Abstract
Eye movement disorder (EMD) is a significant cause of visual impairment and disruption of daily activities. Drug-induced EMD is an underrecognized but critical risk factor for patients. This study aimed to explore the relationship between medications and EMD using real-world data to improve patient safety and clinical decision-making. A total of 6,158 reports of drug-induced EMD, involving 1,834 unique drugs, were analyzed from the FDA Adverse Event Reporting System (FAERS) spanning Q1 2004 to Q4 2024. Disproportionality analysis revealed 30 drugs with significant associations with EMD, primarily within the central nervous system drug class, including antiseizure medications (vigabatrin, topiramate), antipsychotics (aripiprazole, risperidone), and antidepressants (sertraline, venlafaxine). The highest risks were observed with zonisamide (BCPNN = 3.03), vigabatrin (BCPNN = 2.82), and tiagabine (BCPNN = 2.79). Acyclovir and donepezil had the shortest onset times (median: 2 days). EMD-related adverse events were most commonly reported in middle-aged and elderly women (55.3%), and the number of cases has been steadily increasing over time. This study provides essential pharmacovigilance insights, identifying high-risk medications and demographic factors associated with drug-induced EMD, which can inform clinical practice and optimize patient care.
Similar content being viewed by others
Introduction
Eye movement disorder (EMD) encompasses a range of pathological conditions characterized by abnormal or restricted eye movements, which can severely impair visual function and disrupt daily activities. These disorders manifest in various clinical forms, including nystagmus, ocular muscle paralysis, restricted eye movements, strabismus, diplopia, and fixation paralysis1,2,3,4,5,6. Such abnormalities often result in symptoms like blurred vision, dizziness, and balance disturbances, while also significantly affecting tasks requiring fine visual acuity, such as reading and driving2,7,8,9.
The pathophysiology of EMD is complex, with multiple potential etiologies. These include neurological conditions such as stroke, brain tumors, and Parkinson’s disease, ocular muscle diseases like ocular muscle paralysis or myasthenia, and thyroid disorders, notably Graves’ disease. These conditions disrupt the intricate mechanisms controlling eye movements, leading to a wide spectrum of movement abnormalities7,9,10,11,12,13. In addition to these intrinsic pathologies, pharmacological agents have become increasingly recognized as significant contributors to EMD. Certain medications, such as antipsychotics, antidepressants, anticonvulsants, and antiarrhythmic drugs, can interfere with neural pathways, ocular muscle function, or brain regions involved in motor control, thereby inducing eye movement abnormalities. These adverse drug reactions tend to be more pronounced with improper use or prolonged treatment, with common manifestations including nystagmus, ocular muscle paralysis, and bradykinesia14,15,16,17.
Given the broad range of drugs associated with EMD, it is crucial to identify these agents and implement strategies to prevent their adverse effects. Large-scale pharmacovigilance systems, such as the FDA Adverse Event Reporting System (FAERS), play a pivotal role in monitoring drug safety by collecting comprehensive reports on adverse events18,19. While previous studies have leveraged the FAERS database to investigate various adverse drug reactions, research specifically examining drugs linked to EMD remains relatively sparse. Analyzing FAERS data offers a valuable opportunity to identify drugs most strongly linked to EMD, providing critical evidence that can guide clinical decision-making and enhance patient care20,21.
This study aims to systematically analyze FAERS reports of adverse events related to EMD from the first quarter of 2004 through the fourth quarter of 2024. By assessing the risks posed by these drugs and examining the demographic characteristics and onset patterns of EMD, we seek to offer reliable safety information that can guide clinical drug selection and treatment strategies. Ultimately, our findings aim to contribute to the development of more precise and evidence-based drug use guidelines, which will help mitigate the risk of drug-induced EMD and improve patients’ overall quality of life.
Methods
Data acquisition and preprocessing
The data for this study were sourced from the FAERS (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html), covering reports submitted from the first quarter of 2004 to the fourth quarter of 2024. FAERS integrates mandatory reports from pharmaceutical companies with voluntary submissions from healthcare professionals, and consumers. The dataset includes demographic information (e.g., sex, age, weight), drug details (e.g., dose, indication, route of administration), adverse event descriptions (e.g., date of onset, seriousness, outcome), and treatment timelines, all standardized according to the Medical Dictionary for Regulatory Activities (MedDRA, version 27.1). This standardization ensures consistency and comparability across reports, making FAERS a vital resource for pharmacovigilance and drug safety signal detection20,22.
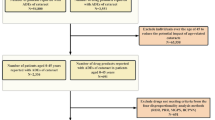
Initially, FAERS contained 22,249,476 records. After removing duplicates, 8,627,667 valid reports remained. To identify cases related to EMD, we selected all reports that included the Preferred Term (PT) Eye Movement Disorder (MedDRA code: 10061129). Eye movement disorder refers to dysfunction of the extraocular muscles, leading to abnormal or restricted eye movements, caused by conditions such as nystagmus, ocular muscle paralysis, and oculomotor nerve damage23.Following FDA guidelines, reports were organized by PRIMARYID, CASEID, and FDA_DT. For identical CASEID and FDA_DT values, the report with the highest PRIMARYID and most recent FDA_DT was selected18,24. To refine classification specificity, a narrow-range MedDRA query (SMQ) was applied, with a focus on reports flagged as “primary suspect.” This approach minimized the inclusion of irrelevant cases, ensuring a more accurate and focused dataset. The use of SMQ helped to filter out extraneous data, concentrating analysis on the most pertinent and reliable reports25,26. This process yielded 6,158 cases, associated with 1,834 distinct drugs. All drug names were standardized using the DrugBank database to ensure consistency27.
