catalyst exhibited higher yields compared to the other copper-based catalysts.
A comparison of the catalytic performance of Cu@ZIF-8 in the synthesis of DHPMs is provided in Table 7. As can be seen, the synthesis of DHMPs in the presence of the introduced catalytic system afforded the corresponding products in higher yield, shorter reaction time and milder reaction conditions.
Conclusion
This research, focusing on the synthesis and performance evaluation of the Cu-Zn@ZIF-8 bimetallic heterogeneous nanocatalyst, achieved significant results in Ullmann cross-coupling reactions and Biginelli multicomponent reactions. The use of metal-organic framework (MOF) structures with high tunability and significant specific surface area paved the way for the development of catalysts with optimized efficiency under mild reaction conditions and minimal environmental impact. The results of this study showed that Cu-ZIF-8 not only provides high yields (93% in the Ullmann reaction and 90% in the Biginelli reaction) but also possesses remarkable structural and chemical stability. Various tests, including XRD, FT-IR, and BET, showed that the crystalline structure of ZIF-8 remained intact after copper doping, and the uniform distribution of copper particles within the framework enhanced catalytic performance. On the other hand, the presence of mesoporous and microporous spaces in the structure of Cu-ZIF-8 improved the adsorption of reactant molecules and increased the reaction rate. This catalyst, with the ability to be recycled and reused without a significant reduction in efficiency over several cycles, effectively addressed the challenges associated with the use of homogeneous catalysts, such as product contamination and difficulty in separation. Moreover, the use of conditions compatible with green chemistry principles, including the use of environmentally friendly solvents such as ethanol and the reduction of reaction temperatures, has made this research a successful model for the development of sustainable technologies. This research not only provides solutions to overcome the limitations of existing catalyst synthesis but also opens new horizons for future research in the field of catalytic chemistry. Utilizing the unique structural and chemical properties of MOFs in combination with various metals can help create more efficient and sustainable industrial processes and pave the way for reducing the environmental impacts of chemical activities.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Hong, K. et al. Palladium nanoparticles on assorted nanostructured supports: applications for Suzuki, heck, and Sonogashira cross-coupling reactions. ACS Appl. Nano Mater. 3, 2070–2103 (2020).
Yashwantrao, G. & Saha, S. Sustainable strategies of C–N bond formation via Ullmann coupling employing Earth abundant copper catalyst. Tetrahedron 97, 132406 (2021).
Alinezhad, H., Ghasemi, S. & Cheraghian, M. MOF nano porous-supported CS cross coupling through one-pot post-synthetic modification. J. Organomet. Chem. 898, 120867 (2019).
Pourshahi, M. et al. Copper (II)-polymer chelate grafted from magnetic mesoporous silica for the O-arylation of phenols via the Ullmann reaction. J. Organomet. Chem. 1014, 123191 (2024).
Ali, A. T. et al. Fe 3 O 4 supported [Cu (ii)(met)(pro-H) 2] complex as a novel nanomagnetic catalytic system for room temperature C–O coupling reactions. RSC Adv. 13, 22538–22548 (2023).
Natarajan, S., Manna, K. & Boruah, R. Zn, cd and Cu coordination polymers for metronidazole sensing and for Ullmann and Chan-Lam coupling reactions. Chemistry–An Asian J., e202400501.
Xu, S. S. et al. Cobalt-Catalyzed Electrochemical Enantioselective Reductive Cross-Coupling of Organohalides, CCS Chemistry, 1–11. (2024).
Fairoosa, J., Neetha, M. & Anilkumar, G. Recent developments and perspectives in the copper-catalyzed multicomponent synthesis of heterocycles. RSC Adv. 11, 3452–3469 (2021).
Chen, Z., Wu, X. F. & Copper-Catalyzed, C. C. Bond formation via carboxylation reactions with CO2, in: CC Cross Couplings with 3d Base Metal Catalysts, Springer, 233–253. (2023).
Alinezhad, H. & Pakzad, K. cross‐coupling reaction using novel and green synthesized CuO nanoparticles assisted by Euphorbia maculata extract. Appl. Organomet. Chem. 33, e5144 (2019).
Fui, C. J., Sarjadi, M. S., Sarkar, S. M. & Rahman, M. L. Recent advancement of Ullmann condensation coupling reaction in the formation of aryl-oxygen (CO) bonding by copper-mediated catalyst. Catalysts 10, 1103 (2020).
