Abstract
The triglyceride–glucose and systemic inflammation index have been linked to mortality outcomes. However, their combined impact on all-cause and cardiovascular mortality remains unclear. This study aimed to evaluate the individual and combined associations of the TyG index and SII with mortality using data from NHANES. Associations between TyG, SII, and mortality were assessed using multivariable Cox proportional hazards models and restricted cubic spline (RCS) regression models. Subgroup and sensitivity analyses were also conducted. Compared to individuals with low TyG and low SII, those with high TyG and high SII had a hazard ratio (HR) of 1.29 (95% CI, 1.16–1.44) for all-cause mortality, 1.54 (95% CI, 1.23–1.94) for cardiovascular mortality, 1.56 (95% CI, 1.20–2.04) for cardiac mortality, and 1.60 (95% CI, 0.83–3.08) for cerebrovascular mortality after full adjustment. The ROC curve analysis indicated that the combination of TyG and SII had an AUC of 0.599 for predicting all-cause mortality and 0.613 for cardiovascular mortality. Subgroup and sensitivity analyses yielded consistent findings. In conclusion, these results underscore the significant combined association of the TyG index and SII on all-cause and cardiovascular mortality, highlighting the importance of jointly assessing these indices to better predict mortality outcomes.
Similar content being viewed by others
Introduction
Cardiovascular disease (CVD) has become the leading cause of death and a major contributor to disability worldwide, encompassing conditions such as ischemic heart disease, stroke, and aortic diseases1. According to estimates from the World Health Organization, approximately 30% of global deaths are attributable to CVD, with around 17.9 million deaths annually2. In 2022, the American Heart Association introduced the “Life’s Essential 8”3, which studies have shown to be associated with reduced mortality risk when higher cardiovascular health levels are achieved4,5.
Insulin resistance (IR) is one of the most critical risk factors for CVD6. The triglyceride–glucose (TyG) index, first proposed in 2008, has emerged as a reliable surrogate marker for IR7,8,9 and has demonstrated predictive value for atherosclerotic changes, microvascular complications, coronary artery calcification, and the progression of cardiovascular and cerebrovascular diseases10,11. Previous research has established a link between the TyG index and both all-cause and CVD mortality, making it a potential tool for predicting CVD onset and progression12,13,14.
In recent years, the prediction of cardiovascular events has increasingly focused on indicators derived from routine blood tests, which are widely available and easily accessible in clinical settings15. Among these, composite inflammatory markers such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and C-reactive protein (CRP) have been validated for their value in predicting slow coronary flow16. However, these biomarkers typically reflect only one or two types of immune-inflammatory cells and may not comprehensively represent the body’s inflammatory status17. The systemic immune-inflammation index (SII), a novel composite inflammation index incorporating three cellular components, provides a more integrated reflection of inflammation and immune balance in the body18. It is easily measurable, stable, and has been widely used in the auxiliary diagnosis and prognosis of malignancies19, coronary artery disease20, myocardial infarction21, and more. Previous studies have confirmed that SII is a prognostic indicator of all-cause mortality and major cardiovascular adverse events in elderly patients aged 65 to 85 years with acute myocardial infarction22.
However, the combined impact of the TyG index and SII on all-cause and cardiovascular mortality has not been adequately studied. Therefore, this study utilized data from the National Health and Nutrition Examination Survey (NHANES) spanning 1999 to 2018 to assess both the individual and combined associations of the TyG index and SII with all-cause and cardiovascular mortality.
Methods
Study design and population
The data utilized in this study were obtained from the 1999–2018 NHANES, which employs a complex, multistage probability sampling method to select a representative sample of the U.S. population. NHANES is designed to assess the health and nutritional status of American adults and children. The research protocol for NHANES was approved by the Ethics Review Board of the National Center for Health Statistics (NCHS), with all participants providing written informed consent. More detailed information can be found at www.cdc.gov/nchs/nhanes/irba98.htm.
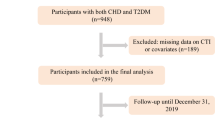
This study analyzed data from ten NHANES cycles conducted between 1999 and 2018, sourced from the National Center for Health Statistics at the Centers for Disease Control and Prevention. The inclusion criteria for the analysis were participants aged 20 years or older who had available TyG and SII data, along with relevant mortality follow-up information. Exclusion criteria included missing baseline TyG or SII data, lack of follow-up data, and absence of essential baseline clinical measurements. After applying these criteria, a total of 20,384 participants were included in the final analysis (Fig. 1).
Flowchart of the selection strategy. TyG triglyceride–glucose index, SII Systemic Inflammation Index, DM diabetes, CVD cardiovascular disease.
Definitions of TyG, SII, and mortality
The TyG index was calculated using the formula ln(fasting triglycerides [mg/dL] × fasting glucose [mg/dL]/2), as established in previous research23. The systemic inflammation index (SII) was defined by dividing the product of peripheral platelet count and neutrophil count by the lymphocyte count24.
For the mortality analysis within NHANES, we determined the vital status of participants using publicly available mortality linkage files, updated through December 31, 2019. These files were linked to the National Death Index (NDI) by the NCHS through a probabilistic matching algorithm. Cause-specific mortality was classified according to the International Classification of Diseases, Tenth Revision (ICD-10). The primary outcomes analyzed in this study were all-cause mortality and cardiovascular mortality, with cardiovascular mortality classified under ICD-10 codes I00-I09, I11, I13, I20-I51, and I60-I69. Additionally, subtypes of cardiovascular mortality, including cardiac mortality (I00-I09, I11, I13, I20-I51) and cerebrovascular mortality (I60-I69), were also examined25.
