Abstract
Tomato (Solanum lycopersicum L.) is a globally important horticultural crop; however, inconsistent germination and weak early seedling vigour remain major bottlenecks to productivity. Seed nanopriming using zinc oxide nanoparticles (ZnO NPs) represents an emerging, eco-compatible strategy for enhancing seed performance by modulating early biochemical and physiological processes. This study investigated the influence of ZnO NPs seed priming on seed quality traits and physio-biochemical responses in three genetically distinct tomato varieties: Pusa Rohini, H-81, and Pusa Prasanskrit. The optimal concentration (50ppm) of ZnO NPs significantly enhanced germination percentage, speed of germination, seedling vigour indices, shoot and root elongation, and biomass accumulation while reducing mean germination time. Biochemically, ZnO nano-primed seedlings exhibited elevated levels of total chlorophyll, proline, and total phenol along with enhanced antioxidant enzyme activities (superoxide dismutase, catalase and peroxidase). A concurrent reduction in malondialdehyde content indicated lower lipid peroxidation and oxidative damage. These effects collectively suggest improved homeostasis and metabolic efficiency. The findings confirm that 50ppm ZnO nanopriming modulates germination-linked signaling and antioxidant defense pathways, accelerating early seedling establishment and enhancing seed quality through improved enzyme activity and seedling development in tomato, while offering a cost effective and environmentally sustainable approach.
Similar content being viewed by others
Introduction
Tomato (Solanum lycopersicum L.) is a highly significant vegetable crop globally, valued for its easy cultivation and rapid growth cycle. Apart from its economic importance as a widely consumed vegetable, it is rich in essential minerals, vitamins (both common and nutraceuticals), and exhibit anticancer properties1,2. Furthermore, tomato plays a crucial role in ensuring food security and generating income. It serves as a valuable resource for studying plant physiology, biochemistry, genetics, genomics, and breeding to enhance desirable agronomic characteristics, such as stress tolerance3.
Germination and seedling establishment are pivotal stages in tomato as it is crucial for plant development and species expansion into new environments4,5. Additionally, rapid and uniform germination is vital for enhancing tomato crop yield and quality, holding significant economic and environmental value in horticulture6 as it facilitates increased automation, simplified weed management, disease prevention, and effective adaptation to environmental stresses. To address issues of low and inconsistent seed germination, seed priming can be employed as a seed enhancement technique7. This pre-germination approach involves partial seed hydration followed by drying to regulate metabolic processes before actual germination, ensuring accelerated, uniform, and synchronized germination. Additionally, it enhances seedling vigour and growth in diverse set of environmental conditions8.
Nanotechnology has advanced agriculture by introducing nanoparticles such as Zn, Ag, Ti, Si, Cu, and Fe, which exhibit unique properties at the nanoscale (1–100 nm) that differ from bulk materials. These NPs are explored as nanofertilizers and nanopesticides to enhance nutrient uptake and crop productivity sustainably9,10. The concept of seed nano-priming introduces an innovative method involving the treatment of seeds with nanoparticles11. Although still an emerging field of research, nanotechnology-based seed priming has shown promising initial results12,13.
Zinc (Zn) is a vital micronutrient involved in enzyme activation14 and protein synthesis and is essential for normal metabolic functions15. ZnO nanoparticles are considered sustainable and cost effective for seed priming due to their small size and high surface area, which facilitate enhanced nutrient absorption and plant growth with minimal environmental impact16.
Although ZnO nanopriming has shown promising results in crops like maize17 radish 18, and cucumber19 etc., recent studies have begun exploring its effect in tomatoes as well. Recent research highlights that the size and concentration of ZnO NPs influence their efficacy in seed priming. For example, Smaller ZnO NPs showed more beneficial effects on tomato seed germination parameters than larger particles with responses varying among cultivars, indicating the importance of cultivar specific optimization20. Additionally, while ZnO NPs can enhance physiological processes such as photosynthesis, nutrient uptake, and root architecture in tomato plants, potential toxicity and its environmental risks must be considered. Some studies report transient toxicity effects on soil microbial biomass at higher ZnO concentrations, suggesting careful management is necessary to balance benefits and risks21,22.
This study aims to address the gap23 in understanding the optimal use of ZnO NPs for seed priming in tomatoes by evaluating their effects on seed quality and physiological-biochemical responses across selected tomato varieties (Pusa Rohini, H-81 and Pusa Prasanskrit). These varieties were chosen due to their agronomic importance and differential responses to environmental stresses, providing insights into the cultivar-specific benefits and mechanisms of ZnO NPs priming. The novelty of this research lies in its focus on optimizing ZnO NPs concentration and particle size for seed priming in these varieties, contributing to sustainable crop productivity enhancement while considering potential risks and mechanistic understanding of nanopriming effects.
Furthermore, unlike prior research, the present study integrates morphological, physiological, and biochemical analyses to provide a comprehensive understanding of how ZnO nanopriming influences early stage tomato growth. This multidimensional evaluation contributes to the novelty of the study and positions it as a significant advancement over earlier findings. While the agricultural applications of nanotechnology have shown clear benefits, it is equally important to consider associated risks23. Nanoparticles, especially in excess, can accumulate in plant tissues or soil and cause phytotoxicity or ecological disturbances. To ensure sustainable adoption, this study also acknowledges these potential risks and emphasizes the need for concentration optimization. Thus, the current study not only investigates the agronomic potential of ZnO nanopriming in tomatoes but also provides a fair and knowledgeable viewpoint, ultimately aiding in the development of safer and more effective nano-enabled seed quality enhancement technique.
