Abstract
To explore the relationship between the incidence of colorectal polyps and related factors such as markers of insulin resistance (IR) , a retrospective analysis was used to collect 2716 patients who underwent physical examinations in Guangzhou Cadre and Talent Health Management Center from June 2020 to June 2023. The patients were classified as colorectal polyp group (n = 1328) and non-polyp group (n = 1388) after colonoscopy. Baseline data of colorectal polyp and non-polyp groups were compared. Non-insulin-based IR markers including Triglyceride Glucose Index (TyG), triglyceride glucose index (TyG)- Body Mass Index(BMI), triglycerides (TG)/high density lipoprotein cholesterol (HDL-C), Metabolic Score for Insulin Resistance (METS-IR), and mean arterial pressure (MAP) were calculated. The risk factors of colorectal polyps were analyzed by binary logistic regression. Receiver operating characteristic (ROC) curves and area under the curve (AUC) were used to evaluate the ability of insulin resistance markers to detect colorectal polyps. The proportion of males in colorectal polyp group was higher than that in non-polyp group. There were significant differences in TyG-BMI, TyG, TG/HDL-C, METS-IR, MAP, and other indicators between the colorectal polyp group and the non-polyp group (p < 0.05). Binary logistic regression analysis showed that elevation in age, BMI, TyG-BMI, TyG, TG/HDL-C, METS-IR, MAP and CEA were positively correlated with the risk of colorectal polyps (p < 0.05). The AUCs of insulin resistance markers and MAP were larger than CEA to detect colorectal polyps. The AUC values of models in combination of age, sex and insulin resistance markers/MAP is higher. TyG, TyG-BMI, TG/HDL-C, and METS-IR could be used as indicators to predict the occurrence of colorectal polyps. These findings provide an important clinical reference for the prevention and treatment of colorectal polyps.
Similar content being viewed by others
Introduction
Colorectal polyps are excrescences of the intestinal mucosa, which are the most common benign disease of the lower digestive tract. According to the pathology, they are classified into adenomatous polyps and non-adenomatous polyps, among which adenomatous polyps are closely related to the occurrence of colorectal cancer. The sequential progression theory of “polyp-adenoma-cancer” has become the current consensus, and the timely detection and removal of colorectal polyps are the main ways to curb the occurrence and development of colorectal cancer. It has been reported that the majority of colorectal cancer originates from adenomatous polyps, and colonoscopy is the most effective method to detect and remove colorectal polyps, thereby reducing the incidence and mortality of colorectal cancer1. However, due to the invasive nature of colonoscopy and people’s psychological fear and reluctance, coupled with poor awareness of physical examinations, colonoscopy is currently difficult to popularize nationwide. Therefore, it is crucial to identify the risk factors for the occurrence of colorectal polyps.
A study has found that age, sex, non-alcoholic fatty liver disease, and type 2 diabetes are independent risk factors for colorectal polyps2. Another study indicated that insulin resistance (IR) was significantly associated with an elevated risk of adenomatous polyps in non-Asian ethnic groups, and higher levels of insulin, C-peptide, and homeostasis model assessment of insulin resistance (HOMA-IR) are significantly associated with an increased risk of colorectal adenoma3. Currently, insulin resistance-related indicators include HOMA-IR4but due to its cumbersome sampling process and high cost, it is difficult to carry out in many clinical practices. Several studies have suggested that non-insulin-based IR markers, such as triglyceride glucose index (TyG)- Body Mass Index(BMI), TyG, triglycerides (TG)/high density lipoprotein cholesterol (HDL-C), Metabolic Score for Insulin Resistance (METS-IR) can effectively assess the degree of insulin resistance5,6. There is evidence indicating an association between insulin resistance and the occurrence of colorectal polyps7but there are fewer studies directly pointing out the correlation between TyG-BMI, TyG, TG/HDL-C, and METS-IR and colorectal polyps. Therefore, this study aims to further explore the relationship between TyG-BMI, TyG, TG/HDL-C, METS-IR, and colorectal polyps, providing a basis for clinical assessment of the risk of colorectal polyp occurrence.
