Abstract
This study was performed to investigate the prognostic value of assessing symptom burden using the Edmonton Symptom Assessment System (ESAS) in older patients with Heart Failure (HF). This prospective cohort study was performed in consecutive patients ≥ 60 years old hospitalized with HF at a university hospital between September 2020 and June 2023. ESAS was used to assess nine common symptoms at hospital discharge (total score: 0–90) with higher scores indicating greater severity. The primary outcome was the combined event of HF readmission and all-cause mortality within 6 months. The median ESAS sum score in the study population consisting of 306 patients (median age, 79 years; 54.9% male) was 13 points. The commonly reported moderate-to-severe symptoms were impaired well-being (34.9%), anxiety (27.5%), drowsiness (25.8%), tiredness (24.9%), and depression (22.2%). In multivariate analyses, ESAS sum score was significantly associated with the combined event (adjusted hazard ratio for each 5-point increase, 1.10 (95% confidence interval: 1.04–1.17; p < 0.001). Inclusion of ESAS sum score in the risk model significantly increased both continuous net reclassification improvement (p = 0.028) and integrated discrimination improvement (p = 0.002) for the primary outcome. Comprehensive assessment of symptom burden with ESAS provided additional prognostic information to conventional risk factors in older patients with HF.
Similar content being viewed by others
Introduction
The incidence of heart failure (HF) is increasing worldwide with the aging of the population, representing a global public health challenge1. In addition to increased morbidity, mortality, and medical costs, HF has been shown to have adverse effects on quality of life (QoL) and to be associated with increased rates of depression, anxiety, and stress2. As optimized disease management requires insight into how patients experience the disease and participate in their own care3 patient-reported outcomes (PROs) have been developed as measure for estimating their objective well-being4.
Along with functional status and health-related QoL, symptom burden represents a general component of patient health status that can have a negative impact on overall QoL5. Patients with HF have extensive symptom burden beyond the typical manifestations of the disease, such as dyspnea and fatigue6. Symptoms often persist despite medical management and device therapy, and older patients with HF commonly show a reduction in QoL over time7. Although a number of tools are available for assessing symptom burden in clinical research, they are not commonly used to guide clinical practice. The Edmonton Symptom Assessment System (ESAS), originally developed to assess comprehensive symptoms in patients with end-stage cancer requiring palliative care8,9 is a simple patient-friendly tool that can be completed quickly and is one of the most commonly cited questionnaires is studies of symptom burden as a PRO measure in HF7. The ESAS is used extensively for screening and monitoring of symptoms in clinical settings worldwide as it enables rapid assessment of multiple symptoms simultaneously, and it has been shown to be useful for predicting prognosis in the fields of palliative care and oncology8. In patients with HF, ESAS score has been shown to be correlated with New York Heart Association (NYHA) functional class and Kansas City Cardiomyopathy Questionnaire (KCCQ) score10 which is frequently used as a measure of global health status in this population. As the associations of ESAS score with prognosis of HF have not been characterized in detail, the present study was performed to investigate the prognostic significance of symptom burden assessed by ESAS in older patients with HF.
Methods
Study design and patient population
Consecutive patients ≥ 60 years old admitted to Nagoya University Hospital due to decompensation of HF diagnosed using the Framingham criteria between September 2020 and June 2023, in whom symptom burden was evaluated at the time of discharge, were included in this prospective observational study. Patients with left ventricular assist device implantation, those undergoing cardiac surgery, or who died during the period of hospitalization were excluded from the study.
Data collection
Baseline findings of physical examinations, blood samples, echocardiography, and medications were obtained before discharge for all patients with hemodynamic stability. The estimated glomerular filtration rate (eGFR) was determined using the Japanese Society of Nephrology formula11 and renal dysfunction was diagnosed based on eGFR < 60 mL/min/1.73 m2. To assess the nutritional score, the geriatric nutritional risk index (GNRI) was calculated using the following formula: 1.489 × serum albumin (g/L) + 41.7 × (body weight [kg]/ideal body weight)12. Ideal body weight was calculated using the formula: 22 × (height m2). Physical and cognitive function were assessed by trained physiotherapists after treatment in the acute phase. Handgrip strength was determined using a Jamar digital dynamometer (MG-4800 digital grip strength meter; Charder Electronic, Taichung, Taiwan) with the patient in a sitting position. Briefly, patients performed two maximal isometric voluntary contractions of both hands for 3 s each, and the highest value, expressed as an absolute value (in kg), was used in the analyses as a measure of physical function. Cognitive function was examined using the Japanese version of the Mini-Mental State Examination (MMSE), which is a brief screening test for cognitive impairment consisting of 11 items designed to measure orientation, registration, attention, calculation, recall, and language13. The maximum total score is 30, with higher scores indicating better performance.