Signal detection and statistical methods
To evaluate the relationship between drugs and EMD, we applied four established disproportionality analysis methods: Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Bayesian Confidence Propagation Neural Network (BCPNN), and Multinomial Gamma-Poisson Shrinkage (MGPS). These methods are widely used for signal detection in pharmacovigilance and offer complementary strengths. The criteria for statistical significance were as follows: (1) ROR: a ≥ 3 and 95% CI lower > 1; (2) PRR: a ≥ 3 and 95% CI lower > 1; (3) BCPNN: IC025 > 0; (4) MGPS: EBGM05 > 2 and a > 0 (Tables 1 and 2). A drug-adverse event relationship was considered significant if it met the thresholds in all four methods25,28,29,30. Each method contributes unique advantages: ROR adjusts for reporting bias, PRR enhances specificity by comparing reporting frequencies, BCPNN effectively addresses reporting distortions, and MGPS excels at detecting rare signals and handling sparse data31. By employing these complementary approaches, we ensured comprehensive signal detection and cross-validated results, reinforcing the reliability of our findings. Statistical analyses and data cleaning, including the removal of duplicates, were conducted using R (version 4.2.2), with additional analyses performed using SPSS (version 26.0) and GraphPad Prism (version 10.1.2).
Subgroup and time-to-onset analysis for drug safety
Further analyses were performed to investigate the influence of patient comorbidities on the development of EMD. To ensure the accuracy of the reported cases, we cross-validated the data by reviewing reports submitted by medical professionals, thereby confirming the consistency of diagnoses with clinical expertise and reducing potential bias from non-professional reporters. Additionally, subgroup analyses were conducted based on gender, age (0–14, 15–24, 25–64, > 65), and indication (including both approved and off-label drug use), to assess variations in the occurrence of EMD across these different groups. A Time-to-Onset (TTO) analysis was also carried out to examine the temporal relationship between drug exposure and the onset of EMD. TTO was defined as the time interval between the onset of the adverse event (EVENT_DT) and the initiation of therapy (START_DT), providing insights into the timing of drug administration relative to the onset of adverse events. These analyses contributed to a comprehensive evaluation of drug safety, highlighting potential clinical implications and revealing broader patterns of drug-induced adverse events. A visual summary of the data cleaning, filtering, and classification process is provided in Fig. 1.
Raw data cleaning flowchart for drug-induced eye movement disorder. Flowchart of patient selection and data cleaning for drug-induced eye movement disorder in the FAERS database. The background map and flowchart were generated using Microsoft Visio 2021 (https://www.microsoft.com/visio) to illustrate the patient selection and data cleaning methodology for analyzing drug-induced eye movement disorder cases within the FAERS database.
Results
Baseline characteristics and gender-age specific patterns in drug-induced eye movement disorder
This study included 6,158 cases of drug-induced EMD adverse events. The cohort had an average age of 44.0 ± 23.9 years, with females representing a larger proportion (55.3%) compared to males (37.7%). Gender-based analysis revealed that females were more prevalent across all age groups, particularly in individuals over 30 years of age and in the elderly population, where the difference was especially pronounced. Conversely, males demonstrated a slight predominance in younger age groups, particularly in the 0–15 age range (Fig. 2A). Time trend analysis indicated a steady increase in the number of cases over the years, with a particularly sharp rise observed after 2012 (Fig. 2B). Regarding the professional background of the reporters, consumers constituted the largest group, accounting for 2,681 cases (43.5%), followed by healthcare professionals, specifically physicians, who reported 1,309 cases (21.3%) (Fig. 2C). In terms of drug administration routes, oral administration was the most frequently reported (2,316 cases, 37.6%), followed by intravenous (548 cases, 8.9%) and subcutaneous (317 cases, 5.1%) routes (Fig. 2D). The most common patient outcome was categorized as “Other serious condition” (3,554 cases, 57.7%), followed by hospitalization (1,655 cases, 26.9%) and life-threatening conditions (405 cases, 6.6%) (Fig. 2E). Geographically, the United States reported the highest number of cases (3,328 cases, 54.0%), followed by the United Kingdom (510 cases, 8.3%), Canada (269 cases, 4.44%), and Germany (222 cases, 3.6%) (Fig. 2F and G). Detailed demographic characteristics and subgroup analysis results are provided in Supplementary Tables 1 and 2.
Distribution of baseline data for patients reporting adverse events of drug-induced eye movement disorder in the FAERS database. The distribution of baseline data for patients reporting adverse events of eye movement disorders in the FAERS database includes various analyses. (A) A pyramid depicting the age distribution of patients reporting adverse events of eye movement disorders by gender. (B) The temporal distribution of eye movement disorder adverse event reports. (C) The distribution of occupations among patients with eye movement disorder adverse events. (D) The distribution of routes of administration for patients with eye movement disorder adverse events. (E) A histogram showing the distribution of outcomes in patients with eye movement disorder adverse events. (F) A histogram representing the distribution of eye movement disorder adverse event reports by country. (G) A heat map illustrating the distribution of reported adverse events of eye movement disorders by country.
Drug classification and risk assessment using ROR algorithm for eye movement disorder associations
To evaluate the association between drugs and EMD, a disproportionality analysis was conducted, identifying 30 drugs that exhibited positive signals, which were subsequently categorized according to their mechanisms of action (Fig. 3). Among these, antipsychotics accounted for the largest proportion of reports (636/1388), with aripiprazole being the most frequently reported (235 cases), followed by risperidone (142 cases). Antiseizure medications were also significantly represented (249/1388), with vigabatrin showing the highest number of reports (86 cases), followed by topiramate (49 cases) and levetiracetam (48 cases). Antidepressants, including sertraline (57 cases) and venlafaxine (24 cases), were also identified as potentially associated with EMD. Within the anxiolytics category, clobazam (14 cases) and lorazepam (11 cases) were also recognized. Although fewer reports were found in the direct acting antivirals category, some notable signals were observed, with acyclovir being the most frequently reported drug (35 cases), followed by ganciclovir (3 cases). Moreover, several other drugs demonstrated significant positive signals related to EMD, notably gabapentin (168 cases), botulinum toxin type A (69 cases), and teriflunomide (47 cases).
