Abstract
Herbicides are the extensively used class of pesticides, which beside the active ingredient, in their formulation accompanying substances such as emulsifiers, surfactants and others is needed. The potential toxicity of these synthetic chemicals could pose serious risks to the human health, nontarget organism and environment. In this work we developed biodegradable lignin nanoparticles (LNPs) as environmentally friendly and controlled release carriers of 2,4-dichlorophenoxyacetic acid 2,4-D and 2-methyl-4-chlorophenoxyacetic acid MCPA. LNPs were synthesized via solvent-free nanoprecipitation, achieving high entrapment efficiencies of 90.7% (2,4-D) and 97.4% (MCPA), confirmed by ultraviolet–visible spectroscopy. In vitro release studies revealed sustained herbicide release in buffer solutions (pH 5.5–7.5), with 68–74% release over 72 h, compared to rapid release from commercial formulations. Bioactivity assays of on Descurainia sophia showed that LNP-encapsulated formulation of herbicides reduced weed dry weight by 62.31% and density by 56.09% compared to untreated controls, statistically matching the weed control efficacy of commercial formulations. Field trials further validated these results. LNP-encapsulated 2,4-D + MCPA reduced Amaranthus blitoides dry weight by 91.10% and density by 65.09%, while this new formulation decreased Chenopodium album dry weight and density by 96.01% and by 66.75%, respectively. Notably, lignin’s inherent biodegradability and non-toxic nature provide a sustainable alternative to conventional synthetic adjuvants, significantly reducing the risks of environmental contamination. Our study highlights the potential of lignin-based nanoencapsulation to preserve weed control efficacy while promoting environmentally friendly and safer herbicide formulations.
Similar content being viewed by others
Introduction
In agricultural ecosystems, weeds compete with crops for essential resources such as nutrients, water, and sunlight, significantly reducing agricultural productivity. Without effective control, weeds can cause drastic yield losses in main crops such as wheat (up to 60%), rice (up to 100%), and maize (up to 93%), threatening food supplies and economic stability1. Therefore, weed management is a critical operation for optimal utilization of cropland potential in crop production and subsequently ensuring food security2. Among the weed control measures, chemical control with herbicides remains indispensable due to their efficiency and cost-effectiveness3. Auxin-like herbicides such as (2,4-D) and (MCPA) are widely used herbicides that selectively target broad-leaf weeds by disrupting plant growth regulators, including ethylene production, cell wall integrity, and other unknown mechanisms4. These herbicides mimic natural plant hormones, causing uncontrolled growth and eventual death in susceptible weeds, making them highly effective for controlling many problematic broadleaf weeds in the main cereal crops (e.g., wheat, barley, rice, maize) and pastures5.
Despite the many benefits of using herbicides as effective weed control measures, conventional herbicide formulations face significant challenges. In the formulations of commercial herbicides, besides the active ingredient, accompanying substances such as such as surfactants, solvents, and stabilizers, emulsifiers, buffering agents, etc. are used to enhance herbicide penetration, retention, and absorption6. While these additives improve herbicide performance, they also contribute to environmental pollution, toxicity to non-target organisms, and groundwater contamination7. For instance, surfactants like alkylphenol ethoxylates have been linked to endocrine disruption in aquatic organisms8. Additionally, the persistence of these chemical compounds in the environment will lead to water and soil pollution, and adversely affecting the sustainability of agricultural practices9. These issues have spurred global interest in developing safer, more environmentally friendly alternatives to conventional herbicide formulations. Nanotechnology offers a promising solution by enabling controlled release and reducing reliance on hazardous adjuvants. Encapsulation systems, such as polymeric nanoparticles, protect active ingredients from premature degradation, extend herbicide persistence on weeds, and minimize off-target effects10. Several controlled release systems for 2,4-D have been reported in the literature, each designed to prolong herbicide efficacy while minimizing environmental impact. For example, Belmokhtar et al.11 developed cellulose derivative-based polymer matrices using the emulsification-solvent evaporation method, which demonstrated a slower, more sustained release profile. The pesticide encapsulated with coumarin polymer has higher thermal stability than free 2,4-D and is slowly washed out. It was also observed that the pesticide encapsulated with coumarin polymer effectively delivers 2,4-D to plant tissues and improves herbicide activity12. Herbicides, such as 2,4-D, play a key role in weed management but also raise environmental concerns due to their potential for leaching, volatilization, and non-target toxicity. To mitigate these issues, several controlled release systems have been developed. For example, Ceolin et al.13 reported the use of a chitosan-tripolyphosphate microparticle system, which achieved high encapsulation efficiency and significantly reduced the volatilization of 2,4-D. Similarly, Akakuru and Onyido14 explored CRFs using chitosan-starch composites, highlighting how crosslinking reduces water absorption and improves release control, especially under variable pH condition. 2,4-dichlorophenoxyacetic acid was integrated into a polymeric system using jute fibers as the polymer matrix and acrylamide as the grafting monomer. The findings indicated that xylene was the most effective solvent for promoting esterification and regulating the herbicide’s release. This approach offers a dual function, serving both as a herbicide and as a fertilizer or soil modifier. This formulation can also be suitable for slow release control of 2,4-dichlorophenoxyacetic acid15.