Ascertainment of covariates
Various demographic and health-related factors were collected through household interviews within the NHANES database, including age, gender, race/ethnicity, education level, household income, marital status, smoking status, alcohol consumption, use of diabetes and hypertension medications, disease status, dyslipidemia, CVD, and hypertension. The diagnoses of hypertension and dyslipidemia were confirmed through physical examinations and laboratory results26,27,28,29. Body mass index (BMI) was calculated by dividing weight in kilograms by the square of height in meters. Race/ethnicity was categorized into Mexican American, non-Hispanic Black, non-Hispanic White, other Hispanic, or other. Education levels were classified as above high school, high school or equivalent, and below high school. Household income and poverty index were grouped into low (0–1.0), medium (1.0–3.0), or high (> 3.0) categories. Smoking status was recorded as never smokers, former smokers, or current smokers. Alcohol consumption was classified into heavy drinkers, moderate drinkers, light drinkers, non-drinkers, or former drinkers.
Statistical analysis
All statistical analyses were conducted following CDC guidelines, taking into account the complex, multistage stratified probability sampling design of NHANES. Sample weights, clustering, and stratification were incorporated into the analysis. The NHANES survey design was defined using the svydesign function of survey package in R, specifying primary sampling units (PSUs; variable: SDMVPSU) to adjust for clustering effects and account for correlations among individuals within the same PSU, stratum variables (SDMVSTRA) to reflect the stratified sampling design, and sample weights (WTMEC2YR, 2-year examination weights for 1999–2018) to correct for differential sampling probabilities, ensuring nationally representative results. Continuous variables were presented as weighted means with standard errors (SEs) to reflect the variability in the estimates while accounting for the complex survey design, and categorical variables were expressed as counts with weighted proportions30.Participants’ baseline characteristics were described based on the joint assessment of the TyG index and SII. TyG and SII values were dichotomized at the median, resulting in four groups: low TyG & low SII, high TyG & low SII, low TyG & high SII, and high TyG & high SII. Weighted linear regression was used for continuous variable comparisons, and design-adjusted chi-square tests, based on the Rao-Scott methodology to account for the complex survey design30,31, were applied for categorical variables.
In the separate analyses of TyG and SII for mortality prognosis, we categorized TyG and SII into tertiles. Survival probabilities were estimated using the Kaplan–Meier method, with participants grouped according to baseline categories (TyG Q1, SII Q1, or low TyG & low SII). Differences between survival curves were assessed using the log-rank test. Cox proportional hazards regression models were employed to first explore the individual associations between TyG and SII with mortality, followed by an assessment of their combined impact on mortality. To test for potential multiplicative effects, an interaction term between TyG and SII (TyG_binary * SII_binary, based on their dichotomized values at medians) was included in Model 3 to evaluate whether the effect of TyG on mortality varies by SII level, or vice versa. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. Four models were estimated: Model 0 was unadjusted, Model 1 adjusted for age and gender, Model 2 further adjusted for race, household income, marital status, and education level, and Model 3 additionally adjusted for alcohol consumption, smoking, BMI, dyslipidemia, hypertension, and CVD history. The proportional hazards (PH) assumption was tested for all variables in each model using Schoenfeld residuals tests, adjusted for the NHANES complex survey design in R. The PH assumption was assessed by testing the correlation between scaled Schoenfeld residuals and time, with p > 0.05 indicating no violation of the assumption. To visualize the linear or non-linear relationship between TyG or SII levels and mortality, restricted cubic spline analysis with four knots was conducted based on multivariable-adjusted Cox regression models. The four knots for TyG and SII were placed at the 5th, 35th, 65th, and 95th percentiles of its distribution.The discriminative ability of TyG, SII, and their combined indices in predicting all-cause and cardiovascular mortality was evaluated using receiver operating characteristic (ROC) curve analysis, with the area under the curve (AUC) calculated. Combined associations on mortality of subgroup analyses were stratified by age (≤ 60 and > 60), gender, education level, education level, BMI, DM, hypertension, and hyperlipidemia based on Model 3. To strengthen the robustness of the findings, four sensitivity analyses were performed: (1) excluding individuals with a history of CVD, (2) excluding participants under 45 years of age, (3) excluding those who died within the first two years of follow-up to mitigate potential reverse causality, and (4) incorporating additional adjustments for the use of antihypertensive and antidiabetic medications in the primary analysis.
Results
Baseline characteristics of the study population
After screening data from ten NHANES cycles, a total of 20,384 individuals aged over 20 years met the inclusion criteria and were included in the analysis, as outlined in Fig. 1. The study population had a mean age of 47.51 years, with 49.9% of participants being male. Among the participants, 26.5% (5,407 individuals) were classified into the low TyG & low SII group, 23.5% (4,786 individuals) into the high TyG & low SII group, 23.5% (4,791 individuals) into the low TyG & high SII group, and 26.5% (5,400 individuals) into the high TyG & high SII group. Participants in the high TyG & high SII group were characterized by a higher likelihood of being older, non-Hispanic White, having an education level below high school, belonging to a middle-income household, having a higher BMI, being current smokers, and having higher prevalences of hypertension and CVD (Table 1 and Supplementary Tables S1A-S1B).