Grouped bubble chart of positive drug signals for eye movement disorders. This figure presents a grouped bubble chart illustrating positive drug signals for eye movement disorders. Different colors represent distinct drug categories, while the size of each bubble corresponds to the number of adverse event reports (n) associated with each drug linked to eye movement disorders.
To further analyze the risk associated with these drugs in relation to EMD, the ROR was used to evaluate potential signals within six predefined therapeutic categories. In the antipsychotic category, haloperidol exhibited the highest ROR for EMD (ROR = 12.86), followed by paliperidone (ROR = 11.72) and cariprazine (ROR = 10.88). Among the antiseizure medications, zonisamide demonstrated the highest ROR (26.03), with vigabatrin (ROR = 22.70) and tiagabine (ROR = 21.97) also presenting positive signals. Regarding antidepressants, sertraline showed the strongest association with EMD (ROR = 6.17), followed by desvenlafaxine (ROR = 5.32). In the anxiolytic category, clobazam (ROR = 7.20) and lorazepam (ROR = 5.47) were associated with positive signals. In the direct acting antivirals category, ganciclovir (ROR = 12.28) and acyclovir (ROR = 6.31) also showed positive signals. Additionally, other drugs such as miglustat (ROR = 15.45), valbenazine (ROR = 8.03), and donepezil (ROR = 7.54) demonstrated significant ROR values, further suggesting an association with EMD. A summary of these findings can be found in Fig. 4 and Supplementary Table 3. Additionally, disproportionality analysis was conducted on reports of the aforementioned 30 drugs from the MD source, all of which yielded positive signals (Supplementary Fig. 1).
Forest plots and heat maps of drugs with positive signals for drug-induced eye movement disorders based on disproportionality analysis methods from the FAERS database. The darker the color of the heat map, the higher its signal value, indicating a correspondingly greater risk of eye movement disorder. ROR reporting odds ratio, PRR proportional reporting ratio, BCPNN Bayesian confidence propagation neural network, MGPS multi-item gamma Poisson shrinker, CI confidence interval.
Subgroup stratification and risk assessment of drug-induced eye movement disorder
Subgroup analysis of 30 drugs comparing their use under indicated versus off-label conditions revealed that all 30 drugs showed positive signals under off-label use. Notably, 27 out of 30 drugs had a higher ROR in off-label use than in indicated use, suggesting that these drugs may independently cause EMD, irrespective of the underlying disease. The remaining three drugs—teriflunomide, donepezil, and botulinum toxin type A—may involve interactions between the drug and the disease itself in the development of EMD, as illustrated in Fig. 5.
Radar plots of indications and off-label uses of positive drugs for drug-induced eye movement disorders from FAERS database. The values represent the ROR values of the 30 drugs, which are positive according to all four algorithms (ROR, PRR, MGPS, BCPNN), under both their indicated and off-label uses. An ROR value of 0 indicates that the drug does not meet the criteria of the four algorithms in that subgroup, implying no statistical significance. ROR reporting odds ratio, PRR proportional reporting ratio, BCPNN Bayesian confidence propagation neural network, MGPS multi-item gamma Poisson shrinker, CI confidence interval.
Further analysis of the distribution of these 30 positive drugs across different age and gender groups revealed a notably higher impact in females, particularly in the 25–64 age group. Among female patients, the most frequently used drugs were aripiprazole, gabapentin, and botulinum toxin type A, reported in 127, 79, and 55 cases, respectively. In contrast, males exhibited a higher incidence of risperidone, paliperidone, and vigabatrin, with 109, 72, and 53 cases, respectively. The distribution of these drugs also varied significantly across age groups. In pediatric patients aged 0–14 years, vigabatrin and risperidone were the most commonly used drugs, reported in 39 and 36 cases, respectively. As patients transitioned into adolescence and young adulthood (ages 15–24), aripiprazole and risperidone were used more frequently, with aripiprazole appearing in 78 cases. In middle-aged patients (ages 25–64), gabapentin and aripiprazole were the most commonly used drugs, appearing in 59 and 43 cases, respectively. Among elderly patients (aged ≥ 65 years), gabapentin was the most frequently prescribed drug, appearing in 28 cases. The specific age- and gender-based distribution of each drug is shown in Fig. 6.
Sunburst chart depicting the age and gender distribution of positive drugs associated with drug-induced eye movement disorders from the FAERS database. The values represent the distribution of 30 positive drugs across gender and age groups. These drugs are identified as positive based on the results from all four algorithms (ROR, PRR, MGPS, and BCPNN). A larger area for each drug or subgroup (such as age or gender) indicates a higher number of reported cases. The drugs included in this analysis show a consistent association with drug-induced eye movement disorders across all four algorithms. For a detailed list of the drugs associated with each number, please see Table 3, where the drugs are listed in order of their corresponding ranking. ROR reporting odds ratio, PRR proportional reporting ratio, BCPNN Bayesian confidence propagation neural network, MGPS multi-item gamma Poisson shrinker, CI confidence interval.
To explore the potential risks associated with these drugs across various gender and age subgroups, a risk assessment analysis was conducted (Fig. 7). The results indicated that zonisamide (ROR = 56.34) and miglustat (ROR = 22.98) exhibited the highest drug risks in females, while vigabatrin (ROR = 26.56) and ganciclovir (ROR = 21.23) were associated with more significant risks in males. Drug risk also varied across age groups. In pediatric patients (ages 0–14), donepezil (ROR = 34.25) and ganciclovir (ROR = 11.71) were identified as higher-risk drugs. In young adults (ages 15–24), valbenazine (ROR = 69.17) and aripiprazole (ROR = 14.28) showed more pronounced risks. Among adults (ages 25–64), vigabatrin (ROR = 25.82) and brexpiprazole (ROR = 17.2) were identified as higher-risk drugs. For elderly patients (aged ≥ 65), zonisamide (ROR = 303.6) and lorazepam (ROR = 22.51) were recognized as the drugs with the highest associated risks.