The primary outcome was the combined event of readmission due to HF and all-cause mortality within 6 months after discharge. After discharge, most patients were followed up in outpatient clinics at least every 3 months in addition to according to medical necessity. HF readmission information and survival status were determined from annual reviews of medical records or telephone- and mail-based surveys. Patients without follow-up data at 6 months after hospital discharge were excluded from the analysis.
Symptom burden
The symptoms of each patient at discharge from hospital were assessed using the self-administered ESAS revised Japanese version questionnaire14 a validated and reliable tool for assessing the severity of nine common symptoms (anxiety, depression, drowsiness, lack of appetite, nausea, pain, shortness of breath, tiredness, and impaired well-being) and an optional 10th symptom (other symptoms) with “now” as the timeframe. According to the previous studies, each symptom was rated on an 11-point numeric scale with scores ranging from 0 (absence of symptom) to 10 (worst possible symptom), yielding a total score between 0 and 90 points (excluding 10th symptom)8,14,15,16. The ESAS scores were classified according to severity as follows: 0, no symptoms; 1–3, mild; 4–6, moderate; and 7–10, severe8.
Statistical analysis
Continuous variables are presented as the median and interquartile range (IQR). Categorical variables are expressed as numbers and percentages. The cohort was divided into three groups according to the ESAS sum score tertiles. Differences between groups were evaluated by the Kruskal-Wallis test for continuous variables and chi-squared or Fisher’s exact test for dichotomous variables as appropriate. The relations of ESAS sum score with each component of ESAS were evaluated by Spearman’s rank correlation analysis.
Event-free survival curves were constructed using the Kaplan-Meier survival method and compared using log-rank statistics. For the outcome of the combination of HF readmission and all-cause mortality, the prognostic predictive capability of ESAS sum score was examined by Cox regression analysis constructing two predictive models as follows: model 1 (baseline model), Meta-Analysis Global Group in Chronic HF (MAGGIC) risk score and (log-transformed) B-type natriuretic peptide (BNP) levels at discharge; and model 2 consisting of model 1 + geriatric assessments (i.e., GNRI score + handgrip strength + MMSE score). We used the MAGGIC risk score as an adjustment variable in multivariate Cox regression analyses because it is a well-validated risk score for Japanese patients with HF17. Based on their clinical importance and previous studies, these measures were selected as adjustment variables in multivariate Cox regression analyses for the combined event of HF readmission and all-cause mortality as outcomes, after confirming the absence of multicollinearity. Multivariable Cox regression models were constructed using multiple imputation to take into account the missing covariate data, excluding ESAS score. A total of 20 data sets were created using a chained-equations procedure18. Parameter estimates were obtained for each data set and subsequently combined to produce an integrated result using the method described by Barnard and Rubin19. Subgroup analyses of symptom burden were performed to examine the potential effect on the association of ESAS sum score with the combined event of HF readmission and mortality within 6 months for an exploratory survey by Cox regression analysis with adjustment for MAGGIC risk score and (log-transformed) BNP level as potential confounders: age (stratified at 75 years), sex, body mass index (BMI, stratified at 18.5 kg/m2), and left ventricular ejection fraction (LVEF, stratified at 50%). Hazard ratios (HRs) are reported with corresponding 95% confidence intervals (CIs).
Logistic regression analysis of ESAS sum score was performed to examine the combined event risk adjusting for MAGGIC risk score and (log-transformed) BNP level. Adjusted odds ratios (ORs) are reported with the corresponding 95% CIs. To evaluate the incremental prognostic value of ESAS sum score, we constructed two models for the outcome of the combined event: a baseline model incorporating preexisting risk factors and the baseline model + ESAS sum score. The baseline model for the composite outcome included MAGGIC risk score and (log-transformed) BNP level. For the outcome, we compared the areas under the curve (AUCs) between the two models, and calculated the net reclassification improvement (NRI) and integrated discrimination improvement (IDI) with addition of the ESAS sum score to the baseline model20. Decision curve analysis was performed to quantify the net benefits at different threshold probabilities to assess the clinical usefulness of the model21.