TyG and SII and mortality
During a median follow-up period of 9.3 years (interquartile range: 5.3 to 13.9 years), a total of 3,328 participants (16.3%) died, including 1,071 deaths due to CVD, of which 880 were cardiac deaths and 191 were cerebrovascular deaths. Additionally, 2,257 participants died from non-CVD causes. Kaplan–Meier survival curves for the different groups are illustrated in Supplementary Figures S1A and S1C.
In the analysis of the association between the TyG index and mortality, Model 3 revealed that the HR for all-cause mortality in the Q3 was 1.26 (95% CI, 1.13–1.04). For CVD mortality, the HR for Q3 was 1.53 (95% CI, 1.24–1.88). In the case of cardiac mortality, the HR for Q3 was 1.42 (95% CI, 1.11–1.82), while for cerebrovascular mortality, the HR for Q2 was 2.71 (95% CI, 1.56–4.73) (Fig. 2 and Supplementary Table S2A).
Forest plot showing the association of TyG and SII with mortality. This forest plot presents the HRs with 95% CIs for mortality according to TyG and SII levels, using Q1 as the reference group. HR hazard ratio, CI confdence interval, TyG triglyceride–glucose index, SII Systemic Inflammation Index. (A) Association of TyG with the risk of mortality; (B) association of SII with the risk of mortality. The event column represents the number of individuals with positive outcomes.
Similarly, in the analysis of SII and mortality, Model 3 demonstrated that the HR for all-cause mortality in Q3 was 1.22 (95% CI, 1.11–1.34). For CVD mortality, the HR was 1.43 (95% CI, 1.20–1.71), and for cardiac mortality, the HR was 1.52 (95% CI, 1.24–1.87). However, the HR for cerebrovascular mortality in Q3 was 1.11 (95% CI, 0.72–1.69) (Fig. 2 and Supplementary Table S2B).
In the combined association of TyG and SII with mortality, compared to the low TyG & low SII group, Model 3 showed the following results: For all-cause mortality, the HRs were 1.29 (95% CI, 1.16–1.44) for the high TyG & high SII group, 1.21 (95% CI, 1.07–1.38) for the low TyG & high SII group, and 1.15 (95% CI, 1.00–1.33) for the high TyG & low SII group. For CVD mortality, the HRs were 1.54 (95% CI, 1.23–1.94), 1.36 (95% CI, 1.08–1.71), and 1.24 (95% CI, 0.95–1.62), respectively. For cardiac mortality, the HRs were 1.56 (95% CI, 1.20–2.04), 1.43 (95% CI, 1.10–1.87), and 1.20 (95% CI, 0.87–1.64). For cerebrovascular mortality, the HRs were 1.60 (95% CI, 0.83–3.08), 0.96 (95% CI, 0.51–1.78), and 1.57 (95% CI, 0.85–2.91), and an increasing trend across different combined groups was observed in Model 3 for mortality outcomes except cerebrovascular mortality (p-trend < 0.001 to < 0.0001) (Fig. 3 and Supplementary Table S2C). To examine potential multiplicative effects between TyG and SII, we included an interaction term (TyG_binary * SII_binary) in the fully adjusted Cox regression model (Model 3). The p-value for the interaction term was 0.4185, indicating no significant multiplicative interaction between TyG and SII in predicting all-cause mortality.
Forest plot showing the association of joint effect with mortality. This forest plot presents the HRs with 95% CIs for mortality,according to the Combined associations of TyG and SII, using the low TyG & low SII group as a reference. HR hazard ratio, CI confdence interval, TyG triglyceride–glucose index, SII Systemic Inflammation Index. The event column represents the number of individuals with positive outcomes.
The RCS analysis revealed a nonlinear relationship between the TyG index and all-cause mortality (p for overall trend < 0.001; p for nonlinearity = 0.001), as well as between SII and all-cause mortality (p for overall trend < 0.001; p for nonlinearity = 0.0003). A nonlinear relationship was also observed between the TyG index and CVD mortality (p for overall trend < 0.001; p for nonlinearity = 0.034), while SII showed a linear association with CVD mortality (p for overall trend < 0.001; p for nonlinearity = 0.4645) (Fig. 4).
Association of TyG and SII with All-Cause and CVD Mortality using RCS. Restricted cubic spline analysis has four knots. In the RCS analysis, adjustments were made for age, sex, ethnicity, marital status, education level, family income, BMI, smoking status, current alcohol consumption, dyslipidemia, hypertension, and CVD. TyG triglyceride–glucose index, SII Systemic Inflammation Index, BMI body mass index, CVD cardiovascular disease. (A) Association of TyG with the risk of mortality; (B) association of SII with the risk of mortality.
Finally, the combined predictive value of TyG and SII was evaluated, with ROC curve analysis indicating that the AUC for predicting all-cause mortality was 0.599, and for CVD mortality, it was 0.613. These findings suggest that the combined index has a modest predictive value for CVD mortality (Fig. 5 and Supplementary Figures S2A and S2B).
Predictive performance of the combined TyG and SII for mortality. The receiver operating characteristic (ROC) curve evaluating the discriminative capabilities by calculating the AUC. AUC area under curve, TyG triglyceride–glucose index, SII Systemic Inflammation Index, CVD cardiovascular disease. (A) Combined association with the All-Cause mortality; (B) Combined association with the CVD mortality.
