Abstract
This study aims to evaluate the association between fasting serum free fatty acids (FFAs) and diabetic retinopathy (DR) in a population of Chinese men with type 2 diabetes mellitus (T2DM). A total of 781 eligible subjects participated in this cross-sectional study. We collected clinical data and information about the patient’s examination and laboratory tests. The Shapiro–Wilk test was employed to assess the normality of continuous variables. For continuous variables with non-normal distributions, the Mann–Whitney U test and the Kruskal-Wallis test was utilized for comparisons. Categorical variable comparisons were performed using the chi-square test. Logistic regression analyses and restricted cubic spline (RCS) curves were conducted to examine the association between FFAs levels and DR. FFAs levels were slightly lower in the DR group compared to the NDR group (0.35 vs. 0.41 mmol/L, p < 0.05). The logistic regression model did not identify a statistically significant linear relationship between FFAs levels and DR incidence (OR = 0.776, 95% CI: 0.308–1.955, p = 0.591). After adjusting for various confounding factors, RCS curves revealed a U-shaped relationship between FFAs and DR. This relationship was validated in subgroup analyses stratified by age, diabetes duration, BMI, and alcohol consumption status. The U-shaped curve was evident in most subgroups, indicating that both low and high levels of FFAs are associated with an increased risk of DR. IIn this study, we found a U-shaped relationship between FFAs and DR in Chinese men with T2DM. Both low and high levels of FFAs were associated with an increased risk of DR.
Similar content being viewed by others
Introduction
Diabetes mellitus is a chronic metabolic disease with rapidly increasing prevalence worldwide, imposing a significant burden on individuals and society. Diabetic retinopathy (DR) is one of the most common microvascular complications of type 2 diabetes mellitus (T2DM) and is a leading cause of blindness1. The global prevalence of DR has been estimated at around 35% among people with diabetes2. DR not only decreases the quality of life for patients but also results in substantial economic burdens1,3. Despite extensive research, the etiology of DR remains unclear. Therefore, evaluating other factors associated with DR is crucial for a better understanding of its occurrence and progression.
Free fatty acids (FFAs) are intermediates in lipid mobilization, primarily generated from lipolysis and triglyceride (TG) breakdown4. FFAs are carboxylic acids with long saturated or unsaturated chains. Most naturally occurring FFAs consist of a straight chain with 4–28 carbon atoms5. Beyond their role as energy sources, FFAs play critical roles in receptor signaling, gene expression, and systemic energy homeostasis under various physiological conditions6,7. However, excessive FFAs can induce endothelial dysfunction through various mechanisms, with elevated plasma FFAs levels significantly contributing to arterial dysfunction. FFAs mediate the regulation of several arterial functions, including endothelial dysfunction, angiogenesis, hypertension, and atherosclerosis8,9,10,11.
The relationship between FFAs and DR has not been thoroughly investigated. In this study, we analyzed the association between DR and FFAs in hospitalized male patients with T2DM.
Methods
Study population
This study included 781 male patients aged 30–80 years old with T2DM who were treated at the Endocrinology Department of Qingdao University Affiliated Hospital between 2016 and 2022. Patients with acute T2DM complications, severe heart failure, significant liver disease, renal insufficiency requiring dialysis, malignant tumors, or those using lipid-lowering medications were excluded from the study. The study protocol was approved by the Ethics Committee of Qingdao University Medical College Affiliated Hospital (Ethics Approval Number: [QYFY WZLL 25757]). As this study was retrospective and the data came from the affiliated hospital of Qingdao University, the Ethics Committee waived patient consent. Clinical trial registration number: ChiCTR2000033114. This research adhered to the STROBE guidelines and the Declaration of Helsinki principles.
Data collection
Data collected for the study included age, height, weight, diabetes duration, alcohol and smoking status, blood pressure, fasting blood glucose (FBG), glycated hemoglobin (HbA1c), fasting C-peptide, fasting insulin, FFAs, low-density lipoprotein cholesterol (LDL), high-density lipoprotein cholesterol (HDL), TG, total cholesterol (TC), serum creatinine (SCr), serum uric acid (UA), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and estimated glomerular filtration rate (eGFR), which was calculated using the CKD-EPI formula. All participants underwent DR assessment using a fundus camera (AFC-330; NIDEK, Kyoto, Japan), slit-lamp microscope (3020 H; Keeler Ltd, Windsor, UK), and non-invasive optical coherence tomography system (5000; Carl Zeiss, Dublin, CA, USA). Patients were diagnosed with DR based on multiple characteristic manifestations: (1) non-proliferative diabetic retinopathy: microaneurysm, hard exudates, cotton-wool spot and so on; (2) proliferative diabetic retinopathy: the formation of neovascularization which can lead to retinal detachment severely. Patients were grouped based on FFA levels into four quartiles: Q1: FFAs < 0.28 mmol/L, Q2: 0.28 ≤ FFAs < 0.4 mmol/L, Q3: 0.4 ≤ FFAs < 0.53 mmol/L, and Q4: FFAs ≥ 0.53 mmol/L.