Lignin, a biodegradable phenolic polymer derived from plant biomass, is an ideal candidate for nanoencapsulation. Its aromatic structure provides chemical stability, while its inherent biodegradability mitigates environmental risks associated with synthetic carriers16,17. Researchers have conducted extensive studies to develop new materials and compounds based on lignin. This biopolymer has been utilized in the production of various products, including antibacterial agents, antioxidant additives, hydrogel-forming molecules, nanoparticles, and solid dosage form18. Lignin-based carriers have been used as encapsulation matrices for the herbicide active ingredient, pelargonic acid. The use of this green technology platform to replace chemical-based herbicides could have potential applications in agricultural fields19. Lignin nanoparticles (LNPs) were prepared for encapsulation of rose essential oil. The analysis showed that REO-LNP had higher thermal stability than free REO. Also, the antioxidant activity of REO was increased. Compared with REO, REO-LNPs showed improved antibacterial properties. In vitro cell viability evaluation showed that the prepared samples were free of cytotoxicity and had excellent biocompatibilit. This study suggests that LNPs can be used as renewable nanocarriers for the protection and controlled release of bioactive compounds in various applications20. Weiss et al.21 reported used Myceliophthora thermophila laccase to optimize the production of lignosulfonate granules. The granules were loaded with the herbicide dicamba, and complete release of the active ingredient was achieved after 48 h. The results showed that lignosulfonates can be effective biodegradable carriers for agricultural applications.
As we discussed, weed management especially in the large-scale crop cultivation relies mainly on herbicides, yet conventional formulations depend on synthetic adjuvants that contribute to environmental pollution and non-target toxicity. To address these challenges, this study explores solvent-free nanoprecipitation as a method to synthesize lignin nanoparticles (LNPs) for encapsulating 2,4-D and MCPA. This approach utilizes lignin’s inherent biodegradability and adhesive properties, thereby eliminating the use of toxic solvents and synthetic additives, aligning with green chemistry principles. Therefore, this work prioritizes optimizing lignin’s physicochemical traits to achieve tunable herbicide release and evaluates bioactivity under field conditions. It seeks to demonstrate the feasibility of an environmentally friendly method for herbicides active ingredient formulation that balances agricultural efficacy with reduced environmental contamination, advancing sustainable alternatives for weed management.
Materials and methods
Materials
2,4-dichlorophenoxyacetic acid (C8H6Cl2O3) and 4-chloro-2-methylphenoxyacetic acid (C9H9ClO3) (95%) were from Karon Co.Ltd,. Lignin and Sodium caprylate were prepared from Sigma Aldrich Co. Ethylbutanoate (C6H12O2), Disodium hydrogen phosphate Na2HPO4, Potassium dihydrogen phosphate KH2PO4, Sodium chloride NaCl, and Potassium chloride KCl were purchased from Merck Chemical Co. The dialysis membrane of Molecular weight cutoff (MWCO) 10,000–14,000 Dalton was purchased from Viskase Co. Ltd. Deionized water was used throughout the research.
Research instrumentation
Hydrodynamic sizing of herbicide-loaded nano micelles was conducted using the ALV-5000 F system from ALV GmbH, Germany, which is equipped with a diode-pumped solid-state laser (2 W, Coherent Innova, USA). Optical analysis and release studies of the microemulsions were performed using the UV-visible spectrophotometer, Specord 210 Plus from Analytik Jena, Germany. A wavelength of λ = 285 nm was used for 2,4-D and λ = 277 nm for MCPA.