Mastalir, Á. & Molnár, Á. A novel insight into the Ullmann homocoupling reactions performed in heterogeneous catalytic systems. Molecules 28, 1769 (2023).
Bagheri, H., Baharfar, R. & Magnetic, M. C. R. F. Graphene oxide complexed with copper Nano-Particles: an efficient and recyclable nanocatalyst for Ullmann C–N coupling reaction. Polycycl. Aromat. Compd. 43, 7580–7596 (2023).
Abdollahi-Alibeik, M. & Ramazani, Z. Core–shell structured magnetic MCM-41-type mesoporous silica-supported Cu/Fe: A novel recyclable nanocatalyst for Ullmann-type homocoupling reactions. Main Group Met. Chem. 45, 190–201 (2022).
Ndolomingo, M. J., Bingwa, N. & Meijboom, R. Review of supported metal nanoparticles: synthesis methodologies, advantages and application as catalysts. J. Mater. Sci. 55, 6195–6241 (2020).
Singh, R. et al. Copper-based metal–organic frameworks (MOFs) as an emerging catalytic framework for click chemistry. Catalysts 13, 130 (2023).
Gorginpour, F. & Zali-Boeini, H. Synergistic effect of copper nanocrystals-nanoparticles incorporated in a porous organic polymer for the Ullmann CO coupling reaction. Mol. Catal. 504, 111460 (2021).
Cheraghian, M., Alinezhad, H. & Ghasemi, S. Post-synthetic modification of UIO-66-NH2 as a highly efficient and recyclable nanocatalyst in the three-component coupling (A3) reaction for the synthesis of Propargylamine derivatives. J. Organomet. Chem. 1002, 122903 (2023).
Najafi, M., Alinezhad, H., Ghasemi, S. & Yeganeh-Salman, E. Synthesis of nanocatalyst Pd immobilized on ZPD as efficient and reusable for Sonogashira cross-coupling reaction. J. Organomet. Chem. 980, 122497 (2022).
Moghadaskhou, F., Tadjarodi, A. & Maleki, A. Synthesis of UiO-66-Pyca-CuO by a simple and novel method: MOF-based metal thin film as heterogeneous catalysts for the synthesis of α-Aminonitriles. ACS Appl. Mater. Interfaces. 16, 41048–41059 (2024).
Sadati, Z., Alinezhad, H. & Tajbakhsh, M. Preparation and characterization of Pd immobilized on the MIL-125-NH2 as an efficient recyclable metal-organic framework in the Suzuki-Miyaura reaction. J. Organomet. Chem., 123466. (2024).
Zheng, Z., Rong, Z., Nguyen, H. L. & Yaghi, O. M. Structural chemistry of zeolitic imidazolate frameworks. Inorg. Chem. 62, 20861–20873 (2023).
Oh, S., Lee, S., Lee, G. & Oh, M. Enhanced adsorption capacity of ZIF-8 for chemical warfare agent simulants caused by its morphology and surface charge. Sci. Rep. 13, 12250 (2023).
Wang, H., Qi, X., Yan, G. & Shi, J. Copper-doped ZIF-8 nanomaterials as an adsorbent for the efficient removal of as (V) from wastewater. J. Phys. Chem. Solids. 179, 111408 (2023).
Wang, X. et al. Peroxidase-like active Cu-ZIF-8 with rich copper (I)-nitrogen sites for excellent antibacterial performances toward drug-resistant bacteria. Nano Res., 1–9. (2024).
Kümbetlioğlu, F., Oskay, K. O., Çıplak, Z. & Ateş, A. Preparation, characterization, and application of metal Oxide-Doped zeolitic imidazolate framework. ACS Omega. 8, 27650–27662 (2023).
Li, J. et al. ZIF-8 derived N-doped porous carbon confined ultrafine PdNi bimetallic nanoparticles for semi-hydrogenation of alkynes. Mol. Catal. 535, 112865 (2023).
Yang, Q., Zhao, Y. & Ma, D. Cu-mediated Ullmann-type cross-coupling and Industrial Applications in Route Design, Process Development, and scale-up of Pharmaceutical and Agrochemical Processes261690–1750 (Organic Process Research & Development, 2022).
Akhtar, R., Zahoor, A. F., Irfan, M. & Bokhari, T. H. A. ul Haq, recent green synthetic approaches toward Ullmann reaction: A review. Chem. Pap. 76, 7275–7293 (2022).