Radar plots of age and gender subgroups for positive drugs associated with drug-induced eye movement disorders from the FAERS database. The values represent the ROR values of the 30 drugs, which are positive according to all four algorithms (ROR, PRR, MGPS, BCPNN), under both their age and gender subgroups. An ROR value of 0 indicates that the drug does not meet the criteria of the four algorithms in that subgroup, implying no statistical significance. ROR reporting odds ratio, PRR proportional reporting ratio, BCPNN Bayesian confidence propagation neural network, MGPS multi-item gamma Poisson shrinker, CI confidence interval.
Risk stratification and latency profiles of drugs based on BCPNN algorithm in eye movement disorder
We applied the BCPNN algorithm to assess the risk of various drugs associated with EMD and classified these risks based on the corresponding BCPNN values. Specifically, BCPNN values ranging from 0 to 1.5 were categorized as low risk, values between 1.5 and 3 as moderate risk, and values greater than 3 as high risk18. Among the 30 drugs evaluated, zonisamide exhibited the highest BCPNN value (3.03), classifying it as a high-risk drug, followed by vigabatrin (2.82) and tiagabine (2.79) (Fig. 8A).
In terms of the latency period for drug-induced EMD, we systematically analyzed the onset times for different drugs and assessed the differences in drug induction times using the quartile method. Our findings revealed that drugs such as acyclovir, donepezil, cariprazine, zonisamide, and gabapentin induced EMD within the shortest latency period, with onset typically occurring within one week. In contrast, the drugs with the longest induction latency included miglustat (1043 days), venlafaxine (312 days), and teriflunomide (234 days) (Fig. 8B).
Ranking of drug-induced eye movement disorders by BCPNN values and induction time. (A) Presents the ranking of drugs associated with eye movement disorders based on BCPNN values. (B) Illustrates the median induction time for eye movement disorders related to each drug. BCPNN Bayesian confidence propagation neural network.
Cumulative risk curve analysis further demonstrated significant differences in the onset times of EMD across drug categories (P < 0.0001) (Supplementary Fig. 2A). Subsequent pairwise comparisons revealed that direct acting antivirals induced EMD in the shortest time (10.73 days), while antidepressants were associated with the longest induction time (436.14 days) (Supplementary Fig. 2B). When categorizing the drugs into neuropsychiatric and non-neuropsychiatric groups, the average induction time for neuropsychiatric drugs was 230.54 days, with a significant difference compared to non-neuropsychiatric drugs (P < 0.001) (Supplementary Fig. 3).
Eye movement disorder associated with different drug categories
We further investigated the association between 30 positive drugs and specific eye movement disorders and found that the most frequent disorders were nystagmus (16/30), opsoclonus-myoclonus (12/30), and diplopia (10/30), followed by gaze palsy (8/30) and strabismus (8/30). These disorders showed varying associations with different drug classes, as indicated by the reporting odds ratios (ROR).
In antipsychotics, oculogyric crisis was most strongly associated with brexpiprazole (ROR = 88.45), followed by cariprazine (ROR = 47.19) and lurasidone (ROR = 44.92). Saccadic eye movement disorder was notably linked to aripiprazole (ROR = 13.2), while gaze palsy was most commonly associated with haloperidol (ROR = 11.12) and paliperidone (ROR = 5.3). Nystagmus was reported with asenapine (ROR = 6.9) and haloperidol (ROR = 2.28), while strabismus showed weaker signals with haloperidol (ROR = 3.59) and risperidone (ROR = 3.11). Similarly, in antiseizure medications, nystagmus was the most frequently observed disorder, with zonisamide showing the highest ROR (19.68), followed by carbamazepine (ROR = 16.82) and vigabatrin (ROR = 12.25). Diplopia was most strongly linked to tiagabine (ROR = 12.99), cenobamate (ROR = 11.74), and zonisamide (ROR = 6.68). Gaze palsy was most strongly associated with carbamazepine (ROR = 10.09) and levetiracetam (ROR = 5.71). Furthermore, levetiracetam (ROR = 24.06) also showed a significant but rare signal for opsoclonus-myoclonus syndrome. Strabismus was notably linked to vigabatrin (ROR = 29.02) and carbamazepine (ROR = 4.24).
For antidepressants, opsoclonus-myoclonus syndrome was most significantly associated with venlafaxine (ROR = 23.32). Oculogyric crisis was frequently reported with fluoxetine (ROR = 9.48) and sertraline (ROR = 5.15), while nystagmus was observed with both fluoxetine (ROR = 5.65) and sertraline (ROR = 3.02). Gaze palsy was noted with fluoxetine (ROR = 4.52) and sertraline (ROR = 2.68), and strabismus was consistently observed with both drugs (fluoxetine ROR = 3.92; sertraline ROR = 3.14). In anxiolytics, similarly, clobazam showed the strongest association with diplopia (ROR = 11.74) and nystagmus (ROR = 11.45). Oculogyric crisis was observed with lorazepam (ROR = 3.4), and strabismus was reported with lorazepam (ROR = 3.76). Furthermore, for direct acting antivirals, acyclovir was associated with nystagmus (ROR = 4.24).
Miscellaneous medications also exhibited a range of eye movement disorders. Specifically, miglustat was strongly associated with gaze palsy (ROR = 33.17), while general eye movement disorder was linked to both gabapentin (ROR = 13.83) and miglustat (ROR = 12.64). Ophthalmoplegia was notably linked to botulinum toxin type A (ROR = 9.61). Furthermore, oculogyric crisis was reported with valbenazine (ROR = 7.33), and diplopia was observed with gabapentin (ROR = 2.78) and teriflunomide (ROR = 1.87), though these signals were weaker. Further details on additional drugs and their specific adverse reactions are provided in Supplementary Table 4.