Fabrication of 2,4-D + MCPA microemulsion with lignin nanoparticles
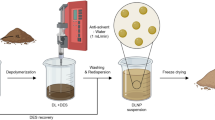
Herbicides -loaded oil-in-water surfactant-based biocompatible nanomicelles were developed according to Varshney et al.22Rahdar et al.23 from dissolving herbicide in 1% (w/w) solutions of ethyl butyrate oil by dissolving an amount of fatty acid sodium caprylate (SC, 0.09 g) and lignin (0.006 g) phosphate-buffered saline (PBS at pH 7.4) under vigorous stirring at a fixed ethyl butyrate-to-surfactant molar ratio and final total volume of 30 mL. The mixture was vigorously stirred in phosphate-buffered saline (PBS, pH 7.4) at a controlled speed of 300 rpm to ensure proper dispersion and micelle formation. The total formulation volume was brought to 30 mL. In our formulation, sodium caprylate (SC) was used as the primary surfactant and stabilizer per 30 mL total formulation volume. SC plays a crucial role in reducing interfacial tension and stabilizing the oil-in-water nanomicelles, ensuring the colloidal stability of the lignin nanoparticles23. The excess of free Herbicides was eliminated by dialysis for 24 h. The structure and components of the synthesized Herbicides -loaded nanomicelles are depicted in (Fig. 1).
Construction of nano herbicides.
Size determination of 2,4-D and MCPA-loaded lignin nanoparticles
A custom ALV-5000 F system from ALV GmbH, Germany, equipped with a diode-pumped solid-state laser (2 W, Coherent Innova, USA), was used to measure the hydrodynamic size of herbicide-loaded nanomicelles. In a nanocolloidal system, the diffusion coefficient of particles is determined by monitoring the temporal variations in light scattering intensity, which reflects the Brownian motion of the particles.
Determination of herbicide encapsulation
The information regarding the measurement of 2,4-D and MCPA using UV spectroscopyincluding detection wavelengths (285 nm for 2,4-D and 277 nm for MCPA), and sample preparation was all carried out following a standard set-up in our laboratory24. To determine encapsulation efficiency, 6 mL of each microemulsion (2,4-D and MCPA) was centrifuged at 12,000 rpm for 30 min. The samples were then filtered through a 0.2 μm CA filter, and the remaining supernatant was separated. UV analysis was conducted for 2,4-D and MCPA. Entrapment efficiency (EE%) was calculated using the following formula (Eq. 1).
In vitro release
The calibration curve for each herbicide served as a reference for quantifying 2,4-D and MCPA. For both herbicides, the calibration curve was obtained as a linear relationship, with correlation coefficients (R² ≥ 0.99). The regression equation for 2,4-D was y = 1.119x + 0.074, while for MCPA, it was y = 1.2241x + 0.0441. The release of 2,4-D and MCPA from microemulsions was performed using the dialysis method. For the in vitro release study, 50 µL of each herbicide sample was placed to dialysis bags, which were then immersed in 120 mL of either distilled water or phosphate-buffered saline (PBS, pH = 7.4). The release study was conducted at 37 °C over a 72-hour period. At predetermined time points (0.16, 0.33, 0.5, 0.66, 1, 2, 4, 6, 24, 48, and 72 h,, 4 mL of the release medium was extracted, and the concentration of the samples was measured using UV spectroscopy. Each sample was tested in triplicate25,26.
Investigating the effect of microemulsions on weeds
Two filed experiments were conducted to evaluate the bioactivity of the new formulation of 2,4-D/MCPA (lignin nanoparticles; LNPs) on broad-leaf weeds including Descurainia sophia (L.) Webb ex Prantl, Amaranthus blitoides S. Watson and Chenopodium album L plants that naturally grow as a broad-leaf weed in the research farm of University of Zanjan. The weed was identified taxonomically by second author, a specimen was kept at plant production & Genetics Department, University of Zanjan, Iran. No permissions or licenses were required for collecting these weed seeds. The experimental site located at 36.68° N latitude and 48.40° E longitude, at an altitude of 1,592 m. The region is characterized by a semi-arid cool climate, with an average annual temperature of 11 °C and average rainfall of 293 mm. The soil type was silty clay loam (37% clay, 45% silt, 18% sand), with 0.111% nitrogen, 16.8 mg/kg available phosphorus, 320 mg/kg potassium, and a pH of 7.45. In the first experiment, wheat seeds (of cv. Mihan was purchased from Khodabandeh County Rural Cooperatives Union) were treated with the systemic fungicide difenoconazole 3% (FS) and then planted on October 17, 2021. The experimental plots measured 6 m² (5 × 1.20 m). The second experiment was conducted in on June 10, 2023. The size of each plot was 2 m² plots (2 × 1 m) at no crop condition. The concentrations of the microemulsions were determined based on the recommended application rates for commercial formulations: 2,4-D (720 g a.i. ha⁻¹) and MCPA (630 g a.i. ha⁻¹). The study compared the effectiveness of 2,4-D + MCPA-loaded lignin-stabilized microemulsions with control treatments (no herbicide application) and a commercial herbicide formulation. A backpack sprayer equipped with flood-jet nozzle tip was used for herbicide application, calibrated to a working pressure of 200 kPa and a flow rate of 300 Lha− 1. The treatments were applied at the 4–6 leaf stage of D. sophia in the first experiment, and at the 2–4 leaf stage of A. blitoides and C. album in the second year. Two weeks after herbicide application, the dry weight and density of D. sophia, A. blitoides and C. album were measured. For density assessment, a 1 m² quadrat was used. After weed samples were harvested, they were dried at 75 °C for 48 h, and the dry weight was determined using a digital scale. The experimental design was randomized complete block with three replicates for each treatment.