Hemmati, S., Ahany Kamangar, S., Yousefi, M., Hashemi Salehi, M. & Hekmati, M. Cu (I)-anchored Polyvinyl alcohol coated‐magnetic nanoparticles as heterogeneous nanocatalyst in Ullmann‐type C–N coupling reactions. Appl. Organomet. Chem. 34, e5611 (2020).
Feng, J., Xi, L. L., Lu, C. J. & Liu, R. R. Transition-metal-catalyzed Enantioselective C–N cross-coupling (Chemical Society Reviews, 2024).
Jagushte, K. U., Ketkar, R. N., Thakkar, C., Choudhury, S. D. & Sadhukhan, N. Convenient synthesis of 6-Amino-2-naphthol by Copper-catalyzed Ullmann reaction. Tetrahedron Lett. 128, 154693 (2023).
Yuan, R. et al. Synthesis of 1, 7-Bis (N-substituted-aminomethyl)-2, 8-dihydroxy-Tröger’s bases and their application in Aldol-Ullmann reaction. Chin. J. Org. Chem. 40, 1017 (2020).
Alizadeh, A., Khalilzadeh, M. A., Alipour, E. & Zareyee, D. Pd (II) Immobilized on Clinoptilolite as a Highly Active Heterogeneous Catalyst for Ullmann Coupling-type S-arylation of Thiols with Aryl Halides23658–666 (Combinatorial Chemistry & High Throughput Screening, 2020).
Zuo, Z., Kim, R. S. & Watson, D. A. Synthesis of axially chiral 2, 2′-bisphosphobiarenes via a nickel-catalyzed asymmetric Ullmann coupling: general access to privileged chiral ligands without optical resolution. J. Am. Chem. Soc. 143, 1328–1333 (2021).
Liu, G. et al. A new highly efficient NHC/Cu (I) catalyst system for the Ullmann-type N-arylation reactions. Mol. Catal. 564, 114284 (2024).
Bagheri, S., Esfanidiary, N. & Yliniemi, J. Porous SB-Cu1 two-dimensional metal-organic framework: the green catalyst towards CN bond-forming reactions. Colloids Surf., A. 637, 128202 (2022).
Ghosh, P., Maity, T., Khatun, N. & Debnath, R. Koner, 2D paddle wheel lanthanide metal-organic framework: synthesis, structure and exploration of catalytic N-arylation reaction. Polyhedron 219, 115789 (2022).
Graziano, G. et al. Multicomponent reaction-assisted drug discovery: A time-and cost-effective green approach speeding up identification and optimization of anticancer drugs. Int. J. Mol. Sci. 24, 6581 (2023).
John, S. E., Gulati, S. & Shankaraiah, N. Recent advances in multi-component reactions and their mechanistic insights: A triennium review. Org. Chem. Front. 8, 4237–4287 (2021).
Younus, H. A. et al. Multicomponent reactions (MCR) in medicinal chemistry: a patent review (2010–2020). Expert Opin. Ther. Pat. 31, 267–289 (2021).
Javahershenas, R., Han, J., Kazemi, M. & JervisP.J. Recent advances in the application of 2-Aminobenzothiazole to the multicomponent synthesis of heterocycles. ChemistryOpen e202400185 (2024).
Buskes, M. J., Coffin, A., Troast, D. M., Stein, R. & Blanco, M. J. Accelerating drug discovery: synthesis of complex chemotypes via multicomponent reactions. ACS Med. Chem. Lett. 14, 376–385 (2023).
Mohlala, R. L., Rashamuse, T. J. & Coyanis, E. M. Multicomponent reactions as an efficient and facile alternative route in organic synthesis and applications, (2023).
Faizan, S., Roohi, T. F., Raju, R. M., Sivamani, Y. & BR, P. K. A century-old one-pot multicomponent Biginelli reaction products still finds a niche in drug discoveries: synthesis, mechanistic studies and diverse biological activities of dihydropyrimidines. J. Mol. Struct. 1291, 136020 (2023).
Fauzi, A., Saifudin, A. & Rullah, K. Synthesis of Dihydropyrimidinone (DHPM) Derivatives through a Multicomponent Reaction (MCR) and Their Biological Activity, (2023).
Ali, L. B. et al. In Silico investigation and biological evaluation viz antimicrobial, genotoxic, and anticancer potentials of new dihydropyrimidinones (DHPMs) synthesized by Biginelli reaction. J. Mol. Struct. 1316, 138758 (2024).
Sarvaiya, B. H., Vaja, P. I., Paghdar, N. A. & Ghelani, S. M. Medicinal perspective of a promising scaffold–dihydropyrimidinones: A review. J. Heterocycl. Chem. 61, 1325–1348 (2024).