Subgroup analyses and sensitivity analyses
The results of the Combined association’s subgroup analyses for mortality are detailed in Table 2 Supplementary Tables S3A-S3C. The subgroup analysis for all-cause mortality revealed that participants in the high TyG & high SII group had significantly higher mortality risks across various subgroups when compared to the low TyG & low SII group. Specifically, this increased risk was observed in participants aged 20–60 years (HR, 1.81; 95% CI, 1.41–2.33) and those aged 60 years and above (HR, 1.27; 95% CI, 1.12–1.43). Both males (HR, 1.69; 95% CI, 1.42–2.01) and females (HR, 2.26; 95% CI, 1.82–2.82) exhibited higher mortality risks. Higher mortality risks were also noted across BMI categories: BMI ≥ 28 (HR 1.55; 95% CI 1.27–1.89), BMI 24–27.9 (HR 1.80; 95% CI 1.42–2.28), and BMI < 23.9 (HR 2.59; 95% CI 2.02–3.31), as did individuals with varying levels of education, including those with education above high school (HR, 1.95; 95% CI, 1.58–2.40), below high school (HR, 1.67; 95% CI, 1.31–2.13), and those with high school or equivalent education (HR, 2.06; 95% CI, 1.61–2.63). The association was also significant among participants without diabetes (HR, 1.77; 95% CI, 1.53–2.04), with hyperlipidemia (HR, 1.58; 95% CI, 1.35–1.84), without hyperlipidemia (HR, 2.88; 95% CI, 2.15–3.86), with hypertension (HR, 1.45; 95% CI, 1.25–1.68), and without hypertension (HR, 1.76; 95% CI, 1.43–2.15). However, no significant association was found among participants with diabetes (HR, 1.15; 95% CI, 0.87–1.51) (Table 2). The results of the Combined association subgroup analyses for CVD mortality (including heart and cerebrovascular deaths) are thoroughly detailed in Supplementary Tables S3A-S3C.
The Supplementary Tables S4A-S4D, S5A-S5D, S6A-S6D, and S7A-S7B, along with Supplementary Figures S3, S4, and S5, display baseline data, outcomes from various models, and the results of Kaplan–Meier sensitivity tests. After excluding participants with CVD, consistent results were observed. Compared to the low TyG & low SII group, Model 3 showed the following HRs for the high TyG & high SII group: 1.31 (95% CI, 1.15–1.51) for all-cause mortality, 1.47 (95% CI, 1.07–2.01) for CVD mortality, 1.45 (95% CI, 1.02–2.06) for cardiac mortality, and 1.81 (95% CI, 0.78–4.21) for cerebrovascular mortality (Supplementary Tables S4A-S4D). Similarly, after excluding participants who died within two years of follow-up, the high TyG & high SII group showed HRs of 1.31 (95% CI, 1.17–1.46) for all-cause mortality, 1.52 (95% CI, 1.21–1.92) for cardiovascular mortality, 1.58 (95% CI, 1.20–2.09) for cardiac mortality, and 1.39 (95% CI, 0.71–2.73) for cerebrovascular mortality (Supplementary Tables S5A-S5D). Excluding participants under 45 years of age yielded HRs of 1.32 (95% CI, 1.18–1.47) for all-cause mortality, 1.52 (95% CI, 1.22–1.90) for cardiovascular mortality, 1.51 (95% CI, 1.16–1.96) for cardiac mortality, and 1.83 (95% CI, 1.04–3.22) for cerebrovascular mortality in the high TyG & high SII group (Supplementary Tables S6A-S6D). Additionally, adjusting for medication use resulted in HRs of 1.25 (95% CI, 1.12–1.39) for all-cause mortality, 1.46 (95% CI, 1.16–1.84) for cardiovascular mortality, 1.49 (95% CI, 1.14–1.94) for cardiac mortality, and 1.53 (95% CI, 0.78–3.03) for cerebrovascular mortality in the high TyG & high SII group (Supplementary Tables S7A-S7B). Statistical differences in survival probabilities among different groups were observed across several sensitivity analyses for mortality (Supplementary Figures S3, S4, and S5).
Discussion
This study, utilizing data from NHANES, investigated the individual and combined associations of the TyG index and the SII with all-cause and cardiovascular mortality. The findings revealed that the combined assessment of TyG and SII significantly enhanced the predictive ability for both all-cause and cardiovascular mortality. This underscores the critical role of insulin resistance and systemic inflammation in determining patient outcomes and highlights the synergistic effect of these two biomarkers.
Specifically, participants in the high TyG & high SII group exhibited a markedly increased risk of mortality, particularly from cardiovascular causes, compared to those in the low TyG & low SII group. Subgroup and sensitivity analyses further confirmed the consistency of these effects across various populations, reinforcing the robustness and reliability of the study’s results.