Data analysis
Data analysis was conducted using Statistical Analysis System (SAS) software (version 9.4). To assess the normality of the data, a normality test was applied. Data that did not follow a normal distribution were transformed using a normalization factor (P < 0.05) to enhance the distribution. Analysis of variance (ANOVA) was performed to compare group means, and Tukey’s post-hoc test was applied for pairwise comparisons at a significance level of 0.05.
Results
Physico-chemical characterization
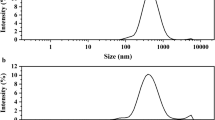
The nanoparticles had a particle size of 29 ± 4 nm (Fig. 2a), and the hydrodynamic size population of bare by the Dynamic Light Scattering (DLS) and herbicide-loaded polymeric nanoparticles, which show a radius about ca. 34 ± 2 nm, is presented in (Fig. 2b). According to observations, the size obtained with Emission Scanning Electron Microscopy (FESEM) and (DLS) are almost similar, and the nanoparticles exhibited a spherical-like morphology. To characterize the hydrodynamic size of nanomicelles from DLS tool with a ALV-5000 F system (ALV GmbH, Germany) equipped with a diode-pumped solid-state laser (2 W, Coherent Innova, USA) was used. According to previous studies on this nanocarrier Rahdar et al.23with the specified PDI of 0.1–0.2 along with morphology of LNPs by FESEM (Fig. 2a) confirmed the stability and size homogeneity of the nanocapsules. In the other words, Morphology of LNPs by FESEM (Fig. 2a) directly reflects the stability and size homogeneity of the nanocapsules in the total microemulsion.
a FESEM image of nanomicelles after 6 months of storage. b Hydrodynamic size population of nanoparticles particles.
Fourier transform infrared spectrum reflection of lignin nanoparticles
The chemical structure of the derivatives of nanoparticles was evaluated by the Fourier transform infrared spectrum (FT-IR), which was acquired on a 102 MB instrument. The IR spectra of the Lignin-encapsulated herbicides both presented the corresponding signals of 2,4-D and MCPA together with the Lignin backbone, clearly confirming the presence of both herbicides in the final nanoparticles. The bands for the hydroxyl acid and carbonyl group of MCPA (red line) were observed at 3450 cm- 1 and 1651 cm- 1, respectively, in the IR spectrum of the Lignin-encapsulated MCPA (Fig. 3), spectrum a, which proved the capturing of MCPA molecules inside the lignin backbone. The corresponding signal of carbonyl group is presenting a meaningful shift to lower wavenumbers which can be attributed to the possible hydrogen binding of herbicide molecules with hydroxyl groups of Lignin. The peaks at 676 cm- 1 ,1439 cm- 1, and 1494 cm- 1 in the MCPA (red line) spectrum correspond to C-Cl stretching vibration, CH2 bending vibrations of alkanes, and C=C vibrations of the aromatic ring, respectively.
a FT-IR spectra of MCPA-loaded Lignin nanoparticles. b FT-IR spectra 2,4-D-loaded Lignin nanoparticles. Red lines are of free 2,4-D and MCPA, Black lines are of 2,4-D and MCPA- loaded Lignin nanoparticles.
Entrapment of particles and release
Encapsulation efficiency is a key characteristic of nanocarriers. The results of this experiment demonstrated that both 2,4-D and MCPA exhibit high entrapment efficiency. The encapsulation efficiency (EE%) of 2,4-D-loaded lignin and MCPA-loaded lignin were 90.7% and 97.4%, respectively. Lignin-based nanoparticles displayed a high release rate in both water and buffer solutions. The release profiles of 2,4-D and MCPA-loaded lignin nanoparticles indicated that approximately 50% of the herbicides were released within the first 35 h (Fig. 4).