A two-tailed p < 0.05 was taken to indicate statistical significance for all analyses except interaction analysis where a threshold of p < 0.10 was used to indicate a significant interaction taking into account the somewhat exploratory nature of the subgroup analyses. Statistical analyses were performed using SPSS version 29.0 (IBM Corp.) and R version 3.2.1 (R Foundation for Statistical Computing).
Results
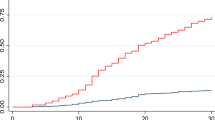
The initial study population consisted of 385 patients ≥ 60 years old hospitalized with HF. Seventy-nine patients were excluded, and the remaining 306 patients (median age 79 years, 54.9% male, 48.7% had LVEF ≥ 50%) for whom complete follow-up data at 6 months were available were included in the analysis (Fig. 1). The median ESAS sum score of the study population was 13 points (IQR 5–23) (Fig. 2A), and the most frequently reported moderate-to-severe symptoms at hospital discharge were impaired well-being (34.9%), anxiety (27.5%), drowsiness (25.8%), tiredness (24.9%), and depression (22.2%) (Fig. 2B). No moderate-to-severe symptoms were reported in 40.5% of the total study population (Fig. 3). Although all components of ESAS were significantly correlated with the ESAS sum score, impaired well-being showed the strongest correlation (r = 0.824, p < 0.001) (Table S1). The sum score of 8 symptoms, not including impaired well-being, was strongly correlated with impaired well-being score (r = 0.751, p < 0.001). Patients with higher ESAS sum score had significantly higher diastolic blood pressure and lower proportion of current smokers, hemoglobin level, handgrip strength, and MMSE score (Table 1). There were no significant differences in age, sex, or body weight reduction during hospitalization, severity of HF (NYHA class, BNP level, and MAGGIC risk score), or length of hospital stay between the groups.
Study flow chart.
Distribution of ESAS sum score (A) and proportion of symptom burden severity (B) at hospital discharge. ESAS Edmonton symptom assessment system.
Total number of moderate-to-severe symptoms.
The mortality and combined event (HF readmission and mortality) rates during 6-month follow-up were 9.5% (n = 29) and 32.4% (n = 99), respectively. The Kaplan-Meier curve plotting ESAS sum score tertiles showed a significant association of higher ESAS sum score with shorter event-free survival period (p = 0.014) (Fig. 4). ESAS sum score was significantly associated with the combined event on multivariate Cox regression analysis after adjusting for MAGGIC risk score and BNP level (adjusted HR for each 5-point increase in ESAS sum score 1.10 [95% CI: 1.04–1.17]; p < 0.001) (Table 2). ESAS sum score remained a significant predictor of the combined event even after adjusting for possible confounding factors, including geriatric assessments (model 2) (adjusted HR for each 5-point increase in ESAS sum score 1.11 [95% CI: 1.05–1.18]; p < 0.001). The ESAS sum score also showed consistent associations with the combined event across various subgroups after adjusting for MAGGIC risk score and BNP level, and there were no significant interactions among subgroups (Figure S1).
Kaplan-Meier curve for the combined event of heart failure readmission and all-cause mortality. ESAS Edmonton symptom assessment system.
ESAS sum score was significantly associated with the combined event on multivariate logistic regression analysis after adjusting for MAGGIC risk score and BNP level (adjusted OR for each 5-point increase in ESAS sum score 1.15 [95% CI: 1.06–1.26]; p = 0.001) (Table S2). We compared the AUCs between the baseline model and the baseline model + ESAS sum score to evaluate the incremental prognostic predictive capability of ESAS sum score for the combined event (Table 3). Although the increase in the AUC with the addition of the ESAS sum score to the baseline model was not significant (0.717 vs. 0.745, respectively; p = 0.079), both continuous NRI (0.264 [95% CI: 0.028–0.499]; p = 0.028) and IDI (0.036 [95% CI: 0.013–0.060]; p = 0.002) were observed for the combined event. The baseline model + ESAS sum score showed a consistently better net benefit than the baseline model with threshold probabilities between 20% and 60% in decision curve analysis (Fig. 5).
Decision curve analysis. BNP B-type natriuretic peptide, ESAS Edmonton symptom assessment system, MAGGIC Meta-analysis global group in chronic heart failure.
Discussion
The prognostic value of symptom burden as a PRO measure was investigated using the ESAS score for the combined event of HF readmission and all-cause mortality within 6 months in older patients hospitalized with HF. The most commonly reported moderate-to-severe symptoms at discharge among patients ≥ 60 years old hospitalized with HF were impaired well-being, anxiety, drowsiness, tiredness, and depression, but not shortness of breath. More than half of the patients had at least one symptom classified as moderate-to-severe at hospital discharge. Higher ESAS sum score was associated with higher rates of all-cause mortality and/or HF readmission within 6 months after discharge. Symptom burden determined using the ESAS sum score provided valuable additional prognostic information to conventional risk factors. These observations suggested that ESAS is useful as a symptom burden screening tool for prediction of prognosis in older patients with HF.