Materials and methods
Study population
The study subjects were selected from individuals who underwent physical examinations and colonoscopy at the Guangzhou Cadre and Talent Health Management Center from June 2020 to June 2023. According to the exclusion and inclusion criteria, a total of 2716 individuals meeting the requirements were screened. All of individuals accomplished the colonoscopy, with a 100% rate of cecal intubation. Among them, there were 2,137 males and 579 females. 1328 patients with colorectal polyps were selected as the observation group, 1388 individuals who showed no obvious abnormalities upon colonoscopy were selected as the control group. Among these 1328 patients with colorectal polyps, 474 patients with colorectal adenomatous polyps and 854 patients with colorectal non-adenomatous polyps were confirmed by pathological examination(As shown in Table 1).
TyG, TyG-BMI, TG/HDL-c, and METS-IR were calculated as follows8:
Inclusion and exclusion criteria
-
Inclusion criteria: (1) age between 20 and 79 years old; (2) complete examination data; (3) the patient has no serious cardiovascular, hepatic, or renal diseases, including but not limited to acute myocardial infarction, heart failure, liver cirrhosis, renal failure, or other systemic disorders such as infectious diseases or malignancies.
-
Exclusion Criteria: (1) inadequate bowel preparation or incomplete endoscopic examination; (2) complicated with severe heart, liver, kidney, or other organ diseases, coagulation disorders, and other underlying diseases or infectious diseases; (3) patients with incomplete medical history examination data; (4) patients in whom polyps were found during colonoscopy but did not undergo pathological examination; (5) patients with two or more pathological types of colorectal polyps; (6) patients have diseases that may cause elevated CEA, such as colorectal cancer, inflammatory bowel disease, lung cancer.
Basic information of study subjects
Sex, age, Body Mass Index (BMI), fasting blood glucose (FBG), systolic blood pressure (SBP), diastolic blood pressure (DBP), cholesterol, triglycerides (TG), low-density lipoprotein (LDL), high-density lipoprotein (HDL), apolipoprotein A-I (ApoA-I), apolipoprotein B (ApoB), carcinoembryonic antigen (CEA), carbohydrate antigen 199 (CA199), urea, creatinine, uric acid, free triiodothyronine (FT3), thyroid stimulating hormone (TSH), free thyroxine (FT4), alanine aminotransferase (ALT), aspartate aminotransferase (AST), total protein (TP), albumin (ALB), total bilirubin (TBIL), and indirect bilirubin (IBIL) and globulin (GLB) were collected from both study groups. Colonoscopy was used to screen whether the subjects had colorectal polyps. TyG-BMI, TyG, TG/HDL-C, METS-IR and MAP were calculated.
Colonoscopy examination
Colonoscopies were conducted by experienced gastroenterologists. Each colonoscopy withdrawal time lasted for at least 6 min to minimize the chance of missing any lesions. Detailed colonoscopy results encompassed the size, quantity, location of polyps, as well as the records of polypectomy procedures. Experienced pathologists confirmed the diagnosis of adenoma through histological examination after colonoscopic polypectomy.
Statistical analysis
Statistical analysis of the data was performed using SPSS 25.0 statistical software and GraphPad Prism 8 software. When the measurement data of the two groups followed a normal distribution, they were expressed as mean ± standard deviation (x ± s) and compared using the t-test. When the measurement data did not follow a normal distribution, they were expressed as median [M(P25, P75)] and compared using the nonparametric rank sum test. Counting data were compared using the χ2 test. Logistic regression analysis was used to analyze the influencing factors of colorectal polyps in healthy individuals undergoing physical examinations. The GraphPad Prism 8 software was used to draw Forest plot. A prediction model for the occurrence of colorectal adenomatous polyps in healthy individuals was constructed, and the receiver operating characteristic curve (ROC) was used to evaluate the predictive value of the model for the onset of colorectal polyps. A p-value < 0.05 was considered statistically significant.