The findings of this study align with previous research that has demonstrated an association between the TyG index and both all-cause and CVD mortality. Liu et al.32 and Chen et al.33 found a nonlinear relationship between the TyG index and all-cause and CVD mortality. Liang et al.34 observed that higher TyG levels were associated with an increased likelihood of CVD events in older adults in the United States. Sun et al.35 reported that elevated TyG levels were linked to an increased risk of all-cause mortality among middle-aged men in the U.S. However, Chen et al.33 noted that, with increasing TyG levels, both all-cause and CVD mortality risks rise in women, whereas men only show an increased risk for all-cause mortality. In Iranian men, higher TyG levels were associated with increased risks for both all-cause and CVD mortality36. There is no consensus on the relationship between TyG index and mortality risk across different genders, which may be influenced by factors such as environmental conditions, biological aging, and heterogeneity within populations. In our study, subgroup analyses revealed that although the combination of high TyG and high SII increases mortality risk for both genders, the effect is more pronounced in women. Postmenopausal women experience a reduction in estrogen, which diminishes cardiovascular protection, leading to increased IR and heightened sensitivity to changes in metabolic and inflammatory states, thereby elevating CVD risk37.
Regarding the SII, Yang et al.20 investigated the predictive value of the SII for major cardiovascular events in patients undergoing coronary heart disease interventions. They found that when the optimal SII cutoff was set at 694.3 × 10^9/L, higher SII levels (≥ 694.3) were independently associated with an increased risk of cardiac death (HR = 2.02; 95% CI: 1.43–2.86) and were also linked to major adverse cardiovascular events and overall major events. SII significantly improved risk stratification for cardiac death in coronary heart disease patients and outperformed traditional risk factors in predicting major cardiovascular events following coronary intervention. Taha Sert et al.38 studied 237 out-of-hospital cardiac arrest (OHCA) patients, with an in-hospital mortality rate of 82.7%. They observed that survivors had significantly lower SII, NLR, and PLR values compared to the non-survivor group. SII was identified as an independent predictor of survival to hospital discharge in OHCA patients. ROC curve analysis further demonstrated that the AUC for SII was higher than that for NLR or PLR, indicating that SII has a superior predictive ability for post-discharge survival in OHCA patients.
The exact mechanisms linking the TyG index to mortality are still being explored. However, several hypotheses have been proposed. Firstly, the TyG index is closely associated with a range of adverse health conditions such as obesity, diabetes, CVD, elevated blood pressure, reduced eGFR, and low HDL-C levels39,40,41. A higher TyG index may thus indicate negative effects on cardiometabolic health. Secondly, endothelial dysfunction might contribute to the relationship between the TyG index and mortality. Unlike other indicators of IR, the TyG index specifically measures IR in muscle tissue and is considered a more accurate marker for peripheral IR. As such, it may be linked to endothelial dysfunction, oxidative stress, and inflammatory responses42,43,44. IR is recognized for initiating the production of glycosylation products and free radicals, which in turn decrease the availability of nitric oxide (NO). This reduction in NO bioavailability adversely affects endothelium-dependent vasodilation45. Chronic inflammation can damage the vascular endothelium, leading to leakage of blood components into surrounding tissues and further vascular damage44. Furthermore, IR activates the mitochondrial electron transport chain, leading to increased oxidative stress and subsequent endothelial damage33. Lastly, insulin might contribute to lipohyalinosis by increasing sympathetic activity, which can result in diffuse hypoperfusion or obstruct small arterioles44,46. Additionally, IR may facilitate the development of atherosclerosis and progression of arterial plaques46.
The exact mechanisms by which the SII increases mortality risk are also not fully understood, but several explanations may be considered. First, elevated SII is associated with increased levels of cytokines, including interleukin-6, interleukin-8, and interleukin-10, which are linked to chronic systemic inflammation47. Increased mortality in individuals with higher SII levels may be due to chronic inflammation, which produces reactive nitrogen and oxygen species, leading to genomic instability and cellular senescence, thereby raising the risk of cardiovascular diseases and mortality48. Additionally, high SII levels could be attributed to elevated neutrophil counts, high platelet counts, and/or low lymphocyte counts49. Different inflammatory components may play varying roles in mortality risk50. Activated neutrophils release extracellular traps, and neutrophil elastase, a major component of these traps, has been linked to increased all-cause and cardiovascular mortality risk in chronic hemodialysis patients51. Thus, we hypothesize that high SII may exacerbate cardiovascular adverse events through enhanced inflammation and oxidative stress, which can burden the heart and damage tissues. However, the exact role of TyG index and SII in mortality still requires further investigation.
Moreover, ROC curve analysis demonstrated that the combined assessment of TyG and SII provided better discriminative ability for predicting all-cause and cardiovascular mortality compared to individual indicators. Although the AUC values indicated only moderate predictive capability, the combined assessment of TyG and SII, when integrated with other risk factors, may still offer significant clinical value. This suggests that incorporating both metabolic and inflammatory markers in risk assessment and early intervention could provide a more comprehensive evaluation of mortality risk.
This study uses the TyG index to estimate insulin resistance, though its correlation with HOMA-IR is weak (r≈0.322), indicating it may not directly reflect insulin resistance. However, the research focuses on how TyG combined with SII predicts mortality risk, not their direct relationship. TyG remains useful for its simplicity and broad use in large-scale studies52,53. While NHANES data allow calculating HOMA or QUICKI, TyG was chosen to match the study’s goals. Future studies could test combining HOMA, QUICKI, SII, or C-reactive protein to expand these findings.
Despite these important findings, several limitations of this study should be acknowledged. Firstly, although we used a large, nationally representative sample, the observational nature of NHANES data precludes causal inferences. Secondly, despite adjusting for multiple confounders in our models, unmeasured confounding factors may still influence the results. Finally, the findings are primarily applicable to the U.S. population and may not be directly generalizable to other regions or ethnic groups.