Materials and methods
Experimental site and seed material
The present research was conducted during 2023-24 at the Division of Seed Science and Technology and the Division of Agricultural Chemicals, ICAR-Indian Agricultural Research Institute (IARI), New Delhi, India. Seeds of three tomato varieties-Pusa Rohini, H-81, and Pusa Prasanskrit were procured from the Division of Vegetable Science, ICAR-IARI, New Delhi. These varieties were selected based on their genotypic variability, agronomic importance, and diverse agroecological zones. Tomato variety namely, Pusa Rohini was used for the optimization of ZnO concentration for priming, while H-81 and Pusa Prasanskrit alongside Pusa Rohini were used for validation.
Synthesis of ZnO nanoparticles
Zinc oxide nanoparticles were synthesized using a solvothermal method with slight modifications as described by Bian et al.24. A 75 ml solution of 0.1 M zinc acetate was prepared by dissolving 1.65 g of zinc acetate in methanol, stirred at 60oC for 45 min at 200 rpm. Separately, a 150 ml solution of 1 M potassium hydroxide (KOH) was prepared by dissolving 8.4 g of KOH in methanol, also stirred for 45 min. The KOH solution was added dropwise into the zinc acetate solution under continuous stirring at 650 rpm and 60oC for 90 min, followed by an additional 2 h of stirring. The resulting precipitate was allowed to settle overnight, then filtered through vacuum filtration, washed three times with distilled water and ethanol, and dried in a hot air oven at 70oC. To ensure purity and avoid contamination, analytical grade reagents were used and multiple wash steps were performed to eliminate unreacted precursors.
Characterization of ZnO nanoparticles
The synthesized ZnO nanoparticles (NPs) were characterized for their structural and morphological properties. X-ray diffraction (XRD) was performed using a Rigaku Miniflex X-ray benchtop diffractometer to confirm the crystalline structure. Dynamic Light Scattering (DLS) was employed to measure the hydrodynamic diameter of the particles, with measurements conducted using a Zetasizer Nano ZS90 (version 7.11) (Malvern Instruments, UK). Transmission Electron Microscopy (TEM) was used to observe particle morphology and size using a FEI TECNAI TF20 microscope operated at 200 keV.
Preparation of nanoparticle solution
A stock solution was prepared by dispersing the nanoparticles in double distilled water to achieve a concentration of 1000 mg/l in a 100 ml conical flask. From the stock, working solutions with concentrations of 25, 50, 100, 250, 500, 750 and 1000ppm were prepared through serial dilution. To ensure homogeneous dispersion of nanoparticles, the solutions were ultrasonicated for 20 min at a frequency of 40 kHz, using a probe sonicator with an energy for input of 8400 J.
Priming of seeds
Tomato seeds (variety Pusa Rohini) were primed using the ZnO nanoparticle solutions at the specified concentrations. The priming process was carried out by maintaining a fixed seed to solution ratio of 1:2, with an optimized soaking duration of 24 h25. Following priming, the nanoparticle solutions were drained, and the seeds were shade-dried at room temperature, along with hydro-primed counterparts. Unprimed seeds served as the control. The experimental setup followed a Completely Randomized Design (CRD) with three replications. The treatment showing the most promising results was selected for further validation on seed quality and physio-biochemical parameters in additional tomato varieties- H-81, Pusa Prasanskrit.
Estimation of germination percentage (%)
The germination test was conducted using the method as per ISTA26 with minor modifications. Fifty seeds in three replications were placed on moist germination paper, and then rolled towels were incubated at 25 ± 1 °C and 90 ± 2% relative humidity. After 14 days, the final count was taken, and seedlings were categorized as normal or abnormal, following the criteria in the “Germination Test” chapter of ISTA (International Rules for Seed Testing). The germination percentage was calculated using the formula: Germination (%) = (Number of normal seedlings observed/ Total number of seeds) × 100. On the 14th day of the test, ten normal seedlings per replication were randomly selected for measurement of root length, shoot length, and total seedling length, with averages calculated and expressed in centimeters. Additionally, the seedlings chosen were dried in a hot air oven at 60 °C for 48 h, and the dry weight of the seedlings was recorded as grams per 10 seedlings.
Estimation of seedling vigour index I and II
Seedling vigour indices were calculated following the method proposed by Abdul-Baki and Anderson27. The formulas used are as follows:
Mean germination time and speed of germination
From the second day onward, seeds with visible radicle emergence (≥ 2 mm) were recorded daily until the final count. Mean Germination Time (MGT) was calculated using the formula proposed by Ellis and Roberts28: MGT= ∑ (Dn)/ ∑(n), where D represents the number of days from the start of germination, and n is the number of seeds germinated on day D. Speed of Germination (SOG) was calculated according to Maguire’s29 formula.
X1—number of seeds germinated at first count. X2—number of seeds germinated at second count. Xn—number of seeds germinated on nth count. Y1—Number of days from sowing to the first count. Y2—number of days from sowing to the second count. Yn—number of days from sowing to nth count.
Seedling emergence percentage (%)
The most effective treatment from laboratory trials was tested under pot conditions to evaluate seedling emergence. On the 15th day after sowing, the number of emerged seedlings was counted, and the emergence percentage was calculated using the formula: (Number of emerged seedlings/ Total number of seeds sown) × 100.
According to ISTA guidelines, the final count of germination was taken on 14th day. Those fourteen-day-old seedlings were then utilized for subsequent physio-biochemical analysis.
Estimation of malondialdehyde content
The malondialdehyde (MDA) content was determined using the method described by Heath and Packer30. Seedling samples were macerated with 0.1% trichloroacetic acid (TCA) and then centrifuged to obtain the supernatant. Then, 0.5 ml of supernatant was mixed with 2 ml of 0.65% thiobarbituric acid solution containing 20% TCA, followed by incubation at 95°C for 30 min. After centrifugation, absorbance was measured at 532 nm and 600 nm, and MDA content was calculated using an absorption coefficient of 155 mmol−1 cm−1.