Taviti, K. S. et al. Design, synthesis and biological evaluation of novel chromones having 3, 4-dihydropyrimidin-2 (1H)-one core at C-8 in combination with Triazoles: new α-glucosidase inhibitors and anti-bacterial agents. Eur. J. Med. Chem. Rep. 12, 100187 (2024).
Nikam, D., Jain, A., Vetale, S., Bhange, A. & Jadhav, S. Synthesis and evaluation of novel thiophene-dhpms designed having anti-breast cancer potential. Curr. Chem. Lett. 13, 717–724 (2024).
Movaheditabar, P., Javaherian, M. & Nobakht, V. Synthesis and catalytic application of a curcumin-based bio‐MOF in one‐pot Preparation of Tetrahydroquinazolinone derivatives via Biginelli reaction. Appl. Organomet. Chem. 36, e6602 (2022).
Zhou, Y. et al. Copper-doped metal–organic frameworks for the controlled generation of nitric oxide from endogenous S-nitrosothiols. J. Mater. Chem. B. 9, 1059–1068 (2021).
Yang, X., Wen, Z., Wu, Z. & Luo, X. Synthesis of ZnO/ZIF-8 hybrid photocatalysts derived from ZIF-8 with enhanced photocatalytic activity. Inorg. Chem. Front. 5, 687–693 (2018).
Parkash, A. Copper doped zeolitic imidazole frameworks (ZIF-8): a new generation of single-atom catalyst for oxygen reduction reaction in alkaline media. J. Electrochem. Soc. 167, 155504 (2020).
Nagarjun, N., Dhakshinamoorthy, A. & Cu-Doped, A. ZIF-8 metal organic framework as a heterogeneous solid catalyst for aerobic oxidation of benzylic hydrocarbons. New J. Chem. 43, 18702–18712 (2019).
Saeedirad, R., Ganjali, S. T., Rashidi, A. & Bazmi, M. Experimental and computational study of organic sulfur removal proficiency of (Ni, Cu, Co)-doped ZIF‐8 adsorbents. ChemistrySelect 5, 231–243 (2020).
Ahmad, A. et al. Cu-doped zeolite imidazole framework (ZIF-8) for effective electrocatalytic CO2 reduction. J. CO2 Utilization. 48, 101523 (2021).
Khodaei, M. M., Alizadeh, A. & Haghipour, M. Preparation and characterization of Isatin complexed with Cu supported on 4-(aminomethyl) benzoic acid-functionalized Fe 3 O 4 nanoparticles as a novel magnetic catalyst for the Ullmann coupling reaction. Res. Chem. Intermed. 45, 2727–2747 (2019).
Mohammadinezhad, A. & Akhlaghinia, B. Co II immobilized on an aminated magnetic metal–organic framework catalyzed C–N and C–S bond forming reactions: a journey for the mild and efficient synthesis of arylamines and arylsulfides. New J. Chem. 43, 15525–15538 (2019).
Khosravi, A., Mokhtari, J., Naimi-Jamal, M. R., Tahmasebi, S. & Panahi, L. Cu 2 (BDC) 2 (BPY)–MOF: an efficient and reusable heterogeneous catalyst for the aerobic Chan–Lam coupling prepared via ball-milling strategy. RSC Adv. 7, 46022–46027 (2017).
Seyedi, N. & Zahedifar, M. Chitosan nanoparticles functionalized poly-2‐hydroxyaniline supported CuO nanoparticles: an efficient heterogeneous and recyclable nanocatalyst for N‐arylation of amines with Phenylboronic acid at ambient temperature. Appl. Organomet. Chem. 35, e6364 (2021).
Said, K. & Salem, R. B. Ultrasonic activation of N-arylation of amines with Aryl halides catalysed by Iron (III). Lett. Org. Chem. 19, 627–635 (2022).
Ma, R., Zhang, R., Xia, H., Wang, L. & Ma, Y. Direct ortho-Selective amination of naphthols with nitroarenes through Transfer‐Hydrogenation. Eur. J. Org. Chem. 27, e202400089 (2024).
Safaei Ghomi, J., Teymuri, R. & Ziarati, A. A green synthesis of 3, 4-dihydropyrimidine-2 (1 H)-one/thione derivatives using nanosilica-supported Tin (II) chloride as a heterogeneous nanocatalyst. Monatshefte Für Chemie-Chemical Monthly. 144, 1865–1870 (2013).
