Abstract
To establish a prognostic stratification nomogram for non-metastatic male breast cancer to determine which patients can benefit from chemotherapy. A population-based study was conducted using data collected from the surveillance, epidemiology, and end results database. Cox proportional hazards analysis identified significant prognostic factors for survival. A prognostic stratification model was developed using R software. Propensity score matching was implemented to balance characteristics between the chemotherapy cohort and the non-chemotherapy cohort. The multivariate analyses indicated that age, race, grade, surgery, primary tumor, marital status, T stage, and N stage were independent prognostic factors for overall survival in non-metastatic metastatic MBC patients who did not receive chemotherapy (all P < 0.05). The C-index was 0.786 (95% CI 0.662–0.870) in the training cohort and 0.763 (95% CI 0.517–0.852) in the validation cohort. The nomogram effectively discriminated between low-risk, moderate-risk, and high-risk groups concerning OS (P < 0.0001). The current study developed the first prognostic stratification nomogram for non-metastatic MBC and identified that patients in the moderate-risk and high-risk groups are more likely to benefit from chemotherapy.
Similar content being viewed by others
Introduction
Male breast cancer (MBC), constituting only 1% of all breast cancer cases, manifests as a rare malignancy affecting the breast tissue in men1,2. Men, diagnosed with MBC, often present with larger, higher-grade tumors and increased lymph node involvement compared to their female counterparts with breast cancer3. The 5-year mortality rate for MBC exceeds that of female breast cancer (FBC), potentially attributed to factors such as older age at diagnosis, diagnostic delays, the presence of comorbidities, or inherent biological distinctions4,5. Invasive ductal carcinoma is the predominant diagnosis for most men with breast cancer, characterized by tumors that are estrogen-receptor (ER) positive, progesterone receptor (PR) positive, and HER2 negative6,7. At the molecular level, the majority of MBC cases exhibit Luminal A-like or Luminal B-like tumors8,9,10. Some studies suggest unique MBC subtypes, M1 and M2, distinct from intrinsic FBC subtypes11,12. Similar to FBC, men with a familial history of BRCA mutations face an increased risk of MBC, with a higher prevalence of BRCA2 mutations compared to BRCA1 mutations in women13. Furthermore, African American men and those with comorbidities such as Klinefelter’s syndrome, hormonal imbalances, liver disease, and obesity have elevated risks of developing breast cancer14,15.
Although MBC shares similarities with certain forms of FBC, the scarcity of data regarding treatment in males complicates the search for tailored therapies16,17,18. Consequently, men afflicted with breast cancer often undergo treatments based on paradigms established for women, despite the potential inefficacy of these approaches. Chemotherapy plays a pivotal role in the comprehensive management of breast cancer. Specifically, chemotherapy regimens incorporating anthracyclines and taxanes have been shown to substantially enhance the survival outcomes of patients with non-metastatic breast cancer19,20.
However, recently, an increasing body of research has indicated that not all breast cancer patients require chemotherapy21,22. This understanding marks a significant shift in the approach to treating breast cancer, moving towards more personalized and targeted therapies. It highlights the importance of accurately identifying the characteristics of the tumor and the individual patient’s profile to determine the most appropriate treatment plan. Several studies have identified gene signatures capable of predicting overall survival or breast cancer recurrence23,24,25. These signatures offer valuable guidance regarding the appropriateness of adopting a more aggressive treatment approach, which can assist doctors in screening which patients require chemotherapy. One such prognostic tool, Oncotype DX®, utilizes a 21-gene panel expression analysis and is widely employed in breast cancer prognosis26,27,28,29. This tailored approach not only minimizes unnecessary exposure to potentially harmful treatments but also improves the overall quality of care for breast cancer patients.
However, currently, the methods used to determine whether patients need chemotherapy mostly rely on data from female breast cancer. For non-metastatic male breast cancer, there is still no effective way to screen which patients are suitable for chemotherapy. This study, based on data from 5656 male breast cancer patients, aimed to comprehensively analyze prognostic factors for non-metastatic male breast cancer. The objectives were to establish a prognostic stratification model for patients and to identify the potential population that could benefit from chemotherapy.