Estimation of total chlorophyll content
For the determination of photosynthetic pigments, the total chlorophyll content was assessed according to the method outlined by Arnon31. Leaf samples were macerated in 80% acetone and centrifuged, and the optical density of the supernatant was measured at 645 nm and 663 nm using a spectrophotometer. Chlorophyll content was expressed in mg/g of fresh leaf and calculated using the formula,
Where V is the volume of extract and W is the weight of leaf tissue.
Estimation of proline content
Proline content in seedling samples was assessed using the method by Bates et al.32. Seedlings were macerated with 3% sulfosalicylic acid, then centrifuged to obtain a supernatant. The supernatant was mixed with glacial acetic acid and acid ninhydrin, incubated at 95 °C for 1 h, and cooled. Color extraction was performed using 2 ml of toluene, followed by measurement of absorbance at 520 nm using a spectrophotometer. Proline concentration was determined using a standard curve based on known proline concentrations.
Estimation of total phenol content
Total phenol content was determined using Folin-Ciocalteu reagent (FCR) as per Singleton and Rossi’s33 method. Seedling samples were macerated with 80% ethanol, and after centrifugation, a reaction mixture of sample supernatant, FCR, and NaHCO3 was incubated at 45 °C for 45 min and absorbance was measured at 765 nm. The phenolic content was quantified using a gallic acid standard curve and expressed as mg of gallic acid equivalent (GAE) per gram fresh weight.
Estimation of antioxidant enzyme activity
For catalase and peroxidase activity analysis, seedling samples were macerated in 0.1 M potassium phosphate buffer (pH 7.8) and centrifuged at 15,000 rpm for 20 min. Catalase activity was analysed by mixing enzyme extract with 0.1 M potassium phosphate buffer (pH 7), H2O2, and water, measuring absorbance at 240 nm over 3 min34. Peroxidase activity was determined by combining enzyme extract with 0.1 M potassium phosphate buffer (pH 6.1), H2O2, guaiacol and water, and measuring absorbance at 470 nm over 3 min35 activities were expressed as units per minute per gram of fresh weight.
Superoxide dismutase (SOD) activity was assessed using the method described by Beauchamp and Fridovich36. Seedling samples were macerated in 50 mM potassium phosphate buffer (pH 7) and centrifuged at 13,000 rpm for 30 min. A reaction mixture containing extract, potassium phosphate buffer (pH 7.8), methionine, nitro blue tetrazolium, EDTA disodium salt, and riboflavin was placed 30 cm below a light bank consisting of two 15 W fluorescent tubes for 10 min. Subsequently, the light was turned off, and the test tubes were covered with a black cloth. The optical density was measured at 560 nm using a spectrophotometer.
Statistical analysis
The mean values for all treatments were calculated based on three replications, and these mean values were used for data analysis. The data analysis was conducted using the software SPSS version 17, following a completely randomized design (CRD). Significant differences among the means were assessed at a significance level of P ≤ 0.05. The calculation of critical difference (CD) and Tukey’s Honest Significant Difference (HSD) test (P = 0.05) was used to compare treatment means. Correlation analysis between seed quality and biochemical parameters was also performed and Pearson’s correlation coefficients were calculated.
Results
Characterization of ZnO nanoparticles
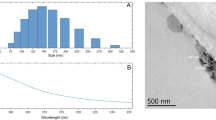
The XRD pattern of the synthesized ZnO nanoparticles (NPs) indicated a high purity wurtzite crystalline structure, as evidenced by the intense and narrow diffraction peaks (Fig. 1a). This observation was further confirmed by comparing the experimental XRD pattern with the standard pattern of ZnO (JCPDS 36-1451). Dynamic Light Scattering (DLS) method was employed to measure the hydrodynamic size of the zinc oxide nanoparticles. The Z-average hydrodynamic diameter of ZnO NPs was observed as 96.5 nm, with a polydispersity index (PDI) of 0.323 (Fig. 1b). The morphological features and size of the synthesized ZnO nanoparticles (NPs) were analyzed using Transmission Electron Microscopy (TEM) (Fig. 1c). The measurements revealed that the diameter of the individual ZnO nanoparticle ranges from 20 to 50 nm.
Characterization results of synthesized ZnO nanoparticles: a X-ray diffraction (XRD) profile indicating phase purity and crystallinity, b Dynamic light scattering (DLS) results showing particle size distribution, and c transmission electron microscopy (TEM) images revealing nanoparticle morphology and size.
Optimization of ZnO nanoparticle concentration for tomato seed priming
Among the various concentrations tested, seed priming with 50ppm ZnO nanoparticles showed significant improvements in seed quality parameters of Pusa Rohini. Compared to hydro-primed and unprimed seeds, ZnO @50ppm enhanced germination percentage, root length and shoot length, total seedling length, seedling dry weight, and seedling vigour index I and II. Additionally, the speed of germination increased while the mean germination time decreased, confirming faster and more uniform germination (Table 1).
Validation of optimized concentration of ZnO nanoparticle priming on seed quality and physio-biochemical responses
ZnO nanopriming improves germination percentage across varieties
Validation across varieties showed significantly higher germination in ZnO@50ppm nanoprimed seeds (Fig. 2a). The average increase in germination percentage was 14.8% and 25.2% over hydroprimed and uprimed seeds, respectively. The effect was more prominent in Pusa Prasanskrit, followed by H-81 and Pusa Rohini.
Effect of ZnO nanoparticles priming (50ppm) on seed quality attributes of tomato compared to hydroprimed and unprimed seeds: a germination (%); b shoot length; c root length and d seedling length in three different varieties. Data presented as mean ± SE (n = 3). Bars with different lowercase letters indicate significant differences among treatment means (p < 0.05, Tukey’s HSD).