Statistical analysis
Statistical analyses were performed using R version 4.3.3. Shapiro-Wilk was used to test the normality of continuous variables. Normally distributed variables were expressed as the mean ± standard deviation, non-normal variables were expressed as the median (interquartile range), while categorical variables were expressed as the percentage (%). Group comparisons were conducted using the Chi-square test or Kruskal-Wallis test, as appropriate. Grouping based on quartiles of FFAs identifies confounders that require correction. Logistic regression analysis was used to examine the association between FFA levels and DR. Additionally, a restricted cubic spline (RCS) curve was utilized to explore the potential non-linear relationship between FFA and DR. Three nodes (knots) were set up based on the method proposed by Harrell, with node positions at the 10th, 50th, and 90th percentiles. A p-value < 0.05 was considered statistically significant.
Results
Baseline characteristics stratified by DR
This study included 781 male patients with T2DM, all of whom were not on lipid-lowering therapy. Patients were divided into two groups based on the presence of DR: the DR group (n = 178) and the non-DR group (NDR, n = 603). The Shapiro-Wilk test demonstrated non-normal distributions across all continuous variable. The baseline characteristics of the two groups are presented in Table 1. The analysis revealed that FFAs levels were slightly lower in the DR group compared to the NDR group (0.35 vs. 0.41 mmol/L, p = 0.034), suggesting a potential association between FFAs levels and the development of DR. Additionally, the DR group had a longer diabetes duration (10 vs. 8 years, p = 0.002) and higher HbA1c levels (8.6% vs. 7.9%, p < 0.001) than the NDR group, which are factors closely related to the onset and progression of retinopathy. Furthermore, fasting C-peptide and fasting insulin levels were significantly lower in the DR group, indicating potentially more severe pancreatic β-cell dysfunction. Notably, although the difference was small, the body mass index (BMI) was slightly lower in the DR group compared to the NDR group (25.6 vs. 26.0 kg/m², p = 0.046). There were no significant differences between the two groups in terms of age, smoking history, alcohol consumption, hypertension history, FBG, eGFR, LDL, HDL, TC, TG, and UA.
Baseline characteristics stratified by FFAs quartiles
Based on FFA quartiles, all patients were divided into four groups, with baseline characteristics detailed in Table 2. As FFAs levels increased, the prevalence of DR initially increased and then decreased, suggesting a potential nonlinear relationship between FFAs and DR. This trend was further validated through RCS regression analysis. Additionally, with higher FFAs levels, there was a significant increase in BMI and a corresponding rise in the prevalence of hypertension. Moreover, FBG, fasting C-peptide, fasting insulin, and TC also showed a progressive upward trend, indicating that FFAs may be closely linked to various factors associated with metabolic syndrome. Conversely, the duration of diabetes tended to shorten as FFAs levels increased, which may reflect varying associations between FFAs levels and disease progression at different stages of diabetes. However, it is noteworthy that there were no significant differences in age, smoking history, alcohol consumption, HbA1c, eGFR, LDL, HDL, and TG across the different FFAs quartiles, suggesting that these factors may not play a dominant role in the variations of FFAs levels.
Logistic regression analysis and RCS curve
In this study, we initially employed logistic regression analysis to investigate the association between FFAs and DR. The logistic regression model did not identify a statistically significant linear relationship between FFAs levels and DR incidence (OR = 0.776, 95% CI: 0.308–1.955, p = 0.591). Based on the results in Table 2 and in conjunction with the risk factors for DR, after adjusting for potential confounders, including age, diabetes duration, hypertension history, smoking status, alcohol consumption, BMI, HbA1c, fasting C-peptide, LDL, HDL, TC, and TG, the association between FFAs and DR remained non-significant (OR = 1.027, 95% CI: 0.380–2.777, p = 0.958) (Table 3).