Patients and methods
Patients’ cohort
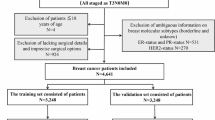
The data of patients diagnosed with non-metastatic breast cancer (MBC) were obtained from the surveillance, epidemiology, and end results (SEER) database. SEER, which compiles cancer incidence data encompassing patient demographics, tumor characteristics, therapy, and follow-up, spans almost 30% of the United States population and undergoes annual updates. The SEER*Stat program (version 8.3.5) was employed to identify pertinent patients between 2000 and 2014. Male patients diagnosed with primary breast cancer according to the third edition of the International Classification of Diseases (ICD-O-3), without distant metastasis (M0), and/or with a confirmed histological diagnosis of breast cancer based on the histological/behavior code (ICD-O-3 Hist/behav, malignant) were included. Patients with bilateral breast cancer, unclear unilateral breast cancer, and/or incomplete follow-up (survival months = 0) were excluded. Furthermore, patients with missing clinical data, including race, grade, treatment details (surgery, radiation therapy, and chemotherapy), TNM stage, estrogen receptor (ER), and progesterone receptor (PR) status, were also excluded.
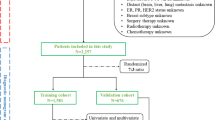
A total of 4445 cases of MBC with non-metastatic were finally included in this study. Among them, 1643 (36.96%) patients received chemotherapy, while 2802 (63.04%) patients did not receive chemotherapy were involved in the non-chemotherapy cohort to construct a nomogram. The detailed flowchart for this study was presented in Fig. 1.
Flowchart of the study design. A total of 4445 patients with non-metastatic male breast cancer were finally involved in our study.
Ethics and statement
This study was approved by the Ethics Committee review of the Xijing Hospital, The Fourth Military Medical University, Xian, China and was in accordance with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical stabdards. This study was conducted using the transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD) statement, which was the reoorting guideline of prediction models30.
Covariates and endpoints
The demographic data (ethnicity, age at the diagnosis and marital status), clinical data (grading, staging, ER and PR status and regional nodes), treatments (surgery, chemotherapy and radiotherapy) and follow-up information were retrieved from the SEER database. Unmarried people were defined as divorced, separated, widowed or single (having a domestic partner or never married). OS was the main primary endpoint, designated as the survival in months to all-cause mortality.
Construction of the nomogram
The categorical variables are presented as proportions and frequencies. Statistical analyses, including the Chi-square and Fisher’s exact tests, were conducted to compare baseline categorical variables between the training and validation cohorts. Continuous variables are reported using the median and interquartile range (IQR). Group comparisons for continuous variables were performed using Student’s t-test and the non-parametric Mann–Whitney U test. Univariate Cox regression analysis identified variables predictive of prognosis, and subsequently, a multivariate Cox regression analysis determined independent prognostic indicators. To predict overall survival (OS) at 3, 5, and 10 years, a nomogram model was constructed by integrating all identified independent prognostic factors.
Nomogram’s discrimination and calibration
The nomogram’s ability to discriminate between different survival groups was estimated through the Concordance index (C-index). Model fitting was done for 1000 bootstraps. The C-index fluctuates from 0.5 to 1, with 1 representing the highest discrimination ability. The time-dependent receiver operating characteristic (tROC) and receiver operating characteristic (ROC) curves were implemented to verify the prediction accuracy.
A calibration curved line reflecting the relevance between the forecasted and the observed survival probability was used to assess the calibration of the model. A straight calibration line threaded through the axis’s starting point with a rake rate of 1 indicates excellent calibration. The predictive capacity of the model correlated with the closeness of the forecasted calibration curved line to the ideal curve. A decision curve analysis (DCA) verified the net benefit and latent clinical effectiveness for different predictive percentage thresholds.
Classification of the risk groups by the model
In the training cohort, the total nomogram score was calculated for each patient. According to a cut-off nomogram score calculated through the X-tile software, patients were stratified into high-, moderate- and low-risk categories. The Kaplan–Meier survival analysis and log-rank test analyzed the different survival between groups.