Seedling growth and vigour are enhanced by ZnO nanopriming
Seedling growth parameters including shoot length, root length, and total seedling length were significantly higher in ZnO@50ppm nanoprimed seeds across all varieties. Compared to hydroprimed and unprimed seeds, mean shoot length increased by 14% and 25.9%, root length by 8.7% and 15.9%, and seedling length by 9.9% and 18.2%, respectively (Fig. 2b–d). Additionally, seedling vigour improved markedly, with seedling vigour index I increased by 26.4% and 47%, and seedling vigour index II increased by 42.8% and 75.3% compared to hydro-primed and unprimed seeds, respectively (Fig. 3a–b).
Effect of ZnO nanoparticles priming (50 ppm) on seed quality attributes of tomato compared to hydroprimed and unprimed seeds: a seedling vigour index I; b seedling vigour index II; c mean germination time (days) and d speed of germination (days) in three different varieties. Data presented as mean ± SE (n = 3). Bars with different lowercase letters indicate significant differences among treatment means (p < 0.05, Tukey’s HSD).
ZnO nanopriming accelerates germination and seedling emergence
The results demonstrated that seed priming with 50ppm ZnO nanoparticles significantly reduced mean germination time while increasing speed of germination and seedling emergence. Specifically, mean germination time decreased by 9.9% and 16.3%, speed of germination increased by 13.7% and 31.1%, (Fig. 3c, d) and seedling emergence improved by 6.4% and 9% over hydroprimed and unprimed seeds, respectively (Fig. 4).
Effect of ZnO nanoparticles priming (50 ppm) on seed quality attributes of tomato compared to hydroprimed and unprimed seeds: a mean seedling emergence percentage; b seedling emergence in tomato variety H-81. Trendline shows the increase of seedling emergence percentage after nanopriming as compared to the control. Data presented as mean ± SE (n = 3). Bars with different lowercase letters indicate significant differences among treatment means (p < 0.05, Tukey’s HSD).
ZnO nanopriming enhances chlorophyll, phenol and proline content
ZnO nanopriming at a concentration of 50 ppm significantly enhanced key biochemical parameters in tomato seedlings. Total chlorophyll content increased by 28.6% and 47.9% compared to hydroprimed and unprimed seeds, respectively, indicating improved photosynthetic capacity. Similarly, total phenol and proline contents were markedly elevated in ZnO-primed seeds, with phenol increasing by 27.7% and 56.3% and proline by 15.6% and 32.5% relative to hydroprimed and unprimed controls. These enhancements contribute to better stress tolerance and seedling vigour (Fig. 5a–c).
Effect of ZnO nanoparticles priming (50 ppm) on physio-biochemical responses in tomato: a total chlorophyll (mg/gFW); b total phenol (mg of GAE/gFW); c proline (µmoles/gFW) and d malondialdehyde content (nmol/mg FW) in three different varieties. Data presented as mean ± SE (n = 3). Bars with different lowercase letters indicate significant differences among treatment means (p < 0.05, Tukey’s HSD).
Nanopriming reduces malondialdehyde (MDA) content in tomato
ZnO@50ppm primed seeds showed a significant decrease in malondialdehyde content, a measure of lipid peroxidation, with average decreases of 11.4% and 20.8% when compared to hydroprimed and unprimed controls. Pusa Prsanskrit and Pusa Rohini showed the highest declines (Fig. 5d).
ZnO nanopriming boosts antioxidant enzyme activities
The seeds treated with ZnO@50 ppm exhibited significantly higher levels of catalase, peroxidase, and superoxide dismutase activities. Compared to hydro-primed and unprimed seeds, nano-primed seeds at a 50 ppm concentration showed increases in catalase activity by 11.7% and 22%, peroxidase activity by 33.7% and 64.8%, and superoxide dismutase content by 14.7% and 33.1%, respectively. Pusa Prasanskrit exhibited the strongest enzymatic responses, but these increases were consistently seen in all three tomato varieties (Fig. 6a–c).
Effect of ZnO nanoparticles priming (50ppm) on biochemical responses in tomato: a catalase (units/min/gFW); b peroxidase (units/min/gFW) and c superoxide dismutase (units/ml/gFW) in three different varieties of tomato. Data presented as mean ± SE (n = 3). Bars with different lowercase letters indicate significant differences among treatment means (p < 0.05, Tukey’s HSD).
Correlation studies of seed quality and biochemical parameters of ZnO nanoparticles primed seeds
Correlation studies of seed quality and biochemical parameters indicate that seed quality parameters such as germination percentage, shoot length, root length, total seedling length, seed vigour index I and II, speed of germination and seedling emergence percentage showed a significantly positive relationship with biochemical parameters such as proline, total phenol, total chlorophyll content and anti-oxidative enzymes such as catalase, peroxidase and super oxide dismutase and negative correlation with malondialdehyde content. In contrast mean germination time showed a significantly positive relationship with malondialdehyde and a negative relationship with all other studied biochemical parameters (Fig. 7).
Correlation coefficients of seed quality and biochemical parameters of ZnO nanoparticle primed seeds.
Discussion
Seed germination and early seedling establishment are crucial stages of plant development, as they improve a plant’s ability to withstand biotic and abiotic stresses. Seed priming is a commonly used strategy to enhance germination and seedling emergence because it accelerates the metabolic processes, which leads to rapid and uniform seedling establishment under field conditions37. This technique has been shown to improve nutrient uptake, water use efficiency, and overall plant performance, potentially resulting in enhanced crop productivity and yield38. Because of the ability to improve the physio-biochemical characteristics and seed quality of various crops, zinc oxide nanoparticles have recently drawn attention.