Results
General characteristics of the study population
Among 1328 patients with colorectal polyps, 1088 were male and 240 were female, with a male-to-female ratio of 4.53:1. Among 1388 patients in the non-polyp group, 1049 were male and 339 were female, with a male-to-female ratio of 3.09:1. When comparing the sex ratios of the two groups using the chi-square test, there was a statistically significant difference (χ2 = 16.322, p < 0.001). It is believed that men are more prone to colorectal polyps than women (Table 2).
Sex ratios were compared between the adenomatous polyp group and the non-adenomatous polyp group. Among 474 patients with adenomatous polyps, 396 were male and 78 were female, with a male-to-female ratio of 5.07:1. Among 854 patients with non-adenomatous polyps, 692 were male and 162 were female, with a male-to-female ratio of 4.27:1. There was no statistically significant difference in sex ratios between the adenomatous polyp group and the non-adenomatous polyp group (χ2 = 1.301, p = 0.254).
As shown in Table 3, the average age of onset for patients with colorectal polyps was 50.03 ± 7.881 years old. Among them, the average age of onset for males was 50.12 ± 7.846 years old, and the average age of onset for females was 49.67 ± 8.044 years old. The incidence rate was high among people over 40 years old, accounting for 89.98% of the total population. Therefore, this study suggests that individuals over 40 years old should complete at least one colonoscopy screening.
Differences in clinical biochemical indicators and related derived indicators were compared between the two groups. All of these indicators are quantitative data. First, a normality test was performed on the data. Among them, BMI, TyG-BMI, diastolic blood pressure, MAP, total cholesterol, LDL-C, ApoB, uric acid, TP, and globulin followed a normal distribution, so t-test was used to analyze. As shown in Table 4, the results showed statistically significant differences in BMI, TyG-BMI, diastolic blood pressure, MAP, and ApoB between the polyp group and the non-polyp group. The BMI, TyG-BMI, diastolic blood pressure, MAP and ApoB were higher in the polyp group than in the non-polyp group (p < 0.05). Age, TyG, TG/HDL-C, METS-IR, FBG, SBP, TG, HDL, ApoA1, CEA, CA199, urea, blood creatinine, FT3, TSH, FT4, ALT, AST, TBIL, IBIL, ALB, alkaline phosphatase, and total bile acids exhibited skewed distribution, thus non-parametric tests were employed. The results revealed statistically significant differences in age, TyG, TG/HDL-C, METS-IR, FBG, SBP, TG, HDL-C, CEA, FT3, total bilirubin, indirect bilirubin, and ALB between the polyp and non-polyp groups (p < 0.05). Age, TyG, TG/HDL-C, METS-IR, FBG, SBP, TG, CEA, FT3, TBIL, and IBIL levels were higher in the polyp group compared to the non-polyp group, whereas HDL and ALB levels were lower in the polyp group compared to the non-polyp group.
Univariate analysis of risk factors for colorectal polyp occurrence
Sixteen factors related to the occurrence of colorectal polyps, identified through univariate analysis, were used as variables for univariate logistic regression analysis. The results showed that sex, age, BMI, FBG, TyG-BMI, TyG, TG/HDL-C, METS-IR, MAP, and CEA were risk factors for the occurrence of colorectal polyps, while HDL-C was a protective factor for the occurrence of colorectal polyps. The above results are shown in Table 5.
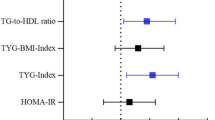
The forest plot shows that there are statistically significant differences in CEA (OR = 1.121, 95% CI: 1.008–1.248, p < 0.05) and HDL-C (OR = 0.746, 95% CI: 0.597–0.932, p < 0.05) between the adenoma group and the non-adenoma group. There are also statistically significant differences in MAP (OR = 1.013, 95% CI: 1.005–1.022, p < 0.05), METS-IR (OR = 1.797, 95% CI: 1.197–2.697, p < 0.05), TG/HDL-C (OR = 1.054, 95% CI: 1.019–1.090, p < 0.05), TyG (OR = 1.536, 95% CI: 1.120–2.108, p < 0.05), TyG-BMI (OR = 1.006, 95% CI: 1.001–1.101, p < 0.05), and Age (OR = 1.045, 95% CI: 1.035–1.056, p < 0.05) between the two groups (as show in Fig. 1).