Discussion
Eye movement disorders significantly impair visual function and present substantial challenges to daily living2,7,8,9. Although therapeutic strategies have advanced, their impact on eye movement disorders remains limited, highlighting the urgent need for early detection and intervention to better manage these disorders and mitigate their effects on patients’ daily lives32,33. This study systematically explored the association between drug use and eye movement disorders using data from the FAERS database. Our findings show that these disorders are most frequently reported in Western countries, particularly among middle-aged and elderly women, with a steadily increasing incidence over time. Using four disproportionate analysis methods, we identified 30 drugs associated with a heightened risk of eye movement disorders, the majority (25 out of 30) being central nervous system medications, such as antiseizure medications, antipsychotics, and antidepressants. Further analysis revealed the risk profiles of these drugs, highlighting their varying roles in the onset of these disorders and differences based on patient demographics and clinical indications. These findings provide crucial real-world data that can guide clinical decision-making and emphasize the importance of closely monitoring drug-induced ocular complications. By identifying key drugs linked to eye movement disorders, this study contributes valuable insights for future research aimed at reducing these adverse effects.
Antiseizure medications (ASMs) are first-line treatments for epilepsy, with approximately two-thirds of patients achieving complete control of seizures after their use34,35. While their therapeutic efficacy is well-established, the ocular motor effects of ASMs were systematically characterized as early as 1980 by Bittencourt et al., who demonstrated quantitative smooth pursuit abnormalities in epileptic patients36. This foundational work, along with subsequent studies, has established that ASMs may disrupt multiple ocular motor pathways - including brainstem, cerebellar, vestibular, and vergence systems - often in dose-dependent patterns16,37. Beyond classical adverse effects like hepatotoxicity and fatigue, these medication-induced eye movement disorders now represent a well-characterized spectrum of neuro-ophthalmic complications, ranging from early-reported smooth pursuit impairments to contemporary understandings of vestibulo-ocular reflex (VOR) disturbances16,37,38,39.
However, the occurrence of ocular motor dysfunction is not solely attributable to drug side effects. The pathological changes associated with epilepsy itself may also influence ocular motor function. For instance, a study by Mrabet et al. on drug-naïve patients with various epileptic syndromes, including juvenile myoclonic epilepsy (JME) and temporal lobe epilepsy, found that 52.2% of patients exhibited saccadic impairments. The most common abnormalities included prolonged horizontal saccadic latencies (32.1%) and smooth pursuit dysfunction (22.6%). These findings highlight that epilepsy itself, independent of medications, can disrupt ocular motor pathways, which are particularly evident in syndrome-specific patterns such as JME. This supports the notion that ocular motor dysfunction in epilepsy may arise from the disease’s neurophysiological impact, rather than solely from drug side effects40,41. Similarly, research by Au Yong et al. in adult patients with refractory focal epilepsy demonstrated that these individuals exhibited impaired spatial accuracy in ocular motor tasks42. This finding further supports the use of ocular motor dysfunction as an indicator of brain dysfunction caused by epilepsy. Taken together, the underlying condition of epilepsy may also be a critical factor in the development of eye movement disorder. Therefore, understanding the interaction between ASMs and the pathophysiology of epilepsy is essential for deciphering the mechanisms behind ocular motor dysfunction. To further explore this issue, we analyzed the effects of ASMs under both their indicated and off-label uses. Our data reveal a significant positive correlation between ASMs and ocular motor dysfunction when used off-label, whereas no such correlation was found when used for their approved indications. This suggests that ASMs may independently cause ocular motor disorders in specific circumstances, beyond the effects of epilepsy itself. Additionally, subgroup analysis indicated that the mechanisms behind ASM-induced ocular dysfunction may not be entirely dose-dependent; some drugs appear to have non-dose-dependent or drug-specific effects, providing important insights into the side effect mechanisms of ASMs16.
Further subgroup analysis highlighted the ocular side effects of vigabatrin in patients with refractory epilepsy and infantile spasms. While previous studies have primarily focused on vigabatrin’s effects on visual field defects43,44. However, our study is the first to reveal a notable gender difference in the incidence of vigabatrin-related eye movement disorder, with a higher risk observed in male patients (ROR: 26.56) compared to females (ROR: 18.11). This observation, along with findings from other drug categories, suggests that the impact of gender on EMD risk is drug-specific, indicating a modulatory rather than universally independent role of gender in EMD development. This gender difference may be related to physiological differences in drug metabolism, pharmacological action, and drug response between males and females. Moreover, the eye movement disorder side effects of vigabatrin are particularly prominent in patients aged 0–14 years, which may be due to delayed myelination of the cortico-mesencephalic vertical eye movement pathways, or immaturity or dysfunction of the extra-geniculocalcarine visual pathways in this age group45. Additionally, we found that both topiramate and carbamazepine were associated with eye movement disorders, which may be related to ocular muscle tone dysfunction, potentially leading to diplopia. These effects were more commonly observed in female patients46. These findings emphasize the importance of considering gender-specific responses to these drugs in clinical practice. Further analysis of cannabidiol revealed its potential to affect nystagmus, with this side effect being more pronounced in females47. An important aspect of our study was the analysis of the onset times for ASM-induced eye movement disorder, which offers crucial insights for clinical monitoring. Our results indicate that Topiramate typically induces ocular dysfunction within 21 days, whereas vigabatrin’s side effects emerge after about 36 days. Carbamazepine’s effects take longer, typically appearing around 127 days post-treatment, and cannabidiol shows a delayed onset, with significant effects usually observed after 166 days. These varying onset times underscore the need for clinicians to closely monitor ocular motor function, particularly during the early stages and prolonged use of ASMs, especially in cases of polypharmacy16,43,46,47.