Symptom burden is similar between advanced HF and advanced cancer22. In the present study, 59.5% of hospitalized patients with HF ≥ 60 years old (median age, 79 years) had at least one symptom classified as moderate-to-severe at the time of discharge from hospital. Consistent with previous reports6,7 these patients showed extensive symptom burden beyond the typical manifestations of HF, such as shortness of breath and fatigue. As patients with advanced HF in developed countries have an average age of more than 75 years at the time of hospitalization6,23 symptoms due to comorbidities are common in addition to the effects of HF itself in this population24,25. Therefore, non–disease-specific PROs may be useful for assessment of symptom burden in older patients with multiple comorbidities, where HF is not the only factor contributing to health status, which may facilitate comparisons among patients with different conditions and between different patient populations3,4. In this study, we used ESAS as a comprehensive non–disease-specific PRO measure that can be applied easily in clinical practice6,7,8. Our results revealed a significant association of higher ESAS sum score with increased risk of the combined event of HF readmission and all-cause mortality even after adjusting for not only conventional risk factors of HF (i.e., BNP and MAGGIC risk score) but also geriatric assessments (i.e., GNRI score, handgrip strength, and MMSE). In addition, ESAS sum score was shown to provide valuable additional prognostic information to other known risk factors. Our results suggested that comprehensive assessment of symptom burden using a simple PRO measure is important in daily clinical practice. In the present study, no significant differences were observed in weight loss during hospitalization, severity of HF (i.e., NYHA class, BNP, and MAGGIC risk score) at hospital discharge, or length of hospital stay according to symptom burden. Therefore, it is important to assess symptom burden using ESAS to identify patients at high risk of short event-free survival.
The results of the present study have implications for both clinical practice and the design of future clinical studies in older patients with HF. Even with no favorable effect on survival or hospitalization rate, the US Food and Drug Administration (FDA) has specified that drugs to treat HF could be approved based on a beneficial effect on symptoms or physical function4. Despite their application in clinical trials regarding HF, PROs have not been widely implemented in HF care, with few centers including such measures in routine assessment26. The Minnesota Living with Heart Failure Questionnaire (MLHFQ), KCCQ, and ESAS are the three most commonly cited questionnaires in studies of symptom burden in HF7. Both the MLHFQ and KCCQ assess HF-specific health status, including symptom burden, and have been shown to be useful for predicting functional and survival outcomes3,27. The MLHFQ and KCCQ have been qualified as Medical Device Development Tools by the US FDA Center for Devices and Radiological Health, with the KCCQ also qualifying as a Clinical Outcomes Assessment tool for use in cardiovascular trials by the FDA Center for Drug Evaluation and Research3,27. However, as these instruments require 5–10 min to complete and assess symptoms over the past 2–4 weeks, they have limitations as measures of symptom burden for older patients hospitalized for HF in a clinical setting. The ESAS, in contrast, can be administered rapidly (~ 1 min), easily, and repeatedly with “now” as the timeframe. In addition, ESAS has been validated and translated into over 20 different languages and is highly accessible through generous licensing agreements8. Therefore, ESAS is a simple and rapid screening tool suitable for clinical use to assess symptom burden in cardiology. Rapid assessment of symptom burden is important in patients with HF to allow timely implementation of suitable interventions, including multidisciplinary management programs8,28,29. Formal assessment can also highlight unmet needs and areas for potential improvement in the delivery of clinical care30. However, whether improvement of ESAS sum score is associated with improved prognosis of patients with HF remains unclear. Similar to the demonstrated benefits in oncology8,31 the incorporation of routine symptom burden measurements in clinical practice will be helpful for both clinicians and patients with HF to discuss individual experiences and to determine whether a given treatment program is fulfilling their expectations. Further detailed studies are required to address these issues and to guide future clinical decision making for the treatment of older patients with HF.