Forest plot of comparison of factors influencing colorectal polyp occurrence.
A binary logistic regression analysis was conducted using four insulin resistance-related indicators and MAP (Table 6). The results showed that TyG, TyG-BMI, and MAP were identified as risk factors for the occurrence of colorectal polyps when analyzing these five indicators, consistent with the results of the univariate analysis. However, it was found that the p-values for TG/HDL-C and METS-IR were greater than 0.05. Considering the influence of other factors, TyG, TyG-BMI, and MAP appeared to have greater predictive value. In Models 2 and 3, after controlling for multiple factors such as sex, age, CEA, FT3, etc., MAP and TyG-BMI were found to have better predictive ability.
Area under the curve of the insulin resistance factors and other related indexes to predict the risk for colorectal polyps
ROC curve analysis results show that in the overall population, the AUC of TyG, TyG-BMI, TG/HDL-C, METS-IR, MAP, CEA, and their combination with sex and age are all greater than 0.5 (p < 0.05), indicating a certain predictive value for the occurrence and development of colorectal polyps. The descending order of AUC values for individual indicators is as follows: TyG-BMI (0.574, 95% CI 0.550–0.598), MAP (0.574, 95% CI 0.550–0.597), TyG (0.567, 95% CI 0.544–0.591), METS-IR (0.563, 95% CI 0.539–0.587), TG/HDL-C (0.561, 95% CI 0.537–0.585), CEA (0.546, 95% CI 0.522–0.570). The descending order of AUC values for combined indicators is as follows: TyG + TyG-BMI + TG/HDL-C + METS-IR + Sex + Age (0.625, 95% CI 0.602–0.648), MAP + Sex + Age (0.624, 95% CI 0.601–0.647), TyG + TyG-BMI + TG/HDL-C + METS-IR (0.577, 95% CI 0.553–0.601), as shown in Fig. 2; Table 7. Based on these results, we conclude that the combined insulin resistance-related indicators, MAP, sex, age, and CEA can better predict the occurrence of colorectal polyps.
Receiver operating characteristic (ROC) curve of makers of insulin resistance and the combination.
Discussion
According to statistics, there were over 1.9 million new cases and 935,000 deaths of colorectal cancer globally in 20209. The incidence rate of colorectal cancer ranks third, but its mortality rate ranks second. Currently, significant progress has been made in detecting colorectal cancer, but simpler and more accurate methods are still lacking. Studies have found that abnormalities in sugar and lipid metabolism also contribute to the occurrence and development of colorectal cancer10. Though previous studies indicated that diabetes increases the probability of developing colorectal polyps2,11neither of them investigated the relationship between insulin resistance indicators and colorectal polyps. In addition, there are currently no reports on the correlation between insulin resistance markers (TyG, TyG-BMI, TG/HDL-C, METS-IR) and colonic polyps. Therefore, this study retrospectively analyzed the association between insulin resistance markers and the presence of colorectal polyps in the population undergoing colonoscopy at our center. To the best of our knowledge, this is the first report on the correlation between insulin resistance markers and the incidence of colonic polyps.
Our study found that there were differences in sex, age, FPG, BMI, TyG, TyG-BMI, TG/HDL-C, METS-IR, MAP, TG, high-density lipoprotein, and CEA between the colorectal polyp group and the non-colorectal polyp group(p < 0.05). Univariate logistic regression analysis indicated that sex, age, BMI, TyG, TyG-BMI, TG/HDL-C, METS-IR, mean arterial pressure, and CEA are risk factors for the occurrence of colorectal polyps. This findings suggests that TyG, TyG-BMI, TG/HDL-C, and METS-IR could be used as indicators to predict the occurrence of colorectal polyps.