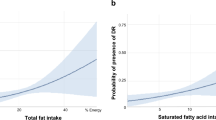
Subsequent analysis using RCS modeling identified a significant U-shaped association between FFAs levels and DR risk (p for nonlinearity = 0.0001; overall p = 0.00063) (Fig. 1). These findings highlight the necessity of accounting for the nonlinear relationship between FFAs and DR across the entire FFAs spectrum when evaluating its influence on DR.
Non-liner relationship between FFAs and DR.
Subgroup analysis
Based on the subgroup analysis results from Fig. 2, a clear U-shaped relationship between FFAs levels and DR risk was observed in most subgroups. Specifically, in the age groups < 60 years vs. ≥60 years (Fig. 2A), diabetes duration groups < 10 years vs. ≥10 years (Fig. 2B), and drinking status groups non-drinkers vs. drinkers (Fig. 2D), a significant nonlinear relationship was evident, with DR risk initially decreasing and then increasing as FFAs levels rose, particularly at higher FFAs levels. However, in patients with a BMI < 25 (Fig. 2C), although the relationship did not reach statistical significance (p = 0.24), this result may be influenced by the limited sample size, and a similar U-shaped trend might still exist. Overall, these findings suggest that the relationship between FFAs levels and DR risk is potentially complex and nonlinear, with consistent trends across different subgroups, indicating that the role of FFAs in the development of DR warrants further investigation.
Non-liner relationship between FFAs and DR(Subgroup analysis).
Discussion
Our study revealed a significant U-shaped relationship between serum FFAs and DR, where both low and high levels of FFAs were associated with increased incidences of DR. This dual role of FFAs underscores the importance of maintaining optimal FFA slevels to prevent the progression of DR in diabetic patients. Notably, extremely low FFAs levels were also linked to a higher risk of DR. There is a strong correlation between dyslipidaemia and DR, and in the Fenofibrate Intervention in Diabetes and Event Lowering (FIELD) and Action to Control Cardiovascular Risk in Diabetes (ACCORD) studies the lipid-lowering drug fenofibrate delayed the progression of retinopathy independently of glycaemic control12,13. Serum levels of saturated fatty acids such as palmitic acid (PA) are significantly elevated in diabetic patients, and together with high glucose levels, PA promotes the expression of DR-associated angiogenic and inflammatory targets, including PTGS2 and CXCL8, in human Müller cells14. These pro-inflammatory cytokines are potent stimulators of retinal endothelial pathology, such as leukocyte stasis, vascular permeability and basement membrane thickening15. In addition, elevated PA promotes apoptosis of retinal microvascular pericytes (PCs) through pathways such as oxidative stress, ceramide synthesis, and NF-κB activation16.
Similarly, other studies have found that low FFAs levels are associated with poor clinical outcomes, such as in out-of-hospital cardiac arrest (OHCA) patients, where lower FFAs levels were significantly linked to worse neurological outcomes17 .These findings collectively suggest that FFAs may have a protective role under certain pathological conditions, while exacerbating disease progression under others. This may be because moderate FFAs serve as an essential energy source for retinal cells, helping to sustain their normal function18. We speculate that in diabetic patients, insulin insufficiency or resistance leads to reduced glucose uptake by cells, particularly during hypoglycemia, exacerbating DR19. At such times, FFAs may act as an alternative energy source, maintaining cellular function and mitigating damage caused by energy deficits, which might explain the negative correlation between FFAs and DR at certain concentrations.
Interestingly, prior studies have also explored the role of specific fatty acids in DR. For example, a study highlighted that higher levels of omega-3 fatty acid DHA were inversely associated with the presence and severity of diabetic retinopathy in patients with type 2 diabetes20. Similarly, a propensity score-matched case-control study highlighted a significant negative association between n-6 PUFA levels and DR21. These findings align with our results, suggesting that not only total FFAs but also specific PUFA subtypes, such as n-6 PUFAs, may play crucial roles in modulating DR risk. Future research could focus on differentiating the effects of various FFAs and PUFAs on DR, which may offer new therapeutic targets for preventing this complication in diabetic patients.