The results of this study indicates that seed priming with zinc oxide nanoparticles (ZnO NPs) led to a significant increase in germination percentage, speed of germination, root and shoot length, seedling vigour indices, and a reduction in mean germination time compared to both hydro primed and unprimed seeds. These improvements can be attributed to the essential role of zinc (Zn) in a series of biochemical transformations that initiate germination. Such transformations include the breaking dormancy, hydrolysis of inhibitors, optimal production of reactive oxygen species (ROS), facilitation of imbibition, and activation of germination related enzymes. Collectively, these processes contribute to the improved germination performance observed in ZnO NP treated seeds39,40. Sharma et al.13 reported that the rice seeds treated with ZnO nanopriming at 20 ppm and 40 ppm exhibited faster germination and earlier radicle protrusion compared to hydro primed seeds. Similar enhancements in germination and seedling vigour have been observed in wheat and maize following ZnO nanoparticle application, largely attributed to increased amylase activity41. Kumar et al.42 found 250ppm ZnO NPs to be optimal for improving germination in chickpea.
The observed increase in radicle and plumule length inthis study may result from zinc’s influence on the biosynthesis of growth hormones such as auxins and gibberellins, which regulate early seedling development43,44. Additionally, zinc’s involvement in carbohydrate and protein metabolism plays a vital role in synchronized germination when used in priming treatments45. Increased activity of indole-3-acetic acid (IAA) likely supports cell division and elongation, leading to longer seedlings at appropriate nanoparticle concentrations. However, as previously reported by Prasad et al.44excessive concentrations of zinc oxide nanoparticles may have phytotoxic effects, including a reduction in plant height due to metabolic disruptions. This study supports earlier findings that low doses of zinc oxide nanoparticles stimulate seedling growth, while higher concentrations can delay development, as noted in chickpea46. Similar beneficial effects on root growth have been observed in peas and rice under optimized ZnO NP treatment47.
In this study, seedling dry matter production was notably higher in ZnO-primed seeds. Similar trends have been reported in rice, where ZnO nanopriming increased fresh and dry weight by 55% and 27%, respectively12 as well as in wheat and maize41. ZnO nanoparticles have been shown to significantly improve root biomass, likely due to zinc’s role in photosynthetic activity, cellular division, and overall growth regulation48. The improvements in seedling length, dry weight, and germination collectively contribute to higher seedling vigour indices I and II, which reflect seedling robustness and establishment potential under diverse field conditions.
Chlorophyll content, a key regulator of photosynthetic efficiency and plant vitality, was significantly elevated in ZnO primed tomato seedlings compared to controls. These results suggest that ZnO nanoparticles may enhance chlorophyll biosynthesis and photochemical performance. Similar trends were reported in maize41and wheat49. This improvement may be linked to the ability of ZnO NPs to enhance the functionality of the water-splitting complex at the donor side of photosystem II (PS II), increasing the number of active reaction centers per chlorophyll molecule. Consequently, light absorption, energy trapping efficiency, and electron transport are improved, thereby supporting overall photosynthetic performance in nanoprimed seedlings12. Proline, a non-essential amino acid, plays multiple protective roles in plants under stress by functioning as an osmoprotectant, a metal chelator, antioxidant, and signaling molecule. In this study, ZnO nanoparticle priming led to increased proline accumulation, this aligns with the findings of Salehi et al.50 in bean and Mirakhorli et al.51 in soybean. This elevation in proline may be due to zinc’s role as a cofactor for the enzyme pyrroline-5-carboxylase synthase (P5CS), a key enzyme in the proline biosynthetic pathway52. Enhanced P5CS activity in the presence of Zn likely contributes to greater proline accumulation, supporting improved stress tolerance and redox regulation in nanoprimed seeds.
Phenolic compounds, another group of protective secondary metabolites, were also significantly increased in ZnO nanoprimed seeds. These compounds contribute to plant defense by scavenging reactive oxygen species (ROS) and reinforcing cell walls. The present results are in agreement with findings in Beta vulgaris and turmeric, where ZnO nanoparticles at 10 mg/l enhanced total phenolic content53. Such biochemical improvements indicate the activation of stress response pathways even under nonstress conditions.
The activities of key antioxidant enzymes-superoxide dismutase (SOD), catalase (CAT), and peroxidase (POX) were significantly elevated in ZnO nanoprimed treatments. The key agents for lowering oxidative damage under stressful conditions are antioxidant enzymes54. Enhanced enzyme activities observed in this study align with earlier reports by Srivastav et al.41 who noted improved antioxidant profiles in wheat and maize following ZnO NP treatment. Amooaghaie et al.55 reported increased SOD activity in nano Zn treated wheat, while Rai Kalal and Jajoo12 found that the CAT activity improved by 47% and 55% with ZnO NPs and ZnSO4, respectively. Similar results were reported in Hordeum vulgare and lupine56,57. Zinc oxide nanoparticle enhance antioxidant enzyme activity in seedlings by releasing Zn2+ ions, which acts as essential cofactors and improve the structural ability and catalytic function58. Also it upregulates genes and transcription factors related to the antioxidant enzymes, thereby strengthening the plant’s response to oxidative stress59.
Malondialdehyde (MDA), a biomarker of lipid peroxidation and oxidative membrane damage, was significantly reduced in ZnO primed seedlings relative to unprimed controls. This reduction suggests a protective effect against oxidative stress, likely due to enhanced enzymatic ROS scavenging. These findings are consistent with those of Burman et al.46 who reported higher MDA levels in Zn deficient chickpeas, whereas nanoprimed plants maintained membrane integrity. The observed effect may be attributed to the higher bioavailability and efficient cellular uptake of ZnO NPs due to their nanoscale size and controlled solubility.
A significant positive correlation was observed between seed quality parameters and biochemical traits such as proline, total phenol, total chlorophyll content, and the activities of antioxidant enzymes including CAT, POD, and SOD. In contrast, a negative correlation was noted with malondialdehyde (MDA) content, indicating reduced lipid peroxidation in ZnO primed seedlings. These associations suggest that ZnO nanoparticle priming enhances the antioxidant defense system, leading to decreased reactive oxygen species (ROS) accumulation and improved redox homeostasis. As a result, membrane stability is maintained, and overall physiological efficiency particularly photosynthetic performance is improved in nanoprimed plants. This favorable physiological state may contribute to the observed rapid and uniform seedling emergence (Fig. 8). However, while these correlations are informative, it is important to note that correlation does not imply causation. More field-based studies are needed to validate these findings under real world conditions. The underlying molecular mechanisms by which ZnO NPs influence these physiological processes remain to be fully elucidated and warrant further investigation.