Our research results suggest that the risk of colorectal polyps increases with age. Logistic regression risk analysis indicated an OR value of 1.045, indicating that the risk of developing colorectal polyps increases by 1.045 times for each age grade increase. We found that the prevalence of colorectal polyps was 89.98% in people over 40 years old, which is consistent with a foreign study12. Therefore, it is recommended that people over 40 years old should undergo colonoscopy as soon as possible. This study found that men are more prone to colorectal polyps than women, consistent with previous reports11,13and the possible mechanisms may be related to unhealthy lifestyles such as smoking14and alcohol consumption15as well as low estrogen levels in males16.
Our research found that as the degree of insulin resistance increases, the risk of developing colorectal polyps also rises. The results of univariate logistic regression analysis showed that METS-IR had the highest risk coefficient (OR 1.797), followed by TyG (OR1.536), while TyG-BMI, TG/HDL-C, and MAP were lower ( OR 1.006, 1.054, and 1.013, respectively). Studies have indicated that insulin resistance can contribute to the occurrence of colorectal polyps17 .The mechanism behind this may be that when insulin resistance is present, elevated insulin levels in the body can promote cell mitosis and the production of insulin-like growth factors, which can inhibit the process of apoptosis when it binds to IGF-1R receptors, and thus contribute to the development of colorectal polyps. Currently, many studies have confirmed that the TyG, TyG-BMI, TG/HDL-C, and METS-IR are reliable and convenient new indicators for reflecting insulin resistance and metabolic syndrome. Previous studies have found that these new indicators of insulin resistance are associated with obesity, hypertension, metabolic syndrome, and diabetes. However, research on the relationship between these new indicators of insulin resistance and the incidence of colorectal polyps has not yet been reported in domestic and foreign studies. This study aims to clarify the predictive value of these four indicators for colorectal polyps, with the hope of providing clinicians with an effective, simple, and convenient prediction method that is easy to promote in clinical practice.
The TyG index is calculated from TG and FPG, the TyG-BMI index is obtained by multiplying BMI based on TG and FPG, the METS-IR index is calculated from FBG, TG, BMI and high-density lipoprotein. TG/HDL-C represents the ratio of TG to HDL-C. These four insulin resistance indicators have certain predictive value for the occurrence of colorectal polyps, reflecting the potential roles of BMI, FPG, TG, and HDL-C in the development and progression of colorectal polyps. They also reveal the roles of lipotoxicity and glucotoxicity in the occurrence of colorectal polyps. Studies have found that there is fat accumulation in the submucosal tissue of patients with colorectal polyps, which is considered to be related to insulin resistance. Under insulin resistance conditions, the supply of free fatty acids to the colonic and rectal tissues increases, leading to TG accumulation and lipid droplet formation18. This study also found that in the polyp group with metabolic syndrome, BMI, FPG, HOMA-IR, and DBP were all higher than those in the control group, while serum HDL-C was lower than that in the control group, which is consistent with the results of our study.
A study has found that HDL, TG, total cholesterol, and LDL are all related to the occurrence of colorectal polyps19. This study noted that in the polyp group, HDL levels are significantly lower than those in the control group, while TG, total cholesterol, and LDL levels are significantly higher. However, our findings differ from those of this study. Total cholesterol and LDL-C are not risk factors for colorectal polyps (p > 0.05). ApoB levels were significantly higher in the polyp group compared with that of the non-polyp group (p < 0.05). Other studies have also indicated that TG and ApoB are risk factors for recurrence after colorectal polyp removal20.