Identify the population benefiting from chemotherapy
To mitigate potential confounding factors and selection bias, propensity score matching (PSM) without replacement was utilized, employing the nearest-neighbor method with a caliper set at 0.231. Standardized mean difference (SMD) was calculated for baseline variables of all independent predictors before and after PSM, where an SMD of < 0.10 indicates relatively minor imbalance32. The MatchIt package facilitated balancing baseline characteristics between the chemotherapy and non-chemotherapy cohorts across different risk groups. By comparing survival outcomes between these cohorts within each risk group, we identified a potential population likely to benefit from chemotherapy. Kaplan–Meier curves were generated using the ggplot2 package, while decision curves were drawn using the Caret and ggDCA packages.
Statistical analysis
We deployed SPSS program (version 26.0), R software (version 3.5.3) and X-tile software to analyze data. We judged a P value below 0.05 as statistically significant.
Results
Characteristics of the training cohort and validation cohort
A cohort of 4445 patients diagnosed with non-metastatic MBC from the SEER database was included in our study. Among these, 1643 patients underwent chemotherapy, while the remaining 2802 did not receive chemotherapy (Fig. 1). Patients who did not receive chemotherapy were divided into different groups using the random split sample method at a ratio of 7:3. The training cohort comprised 1961 patients, while the validation cohort consisted of an additional 841 patients (Fig. 1). Characteristics of the two cohorts are presented in Table 1, showing comparable attributes. All patients were divided into two prognostic groups based on the cutoff value of age using R software (Fig. 2B): < 75 group and ≥ 75 group.
Development of a prognostic stratification nomogram and validation of the proposed nomogram. (A) A prognostic stratification nomogram to accurately predict overall survival for non-metastatic MBC. (B) Identify the optimal cutoff value for age using R software.
The median follow-up for the total population was 60 months (IQR 33–99 months), with 3-year, 5-year, and 10-year overall survival (OS) rates of 0.812 (95% CI 0.798–0.827), 0.691 (95% CI 0.673–0.710), and 0.461 (95% CI 0.437–0.485), respectively. The median follow-up for the training cohort and validation cohort were 60 months (IQR 33–100 months) and 62 months (IQR 35–98 months), respectively. The 3-year, 5-year, and 10-year overall survival (OS) rates of the training cohort were 0.806 (95% CI 0.788–0.824), 0.688 (95% CI 0.667–0.711), and 0.453 (95% CI 0.425–0.482), respectively. The 3-year, 5-year, and 10-year overall survival (OS) rates of the validation cohort were 0.827 (95% CI 0.802–0.854), 0.697 (95% CI 0.665–0.731), and 0.481 (95% CI 0.439–0.527), respectively.
Independent prognostic variables in the training cohort and establishment of the nomogram of prognosis
The univariable and multivariable results were presented in Table 2. Age, race, grade, surgery, primary tumor, marital status, T stage and N stage were independent prognostic factors for OS in non-metastatic MBC (all P < 0.05). The nomogram forecasting survival time based on the 8 independent risk factors is illustrated in Fig. 2A. The risk score for each independent prognostic indicator was calculated by sketching a vertical line from the independent variable. The total risk was then calculated by considering the scores obtained for each variable. The OS at 3, 5 and 10 years was calculated by sketching a line from the total points axis to the corresponding survival axis.
Verification of the model’s performance
For overall survival (OS), the concordance index (C-index) demonstrated robust prognostic discrimination in both the training (0.786, 95% CI 0.662–0.870) and validation cohorts (0.763, 95% CI 0.517–0.852). Area under the curve (AUC) values at 3, 5, and 10 years were 0.738 (95% CI 0.711–0.766), 0.764 (95% CI 0.740–0.788), and 0.791 (95% CI 0.761–0.822) respectively for the training cohort. For the validation cohort, AUC values at the corresponding years were 0.733 (95% CI 0.688–0.777), 0.768 (95% CI 0.731–0.805), and 0.789 (95% CI 0.744–0.834) (Fig. 3A,B). The time-dependent AUC values demonstrated excellent performance and discrimination, particularly evident in Fig. 3C and D. Calibration curves confirmed a satisfactory match between predicted and observed OS outcomes in both cohorts, as illustrated in Fig. 3E and F. Decision curve analysis over 3, 5, and 10 years revealed that, compared to single-factor models encompassing AJCC stage, grade, and N stage, the nomogram provided a substantial net benefit across all threshold probabilities, as detailed in Fig. 4.