Meng, F. J., Shi, L., Feng, G. S., Sun, L. & Zhou, Y. G. Enantioselective synthesis of 3, 4-dihydropyrimidin-2 (1 H)-ones through organocatalytic transfer hydrogenation of 2-hydroxypyrimidines. J. Org. Chem. 84, 4435–4442 (2019).
Khaksar, S., Vahdat, S. M. & Moghaddamnejad, R. N. Pentafluorophenylammonium triflate: an efficient, practical, and cost-effective organocatalyst for the Biginelli reaction. Monatshefte Für Chemie-Chemical Monthly. 143, 1671–1674 (2012).
Kakati, P., Singh, P., Yadav, P. & Awasthi, S. K. Aiding the versatility of simple ammonium ionic liquids by the synthesis of bioactive 1, 2, 3, 4-tetrahydropyrimidine, 2-aminothiazole and Quinazolinone derivatives. New J. Chem. 45, 6724–6738 (2021).
Nasr-Esfahani, M. & Abdizadeh, T. Vanadatesulfuric acid: A novel, recyclable, and heterogeneous catalyst for the one-pot synthesis of dihydropyrimidinones and dihydropyrimidinthiones under solvent-free conditions. Phosphorus Sulfur Silicon Relat. Elem. 188, 596–608 (2013).
Safari, J. & Gandomi-Ravandi, S. Titanium dioxide supported on MWCNTs as an eco-friendly catalyst in the synthesis of 3, 4-dihydropyrimidin-2-(1 H)-ones accelerated under microwave irradiation. New J. Chem. 38, 3514–3521 (2014).
Mashhoori, M. S., Sandaroos, R., Zeraatkar, A. & Moghaddam Polymeric imidazolium ionic liquid-tagged manganese schiff base complex: an efficient catalyst for the Biginelli reaction. Res. Chem. Intermed. 46, 4939–4954 (2020).
Karami, S., Karami, B. & Khodabakhshi, S. Solvent-free synthesis of novel and known octahydroquinazolinones/thiones by the use of ZrOCl2. 8H2O as a highly efficient and reusable catalyst. J. Chin. Chem. Soc. 60, 22–26 (2013).
Kiyani, H. & Ghiasi, M. Solvent-free efficient one-pot synthesis of Biginelli and Hantzsch compounds catalyzed by potassium phthalimide as a green and reusable organocatalyst. Res. Chem. Intermed. 41, 5177–5203 (2015).
Alinezhad, H. & Pakzad, K. Green synthesis of copper oxide nanoparticles with an extract of Euphorbia Maculata and their use in the Biginelli reaction. Org. Prep. Proced. Int. 52, 319–327 (2020).
Hedayati, V. R. & Ghasemzadeh, M. A. Efficient, one-pot synthesis of polyfunctionalised octahydroquinazolin-2, 5-diones catalysed by Fe3O4 nanoparticles. J. Chem. Res. 39, 56–61 (2015).
Akbarzadeh, P. & Koukabi, N. Synthesis and characterization of the immobilized polythiophene on magnetic carbon nanotube as a prominent catalyst for the synthesis of Dihydropyrimidinone and Octahydroquinazolinone derivatives. Res. Chem. Intermed. 46, 4955–4969 (2020).
Esrafili, L., Morsali, A., Dehghani Firuzabadi, F. & Retailleau, P. Development of porous cobalt-/copper-doped carbon nanohybrids derived from functionalized MOFs as efficient catalysts for the Ullmann cross-coupling reaction: insights into the active centers. ACS Appl. Mater. Interfaces. 12, 43115–43124 (2020).
Sonawane, R. B., Rasal, N. K., Bhange, D. S. & Jagtap, S. V. Copper-(II) catalyzed N‐formylation and N‐acylation of aromatic, aliphatic, and heterocyclic amines and a preventive study in the C‐N cross coupling of amines with Aryl halides. ChemCatChem 10, 3907–3913 (2018).
Du, F. et al. Copper (II)-Catalyzed C–N Coupling of Aryl Halides and N-Nucleophiles Promoted by Quebrachitol or Diethylene Glycol. Synlett 30, 2161–2168. (2019).
Pourshahi, M. A., Mansoori, Y., Ghahramani, F., Bezaatpour, A. & John, M. Poly (N-2‐Aminoethylacrylamide)‐Cu (I) chelate grafted from Fe3O4@ SiO2 Core‐Shells for the selective monoarylation of anilines via the Ullmann reaction. ChemistrySelect 9, e202400209 (2024).