In our study, we found that MAP in the colorectal polyp group was significantly higher than that in the control group. Logistic regression analysis also showed that MAP is a risk factor for the occurrence of colorectal polyps. Previously, there have been few studies on the relationship between hypertension and the risk of colorectal polyps, and the conclusions were inconsistent. One study on a Chinese population found that blood pressure in the non-colorectal polyp group was lower than that in the polyp group21. Another study found that hypertension is a predictor of colorectal polyp recurrence in the elderly22. However, a study in Japan suggested that the use of antihypertensive medications may be a risk factor for colorectal polyps23.Therefore, the relationship between blood pressure levels, duration of hypertension, and the progression of colorectal polyps needs to be further investigated.
The development of colorectal cancer typically follows an evolutionary process of “polyp - adenoma - carcinogenesis”24. CEA testing provides a certain degree of diagnostic capability for polyp carcinogenesis. A study found that an elevated CEA level (> 1.435 ng/mL) is an independent risk factor for polyps25. Our research also revealed that CEA levels were significantly higher in the colorectal polyp group than.
that in the non-polyp group, suggesting a correlation between CEA and the occurrence of colorectal polyps. ROC curve analysis indicated that the optimal cutoff value for CEA is 1.22 ng/mL. In the present study, we also discovered that the predictive ability of all four insulin resistance-related indicators is stronger than that of CEA. The AUC values were even higher when considering the four insulin resistance indices in combination with age and sex. During ROC curve analysis, we also observed that MAP has a certain predictive ability. The predictive effectiveness of insulin resistance indicators and MAP for the occurrence of colorectal polyps is higher than that of CEA, and the predictive efficacy of combining insulin resistance indicators and MAP with sex and age was even higher. This suggests that while indicators of insulin resistance are useful for risk stratification, they should be combined with other clinical indicators such as blood pressure, sex, age, and CEA. This further illustrates that using simple and convenient insulin resistance indicators, along with consideration of blood pressure, may more accurately reflect the probability of colorectal polyp occurrence. Therefore, a comprehensive consideration of factors such as sex, age, CEA, blood pressure, dyslipidemia, and insulin resistance provides even more convincing evidence.
There are shortcomings in our study. Firstly, our study belongs to a single-center retrospective study, this study is mainly based on healthy people physical examination of the population. Due to the high health literacy samples, so it is easy to bias the results of the study, and it is necessary to further expand the scope of the study, and to unite more centers to carry out the study. Secondly, due to the observational cross-sectional design employed in this study, its inherent limitations restrict the ability to infer causal relationships between of insulin resistance markers and colorectal polyps. Thirdly, some genetic factors promote the development of colorectal polyps, such as the hereditary mixed polyposis syndrome, Lynch syndrome, etc26. Some studies have found that sugar and sugar-containing soft drink consumption in adulthood is not associated with the risk of adenomas27 and carbohydrate intake is not associated with the risk of colorectal polyps28. However, some studies have also shown dietary polyamine exposure increased odds of colorectal adenoma29,and higher flavonol intakes were inversely associated with high risk adenoma recurren30. Therefore, we will expand the sample size and collect dietary and genetic information in future studies to rule out the influence of these two factors on the results. The current findings could be regarded as preliminary exploratory discoveries, and further longitudinal cohort study is needed to validate the causality between the two by tracking changes in insulin resistance marker levels and the temporal relationship with the occurrence/development of colorectal polyps.
In summary, middle-aged and elderly individuals, males, those with high blood pressure, insulin resistance, dyslipidemia (especially high TG and low HDL), and elevated CEA levels are independent risk factors for the onset of colorectal polyps. A risk prediction model constructed based on these factors can effectively predict individuals at risk of developing colorectal polyps. By combining colonoscopy with other screening methods, the occurrence of colorectal polyps can be effectively screened and prevented, thereby minimizing the risk of colorectal cancer.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Mareth, K., Gurm, H. & Madhoun, M. F. Endoscopic recognition and classification of colorectal polyps. Gastrointest. Endosc. Clin. N. Am. 32, 227–240. https://doi.org/10.1016/j.giec.2021.12.003 (2022).