Validation of the proposed nomogram. (A,B) ROC curves for predicting the overall survival in the training and validation cohorts at 3-year, 5-year, and 10-year, respectively. (C,D) the time-dependent AUC values of nomogram in the training cohort and validation cohort. (E,F) the calibration curves for predicting patients’ overall survival in the training and validation cohorts at 3-year, 5-year, and 10-year, respectively.
Decision curve analysis for non-metastatic MBC. Decision curve analysis of the nomogram and single independent predictors for predicting the 3-year OS (A,D), 5-year OS (B,E) and 10-year OS (C,F) in the training cohort and validation cohort, respectively.
Risk stratification
MBC patients who received chemotherapy exhibited a better prognosis compared to those who did not receive chemotherapy, as shown in Fig. 5D. A total of 1961 MBC patients who did not undergo chemotherapy were classified into three prognostic groups based on their total scores, calculated using X-tile software (Fig. 5A–C): the low-risk group (891 patients; total score ≤ 104.2), the moderate-risk group (859 patients; 104.2 < total score ≤ 224.9), and the high-risk group (211 patients; total score > 224.9). The Kaplan–Meier survival curves demonstrated excellent discrimination among these groups (P < 0.001, Fig. 5E).
Determination of best cut-off value of total sum score by the X-tile software (A–C). The cut-point distribution histogram illustrates the distribution of patient numbers corresponding to each threshold value across the range of cut-off points (A). The cut-point selection plot, generated by X-tile, visually presents the distribution of low-population and high-population patient groups across various threshold values, assisting researchers in identifying an optimal cutoff that is both statistically significant and clinically meaningful (B). The survival curve plot illustrates the differences in survival between patient groups stratified by a specific cutoff value, aiding in the evaluation of the clinical and statistical significance of the selected threshold (C). Kaplan–Meier survival curves between the chemotherapy cohort and non-chemotherapy cohort for the entire group before PSM (D) and after PSM (F). Survival curves in the entire cohort stratified by the total score of the nomogram (E).
Identify patients with non-metastatic MBC who could derive the most benefit from chemotherapy, based on PSM
Propensity score matching (PSM) was employed to balance independent prognostic factors—specifically, age, race, grade, type of surgery, primary tumor, marital status, T stage, and N stage—between the chemotherapy cohort and the non-chemotherapy cohort. After PSM, each cohort comprised 1051 cases (Table 3). The 10-year overall survival (OS) rate was 57.5% in the chemotherapy cohort and 49.2% in the non-chemotherapy cohort. This finding indicates that chemotherapy significantly improves 10-year OS (P < 0.001; HR 2.465; 95% CI 2.192–2.772; Fig. 5F). Subsequent PSM analyses within three different prognostic groups—low-risk, moderate-risk, and high-risk—revealed 447, 410, and 59 cases of male breast cancer, respectively. As shown in Table 3, standardized mean differences (SMDs) for all variables were less than 0.1. Before PSM, chemotherapy improved OS in all three groups (Fig. 6A–C). However, after PSM, chemotherapy improved OS in the high-risk and moderate-risk groups but did not significantly enhance OS in the low-risk group (Fig. 6D–F). In the low-risk group, the 10-year OS was nearly equivalent between cohorts (P = 0.52; HR 0.92; 95% CI 0.71–1.18), with rates of 89.6% in the chemotherapy cohort and 86.1% in the non-chemotherapy cohort. In the moderate-risk group, chemotherapy significantly improved 10-year OS (P < 0.0001; HR 0.56; 95% CI 0.46–0.69), with rates of 78.3% in the chemotherapy cohort and 49.4% in the non-chemotherapy cohort. In the high-risk group, chemotherapy also significantly improved 10-year OS (P = 0.034; HR 0.61; 95% CI 0.38–0.97), with rates of 25.0% in the chemotherapy cohort and 16.4% in the non-chemotherapy cohort. This study concludes that chemotherapy significantly improves OS in non-metastatic male breast cancer patients within the moderate and high-risk groups.
Kaplan–Meier survival curves of OS between the chemotherapy cohort and non-chemotherapy cohort for the low-risk group (A,D), moderate-risk group (B,E) and high-risk group (C,F) before PSM and after PSM.