Antipsychotic medications are commonly prescribed for conditions like schizophrenia and bipolar disorder, effectively managing symptoms such as hallucinations, delusions, and mood instability, while preventing relapse48. However, they are associated with several side effects, including ocular motor disturbances, particularly ocular motor crises (OGC). These crises, characterized by acute extraocular muscle dystonia and upward eye deviation, can last from minutes to hours and significantly impair quality of life49. The mechanism underlying ocular motor disturbances is linked to antipsychotics’ effects on dopamine and serotonin receptors, particularly the blockade of dopamine D2 receptors. Dopamine plays a key role in eye movement, and antipsychotics may disrupt the coordination of extraocular muscles46,50. First-generation antipsychotics (FGAs), like haloperidol, are particularly prone to inducing these crises due to their strong extrapyramidal side effects (EPS). While second-generation antipsychotics (SGAs) are favored for their lower EPS risk, some, such as aripiprazole, have been linked to tardive dystonia51,52. Additionally, asenapine, paliperidone, and risperidone have also been associated with ocular motor crises17,53,54,55. Given the long-term nature of antipsychotic treatment, our study found that they represent the largest proportion of drugs linked to ocular motor disturbances (636/1388), with an onset averaging 214.93 days. This emphasizes the need for regular ophthalmologic monitoring during prolonged therapy56.
Antidepressant medications, by modulating the activity of neurotransmitters such as serotonin, norepinephrine, and dopamine, are effective in alleviating depressive symptoms. However, despite their significant mood-improving effects, these drugs are also associated with a range of movement disorders, including akathisia, bruxism, and dystonia, which pose considerable challenges in clinical practice57,58. Our study further underscores the potential risk of antidepressant-induced ocular movement disorders, particularly among adults aged 25 years and older. This finding is consistent with a retrospective study by Leat et al., which demonstrated that both age and the use of antidepressants are linked to ocular motor disorders, with antidepressant use showing a stronger association than age itself59. The movement disorders induced by these drugs are often related to non-specific adverse effects in the basal ganglia and cerebellum. Given this, future research should not only focus on developing more selective medications with fewer side effects but should also pay closer attention to the neurochemical characteristics of the affected functional systems. Such an approach could provide deeper insights into the pathophysiological mechanisms underlying these adverse effects and help mitigate them in clinical settings60.
In addition to the concerns regarding antidepressants, our study also identified potential risks associated with the anxiolytic drugs clobazam and lorazepam in inducing ocular movement disorders. These findings corroborate those of Sarah E. Aylett, who observed that children with epilepsy may exhibit eye-rolling symptoms when intoxicated with clobazam, with dosage reduction alleviating this side effect61. Similarly, Katharina Bey and colleagues reported that lorazepam can impair ocular saccadic function, which further supports our conclusions62. Furthermore, our study highlights the need for further investigation into the relationship between antiviral medications, such as ganciclovir and acyclovir, and ocular movement disorders. Our findings suggest that these drugs tend to induce ocular movement disorders approximately 10.73 days after administration. Supporting this, reports from Kim and Openshaw indicate that ocular movement disorders often present acutely but resolve gradually upon discontinuation of the medication. These findings emphasize the importance of closely monitoring the use of antiviral drugs, especially during the critical period when the risk of ocular movement disorders is elevated63,64.
In conclusion, this study systematically assessed the association between drug exposure and EMD, delineating distinct risk profiles and time-to-onset patterns across various pharmacological classes. These findings provide valuable pharmacovigilance insights that may inform risk mitigation strategies in clinical settings. To reduce the likelihood of drug-induced EMD, careful drug selection, individualized dosing, and appropriate treatment durations are recommended. Despite these contributions, several limitations warrant consideration. First, disproportionality analyses can identify potential safety signals but do not establish causality. Although we employed subgroup analyses and multiple signal detection methods to enhance robustness, residual confounding—particularly protopathic bias—may persist. Second, inherent limitations of spontaneous reporting systems, including underreporting, selective reporting, and incomplete dosage documentation, restrict detailed evaluation of dose–response relationships. Third, the absence of patient-level clinical information (e.g., comorbidities, concomitant medications) hinders assessment of individual susceptibility and drug–drug interactions. Lastly, limited clinical granularity within the FAERS database constrains accurate subtyping of EMD25. Future studies should seek to validate these findings using independent pharmacovigilance databases, investigate high-risk populations in real-world contexts, and explore the neuropharmacological mechanisms underlying drug-induced eye movement disorders. Such efforts will be essential for refining clinical practice and enhancing medication safety in neurologically vulnerable populations.
Conclusion
In conclusion, this study provides a comprehensive analysis of drug-induced eye movement disorder, identifying drugs linked to these eye movement disorders and examining their distribution patterns and pharmacological targets. Through the systematic classification of 30 drugs, coupled with subgroup analyses (based on age, gender, and indications), and an evaluation of the duration of induced eye movement disorders, we have revealed the intricate relationship between drug use and eye movement disorder. Our findings underscore the epidemiological characteristics of high-risk drugs and highlight the critical importance of proactive pharmacovigilance. These results not only enhance our understanding of drug safety but also offer valuable insights to guide clinical decisions regarding drug selection, dosage adjustments, and ongoing monitoring, ultimately optimizing patient care and minimizing potential adverse effects.
Data availability
The datasets used in this study are publicly available from the FDA Adverse Event Reporting System (FAERS). FAERS provides open access to its data without the requirement for accession numbers or user accounts. Data can be directly accessed and downloaded from the FAERS Public Dashboard (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html).
References
Self, J. E. et al. Management of nystagmus in children: a review of the literature and current practice in UK specialist services. Eye (Lond). 34, 1515–1534 (2020).
Leigh, R. J. & Tomsak, R. L. Drug treatments for eye movement disorders. J. Neurol. Neurosurg. Psychiatry. 74, 1–4 (2003).
Yoo, Y. J., Yang, H. K., Kim, N. & Hwang, J. M. Pediatric orbital wall fractures: prognostic factors of diplopia and ocular motility limitation. PLoS One. 12, e0184945 (2017).
Weidauer, S., Hofmann, C., Wagner, M. & Hattingen, E. Neuroradiological and clinical features in ophthalmoplegia. Neuroradiology 61, 365–387 (2019).
Ranjan, R. et al. Infectious ophthalmoplegias. J. Neurol. Sci. 427, 117504 (2021).