Schematic diagram illustrating the effect of ZnO nanoparticles priming on physio-biochemical responses promoting germination and seedling vigour in tomato. CAT Catalase, POX peroxidase, SOD superoxide dismutase, PR proline, TP total phenol, CHL total chlorophyll, MDA malondialdehyde content, GER germination (%), SVI I seedling vigour index I, SVI II seedling vigour index II, MGT mean germination time, SOG speed of germination.
Conclusion
This study demonstrated that seed priming with zinc oxide (ZnO) nanoparticles, particularly at a concentration of 50ppm, significantly improved seed quality and early seedling performance in tomatoes. Enhanced outcomes included accelerated germination, improved seedling vigour, increased seedling length and biomass, reduced mean germination time, elevated chlorophyll and osmoprotectant levels (such as proline and phenols), and reduced oxidative stress, as evidenced by lower malondialdehyde levels and increased antioxidant enzyme activity across all the varieties. Applying ZnO nanoparticles at low concentrations enhances the cost-effectiveness of seed priming by minimizing material requirements. Moreover, the reduced dosage decreases environmental risks by limiting nanoparticle accumulation and toxicity to non-target organisms. These factors contribute to the method’s sustainability and potential for large-scale implementation. Therefore, ZnO nanoparticle-based seed priming emerges as a remarkable method for enhancing seed quality by hastening seedling emergence due to enhanced seed vigour by changing morpho-physiological and biochemical mechanisms during tomato seed germination. However, further research is needed to elucidate the underlying molecular mechanisms and validate these findings under field conditions.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Canene-Adams, K., Campbell, J. K., Zaripheh, S., Jeffery, E. H. & Erdman, J. W. The tomato as a functional food. J. Nutr. https://doi.org/10.1093/jn/135.5.1226 (2005). 135,1226.
Tan, H. L. et al. Tomato-based food products for prostate cancer prevention: what have we learned? Cancer Metastasis Rev. 29, 553. https://doi.org/10.1007/s10555-010-9246-z (2010).
Kissoudis, C. et al. Combined biotic and abiotic stress resistance in tomato. Euphytica 202, 317. https://doi.org/10.1007/s10681-015-1363-x (2015).
Bewley, J. D. Seed germination and dormancy. Plant Cell 9, 1055. https://doi.org/10.1105/tpc.9.7.1055 (1997).
Ogbaji, P. O., Shahrajabian, M. H. & Xue, X. Changes in germination and primarily growth of three cultivars of tomato under diatomite and soil materials in auto-irrigation system. Int. J. Biol. 5, 80. https://doi.org/10.5539/ijb.v5n3p80 (2013).
Zhang, M. et al. Osmopriming improves tomato seed vigor under aging and salinity stress. Afr. J. Biotechnol. 11, 6305. https://doi.org/10.1039/C8EE02656D (2012).
Poomani, S. et al. Seed priming with humic acid modifies seedling vigour and biochemical response of lentil under heat stress conditions. Turk. J. Agric. For. 47, 6. https://doi.org/10.55730/1300-011X.3147 (2023).
Farooq, M. et al. Seed priming in field crops: potential benefits, adoption and challenges. Crop Pasture Sci. 70, 731. https://doi.org/10.1071/CP18604 (2019).
Chhipa, H. Nanofertilizers and nanopesticides for agriculture. Environ. Chem. Lett. 15 https://doi.org/10.1007/s10311-016-0600-4 (2017).
Włodarczyk, K. & Smolinska, B. The effect of nano-ZnO on seeds germination parameters of different tomatoes (Solanum lycopersicum L.) cultivars. Molecules 27, 4963. https://doi.org/10.3390/molecules27154963 (2022).
Mahakham, W., Sarmah, A. K., Maensiri, S. & Theerakulpisut, P. Nanopriming technology for enhancing germination and starch metabolism of aged rice seeds using phytosynthesized silver nanoparticles. Sci. Rep. 7, 8263. https://doi.org/10.1038/s41598-017-08669-5 (2017).
Rai-Kalal, P. & Jajoo, A. Priming with zinc oxide nanoparticles improve germination and photosynthetic performance in wheat. Plant. Physiol. Biochem. 160, 341. https://doi.org/10.1016/j.plaphy.2021.01.032 (2021).
Sharma, D., Afzal, S. & Singh, N. K. Nanopriming with phytosynthesized zinc oxide nanoparticles for promoting germination and starch metabolism in rice seeds. J. Biotechnol. 336, 64. https://doi.org/10.1007/s12298-012-0139-1 (2021).
Marschner, H. Mineral Nutrition of Higher Plants (2nd Ed.) Academic Press, London 889. https://doi.org/10.1006/anbo.1996.0155
Sharma, A., Patni, B., Shankhdhar, D. & &Shankhdhar, S. C. Zinc—an indispensable micronutrient. Physiol. Mol. Biol. Plants. 19, 11. https://doi.org/10.1007/s12298-012-0139-1 (2013).
Malik, A. et al. Bio stimulant-treated seedlings under sustainable agriculture: a global perspective facing climate change. Agronomy 11, 14. https://doi.org/10.3390/agronomy11010014 (2020).
Prajapati, B. J., Patel, K. C., Kumar, D., Patel, S. H. & Patel, V. J. Impact assessment of zinc oxide nanoparticles (ZnO-NPs) on seed germination and seedling development of maize. Int. J. Plant. Sci. 36, 9. https://doi.org/10.9734/ijpss/2024/v36i95030 (2024).