Discussion
Based on the data of 4445 MBC patients with non-metastatic from the real-world, we determined its independent prognostic factors, developed a prognostic stratification model that can predict individual prognosis with favorable accuracy and discrimination, and applied the model to stratify the entire cohort into different risk groups to identify the optimal candidates benefiting from chemotherapy. In the entire cohort, chemotherapy did improve 10-year OS of non-metastatic MBC significantly. In the low-risk group, 10-year OS rates of the chemotherapy cohort and non-chemotherapy cohort were not significantly different, while chemotherapy can significantly improve the 10-year OS rate in the high-risk group and moderate-risk group. The innovation and advantage of this research lies in: (a) conducting the study based on a large sample size; (b) building up the first prognostic stratification nomogram specially for non-metastatic MBC based on multi-ethnic population; (c) validating excellent performance of risk stratification of nomogram by using Kaplan–Meier method and decision curve analysis; (d) applying PSM to balance the baseline characteristics between the chemotherapy cohort and non-chemotherapy cohort in prognostic stratification groups to minimize the confounding factors of independent features; (e) finding that chemotherapy could effectively improve the OS of patients in the high-risk group and moderate-risk group after PSM, with 10-year OS absolute improvement by 8.6% and 28.9%.
In this study, age, race, grade, surgical history, primary tumor, marital status, T stage, and N stage were identified as independent prognostic factors for non-metastatic MBC. Consistent with previous research, older age, grade III/IV tumors, stage IV disease, lack of surgical intervention, and unmarried status were associated with significantly shorter overall survival (OS)33,34,35. Furthermore, our findings indicated that patients with non-primary male breast cancer and those of African descent often exhibit a substantially higher prognostic risk. Notably, our analysis revealed that the age group ≥ 70 years represented an independent risk factor for OS compared to the age group < 70 years, which partially contrasts with earlier studies on female breast cancer that designated individuals aged 40 years or younger as a significant high-risk factor36,37. This discrepancy aligns with the observations of Elomrani et al.38, which suggest that individuals aged ≥ 70 are more likely to have comorbidities, such as cardiovascular diseases and chronic obstructive pulmonary disease, leading to an increased incidence of all-cause mortality compared with those aged < 70. Li et al.3 reported a median age of 66.8 for MBC, notably higher than the 61.6 median age for female breast cancer. In our study, the median age was 68, corroborating Li et al.’s findings and indicating a later onset of MBC. Moreover, our study revealed a higher prevalence of hormone receptor positivity in MBC, with 96.89% of patients being hormone receptor positive (HR+) and 88.12% being progesterone receptor positive (PR+), surpassing rates found in studies of female breast cancer3,39,40,41. Due to the high prevalence of HR+ tumors, patients with MBC may experience a significantly better relative survival rate than those with female breast cancer. However, a study by Bhardwaj et al. revealed that the outcomes for male breast cancer patients, when matched by stage, are worse compared to a similar subtype of female breast cancer42.
In this study, we report on the efficacy of chemotherapy in non-metastatic MBC patients. To our knowledge, this represents the largest series describing the effectiveness of chemotherapy and introduces the first stratification model to accurately identify patients who may benefit from chemotherapy and those who may be exempt from it within this population. MBC is a disease predominantly affecting elderly men, who are at a heightened risk of harm from chemotherapeutic agents, particularly due to the frequently co-existing comorbidities43. Therefore, it is crucial to identify which patients can genuinely benefit from chemotherapy. This study demonstrated that chemotherapy significantly improves the prognosis of patients with MBC (chemotherapy vs non-chemotherapy, P < 0.001, HR 0.680, 95% CI 0.616–0.752). Even after propensity score matching (PSM) to balance independent prognostic factors—including age, race, tumor grade, type of surgery, primary tumor, marital status, T stage, and N stage—the chemotherapy group consistently displayed better survival outcomes (chemotherapy vs. non-chemotherapy, P < 0.001, HR 0.729, 95% CI 0.637–0.835).