Conclusion
Overall, the findings of this study underscore the importance of the combined assessment of the TyG index and SII in clinical practice. This approach not only enhances the accuracy of predicting all-cause and cardiovascular mortality but also provides a basis for developing personalized prevention and intervention strategies. Future research should focus on optimizing the predictive capability of these indices across different populations and exploring the underlying mechanisms to better understand the roles of metabolism and inflammation in mortality risk.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CVD:
-
Cardiovascular diseases
- IR:
-
Insulin resistance
- TyG:
-
Triglyceride–glucose
- NLR:
-
Neutrophil-to-lymphocyte ratio
- PLR:
-
Platelet-to-lymphocyte ratio
- CRP:
-
C-reactive protein
- SII:
-
Systemic immune-inflammation index
- NHANES:
-
National Health and Nutrition Examination Survey
- NCHS:
-
National Center for Health Statistics
- ICD-10:
-
Tenth revision
- BMI:
-
Body mass index
- HR:
-
Hazard ratio
- CI:
-
Confidence intervals
- ROC:
-
Operating characteristic curves
- AUC:
-
Area under the curve
References
Zhao, D., Liu, J., Wang, M., Zhang, X. & Zhou, M. Epidemiology of cardiovascular disease in China: Current features and implications. Nat Rev Cardiol. 16(4), 203–212. https://doi.org/10.1038/s41569-018-0119-4 (2019).
Brookes, A. Global Epidemiology of Cardiovascular Disease.
Lloyd-Jones, D. M. et al. Life’s essential 8: Updating and enhancing the American Heart Association’s construct of cardiovascular health: A presidential advisory from the American Heart Association. Circulation 146(5), e18–e43. https://doi.org/10.1161/cir.0000000000001078 (2022).
Sun, J. et al. Association of the American Heart Association’s new “Life’s Essential 8” with all-cause and cardiovascular disease-specific mortality: prospective cohort study. BMC Med. 21(1), 116. https://doi.org/10.1186/s12916-023-02824-8 (2023).
Yi, J., Wang, L., Guo, X. & Ren, X. Association of Life’s Essential 8 with all-cause and cardiovascular mortality among US adults: A prospective cohort study from the NHANES 2005–2014. Nutr. Metab. Cardiovasc. Dis. 33(6), 1134–1143. https://doi.org/10.1016/j.numecd.2023.01.021 (2023).
Hill, M. A. et al. Insulin resistance, cardiovascular stiffening and cardiovascular disease. Metabolism 119, 154766. https://doi.org/10.1016/j.metabol.2021.154766 (2021).
Simental-Mendía, L. E., Rodríguez-Morán, M. & Guerrero-Romero, F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat Disord. 6(4), 299–304. https://doi.org/10.1089/met.2008.0034 (2008).
Huang, Y. et al. Inflammatory markers link triglyceride-glucose index and obesity indicators with adverse cardiovascular events in patients with hypertension: insights from three cohorts. Cardiovasc. Diabetol. 24(1), 11. https://doi.org/10.1186/s12933-024-02571-x (2025).
Mao, Q., Zhu, X., Zhang, X. & Kong, Y. Triglyceride-glucose Index and Its combination with obesity indicators mediating the association between 2-hydroxyfluorene and the prevalence of cardiovascular disease: Evidence from the NHANES (2005–2018). Ecotoxicol. Environ. Saf. 287, 117283. https://doi.org/10.1016/j.ecoenv.2024.117283 (2024).
Mirr, M., Skrypnik, D., Bogdański, P. & Owecki, M. Newly proposed insulin resistance indexes called TyG-NC and TyG-NHtR show efficacy in diagnosing the metabolic syndrome. J. Endocrinol. Investig. 44(12), 2831–2843. https://doi.org/10.1007/s40618-021-01608-2 (2021).
Park, K. et al. Elevated TyG index predicts progression of coronary artery calcification. Diabetes Care 42(8), 1569–1573. https://doi.org/10.2337/dc18-1920 (2019).
Tao, L. C., Xu, J. N., Wang, T. T., Hua, F. & Li, J. J. Triglyceride-glucose index as a marker in cardiovascular diseases: Landscape and limitations. Cardiovasc. Diabetol. 21(1), 68. https://doi.org/10.1186/s12933-022-01511-x (2022).
Liu, Q. et al. Triglyceride-glucose index associated with the risk of cardiovascular disease: The Kailuan study. Endocrine 75(2), 392–399. https://doi.org/10.1007/s12020-021-02862-3 (2022).
Li, S. et al. The role of the triglyceride (triacylglycerol) glucose index in the development of cardiovascular events: A retrospective cohort analysis. Sci. Rep. 9(1), 7320. https://doi.org/10.1038/s41598-019-43776-5 (2019).
Kim, Y., Sohn, J. H., Kim, C., Park, S. Y. & Lee, S. H. The clinical value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio for predicting hematoma expansion and poor outcomes in patients with acute intracerebral hemorrhage. J. Clin. Med. https://doi.org/10.3390/jcm12083004 (2023).
Xiong, Y., Zhong, Q., Zhang, Y., Qin, F. & Yuan, J. The association between the platelet to white blood cell ratio and chronic kidney disease in an aging population: a four-year follow-up study. J. Clin. Med. https://doi.org/10.3390/jcm12227073 (2023).