However, as FFAs levels continue to rise, their relationship with retinopathy gradually shifts to a positive correlation, a finding that aligns closely with the results of Lakshmi Kanta Mondal et al. Their study conducted an in-depth cross-sectional analysis of lipid profiles in patients with T2DM and DR, revealing that blood levels of FFAs were significantly elevated in patients with DR22. In contrast, our study not only included a larger sample size of male patients but also identified and established for the first time a U-shaped relationship between FFAs and DR. Although studies directly linking total FFAs to DR are relatively scarce, the detrimental effects of excessive fatty acids on metabolic systemic diseases have been well documented in the literature. For instance, research has clearly demonstrated that FFAs are significantly associated with insulin resistance23 and the development of T2DM24, and they also serve as an independent and potent risk factor for hypertension25. The potential mechanisms underlying the positive correlation between high FFAs levels and retinopathy may include several aspects: Firstly, FFAs can activate multiple inflammatory pathways, leading to a significant increase in the expression of inflammatory mediators such as tumor necrosis factor-α (TNF-α), monocyte chemoattractant protein-1 (MCP-1), and interleukin-6 (IL-6), and promoting the secretion of adhesion molecules such as ICAM-1 and VCAM-1, thereby initiating and exacerbating endothelial dysfunction8,10,26,27. Additionally, FFAs are a major source of reactive oxygen species (ROS); excessive FFAs can increase ROS production by activating pathways involving protein kinase C (PKC) and NADPH oxidase, thereby inducing oxidative stress25,28,29, which further damages tissues and cells. Moreover, high levels of FFAs may induce endothelial cell apoptosis and exacerbate endothelial damage through the protein phosphatase 2Cb (PP2Cb) and TLR4 signaling pathways30,31. In summary, this U-shaped relationship suggests that maintaining appropriate FFA levels may have significant clinical implications for the long-term management of diabetes, not only in delaying or preventing the onset of DR but also in improving overall metabolic outcomes and prognosis in patients.
This study has several limitations. First, as a cross-sectional study, we cannot infer causality. Second, all recruited patients were hospitalized in the same region, so the results may not be generalizable to other populations. Third, there is currently a lack of sufficient experimental evidence to explain the relationship between FFAs and DR.
In the future, we aim to explore new research areas regarding the relationship between FFAs and DR. The potential role of FFAs in the treatment of DR still needs to be carefully and comprehensively evaluated. Large-scale prospective cohort studies are necessary to determine the causal relationship between FFAs and DR. Furthermore, our current measurements are of total FFAs, without differentiating their specific types. Future research needs to clarify the specific relationships between different types of FFAs and DR.
Conclusion
In this study, we found a U-shaped relationship between FFAs and DR in Chinese men with T2DM. Both low and high levels of FFAs were associated with an increased risk of DR. Future, more complex prospective studies are needed to confirm these findings and explore their causal relationships. This suggests that monitoring and managing FFA levels may be crucial in reducing the incidence of DR in T2DM patients.
Data availability
Clinical data for this study can be obtained by contacting the corresponding author.
References
Cheung, N., Mitchell, P. & Wong, T. Y. Diabetic retinopathy. Lancet 376 (9735), 124–136 (2010).
Leasher, J. L. et al. Global estimates on the number of people blind or visually impaired by diabetic retinopathy: A Meta-analysis from 1990 to 2010. Diabetes Care. 39 (9), 1643–1649 (2016).
Brown, M. M. et al. Utility values and diabetic retinopathy. Am. J. Ophthalmol. 128 (3), 324–330 (1999).
Zechner, R. et al. Lipolysis: pathway under construction. Curr. Opin. Lipidol. 16 (3), 333–340 (2005).
Kimura, I. et al. Free fatty acid receptors in health and disease. Physiol. Rev. 100 (1), 171–210 (2020).
Blad, C. C., Tang, C. & Offermanns, S. G protein-coupled receptors for energy metabolites as new therapeutic targets. Nat. Rev. Drug Discov. 11 (8), 603–619 (2012).
Hara, T. et al. Free fatty acid receptors and their role in regulation of energy metabolism. Rev. Physiol. Biochem. Pharmacol. 164, 77–116 (2013).
Iantorno, M. et al. Obesity, inflammation and endothelial dysfunction. J. Biol. Regul. Homeost. Agents. 28 (2), 169–176 (2014).
Virdis, A. Endothelial dysfunction in obesity: role of inflammation. High Blood Press. Cardiovasc. Prev. 23 (2), 83–85 (2016).
Wang, Y. H. et al. Physcion prevents high-fat diet-induced endothelial dysfunction by inhibiting oxidative stress and endoplasmic reticulum stress pathways. Eur. J. Pharmacol. 943, 175554 (2023).
Yu, F. et al. Free fatty acids and free fatty acid receptors: role in regulating arterial function. Int. J. Mol. Sci. 25 (14) (2024).