The prognostic model stratifies patients’ prognosis into three distinct risk groups. Our analysis suggests that individuals in the high-risk and moderate-risk categories may constitute a population that could benefit from chemotherapy. Patients classified in the high-risk and moderate-risk groups exhibit a greater number of independent risk factors and potentially experience higher all-cause mortality rates compared to those in the low-risk group. The escalation of treatment has provided these patients with improved tumor control and prolonged survival, particularly for those at higher risk of mortality. Recent studies have sought to identify treatments that can influence the prognosis of MBC. While these studies primarily focus on hormonal therapy and patients with metastatic disease, they typically consider only tumor burden—defined as T stage and N stage—while neglecting other critical factors43,44,45. Our study comprehensively incorporated all independent factors into a prognostic nomogram and compared the net benefits of risk stratification nomogram-assisted decisions with those of single-factor-assisted decisions. Notably, the net benefits derived from decisions based on our model and its risk stratification significantly exceeded those based on the AJCC stage, grade and N stage.
The present study developed a novel risk stratification model for non-metastatic MBC that accurately stratifies patient prognosis into distinct risk groups and identifies the optimal candidates who may benefit from chemotherapy. In principle, our nomogram can assess whether non-metastatic MBC patients represent a population that could benefit from chemotherapy. Despite the strengths of our study, several limitations must be acknowledged: (a) the SEER database lacks detailed information on radiotherapy, targeted therapy, and the dosage, regimen, and associated complications of chemotherapy, which may introduce confounding factors; (b) the database does not contain data on recurrence and disease-free survival, limiting our ability to evaluate the impact of chemotherapy on recurrence and disease-free survival; (c) while this study’s retrospective design presents inherent limitations, the propensity score matching method was employed to mitigate the influence of confounding variables; (d) our prognostic stratification model has not been validated across other centers or databases, such as the National Cancer Database or SEER-Medicare.
Conclusions
This study developed a novel prognostic stratification model for non-metastatic male breast cancer, aimed at identifying the population that may benefit from chemotherapy. The model is designed to facilitate personalized treatment approaches, ensuring that patients with non-metastatic male breast cancer receive optimal chemotherapy tailored to their specific needs.
Data availability
Publicly available datasets were analyzed in this study. This data can be found here: https://seer.cancer.gov/data/.
References
American Cancer Society. Cancer Facts and Figures 2022 (National Home Office, 2022).
Miao, H. et al. Incidence and outcome of male breast cancer: An international population-based study. J. Clin. Oncol. 29(33), 4381–4386 (2011).
Li, X. et al. Hormone receptor-positive breast cancer has a worse prognosis in male than in female patients. Clin. Breast Cancer 17, 356–366. https://doi.org/10.1016/j.clbc.2017.03.005 (2017).
Leone, J. et al. Tumor subtypes and survival in male breast cancer. Breast Cancer Res. Treat. 188, 695–702. https://doi.org/10.1007/s10549-021-06182-y (2021).
Wang, F. et al. Overall mortality after diagnosis of breast cancer in men vs women. JAMA Oncol. 5, 1589–1596. https://doi.org/10.1001/jamaoncol.2019.2803 (2019).
Brcic, I. et al. Tumor microenvironment in male breast carcinoma with emphasis on tumor infiltrating lymphocytes and PD-L1 expression. Int. J. Mol. Sci. 24(1), 818 (2023).
Blum, J. L. et al. Outcomes of male patients with HR+/HER2− advanced breast cancer receiving palbociclib in the real-world POLARIS study. Breast Cancer Res. Treat. https://doi.org/10.1007/s10549-023-07145-1 (2023).
Sun, H. et al. Single-cell transcriptome analysis indicates fatty acid metabolism-mediated metastasis and immunosuppression in male breast cancer. Nat. Commun. 14, 5590. https://doi.org/10.1038/s41467-023-41318-2 (2023).
Kim, H. et al. Single-cell transcriptional and epigenetic profiles of male breast cancer nominate salient cancer-specific enhancers. Int. J. Mol. Sci. 24, 13053. https://doi.org/10.3390/ijms241713053 (2023).
Chatterji, S. et al. Prediction models for hormone receptor status in female breast cancer do not extend to males: Further evidence of sex-based disparity in breast cancer. NPJ Breast Cancer 9, 91. https://doi.org/10.1038/s41523-023-00599-y (2023).
Johansson, I. et al. Gene expression profiling of primary male breast cancers reveals two unique subgroups and identifies N-acetyltransferase-1 (NAT1) as a novel prognostic biomarker. Breast Cancer Res. 14, R31. https://doi.org/10.1186/bcr3116 (2012).