Mullick, K. et al. Ullmann reaction catalyzed by heterogeneous mesoporous Copper/manganese oxide: a kinetic and mechanistic analysis. Inorg. Chem. 56, 10290–10297 (2017).
Guo, X., Rao, H., Fu, H., Jiang, Y. & Zhao, Y. An inexpensive and efficient copper catalyst for N-Arylation of amines, amides and Nitrogen‐Containing heterocycles. Adv. Synth. Catal. 348, 2197–2202 (2006).
Veisi, H. et al. Copper nanoparticle anchored biguanidine-modified Zr-UiO-66 MOFs: a competent heterogeneous and reusable nanocatalyst in Buchwald–Hartwig and Ullmann type coupling reactions. RSC Adv. 11, 22278–22286 (2021).
Babapoor, A., Asgharzadeh, R. & Pourshahi, M. Cu (I) Schiff Base Complex Stabilized on Magnetic Graphene Oxide as a Recoverable Heterogeneous Catalyst for N-arylation of Amines Under Mild Conditions and With High Efficiency, Chemistry Africa, 1–13. (2024).
Güell, I. & Ribas, X. Ligand-Free Ullmann‐Type C–Heteroatom couplings under practical conditions. Eur. J. Org. Chem. 2014, 3188–3195 (2014).
Kidwai, M. et al. Cu nanoparticles in PEG: A new recyclable catalytic system for N-Arylation of amines with Aryl halides. ChemCatChem 2, 1312–1317 (2010).
Najafi, M. et al. Decorated Cu nanoparticles on ZPD as a novel and highly proficient nanocatalyst for synthesis of Chromene and Biginelli reactions. Res. Chem. Intermed. 50, 1177–1209 (2024).
Zhao, S. Y., Chen, Z. Y., Wei, N., Liu, L. & Han, Z. B. Highly efficient cooperative catalysis of single-site Lewis acid and Brønsted acid in a metal–organic framework for the Biginelli reaction. Inorg. Chem. 58, 7657–7661 (2019).
Krishna, B., Payra, S. & Roy, S. Synthesis of dihydropyrimidinones via multicomponent reaction route over acid functionalized Metal-Organic framework catalysts. J. Colloid Interface Sci. 607, 729–741 (2022).
Santos, M. C., Uemi, M., Gonçalves, N. S., Bizeto, M. A. & Camilo, F. F. Niobium chloride in 1 – n– butyl – 3– Methylimidazolium chloride ionic liquid as a catalyst for Biginelli reaction. J. Mol. Struct. 1220, 128653 (2020).
Pal, T. K., De, D., Senthilkumar, S., Neogi, S. & Bharadwaj, P. K. A partially fluorinated, water-stable Cu (II)–MOF derived via transmetalation: significant gas adsorption with high CO2 selectivity and catalysis of Biginelli reactions. Inorg. Chem. 55, 7835–7842 (2016).
Grami, M. & Rafiee, Z. Fabrication of magnetic carbohydrate-modified iron oxide nanoparticles (Fe 3 O 4/pectin) decorated with bimetallic Co/Cu-MOF as an effective and recoverable catalyst for the Biginelli reaction. RSC Adv. 14, 24175–24184 (2024).
Houshiar, S., Rafiee, Z. & Grami, M. Polymer/ZIF-67 composite as an effective and recyclable nanocatalyst for Biginelli reaction. Appl. Organomet. Chem. 36, e6800 (2022).
Saikia, M., Bhuyan, D. & Saikia, L. Keggin type phosphotungstic acid encapsulated chromium (III) terephthalate metal organic framework as active catalyst for Biginelli condensation. Appl. Catal. A. 505, 501–506 (2015).
Acknowledgements
The authors acknowledge the Research Council of the University of Mazandaran.
Author information
Authors and Affiliations
Contributions
[Zahra Doraghi]: Conceptualization, methodology, data collection, formal analysis, and manuscript writing.[Arezoo Ahmadi]: Data analysis, validation, manuscript review, and editing.[Heshmatollah Alinezhad]: Supervision, project administration, funding acquisition, and final approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This work does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Doraghi, Z., Alinezhad, H. & Ahmadi, A. A copper-doped ZIF-8: a high-performance catalyst for eco-friendly Ullmann and Biginelli reactions. Sci Rep 15, 24432 (2025). https://doi.org/10.1038/s41598-025-03827-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03827-6