Release of 2,4-D and MCPA-loaded lignin nanoparticles. Blue lines are the release of 2,4-D and MCPA- loaded lignin nanoparticles inside water. Red lines are the release of 2,4-D and MCPA- loaded lignin nanoparticles inside buffer (PH = 7.4).
Weed control efficacy of nano-herbicide
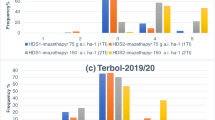
The analysis of variance showed that the stabilized 2,4-D and MCPA- loaded lignin and the commercial 2,4-D + MCPA significantly affected the growth D. sophia, A. blitoides and C. album. Without using herbicide spraying D. sophia, A. blitoides and C. album dry weight were 17.33, 24.97 and 14.46 g m− 2, while using commercial herbicide and the new microemulsion formulation it decreased in D. sophia 4.44 and 6.53 g m− 2, respectively, two weeks after spraying (Fig. 5). While using commercial herbicide and the new microemulsion formulation it decreased in A. blitoides 1.23 and 2.22 g m− 2, respectively, and it decreased in C. album 0.41 and 0.57 g m− 2, respectively, two weeks after spraying (Fig. 5). Using commercial herbicide and the new microemulsion formulation decreased in D. sophia density 7 and 9.66 weed m− 2, two weeks after spraying respectively, (Fig. 5). Also, using commercial herbicide and the new microemulsion formulation decreased in A. blitoides density 14.33 and 19.66 weed m− 2, two weeks after spraying respectively, (Fig. 5). Without using herbicide spraying C. album density was 4 weed m− 2, this reduction in density in commercial and new microemulsion of 2,4-D and MCPA had a similar trend and in both treatments, 1.33 weed m− 2, was obtained (Fig. 5).
The effect of commercial herbicides and 2,4-D and MCPA-loaded lignin nanoparticles on the a Dry weight, b Density respectively of the D. Sophia. c Dry weight, d Density respectively of the A. blitoides. e Dry weight, f Density respectively of the C. album. Columns with common letters are not significantly different based on Tukey’s test (P < 0.05) (mean ± SD).
Discussion
Colloidal stability aligns with prior studies on lignin nanoparticles27confirming the suitability of solvent-free nanoprecipitation for scalable synthesis. Slight swelling during storage highlights a trade-off between environmental responsiveness and structural integrity, though preserved morphology suggests practical shelf stability. FTIR spectral shifts (e.g., carbonyl redshift) provide direct evidence of hydrogen bonding between herbicides and lignin. This interaction likely stabilizes the nanoemulsion, delaying premature release. Same signals were also observed for 2,4 D-loaded Lignin exhibiting the sign of successful encapsulation of 2,4-D inside the Lignin chain28. The lignin particles effectively play a role in stabilizing emulsions and contribute to stability29. Our results suggest that lignin nanoparticles are more stable under suitable pH conditions (around 7 to 9), effectively preventing aggregation or dissolution. It has also been reported that lignosulfonate nanoparticles have high efficiency and controlled release, which can be effective in agricultural applications30. Lignin-poly (lactic acid) (AL-PLA) composite microspheres were used for loading and encapsulating 2,4-D. These microspheres were able to effectively encapsulate 87.87%. The hydrogen bonding between lignin and poly (lactic acid) played a crucial role in stabilizing 2,4-D and enabling controlled release from the microspheres. These characteristics enhanced the encapsulation efficiency, making it a promising method for reducing pesticide usage and protecting the environment, particularly in agricultural applications31. The slower release in water versus buffer solutions suggests pH sensitive behavior, advantageous for targeted delivery in specific soil conditions. The ion exchange mechanism in buffers 7 underscores the formulation’s adaptability to environmental pH, though prolonged herbicide persistence may require further study to avoid off-target effects32.
The lignin-based nanoencapsulation of 2,4-D and MCPA demonstrated comparable weed control efficacy to commercial herbicides for A. blitoides (91.10% vs. 95.07% dry weight reduction) and Ch. album (96.01% vs. 97.14%), D. sophia (62.31% vs. 74.37% reduction), demonstrating its potential for field applications.