Johansson, I., Ringnér, M. & Hedenfalk, I. The landscape of candidate driver genes differs between male and female breast cancer. PLoS ONE 8, e78299. https://doi.org/10.1371/journal.pone.0078299 (2013).
Shimomura, A. et al. Clinicopathological features, genetic alterations, and BRCA1 promoter methylation in Japanese male patients with breast cancer. Breast Cancer Res. Treat. 197, 593–602. https://doi.org/10.1007/s10549-022-06822-x (2023).
DeSantis, C. E. et al. Breast cancer statistics, 2019. CA Cancer J. Clin. 69, 438–451. https://doi.org/10.3322/caac.21583 (2019).
D’Angelo, A. et al. Male breast: A review of the literature and current state of the art of diagnostic imaging work-up. Diagnostics (Basel) 13, 3620. https://doi.org/10.3390/diagnostics13243620 (2023).
Accomasso, F. et al. Clinical, pathological, and prognostic features of male breast cancer: A multicenter study. Curr. Oncol. 30, 9860–9871. https://doi.org/10.3390/curroncol30110716 (2023).
Fox, S., Speirs, V. & Shaaban, A. M. Male breast cancer: An update. Virchows Arch. 480, 85–93. https://doi.org/10.1007/s00428-021-03190-7 (2022).
Zheng, G. & Leone, J. P. Male breast cancer: An updated review of epidemiology, clinicopathology, and treatment. J. Oncol. 2022, 1734049. https://doi.org/10.1155/2022/1734049 (2022).
Braybrooke, J. et al. Anthracycline-containing and taxane-containing chemotherapy for early-stage operable breast cancer: A patient-level meta-analysis of 100 000 women from 86 randomised trials. Lancet 401, 1277–1292. https://doi.org/10.1016/S0140-6736(23)00285-4 (2023).
Yu, K. et al. Personalized chemotherapy selection for breast cancer using gene expression profiles. Sci. Rep. 7, 43294. https://doi.org/10.1038/srep43294 (2017).
Mittal, A. & Batra, A. Adjuvant chemotherapy in early breast cancer: Are we over-treating patients. Natl. Med. J. India 32, 230–231. https://doi.org/10.4103/0970-258X.291305 (2019).
Eng-Wong, J. & Isaacs, C. Prediction of benefit from adjuvant treatment in patients with breast cancer. Clin. Breast Cancer 10(Suppl 1), E32–E37. https://doi.org/10.3816/CBC.2010.s.005 (2010).
Foekens, J. A. et al. Multicenter validation of a gene expression-based prognostic signature in lymph node-negative primary breast cancer. J. Clin. Oncol. 24, 1665–1671. https://doi.org/10.1200/JCO.2005.03.9115 (2006).
Harbeck, N. et al. Ten-year analysis of the prospective multicentre chemo-N0 trial validates American Society of Clinical Oncology (ASCO)-recommended biomarkers uPA and PAI-1 for therapy decision making in node-negative breast cancer patients. Eur. J. Cancer 49, 1825–1835. https://doi.org/10.1016/j.ejca.2013.01.007 (2013).
Hatzis, C. et al. A genomic predictor of response and survival following taxane-anthracycline chemotherapy for invasive breast cancer. JAMA 305, 1873–1881. https://doi.org/10.1001/jama.2011.593 (2011).
Ji, J. H. et al. Prediction of a multi-gene assay (oncotype DX and mammaprint) recurrence risk group using machine learning in estrogen receptor-positive, HER2-negative breast cancer-the BRAIN study. Cancers (Basel) 16, 774. https://doi.org/10.3390/cancers16040774 (2024).
Lux, M. P. et al. Budget impact of the oncotype DX breast recurrence score(®) test in patients with early primary hormone-receptor-positive, HER2-negative, node-positive breast cancer in Germany. Breast Care (Basel) 19, 27–33. https://doi.org/10.1159/000534096 (2024).
Xie, S. J., Wang, R. J., Wu, S. G. & Zhang, F. X. 21-gene recurrence score in predicting the outcome of postoperative radiotherapy in T1–2N1 luminal breast cancer after breast-conserving surgery. Breast 74, 103679. https://doi.org/10.1016/j.breast.2024.103679 (2024).