Wang, Z. C. et al. Systemic immune-inflammation index independently predicts poor survival of older adults with hip fracture: A prospective cohort study. BMC Geriatr. 21(1), 155. https://doi.org/10.1186/s12877-021-02102-3 (2021).
Ma, M., Yu, N. & Wu, B. High systemic immune-inflammation index represents an unfavorable prognosis of malignant pleural mesothelioma. Cancer Manag. Res. 11, 3973–3979. https://doi.org/10.2147/cmar.S201269 (2019).
Zhang, Y., Sun, Y. & Zhang, Q. Prognostic value of the systemic immune-inflammation index in patients with breast cancer: A meta-analysis. Cancer Cell Int. 20, 224. https://doi.org/10.1186/s12935-020-01308-6 (2020).
Yang, Y. L. et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur. J. Clin. Investig. 50(5), e13230. https://doi.org/10.1111/eci.13230 (2020).
Wei, X., Zhang, Z., Wei, J. & Luo, C. Association of systemic immune inflammation index and system inflammation response index with clinical risk of acute myocardial infarction. Front. Cardiovasc. Med. 10, 1248655. https://doi.org/10.3389/fcvm.2023.1248655 (2023).
Huang, J. et al. Systemic immune-inflammatory index predicts clinical outcomes for elderly patients with acute myocardial infarction receiving percutaneous coronary intervention. Med. Sci. Monit. 25, 9690–9701. https://doi.org/10.12659/msm.919802 (2019).
Alizargar, J., Bai, C. H., Hsieh, N. C. & Wu, S. V. Use of the triglyceride-glucose index (TyG) in cardiovascular disease patients. Cardiovasc. Diabetol. 19(1), 8. https://doi.org/10.1186/s12933-019-0982-2 (2020).
Zeng, Q. Y. et al. Systemic immune-inflammation index and all-cause and cause-specific mortality in sarcopenia: A study from National Health and Nutrition Examination Survey 1999–2018. Front. Immunol. 15, 1376544. https://doi.org/10.3389/fimmu.2024.1376544 (2024).
World Health O. International Statistical Classification of Diseases and related health problems: Alphabetical index: World Health Organization (2004).
Williams, B. et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 39(33), 3021–3104. https://doi.org/10.1093/eurheartj/ehy339 (2018).
ElSayed, N. A. et al. 2. Classification and diagnosis of diabetes: standards of care in diabetes-2023. Diabetes Care 46(Suppl 1), S19–S40. https://doi.org/10.2337/dc23-S002 (2023).
Zhou, Z. et al. Impact of blood lipids on 10-year cardiovascular risk in individuals without dyslipidemia and with low risk factor burden. Mayo Clin. Proc. 97(10), 1883–1893. https://doi.org/10.1016/j.mayocp.2022.03.025 (2022).
Delanaye, P. et al. CKD: A call for an age-adapted definition. J. Am. Soc. Nephrol. 30(10), 1785–1805. https://doi.org/10.1681/asn.2019030238 (2019).
Wang, Y., Wang, H., Cheng, B. & Xia, J. Associations between triglyceride glucose index-related obesity indices and anxiety: Insights from the National Health and Nutrition Examination Survey 2007–2012. J. Affect. Disord. 382, 443–452. https://doi.org/10.1016/j.jad.2025.04.134 (2025).
Rao, J. N. K. & Scott, A. J. On simple adjustments to chi-square tests with sample survey data. Ann. Stat. 15(1), 385–397 (1987).
Liu, X. C., He, G. D., Lo, K., Huang, Y. Q. & Feng, Y. Q. The triglyceride–glucose index, an insulin resistance marker, was non-linear associated with all-cause and cardiovascular mortality in the general population. Front. Cardiovasc. Med. 7, 628109. https://doi.org/10.3389/fcvm.2020.628109 (2020).
Chen, J., Wu, K., Lin, Y., Huang, M. & Xie, S. Association of triglyceride glucose index with all-cause and cardiovascular mortality in the general population. Cardiovasc. Diabetol. 22(1), 320. https://doi.org/10.1186/s12933-023-02054-5 (2023).
Liang, D., Liu, C. & Wang, Y. The association between triglyceride-glucose index and the likelihood of cardiovascular disease in the US population of older adults aged ≥ 60 years: a population-based study. Cardiovasc. Diabetol. 23(1), 151. https://doi.org/10.1186/s12933-024-02248-5 (2024).
Sun, M., Guo, H., Wang, Y. & Ma, D. Association of triglyceride glucose index with all-cause and cause-specific mortality among middle age and elderly US population. BMC Geriatr. 22(1), 461. https://doi.org/10.1186/s12877-022-03155-8 (2022).
Alavi Tabatabaei, G. et al. Association of the triglyceride glucose index with all-cause and cardiovascular mortality in a general population of Iranian adults. Cardiovasc. Diabetol. 23(1), 66. https://doi.org/10.1186/s12933-024-02148-8 (2024).
Ma, M., Jiang, T., Wen, Z., Zhang, D. & Xiu, L. Gender differences in relation to body composition, insulin resistance, and islet beta cell function in newly diagnosed diabetic or pre-diabetic patients. Diabetes Metab. Syndr. Obes. 16, 723–732. https://doi.org/10.2147/dmso.S397528 (2023).
Taha Sert, E., Kokulu, K., Mutlu, H., Gül, M. & Uslu, Y. Performance of the systemic immune-inflammation index in predicting survival to discharge in out-of-hospital cardiac arrest. Resusc. Plus 14, 100382. https://doi.org/10.1016/j.resplu.2023.100382 (2023).