Xu, J., He, W., Zhang, N., Sang, N. & Zhao, J. Risk factors and correlation of colorectal polyps with type 2 diabetes mellitus. Annals Palliat. Med. 11, 647–654. https://doi.org/10.21037/apm-21-3943 (2022).
Yoon, Y. S., Keum, N., Zhang, X., Cho, E. & Giovannucci, E. L. Hyperinsulinemia, insulin resistance and colorectal adenomas: A meta-analysis. Metab. Clin. Exp. 64, 1324–1333. https://doi.org/10.1016/j.metabol.2015.06.013 (2015).
Zeng, P., Cai, X., Yu, X., Huang, L. & Chen, X. HOMA-IR is an effective biomarker of non-alcoholic fatty liver disease in non-diabetic population. J. Int. Med. Res. 51, 3000605231204462. https://doi.org/10.1177/03000605231204462 (2023).
Rokicka, D. et al. The prognostic impact of insulin resistance surrogates in patients with acute myocardial infarction with and without type 2 diabetes. Cardiovasc. Diabetol. 23, 147. https://doi.org/10.1186/s12933-024-02240-z (2024).
Son, M., Moon, S. Y., Koh, M., Kang, Y. & Lee, J. Y. Association between surrogate markers of insulin resistance and the incidence of colorectal Cancer in korea: A nationwide Population-Based study. J. Clin. Med. https://doi.org/10.3390/jcm13061628 (2024).
Qin, M. et al. Relationship between insulin resistance, serum VCAM-1, FGF19, IGF-1 and colorectal polyps. Zhonghua Zhong Liu Za Zhi [Chinese J. Oncology]. 43, 553–562. https://doi.org/10.3760/cma.j.cn112152-20210219-00146 (2021).
Zeng, P., Cai, X., Yu, X. & Gong, L. Markers of insulin resistance associated with non-alcoholic fatty liver disease in non-diabetic population. Sci. Rep. 13, 20470. https://doi.org/10.1038/s41598-023-47269-4 (2023).
Sung, H. et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Goodarzi, G. et al. Metabolic phenotypes and risk of colorectal cancer: a systematic review and meta-analysis of cohort studies. BMC cancer. 22, 89. https://doi.org/10.1186/s12885-021-09149-w (2022).
Sninsky, J. A., Shore, B. M., Lupu, G. V. & Crockett, S. D. Risk factors for colorectal polyps and Cancer. Gastrointest. Endosc. Clin. N. Am. 32, 195–213. https://doi.org/10.1016/j.giec.2021.12.008 (2022).
Ejtehadi, F. et al. Prevalence of colonic polyps detected by colonoscopy in symptomatic patients and comparison between different age groups. What age should be considered for investigation?? Polski Przeglad Chirurgiczny. 96, 15–21. https://doi.org/10.5604/01.3001.0053.3997 (2023).
Jeong, Y. H. et al. Risk factors of advanced adenoma in small and diminutive colorectal polyp. J. Korean Med. Sci. 31, 1426–1430. https://doi.org/10.3346/jkms.2016.31.9.1426 (2016).
Hoffmeister, M. et al. Male sex and smoking have a larger impact on the prevalence of colorectal neoplasia than family history of colorectal cancer. Clin. Gastroenterol. Hepatol. 8, 870–876 (2010).
Williams, C., DiLeo, A., Niv, Y. & Gustafsson, J. Estrogen receptor beta as target for colorectal cancer prevention. Cancer Lett. 372, 48–56. https://doi.org/10.1016/j.canlet.2015.12.009 (2016).
Stevanato Filho, P. R. et al. Estrogen receptor β as a prognostic marker of tumor progression in colorectal Cancer with Familial adenomatous polyposis and sporadic polyps. Pathol. Oncol. Res. 24, 533–540. https://doi.org/10.1007/s12253-017-0268-5 (2018).
Shin, H. S. & Cho, Y. J. Insulin levels are associated with risk of colon adenoma and not nonadenomatous polyps: A retrospective, hospital-based study. Medicine 101, e30200. https://doi.org/10.1097/md.0000000000030200 (2022).