Record, S. M. et al. Anatomy versus biology: What guides chemotherapy decisions in older patients with breast cancer. J. Surg. Res. 296, 654–664. https://doi.org/10.1016/j.jss.2024.01.037 (2024).
Heus, P. et al. Transparent reporting of multivariable prediction models in journal and conference abstracts: TRIPOD for abstracts. Ann. Intern. Med. https://doi.org/10.7326/M20-0193 (2020).
Austin, P. C. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm. Stat. 10, 150–161. https://doi.org/10.1002/pst.433 (2011).
Bangalore, S. et al. Everolimus-eluting stents or bypass surgery for multivessel coronary disease. N. Engl. J. Med. 372, 1213–1222. https://doi.org/10.1056/NEJMoa1412168 (2015).
Leone, J., Zwenger, A. O., Leone, B. A., Vallejo, C. T. & Leone, J. P. Overall survival of men and women with breast cancer according to tumor subtype: A population-based study. Am. J. Clin. Oncol. 42, 215–220. https://doi.org/10.1097/COC.0000000000000497 (2019).
Leone, J. P. et al. Prognostic factors in male breast cancer: A population-based study. Breast Cancer Res. Treat. 156, 539–548. https://doi.org/10.1007/s10549-016-3768-1 (2016).
Leone, J. P. et al. Prognostic significance of tumor subtypes in male breast cancer: A population-based study. Breast Cancer Res. Treat. 152, 601–609. https://doi.org/10.1007/s10549-015-3488-y (2015).
Johnson, R. H., Anders, C. K., Litton, J. K., Ruddy, K. J. & Bleyer, A. Breast cancer in adolescents and young adults. Pediatr. Blood Cancer 65, e27397. https://doi.org/10.1002/pbc.27397 (2018).
Copson, E. et al. Prospective observational study of breast cancer treatment outcomes for UK women aged 18–40 years at diagnosis: The POSH study. J. Natl. Cancer Inst. 105, 978–988. https://doi.org/10.1093/jnci/djt134 (2013).
Elomrani, F. et al. Management of early breast cancer in older women: From screening to treatment. Breast Cancer (Dove Med Press) 7, 165–171. https://doi.org/10.2147/BCTT.S87125 (2015).
Teng, L. et al. A novel nomogram and survival analysis for different lymph node status in breast cancer based on the SEER database. Breast Cancer https://doi.org/10.1007/s12282-024-01591-5 (2024).
Shi, W. & Li, W. An incidental male breast cancer of (18)F-FDG PET/CT scanning for esophageal cancer. Asian J. Surg. 46, 1680–1681. https://doi.org/10.1016/j.asjsur.2022.09.121 (2023).
Fanale, D. et al. BRCA-associated hereditary male cancers: Can gender affect the prevalence and spectrum of germline pathogenic variants. Front. Oncol. 14, 1414343. https://doi.org/10.3389/fonc.2024.1414343 (2024).
Bhardwaj, P. V., Gupta, S., Elyash, A. & Teplinsky, E. Male breast cancer: A review on diagnosis, treatment, and survivorship. Curr. Oncol. Rep. 26, 34–45. https://doi.org/10.1007/s11912-023-01489-z (2024).
Di Lauro, L. et al. Efficacy of chemotherapy in metastatic male breast cancer patients: A retrospective study. J. Exp. Clin. Cancer Res. 34, 26. https://doi.org/10.1186/s13046-015-0143-8 (2015).
Corti, C., Crimini, E., Criscitiello, C., Trapani, D. & Curigliano, G. Adjuvant treatment of early male breast cancer. Curr. Opin. Oncol. 32, 594–602. https://doi.org/10.1097/CCO.0000000000000678 (2020).
Eggemann, H. et al. Tamoxifen treatment for male breast cancer and risk of thromboembolism: Prospective cohort analysis. Br. J. Cancer 120, 301–305. https://doi.org/10.1038/s41416-018-0369-2 (2019).
Author information
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study was approved by the ethics committee of the local hospital (Xijing hospital), and because it was a retrospective study, permission for the exemption of informed consent was obtained.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, M., Yao, Q., Qin, Y. et al. Development, verification, and comparison of a risk stratification model to identify potential population benefiting from chemotherapy in non-metastatic male breast cancer. Sci Rep 15, 24301 (2025). https://doi.org/10.1038/s41598-025-08440-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08440-1