Lin, D. & Xing, B. Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ. Pollut. 150, 243. https://doi.org/10.1016/j.envpol.2007.01.016 (2007).
Zhao, L. et al. CeO2 and ZnO nanoparticles change the nutritional qualities of cucumber (Cucumis sativus). J. Agric. Food Chem. 62, 2752. https://doi.org/10.1021/jf405476u (2014).
Włodarczyk, K. & Smolińska, B. The effect of nano-ZnO on seeds germination parameters of different tomatoes (Solanum lycopersicum L.) cultivars. Molecules 27, 4963. https://doi.org/10.3390/molecules27154963 (2022).
Haq, I. U. et al. Interactions between nanoparticles and tomato plants: influencing host physiology and the tomato leafminer’s molecular response. Nanomaterials 14, 1788. https://doi.org/10.3390/nano14221788 (2024).
Shemawar, Mahmood, A. et al. Toxicity of biogenic zinc oxide nanoparticles to soil organic matter cycling and their interaction with rice-straw derived biochar. Sci. Rep. 11, 8429. https://doi.org/10.1038/s41598-021-88016-x (2020).
Rhaman, M. S. et al. Seed priming with nanoparticles: an emerging technique for improving plant growth, development, and abiotic stress tolerance. J. Soil. Sci. Plant. Nutr. 22, 4047. https://doi.org/10.1007/s42729-022-01007-3 (2022).
Bian, S. W., Mudunkotuwa, I. A., Rupasinghe, T. & Grassian, V. H. Aggregation and dissolution of 4 Nm ZnO nanoparticles in aqueous environments: influence of pH, ionic strength, size, and adsorption of humic acid. Langmuir 27, 6059. https://doi.org/10.1021/la200570n (2011).
Sushma, M. K. et al. Hydro-priming as a sustainable approach for improving germination and seedling growth in tomato (Solanum lycopersicum L). Seed Res. 51, 11–17. https://doi.org/10.56093/sr.v51i1.154654 (2024).
International Seed Testing Association. International rules for seed testing: rules, ISTA. Full Issue I 19, 276 (2022).
Abdul-Baki, A. A. & Anderson, J. D. Vigor determination in soybean seed by multiple criteria 1. Crop Sci. 13, 630. https://doi.org/10.2135/cropsci1973.0011183X001300060013x (1973).
Ellis, R. H., Covell, S., Roberts, E. H. & Summerfield, R. J. The influence of temperature on seed germination rate in grain legumes: II. Intraspecific variation in Chickpea (Cicer arietinum L.) at constant temperatures. J. Exp. Bot. 37, 1503. https://doi.org/10.1093/jxb/37.5.705 (1986).
Maguire, J. D. Speed of germination-aid in selection and evaluation for seedling emergence and vigour. Crop Sci. 2, 176. https://doi.org/10.2135/cropsci1962.0011183X000200020033x (1962).
Heath, R. L. & Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 189. https://doi.org/10.1016/0003-9861(68)90654-1 (1968).
Arnon, D. I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant. Physiol. 24, 1. https://doi.org/10.1104/pp.24.1.1 (1949).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant. Soil. 39, 205. https://doi.org/10.1007/BF00018060 (1973).
Singleton, V. L. & Rossi, J. A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. AJEV 16, 144. https://doi.org/10.5344/ajev.1965.16.3.144 (1965).
Braber, J. M. Catalase and peroxidase in primary bean leaves during development and senescence, Zeitschrift Pflanzenphysiologie 97, 135. https://doi.org/10.1016/S0044-328X(80)80027-4 (1980).
Castillo, F. J., Penel, C. & Greppin, H. Peroxidase release induced by Ozone in Sedum album leaves: involvement of Ca2+. Plant. Physiol. 74 846. https://doi.org/10.1104/2Fpp.74.4.846 (1984).
Beauchamp, C. & Fridovich, I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 44, 276. https://doi.org/10.1016/0003-2697(71)90370-8 (1971).
Rao, D. et al. Silicic and humic acid priming improves micro-and macronutrient uptake, salinity stress tolerance, seed quality, and physio-biochemical parameters in lentil (Lens culinaris spp.). Plants 12, 3539. https://doi.org/10.3390/plants12203539 (2023).
Devika, O. S. et al. Seed priming: a potential supplement in integrated resource management under fragile intensive ecosystems. Front. Sustain. Food Syst. 5, 654001. https://doi.org/10.3389/fsufs.2021.654001 (2021).
Harris, D., Rashid, A., Miraj, G., Arif, M. & Shah, H. On-farm’seed priming with zinc sulphate solution—a cost-effective way to increase the maize yields of resource-poor farmers. Field Crops Res. 102, 119–127. https://doi.org/10.1016/j.fcr.2007.03.005 (2007).
Samad, A., Khan, M. J., Shah, Z. & Jan, M. T. Determination of optimal duration and concentration of zinc and phosphorus for priming wheat seed. Sarhad J. Agric. 30, 27. https://doi.org/10.14741/ijcet/v.12.4.7 (2014).
Srivastav, A. et al. Effect of ZnO nanoparticles on growth and biochemical responses of wheat and maize. Plants 10, 2556. https://doi.org/10.3390/plants10122556 (2021).
Kumar, S. M. et al. Preliminary studies on the effect of nanoparticle seed treatments on seed quality attributes and yield in Chickpea. Turk. J. Agric. For. 47, 100. https://doi.org/10.55730/1300-011X.3067 (2023).
Cakmak, I. Enrichment of cereal grains with zinc: agronomic or genetic biofortification? Plant. Soil. 302 (1). https://doi.org/10.1007/s11104-007-9466-3 (2008).
Prasad, T. N. V. K. V. et al. Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J. Plant Nutr. 35, 905. https://doi.org/10.1080/01904167.2012.663443 (2012).