Pollock, A. et al. Interventions for disorders of eye movement in patients with stroke. Cochrane Database Syst. Rev. CD008389 https://doi.org/10.1002/14651858.CD008389.pub2 (2011).
Rossor, T. et al. Diagnosis and management of Opsoclonus-Myoclonus-Ataxia syndrome in children: an international perspective. Neurol. Neuroimmunol. Neuroinflamm. 9, e1153 (2022).
Rowe, F. J. et al. Interventions for eye movement disorders due to acquired brain injury. Cochrane Database Syst. Rev. 3, CD011290 (2018).
Rowe, F. et al. Reading difficulty after stroke: ocular and Non ocular causes. Int. J. Stroke. 6, 404–411 (2011).
Almer, Z., Klein, K. S., Marsh, L., Gerstenhaber, M. & Repka, M. X. Ocular motor and sensory function in parkinson’s disease. Ophthalmology 119, 178–182 (2012).
Willard, A. & Lueck, C. J. Ocular motor disorders. Curr. Opin. Neurol. 27, 75–82 (2014).
McClelland, C., Manousakis, G. & Lee, M. S. Progressive external ophthalmoplegia. Curr. Neurol. Neurosci. Rep. 16, 53 (2016).
Campi, I. et al. Quantification of global ocular motility impairment in graves’ orbitopathy by measuring eye muscle ductions. Thyroid 31, 280–287 (2021).
Kaur, P. et al. Amiodarone-Induced nystagmus and ataxia: case report and systematic review of case reports. J. Clin. Pharmacol. 63, 1324–1329 (2023).
Murty, S. Antiepileptic overdose. Indian J. Crit. Care Med. 23, S290–S295 (2019).
Hamed, S. A. Ocular dysfunctions and toxicities induced by antiepileptic medications: types, pathogenic mechanisms, and treatment strategies. Expert Rev. Clin. Pharmacol. 12, 309–328 (2019).
Gardner, D. M. et al. Incidence of oculogyric crisis and Long-Term outcomes with Second-Generation antipsychotics in a First-Episode psychosis program. J. Clin. Psychopharmacol. 35, 715–718 (2015).
Chen, X. D., Xiao, K. H. & Zhou, C. B. Drug-induced retinal vein occlusion: a disproportionality analysis from the FDA adverse event reporting system (2004–2023). Front. Pharmacol. 15, 1480269 (2024).
Carlson, J., McBride, K. & O’Connor, M. Drugs associated with cataract formation represent an unmet need in cataract research. Front. Med. (Lausanne). 9, 947659 (2022).
Li, D. et al. Drug-Induced acute pancreatitis: A Real-World pharmacovigilance study using the FDA adverse event reporting system database. Clin. Pharmacol. Ther. 115, 535–544 (2024).
Yu, R. J., Krantz, M. S., Phillips, E. J. & Stone, C. A. Emerging causes of Drug-Induced anaphylaxis: A review of Anaphylaxis-Associated reports in the FDA adverse event reporting system (FAERS). J. Allergy Clin. Immunol. Pract. 9, 819–829 (2021).
Sakaeda, T., Tamon, A., Kadoyama, K. & Okuno, Y. Data mining of the public version of the FDA adverse event reporting system. Int. J. Med. Sci. 10, 796–803 (2013).
Mozzicato, P. Standardised MedDRA queries: their role in signal detection. Drug Saf. 30, 617–619 (2007).
Yin, Y., Shu, Y., Zhu, J., Li, F. & Li, J. A real-world pharmacovigilance study of FDA adverse event reporting system (FAERS) events for osimertinib. Sci. Rep. 12, 19555 (2022).
Wu, S. N. et al. Drug-associated glaucoma: A real-world study based on the food and drug administration adverse event reporting system database. Clin. Exp. Ophthalmol. https://doi.org/10.1111/ceo.14454 (2024).
Li, D. et al. Severe cutaneous adverse reactions to drugs: A real-world pharmacovigilance study using the FDA adverse event reporting system database. Front. Pharmacol. 14, 1117391 (2023).
Wishart, D. S. et al. DrugBank 5.0: a major update to the drugbank database for 2018. Nucleic Acids Res. 46, D1074–D1082 (2018).
Wu, S. N. et al. Drug-Related keratitis: A Real-World FDA adverse event reporting system database study. Transl. Vis. Sci. Technol. 13, 17 (2024).
Wu, S. N. et al. Real-World large sample assessment of Drug-related dry eye risk: based on the FDA adverse event reporting system database. Asia Pac. J. Ophthalmol. (Phila). 13, 100104 (2024).
Chen, X. et al. Real world pharmacovigilance assessment of drug related macular degeneration risks. Sci. Rep. 15, 1220 (2025).
Jiang, Y. et al. Safety assessment of brexpiprazole: Real-world adverse event analysis from the FAERS database. J. Affect. Disord. 346, 223–229 (2024).
Lee, S. K. & Lee, M. S. Ocular neuromyotonia: a review of diagnosis and treatment. Curr. Opin. Ophthalmol. 33, 465–470 (2022).
Strupp, M. et al. Pharmacotherapy of vestibular and ocular motor disorders, including nystagmus. J. Neurol. 258, 1207–1222 (2011).
Perucca, P., Scheffer, I. E. & Kiley, M. The management of epilepsy in children and adults. Med. J. Aust. 208, 226–233 (2018).
Asadi-Pooya, A. A., Brigo, F., Lattanzi, S. & Blumcke, I. Adult epilepsy. Lancet 402, 412–424 (2023).
Bittencourt, P. R., Gresty, M. A. & Richens, A. Quantitative assessment of smooth-pursuit eye movements in healthy and epileptic subjects. J. Neurol. Neurosurg. Psychiatry. 43, 1119–1124 (1980).
Veerapandiyan, A. et al. Oculogyric crises secondary to lamotrigine overdosage. Epilepsia 52, e4–6 (2011).