Zhang, M. et al. Cumulative increased risk of incident type 2 diabetes mellitus with increasing triglyceride glucose index in normal-weight people: The Rural Chinese Cohort Study. Cardiovasc. Diabetol. 16(1), 30. https://doi.org/10.1186/s12933-017-0514-x (2017).
Zhu, B. et al. A high triglyceride glucose index is more closely associated with hypertension than lipid or glycemic parameters in elderly individuals: A cross-sectional survey from the Reaction Study. Cardiovasc. Diabetol. 19(1), 112. https://doi.org/10.1186/s12933-020-01077-6 (2020).
Cho, Y. R. et al. Association between insulin resistance, hyperglycemia, and coronary artery disease according to the presence of diabetes. Sci. Rep. 9(1), 6129. https://doi.org/10.1038/s41598-019-42700-1 (2019).
Petersen, M. C. & Shulman, G. I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 98(4), 2133–2223. https://doi.org/10.1152/physrev.00063.2017 (2018).
Ormazabal, V. et al. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 17(1), 122. https://doi.org/10.1186/s12933-018-0762-4 (2018).
Han, T. et al. Changes in triglycerides and high-density lipoprotein cholesterol may precede peripheral insulin resistance, with 2-h insulin partially mediating this unidirectional relationship: A prospective cohort study. Cardiovasc. Diabetol. 15(1), 154. https://doi.org/10.1186/s12933-016-0469-3 (2016).
Molina, M. N., Ferder, L. & Manucha, W. Emerging role of nitric oxide and heat shock proteins in insulin resistance. Curr. Hypertens. Rep. 18(1), 1. https://doi.org/10.1007/s11906-015-0615-4 (2016).
Bornfeldt, K. E. & Tabas, I. Insulin resistance, hyperglycemia, and atherosclerosis. Cell Metab. 14(5), 575–585. https://doi.org/10.1016/j.cmet.2011.07.015 (2011).
Çavuşoğlu Türker, B. et al. Systemic immune-inflammation and systemic inflammation response indices are predictive markers of mortality in inpatients internal medicine services. Int. J. Gen. Med. 16, 3163–3170. https://doi.org/10.2147/ijgm.S420332 (2023).
Zhong, Q. et al. Protein posttranslational modifications in health and diseases: Functions, regulatory mechanisms, and therapeutic implications. MedComm 4(3), e261. https://doi.org/10.1002/mco2.261 (2020).
Elbeyli, A. & Kurtul, B. E. Systemic immune-inflammation index, neutrophil-to-lymphocyte ratio, and platelet-to-lymphocyte ratio levels are associated with keratoconus. Indian J. Ophthalmol. 69(7), 1725–1729. https://doi.org/10.4103/ijo.IJO_3011_20 (2021).
Zimmermann, J., Herrlinger, S., Pruy, A., Metzger, T. & Wanner, C. Inflammation enhances cardiovascular risk and mortality in hemodialysis patients. Kidney Int. 55(2), 648–658. https://doi.org/10.1046/j.1523-1755.1999.00273.x (1999).
Li, H., Lu, X., Xiong, R. & Wang, S. High neutrophil-to-lymphocyte ratio predicts cardiovascular mortality in chronic hemodialysis patients. Mediators Inflamm. 2017, 9327136. https://doi.org/10.1155/2017/9327136 (2017).
Li, C., Lin, Q., Wan, C. & Li, L. Nonlinear relationships between the triglyceride glucose-body mass index and cardiovascular disease in middle-aged and elderly women from NHANES (1999–2018). Sci Rep. 15(1), 10953. https://doi.org/10.1038/s41598-025-95677-5 (2025).
Fang, C. et al. The association between TyG index and cardiovascular mortality is modified by antidiabetic or lipid-lowering agent: a prospective cohort study. Cardiovasc. Diabetol. 24(1), 65. https://doi.org/10.1186/s12933-025-02620-z (2025).
Acknowledgements
The authors sincerely thank the authors who shared the original dataset in this study. Thanks to Zhang Jing (Second Department of Infectious Disease, Shanghai Fifth People’s Hospital, Fudan University) for his work on the NHANES database. His outstanding work, nhanesR package and webpage, makes it easier for us to explore NHANES database.
Funding
This work was supported by the This study was supported by Community Health Association of China (Grant number 2021-2-045) and National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University (Grant number Z2021JC005).
Author information
Authors and Affiliations
Contributions
Conceptualization: QZ, GL,ZA, and SL. Data curation: QZ, RZ and QZ. Formal analysis: QZ . Writing-original draft: QZ . Writing-review & editing: ZA and SL. Supervision: ZA and SL. All authors actively participated in the research process made substantial contributions to manuscript revisions, and carefully reviewed and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was performed according to the guidelines of the Helsinki Declaration. Consent to participate was obtained and the National Center for Health Statistics ethics committee approved the protocol of the NHANES study. The data is publicly available, therefore, the ethical approval statement and the requirement for informed consent were waived for this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, G., Zheng, R., Zeng, Q. et al. Combined association of triglyceride–glucose index and systemic inflammation index on all-cause and cardiovascular mortality. Sci Rep 15, 21464 (2025). https://doi.org/10.1038/s41598-025-06770-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06770-8