Lignin nanoparticles prioritize sustained herbicide delivery over immediate efficacy, which may reduce environmental persistence but necessitates optimization for weeds requiring faster action. A key advantage of this formulation is the elimination of synthetic adjuvants, replacing them with biodegradable lignin to avoid compounds linked to water contamination and non-target toxicity. While lignin’s slow degradation and natural abundance suggest ecological benefits, direct assessments of its environmental fate such as impacts on soil microbiota or non-target organisms-are needed to validate these results. Future refinements should focus on enhancing foliar adhesion through lignin functionalization (e.g., sulfonation) and tailoring release kinetics to improve efficacy for recalcitrant weed species.
Traditional agricultural methods in pesticide delivery, due to the lack of control over the release process and the need for repeated high-dose applications, not only have limited effectiveness but also cause environmental problems and resistance in pests. This highlights the need for innovative approaches such as controlled and sustained release systems, which can help mitigate these issues33. Nanoherbicides can play a crucial role in reducing environmental risks and minimizing toxic effects34. The application of nanotechnology holds significant potential for enhancing human health preservation35,36. Extensive research has been conducted on nanoherbicides, driven by their advantages in environmental protection37,38.
While lignin is a biodegradable and naturally abundant biopolymer, certain factors may limit its effectiveness in real-world agricultural settings. For instance, chemical modifications such as cross-linking introduced during nanoparticle synthesis can reduce its biodegradability by hindering the activity of microbial enzymes that typically act on phenolic hydroxyl groups39. The challenges of lignin nanoparticles (LNPs) include issues such as instability in organic solvents and pH variations, which lead to the dissolution or aggregation of the particles in acidic or basic environments. These problems create limitations in the chemical functionality and applications of these nanoparticles. Additionally, the need for organic solvents during the production process, incompatibility with hydrophobic polymeric matrices are other challenges that need to be addressed40. Despite significant progress and considerable efforts in the development of lignin-based controlled-release fertilizers, these fertilizers still face several challenges. When used as a coating material, lignin tends to have an uneven coating, higher surface porosity, and is prone to fractures due to its inherent structural variability, which results in unstable performance in controlled nutrient release41. Most of the existing studies have been conducted in laboratory-scale experiments under controlled conditions, so their results may not directly applay to real-field agricultural settings. Additionally, the mechanisms of nanoparticle uptake and translocation in plants are not fully understood, and their effects may vary depending on the plant species. Furthermore, the application method (e.g., foliar spraying vs. soil application) could influence their interactions with soil biota and plant42.
Future research should expand on this work by investigating the release kinetics of lignin-based nanoparticles under a range of agro-environmental conditions. Such studies will provide deeper insights into the behavior of the formulation in diverse environments and enable the optimization of the system for specific agricultural applications. Additionally, given the biocompatible nature of the materials used, such as lignin and sodium caprylate, it is anticipated that the formulation will pose minimal toxicity risks to the environment. To further ensure the safety of these formulations, future research will also assess the potential impacts of lignin-based nanocarriers on soil biota, beneficial insects, and aquatic life.
Long-term studies on lignin nanoparticle degradation in diverse ecosystems will also be critical to confirming their sustainability profile. Despite these gaps, this work establishes lignin-based nanoencapsulation as a viable alternative to conventional herbicides, offering a pathway to reduce reliance on toxic additives without compromising agricultural productivity. By bridging the gap between controlled release and practical efficacy, this approach highlights the potential of bio-based materials to advance environmentally sustainable weed management.
Conclusion
The environmental risks associated with conventional herbicides including water and soil contamination and threats to non-target organisms highlight the need for sustainable alternatives. This study demonstrates that lignin-based nanoencapsulation of 2,4-D and MCPA offers a promising strategy to address these challenges. In field trials, the lignin nanoformulation achieved 91.10–96.01% dry weight reduction in A.blitoides and Ch. album, matching the efficacy of commercial herbicides. By replacing synthetic adjuvants with biodegradable lignin, this formulation eliminates additives linked to environmental toxicity while leveraging lignin’s natural adhesion and slow degradation to prolong herbicide retention on weeds. These design advantages position lignin nanoparticles as a practical alternative for reducing reliance on conventional formulations, which are optimized for rapid action but often contain ecologically harmful components. Future work should evaluate the long-term environmental fate of lignin nanoparticles and their impacts on non-target organisms to validate the proposed sustainability benefits. Overall, this study establishes lignin-based nanoencapsulation as a critical step toward environmentally-friendly herbicide delivery, demonstrating that sustainable agricultural practices can align with effective weed management.
Data availability
Data are available from the corresponding author.