Broadley, M. R., White, P. J., Hammond, J. P., Zelko, I. & Lux, A. Zinc in plants. New. Phytol. 173, 677. https://doi.org/10.1111/j.1469-8137.2007.01996.x (2007).
Burman, U., Saini, M. & Kumar, P. Effect of zinc oxide nanoparticles on growth and antioxidant system of chickpea seedlings. Toxicol. Environ. Chem. 95, 605. https://doi.org/10.1080/02772248.2013.803796 (2013).
Cyriac, J., Melethil, K., Thomas, B., Sreejit, M. & Varghese, T. Synthesis of biogenic ZnO nanoparticles and its impact on seed germination and root growth of Oryza sativa L. and Vigna unguiculata L. Mater. Today Proc. 25, 224. https://doi.org/10.1016/j.matpr.2020.01.107 (2020).
Torabian, S., Zahed, M. & Khoshgoftar, A. H. Effects of foliar spray of two kinds of zinc oxide on the growth and ion concentration of sunflower cultivars under salt stress. J. Plant. Nutr. 39, 172. https://doi.org/10.1080/01904167.2015.1009107 (2016).
Abou-Zeid, H. M., Ismail, G. S. M. & Abdel-Latif, S. A. Influence of seed priming with ZnO nanoparticles on the salt-induced damages in wheat (Triticum aestivum L.) plants. J. Plant. Nutr. 44, 629–643. https://doi.org/10.1080/01904167.2020.1849288 (2021).
Salehi, H. et al. Exogenous application of ZnO nanoparticles and ZnSO4 distinctly influence the metabolic response in Phaseolus vulgaris L. Sci. Total Environ. 778 https://doi.org/10.1016/j.scitotenv.2021.146331 (2021).
Mirakhorli, T., Ardebili, Z. O., Ladan-Moghadam, A. & &Danaee, E. Bulk and nanoparticles of zinc oxide exerted their beneficial effects by conferring modifications in transcription factors, histone deacetylase, carbon and nitrogen assimilation, antioxidant biomarkers, and secondary metabolism in soybean. Plos One 16, e0256905. https://doi.org/10.17660/ActaHortic.2023.1375.51 (2021).
Mushtaq, N. U. et al. Exogenous zinc mitigates salinity stress by stimulating proline metabolism in proso millet (Panicum miliaceum L). Front. Plant. Sci. 14, 1053869. https://doi.org/10.3389/fpls.2023.1053869 (2023).
Miliauskiene, J., Brazaitytė, A., Sutulienė, R., Urbutis, M. & Tučkutė, S. ZnO nanoparticle size-dependent effects on Swiss Chard growth and nutritional quality. Agriculture 12, 1905. https://doi.org/10.3390/agriculture12111905 (2022).
Amooaghaie, R., Norouzi, M. & Saeri, M. Impact of zinc and zinc oxide nanoparticles on the physiological and biochemical processes in tomato and wheat. Botany 95, 441. https://doi.org/10.1139/CJB-2016-0194 (2017).
Sultana, S. et al. Foliar and root applications of salicylic acid alleviate salinity stress by modulating morpho-physiological and biochemical aspects in tomato (Solanum lycopersicum). Discov. Plants. 2, 36. https://doi.org/10.1007/s44372-025-00117-3 (2025).
Azarin, K. et al. Effects of ZnO nanoparticles and its bulk form on growth, antioxidant defense system and expression of oxidative stress related genes in Hordeum vulgare L. Chemosphere 287, 132167. https://doi.org/10.1016/j.chemosphere.2021.132167 (2022).
Abdel Latef, A. A. H., Alhmad, A. & Abdelfattah, M. F. The possible roles of priming with ZnO nanoparticles in mitigation of salinity stress in lupine (Lupinus termis) plants. J. Plant Growth Regul. 36, 60. https://doi.org/10.1007/s00344-016-9618-x (2017).
Guillén-Enríquez, R. R. et al. ZnO nanoparticles improve bioactive compounds, enzymatic activity and zinc concentration in grapevine. Not Bot. HortiAgrobo. 51, 13377. https://doi.org/10.15835/nbha51413377 (2023).
Song, Y., Jiang, M., Zhang, H. & Li, R. Zinc oxide nanoparticles alleviate chilling stress in rice (Oryza sativa L.) by regulating antioxidative system and chilling response transcription factors. Molecules 26, 2196. https://doi.org/10.3390/molecules26082196 (2021).
Acknowledgements
The authors would like to express their sincere gratitude to ICAR-Indian Agricultural Research Institute, New Delhi, for providing the essential facilities, necessary for conducting these studies.
Funding
This research received financial support from the ICAR-IARI fellowship and the Division of Seed Science and Technology in New Delhi, India.
Author information
Authors and Affiliations
Contributions
SMK: Writing—original draft, Writing—review & editing, Formal analysis, Investigation. SY: Writing—Review & Editing, Supervision, Conceptualization, Methodology, Validation, Resources. RC: Writing—Review & Editing, Visualization, Validation. AA: Formal analysis, Investigation. AY: Formal analysis, Investigation. ZH: Methodology, Validation, Supervision, Resources. PKS: Methodology, Validation, Resources. SPS: Methodology, Validation, Resources, Supervision. AM: Methodology, Supervision. NS: Resources, Validation. SS: Writing—Review & Editing, SE: Writing—Review & Editing. US: Writing—Review & Editing. SKY: Writing—Review & Editing, Supervision, Conceptualization, Methodology, Validation, Resources.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kumar, S.M., Yadav, S., Choudhary, R. et al. Nanopriming with zinc oxide nanoparticle boosts seed vigour, photosynthesis, osmolytes accumulation and antioxidant activity in tomato. Sci Rep 15, 37375 (2025). https://doi.org/10.1038/s41598-025-09269-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09269-4