This study had several limitations in that this was a single-center study with a small sample size, and the small number of events may have increased the risk of false positives. Censored patients without follow-up data were excluded from the analysis, but complete follow-up data at 6 months were available in most cases. In addition, the study population consisted only of Asian patients limiting generalizability of the results, and the substantially longer hospital stay in Japan compared to Western countries may have affected the results with no other PRO measure of symptom burden used in the evaluation. The long-term prognostic capability of ESAS and its additive value on the predictive ability of well-validated risk scores other than the MAGGIC score are also unknown. Further studies are needed to validate use of the ESAS in other ethnic populations and against HF-specific PRO of symptom burden. Finally, symptom burden was not assessed after discharge from hospital in this study. The ESCAPE trial showed that more than half of patients with advanced HF were willing to trade survival time for improved symptom control shortly after discharge, but the majority prioritized survival once their symptoms had stabilized beyond 6 months32. In a meta-analysis of the TOPCAT and HF-ACTION trials, the most recent KCCQ score rather than the change was associated with subsequent death and cardiovascular hospitalization in patients with HF33. Further studies are required as there is still uncertainty regarding the benefits of one-time vs. serial PRO measurements and whether a serial approach is preferable in older patients with HF.
Conclusions
Symptom burden was significant in older patients with HF at hospital discharge, and greater symptom burden as determined by ESAS was associated with poorer prognosis. Comprehensive evaluation of symptom burden is important in older patients with HF, and is crucial not only for prediction of prognosis but also to ensure personalized therapeutic decision making.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AUC:
-
Area under the curve
- BNP:
-
B-type natriuretic peptide
- eGFR:
-
Estimated glomerular filtration rate
- ESAS:
-
Edmonton symptom assessment system
- FDA:
-
Food and drug administration
- GNRI:
-
Geriatric nutritional risk index
- HF:
-
Heart failure
- IDI:
-
Integrated discrimination improvement
- KCCQ:
-
Kansas city cardiomyopathy questionnaire
- MAGGIC:
-
Meta-analysis global group in chronic heart failure
- MLHFQ:
-
Minnesota living with heart failure questionnaire
- MMSE:
-
Mini-mental state examination
- NRI:
-
Net reclassification improvement
- NYHA:
-
New york heart association
- QoL:
-
Quality of life
- PRO:
-
Patient-reported outcome
References
Ambrosy, A. P. et al. The global health and economic burden of hospitalizations for heart failure: lessons learned from hospitalized heart failure registries. J. Am. Coll. Cardiol. 63, 1123–1133 (2014).
Savarese, G. et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc. Res. 118, 3272–3287 (2023).
Savarese, G. et al. Use of patient-reported outcomes in heart failure: from clinical trials to routine practice. Eur. J. Heart Fail. 25, 139–151 (2023).
Moons, P. et al. Placing patient-reported outcomes at the centre of cardiovascular clinical practice: implications for quality of care and management. Eur. Heart J. 44, 3405–3422 (2023).
Rumsfeld, J. S. et al. Cardiovascular health: the importance of measuring patient-reported health status: a scientific statement from the American heart association. Circulation 127, 2233–2249 (2013).
Alpert, C. M., Smith, M. A., Hummel, S. L. & Hummel, E. K. Symptom burden in heart failure: assessment, impact on outcomes, and management. Heart Fail. Rev. 22, 25–39 (2017).
Koshy, A. O. et al. Prioritizing symptom management in the treatment of chronic heart failure. ESC Heart Fail. 7, 2193–2207 (2020).
Hui, D. & Bruera, E. The Edmonton symptom assessment system 25 years later: past, present, and future developments. J. Pain Symptom Manage. 53, 630–643 (2017).
Richardson, L. A. & Jones, G. W. A review of the reliability and validity of the Edmonton symptom assessment system. Curr. Oncol. 16, 55 (2009).
Ezekowitz, J. A., Thai, V., Hodnefield, T. S., Sanderson, L. & Cujec, B. The correlation of standard heart failure assessment and palliative care questionnaires in a multidisciplinary heart failure clinic. J. Pain Symptom Manage. 42, 379–387 (2011).
Ando, Y. et al. CKD clinical practice guidebook. The essence of treatment for CKD patients. Clin. Exp. Nephrol. 13, 191–248 (2009).
Bouillanne, O. et al. Geriatric nutritional risk index: a new index for evaluating at-risk elderly medical patients. Am. J. Clin. Nutr. 82, 777–783 (2005).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr Res. 12, 189–198 (1975).
Yokomichi, N. et al. Validation of the Japanese version of the Edmonton symptom assessment System-Revised. J. Pain Symptom Manage. 50, 718–723 (2015).
Hallet, J. et al. Association of Patient-Reported outcomes with subsequent nonfatal Self-injury after a new Cancer diagnosis. JAMA Oncol. 8, e220203 (2022).