Wada, S. et al. Submucosal fat accumulation in human colorectal tissue and its association with abdominal obesity and insulin resistance. United Eur. Gastroenterol. J. 6, 1065–1073. https://doi.org/10.1177/2050640618766926 (2018).
Wang, X. et al. The relationship between serum lipid levels and colorectal serrated lesions: A systematic review and meta-analysis. Front. Physiol. 13, 984586. https://doi.org/10.3389/fphys.2022.984586 (2022).
Du, J. Y. et al. High levels of triglycerides, Apolipoprotein B, and the number of colorectal polyps are risk factors for colorectal polyp recurrence after endoscopic resection: a retrospective study. J. Gastrointest. Oncol. 13, 1753–1760. https://doi.org/10.21037/jgo-22-491 (2022).
Liu, C. S. et al. Central obesity and atherogenic dyslipidemia in metabolic syndrome are associated with increased risk for colorectal adenoma in a Chinese population. BMC Gastroenterol. 10, 51. https://doi.org/10.1186/1471-230x-10-51 (2010).
Saiken, A. & Gu, F. Lifestyle and lifestyle-related comorbidities independently associated with colorectal adenoma recurrence in elderly Chinese people. Clin. Interv. Aging. 11, 801–805. https://doi.org/10.2147/cia.s105472 (2016).
Watanabe, Y. et al. Association between colorectal polyps and hypertension treatment. J. Dig. Dis. 16, 649–655. https://doi.org/10.1111/1751-2980.12289 (2015).
Morson, B. C. Evolution of cancer of the colon and rectum. Cancer 34, 845–849 (1974).
Zhang, C. et al. Predictors for colorectal polyps in an asymptomatic population undergoing medical Check-ups. Surgical laparoscopy. Endoscopy Percutaneous Techniques. 33, 108–114. https://doi.org/10.1097/sle.0000000000001152 (2023).
Valle, L. & Monahan, K. J. Genetic predisposition to Gastrointestinal polyposis: syndromes, tumour features, genetic testing, and clinical management. Lancet Gastroenterol. Hepatol. 9(1), 68–82. https://doi.org/10.1016/S2468-1253(23)00240-6 (2024).
Joh, H. K. et al. Simple sugar and sugar-Sweetened beverage intake during adolescence and risk of colorectal Cancer precursors. Gastroenterology 161(1), 128–142e20. https://doi.org/10.1053/j.gastro.2021.03.028 (2021).
Coleman, H. G., Ness, R. M., Smalley, W. E., Zheng, W. & Shrubsole, M. J. Aspects of dietary carbohydrate intake are not related to risk of colorectal polyps in the Tennessee colorectal polyp study. Cancer Causes Control. 26(8), 1197–1202. https://doi.org/10.1007/s10552-015-0605-5 (2015).
Vargas, A. J. et al. Dietary polyamine intake and risk of colorectal adenomatous polyps. Am. J. Clin. Nutr. 96(1), 133–141. https://doi.org/10.3945/ajcn.111.030353 (2012).
Bobe, G. et al. Interleukin-6 as a potential indicator for prevention of high-risk adenoma recurrence by dietary flavonols in the polyp prevention trial. Cancer Prev. Res. (Phila). 3(6), 764–775. https://doi.org/10.1158/1940-6207.CAPR-09-0161 (2010).
Author information
Authors and Affiliations
Contributions
All authors contributed to conceptualizing, drafting, and revising the manuscript. All authors have read and agreed to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All experiments were approved and carried out following the guidelines of the Ethics Committee of Guangzhou Cadre Health Management Center (approval number: JGZX-2024-01). All methods were performed following the relevant guidelines and regulations. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, Y., He, H., Ye, Z. et al. Association of incidence of colorectal polyp with markers of insulin resistance and other related factors: a cross-sectional study. Sci Rep 15, 23969 (2025). https://doi.org/10.1038/s41598-025-08505-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08505-1