References
Sardana, V., Mahajan, G., Jabran, K. & Chauhan, B.S. Role of competition in managing weeds: An introduction to the special issue. Crop Prot. 1-7 (2016) http://dx.doi.org/10.1016/j.cropro.2016.09.011
Sardana, V., Mahajan, G., Jabran, K. & Chauhan, B. S. Role of competition in managing weeds: an introduction to the special issue. J. Crop Prot. 1–7. https://doi.org/10.1016/j.cropro.2016.09.011 (2016).
Paul, S.K., Mazumder, S., & Naidu, R. Herbicidal weed management practices: History and future prospects of nanotechnology in an eco-friendly crop production system. Heliyon. 10, e26527 (2024) https://doi.org/10.1016/j.heliyon.2024.e26527
Wei, Y. D., Zheng, H. G. & Hall, J. C. Role of auxinic herbicide-induced ethylene on hypocotyl elongation and root/hypocotyl radial expansion. Pest Manag Sci. 56, 377–387. https://doi.org/10.1002/(SICI)1526-4998(200005)56:5<377:AIDPS154>3.3.CO;2-D (2000).
Busi, R. et al. Weed resistance to synthetic auxin herbicides. Pest Manag Sci. 74, 2265–2276. https://doi.org/10.1002/ps.4823 (2018).
Kulenkampff, J. et al. Time-Lapse 3D imaging by positron emission tomography of Cu mobilized in a soil column by the herbicide MCPA. Sci. Rep. 8, 7091. https://doi.org/10.1038/s41598-018-25413-9 (2018).
Mesnage, R. & Antoniou, M. N. Ignoring Adjuvant Toxicity Falsifies the Safety Profile of Commercial Pesticides. Front Public Health. 5, 361 (2018) https://doi.org/10.3389/fpubh.2017.00361
Lin, Q. et al. SPE-UPLC-MS/MS for determination of 36 monomers of alkylphenol ethoxylates in tea. Molecules 28, 3216. https://doi.org/10.3390/molecules28073216 (2023).
He, J., Li, J., Gao, Y., He, X. & Hao, G. Nano-based smart formulations: A potential solution to the hazardous effects of pesticide on the environment. J. Hazard. Mater. 456, 131599. https://doi.org/10.1016/j.jhazmat.2023.131599 (2023).
Mattos, B. D., Tardy, B. L., Magalhães, W. L. E. & Rojas, O. J. Controlled release for crop and wood protection: recent progress toward sustainable and safe nanostructured biocidal systems. J. Control Release. 262, 139–150. https://doi.org/10.1016/j.jconrel.2017.07.025 (2017).
Belmokhtar, F. Z., Elbahri, Z. & Elbahri, M. Preparation and optimization of agrochemical 2,4-D controlled release microparticles using designs of experiments. J. Mex Chem. Soc. 62 https://doi.org/10.29356/jmcs.v62i1.579 (2018).
Atta, S. et al. Photoresponsive polymers based on a coumarin moiety for the controlled release of pesticide 2,4-D. RSC Adv. 5 https://doi.org/10.1039/C5RA18944F (2015).
Ceolin, B. C., Nörnberg, A. B., Aranha, B., Fajardo, A. R. & Camargo, E. R. Controlled release and mitigation of the volatility of 2,4-dichlorophenoxyacetic acid herbicide using biopolymer microparticles as carriers. Colloids Surf. A: Physicochem Eng. Asp. 699, 134672. https://doi.org/10.1016/j.colsurfa.2024.134672 (2024).
Akakuru, O. U. & Onyido, I. Controlled release formulations of 2,4-dichlorophenoxyacetic acid with ecofriendly matrices for agricultural and environmental sustainability. Macromol. Res. 29, 40–53 https://doi.org/10.1007/s13233-021-9004-9 (2021).
Islam, S., Kamruzzaman, M., Ray, S. K. & Mustafizur Rahman, A. F. M. Controlled release of 2,4-Dichlorophenoxyacetic acid herbicide from acrylamide grafted jute polymer. Dhaka Univ. J. Sci. 68, 105–110. https://doi.org/10.3329/dujs.v68i2.54606 (2020).
Duval, A. & Lawoko, M. A review on lignin-based polymeric, micro-and nano-structured materials. React. Funct. Polym. 85, 78–96. https://doi.org/10.1016/J.REACTFUNCTPOLYM.2014.09.017 (2014).
Ragauskas, A. J. et al. Lignin valorization: improving lignin processing in the biorefinery. Science 344, 1246843. https://doi.org/10.1126/science.1246843 (2014).
Creteanu, A., Lungu, C. N. & Lungu, M. Lignin: an adaptable biodegradable polymer used in different formulation processes. Pharmaceuticals 17, 1406. https://doi.org/10.3390/ph17101406 (2024).