A, S. & G, D. S. L, G., E, R. A review on antiepileptic drugs-dependent fatigue: pathophysiological mechanisms and incidence. Eur. J. Pharmacol. 718 (2013).
Vidaurre, J., Gedela, S. & Yarosz, S. Antiepileptic drugs and liver disease. Pediatr. Neurol. 77, 23–36 (2017).
Lunn, J. et al. Saccadic eye movement abnormalities in children with epilepsy. PLoS One. 11, e0160508 (2016).
Mrabet, S. et al. Study of eye movements abnormalities in epilepsy. Neuroophthalmology 49, 137–146 (2025).
Au Yong, H. M. et al. Ocular motility as a measure of cerebral dysfunction in adults with focal epilepsy. Epilepsy Behav. 141, 109140 (2023).
Jm, W., Pem, S. & C, K. Objective derivation of the morphology and staging of visual field loss associated with Long-Term Vigabatrin therapy. CNS Drugs 33, (2019).
Mt, M., Oc, T. W. B. M. & Ca, W. S. Retinal defect in children with infantile spasms of varying etiologies: an observational study. Neurology 94 (2020).
Rodan, L. H., Tein, I. & Buncic, R. Clinical reasoning: a case of abnormal eye movements in an infant: more than Meets the eye. Neurology 81, e112–115 (2013).
Richa, S. & Yazbek, J. C. Ocular adverse effects of common psychotropic agents: a review. CNS Drugs. 24, 501–526 (2010).
Mtm, W. & Hv, D. M. Cannabinoids and the eye. Survey Ophthalmol. 66 (2021).
Schneider-Thoma, J. et al. Comparative efficacy and tolerability of 32 oral and long-acting injectable antipsychotics for the maintenance treatment of adults with schizophrenia: a systematic review and network meta-analysis. Lancet 399, 824–836 (2022).
M, V. B., Ha, P., St, C. & Fe, C. Ziprasidone-related oculogyric crisis in an adult. Clin. Neurol. Neurosurg. 111 (2009).
O, U. & A, D. Tardive oculogyric crisis during treatment with clozapine: report of three cases. Clin. Drug Investig. 27 (2007).
S, L. et al. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet 373 (2009).
C, R. K. et al. Second-generation antipsychotic drugs and extrapyramidal side effects: a systematic review and meta-analysis of head-to-head comparisons. Schizophr. Bull. 38 (2012).
M, S. & J, K. Oculogyric Crises. Tremor and other hyperkinetic movements 7 (2017).
Hk, L., Cu, P., C, L. & Cu, L. Tardive dystonic symptoms associated with Aripiprazole treatment. Progr. Neuro Psychopharmacol. Biol. Psychiatry 32 (2008).
Darling, A., Poo, P., Perez-Duenas, B. & Campistol, J. [Medication-related oculogyric crises: a description of four cases and a review of the literature]. Rev. Neurol. 56, 152–156 (2013).
Rubio, J. M. et al. Long-term continuity of antipsychotic treatment for schizophrenia: A nationwide study. Schizophr. Bull. 47, 1611–1620 (2021).
Blier, P. Neurobiology of depression and mechanism of action of depression treatments. J. Clin. Psychiatry. 77, e319 (2016).
Revet, A. et al. Antidepressants and movement disorders: a postmarketing study in the world pharmacovigilance database. BMC Psychiatry. 20, 308 (2020).
Leat, S. J. et al. Binocular vision and eye movement disorders in older adults. Invest. Ophthalmol. Vis. Sci. 54, 3798–3805 (2013).
Zádori, D., Veres, G., Szalárdy, L., Klivényi, P. & Vécsei, L. Drug-induced movement disorders. Expert Opin. Drug Saf. 14, 877–890 (2015).
Aylett, S. E., Cross, H. & Berry, D. Eye rolling as a manifestation of clobazam toxicity in a child with epilepsy. Dev. Med. Child. Neurol. 48, 612–615 (2006).
Bey, K., Lippold, J. V., Aslan, B., Hurlemann, R. & Ettinger, U. Effects of lorazepam on prosaccades and saccadic adaptation. J. Psychopharmacol. 35, 91–99 (2021).
Openshaw, H., Slatkin, N. E. & Smith, E. Eye movement disorders in bone marrow transplant patients on cyclosporin and ganciclovir. Bone Marrow Transpl. 19, 503–505 (1997).
Kim, J. H., Lee, S. J. & Kim, M. External ophthalmoplegia with orbital myositis in an adult patient after chickenpox infection. BMJ Case Rep bcr2013202415 (2014).
Funding
This study was supported by the Zhejiang Provincial County-Level Traditional Chinese Medicine Advantageous Discipline Construction Plan Project (grant NO. 2023-XK-D017).
Author information
Authors and Affiliations
Contributions
Si-Yuan Hong and Peng-He Wang conceived the research idea. Chao-Bing Zhou and Xiao-Dong Chen performed data cleaning and conducted the literature review. Si-Yuan Hong, Chao-Bing Zhou, and Xiao-Dong Chen contributed to drafting the manuscript and making critical revisions to its content. Guang-Ping Zhu and Chun-Hong Gao conducted the analysis and prepared the figures and tables. Si-Yuan Hong and Peng-He Wang provided a critical review of the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hong, SY., Zhou, CB., Chen, XD. et al. Pharmacovigilance study of drug-induced eye movement disorder based on FDA adverse event reports from 2004 to 2024. Sci Rep 15, 21341 (2025). https://doi.org/10.1038/s41598-025-05709-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05709-3
Keywords
This article is cited by
-
Evaluating adverse events reported for non-steroidal anti-inflammatory drugs in osteoarthritis: a real-world pharmacovigilance study
Inflammopharmacology (2026)
-
Neonatal-Onset Developmental Epileptic Encephalopathy due to 2q24.3 Microduplication Associated with Incidental Finding of RLBP1-Related Retinopathy
Indian Journal of Pediatrics (2025)