Wong, F. K. et al. Effects of a transitional palliative care model on patients with end-stage heart failure: a randomised controlled trial. Heart 102, 1100–1108 (2016).
Sawano, M. et al. Performance of the MAGGIC heart failure risk score and its modification with the addition of discharge natriuretic peptides. ESC Heart Fail. 5, 610–619 (2018).
van Buuren, S., Boshuizen, H. C. & Knook, D. L. Multiple imputation of missing blood pressure covariates in survival analysis. Stat. Med. 18, 681–694 (1999).
Barnard, J., Rubin, D., Miscellanea & 19 & Small-sample degrees of freedom with multiple imputation. Biometrika 86, 948–955 (1999).
Pencina, M. J., D’Agostino, R. B., Steyerberg, E. W. & Sr. & Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat. Med. 30, 11–21 (2011).
Kerr, K. F., Brown, M. D., Zhu, K. & Janes, H. Assessing the clinical impact of risk prediction models with decision curves: guidance for correct interpretation and appropriate use. J. Clin. Oncol. 34, 2534–2540 (2016).
Bekelman, D. B. et al. Symptom burden, depression, and spiritual well-being: a comparison of heart failure and advanced cancer patients. J. Gen. Intern. Med. 24, 592–598 (2009).
Jaarsma, T. et al. Palliative care in heart failure: a position statement from the palliative care workshop of the heart failure association of the European society of cardiology. Eur. J. Heart Fail. 11, 433–443 (2009).
Yang, M. et al. Impact of comorbidities on health status measured using the Kansas City cardiomyopathy questionnaire in patients with heart failure with reduced and preserved ejection fraction. Eur. J. Heart Fail. 25, 1606–1618 (2023).
Yang, M. et al. Dapagliflozin and quality of life measured using the EuroQol 5-dimension questionnaire in patients with heart failure with reduced and mildly reduced/preserved ejection fraction. Eur J. Heart Fail (2024).
Eliya, Y. et al. Temporal trends and factors associated with the inclusion of Patient-Reported outcomes in heart failure randomized controlled trials: A systematic review. J. Am. Heart Assoc. 10, e022353 (2021).
Kelkar, A. A. et al. Utility of Patient-Reported outcome instruments in heart failure. JACC Heart Fail. 4, 165–175 (2016).
Ghobadi, P. et al. Effects of a multidisciplinary management program on symptom burden and medication adherence in heart failure patients with comorbidities: A randomized controlled trial. BMC Nurs. 21, 346 (2022).
Saleh, Z. T. et al. Effect of a Home-Based mobile health app intervention on physical activity levels in patients with heart failure: A randomized controlled trial. J. Cardiovasc. Nurs. 38, 128–139 (2023).
Yang, M. et al. Knowledge about self-efficacy and outcomes in patients with heart failure and reduced ejection fraction. Eur. J. Heart Fail. 25, 1831–1839 (2023).
Basch, E. et al. Symptom monitoring with Patient-Reported outcomes during routine Cancer treatment: A randomized controlled trial. J. Clin. Oncol. 34, 557–565 (2016).
Stevenson, L. W. et al. Changing preferences for survival after hospitalization with advanced heart failure. J. Am. Coll. Cardiol. 52, 1702–1708 (2008).
Pokharel, Y. et al. Association of serial Kansas City cardiomyopathy questionnaire assessments with death and hospitalization in patients with heart failure with preserved and reduced ejection fraction: A secondary analysis of 2 randomized clinical trials. JAMA Cardiol. 2, 1315–1321 (2017).
Acknowledgements
They thank all the Nagoya University Hospital for their support.
Funding
This work was supported by the Japan Society for the Promotion of Science Grant-in-Aid (JSPS KAKENHI, Grant No. 24K20527).
Author information
Authors and Affiliations
Contributions
ST and TO contributed to the conception or design of the work. TI, AM, KK, DT, KH, NY, ST, YT, TI, MN, RM, TK, HH, and SK contributed to the acquisition, analysis, or interpretation of data for the work. ST drafted the manuscript. YN, TO, and TM critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Institutional Review Board of Nagoya University Hospital. All participants were informed that they were free to opt out of participation in the study at any time.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tanaka, S., Imaizumi, T., Morohashi, A. et al. Prognostic value of symptom burden as a simple patient-reported outcome measure in older patients with heart failure. Sci Rep 15, 22954 (2025). https://doi.org/10.1038/s41598-025-06615-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06615-4