Keech, A. C. et al. Effect of Fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): a randomised controlled trial. Lancet 370 (9600), 1687–1697 (2007).
Wright, A. D. Dodson. Medical management of diabetic retinopathy: Fenofibrate and ACCORD eye studies. Eye (Lond). 25 (7), 843–849 (2011).
Capozzi, M. E. & Giblin, M. J. Penn. Palmitic acid induces Muller cell inflammation that is potentiated by Co-treatment with glucose. Sci. Rep. 8 (1), 5459 (2018).
Capozzi, M. E. et al. The peroxisome proliferator-activated receptor-beta/delta antagonist GSK0660 mitigates retinal cell inflammation and leukostasis. Exp. Eye Res. 190, 107885 (2020).
Cacicedo, J. M. et al. Activation of AMP-activated protein kinase prevents lipotoxicity in retinal pericytes. Invest. Ophthalmol. Vis. Sci. 52 (6), 3630–3639 (2011).
Lee, D. et al. The association between serum free fatty acid levels and neurological outcomes in out-of-hospital cardiac arrest patients: A prospective observational study. Med. (Baltim). 103 (27), e38772 (2024).
Calder, P. C. Functional roles of fatty acids and their effects on human health. JPEN J. Parenter. Enter. Nutr. 39 (1 Suppl), 18S–32S (2015).
Guo, C. et al. HIF-1alpha accumulation in response to transient hypoglycemia May worsen diabetic eye disease. Cell. Rep. 42 (1), 111976 (2023).
Weir, N. L. et al. A combined analysis of MESA and GOLDR cohorts. Retina 43 (6), 984–991 (2023).
Li, J. S. et al. Association of n-6 PUFAs with the risk of diabetic retinopathy in diabetic patients. Endocr. Connect. 9 (12), 1191–1201 (2020).
Mondal, L. K. et al. Do different lipid components accelerate the pathogenesis and severity of diabetic retinopathy??. Int. J. Retina Vitreous. 8 (1), 39 (2022).
Alipoor, E. et al. The relationship between serum adipokines and glucose homeostasis in normal-weight and obese patients on hemodialysis: a preliminary study. Int. Urol. Nephrol. 52 (11), 2179–2187 (2020).
Huang, X. et al. Interactive association of lipopolysaccharide and free fatty acid with the prevalence of type 2 diabetes: A community-based cross-sectional study. J. Diabetes Investig. 10 (6), 1438–1446 (2019).
Fagot-Campagna, A. et al. High free fatty acid concentration: an independent risk factor for hypertension in the Paris prospective study. Int. J. Epidemiol. 27 (5), 808–813 (1998).
Ishida, T. et al. Eicosapentaenoic acid prevents saturated fatty acid-Induced vascular endothelial dysfunction: involvement of Long-Chain Acyl-CoA synthetase. J. Atheroscler Thromb. 22 (11), 1172–1185 (2015).
Boden, G. Obesity, insulin resistance and free fatty acids. Curr. Opin. Endocrinol. Diabetes Obes. 18 (2), 139–143 (2011).
Inoguchi, T. et al. High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C–dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes 49 (11), 1939–1945 (2000).
Cury-Boaventura, M. F. & Curi, R. Regulation of reactive oxygen species (ROS) production by C18 fatty acids in Jurkat and Raji cells. Clin. Sci. (Lond). 108 (3), 245–253 (2005).
Hufnagel, B. et al. Unsaturated fatty acids isolated from human lipoproteins activate protein phosphatase type 2Cbeta and induce apoptosis in endothelial cells. Atherosclerosis 180 (2), 245–254 (2005).
Zhang, Y. et al. Palmitate induces VSMC apoptosis via toll like receptor (TLR)4/ROS/p53 pathway. Atherosclerosis 263, 74–81 (2017).
Funding
Project ZR2023MH316 supported by Shandong Provincial Natural Science Foundation.
Author information
Authors and Affiliations
Contributions
Conception and design: Jintao Chen, Yangang Wang. Data collection: Jintao Chen, Chuanfeng Liu, Yajing Huang. Writing the manuscript: Jintao Chen, Yajing Hang,. Review, and revision of the manuscript: Yangang WANG, Lili Xu, Jingwei Chi. All authors read and approved the final draft of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, J., Huang, Y., Liu, C. et al. Relationship between free fatty acids and diabetic retinopathy in male patients with type 2 diabetes. Sci Rep 15, 20713 (2025). https://doi.org/10.1038/s41598-025-07001-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07001-w