Restu, W. K. et al. The Preparation of nanoemulsion formulation from pelargonic acid in lignin carrier and its application to weeds. J. Inst. Eng. (India): E. 104, 297–303. https://doi.org/10.1007/s40034-023-00272-w (2023).
Khodadadi, F., Nikzad, M. & Hamedi, S. Lignin nanoparticles as a promising nanomaterial for encapsulation of Rose damascene essential oil: physicochemical, structural, antimicrobial and in-vitro release properties. Colloids Surf. A: Physicochem Eng. Asp. 687, 133580. https://doi.org/10.1016/j.colsurfa.2024.133580 (2024).
Weiss, R., Ghitti, E., Sumetzberger-Hasinger, M., Guebitz, G. M. & Nyanhongo, G. S. Lignin-Based pesticide delivery system. ACS Omega. 5, 4322–4329. https://doi.org/10.1021/acsomega.9b04275 (2020).
Varshney, M. et al. Pluronic microemulsions as nanoreservoirs for extraction of bupivacaine from normal saline. J. Am. Chem. Soc. 126, 5108–5112. https://doi.org/10.1021/ja0394479 (2004).
Rahdar, A. et al. Lignin-stabilized doxorubicin microemulsions: synthesis, physical characterization, and in vitro assessments. Polymers 13, 641. https://doi.org/10.3390/polym13040641 (2021).
Heydari, M. et al. Microemulsion of tribenuron-methyl using pluronic F127: Phsico-chemical characterization and efficiency on wheat weed. J. Mol. Liq. 326, 115263. https://doi.org/10.1016/j.molliq.2020.115263 (2021).
Virzì, N. F. et al. Berberine-styrene-co-maleic acid nanomicelles: unlocking opportunities for the treatment and prevention of bacterial infections. RSC Adv. 14, 34066–34080. https://doi.org/10.1039/D4RA04457F (2024).
Yu, M. et al. Development of functionalized abamectin Poly (lactic acid) nanoparticles with regulatable adhesion to enhance foliar retention. RSC Adv. 7, 11271–11280. https://doi.org/10.1039/C6RA27345A (2017).
Machado, T. O. et al. Bio-based lignin nanocarriers loaded with fungicides as a versatile platform for drug delivery in plants. Biomacromol 21, 2755–2763. https://doi.org/10.1021/acs.biomac.0c00487 (2020).
Jiang, T. Wang, Y. Yu, Z. & Du, L. Synthesis, characterization of chitosan/tripolyphosphate nanoparticles loaded with 4-chloro-2-methylphenoxyacetate sodium salt and its herbicidal activity against Bidens pilosa L. Sci Rep. 14, 18754 (2024) https://doi.org/10.1038/s41598-024-69438-9
Chen, Q. et al. Pickering emulsion via interfacial assembly of lignin particles and cationic surfactant: formation of robust anchoring layer. Colloids Surf. A: Physicochem Eng. Asp. 701, 134893. https://doi.org/10.1016/j.colsurfa.2024.134893 (2024).
Beckers, S., Peil, S. & Wurm, F. R. Pesticide-Loaded nanocarriers from lignin Sulfonates—A promising tool for sustainable plant protection. ACS Sustain. Chem. Eng. 8, 18468–18475. https://doi.org/10.1021/acssuschemeng.0c05897 (2020).
Jiang, F. et al. In situ Preparation of monodisperse lignin-poly (lactic acid) microspheres for efficient encapsulation of 2,4-dichlorophenoxyacetic acid and controlled release. React. Funct. Polym. 184, 105517. https://doi.org/10.1016/j.reactfunctpolym.2023.105517 (2023).
Gao, Y. et al. Controlled release of herbicides by 2,4-D-, MCPA-, and bromoxynil-intercalated hydrotalcite nanosheets. Green. Chem. 23, 4560. https://doi.org/10.1039/d1gc01349a (2021).
Acharya, A. & Pal, P. K. Agriculture nanotechnology: translating research outcome to field applications by influencing environmental sustainability. Nanolmpact 19, 100232. https://doi.org/10.1016/j.impact.2020.100232 (2020).
Oliveira, H. et al. (ed, C.) Nanoencapsulation enhances the post-emergence herbicidal activity of atrazine against mustard plants. PLoS One 10 e0132971 https://doi.org/10.1371/journal.pone.0132971 (2015).
Jacques, M. T., Oliveira, J. L., Campos, E. V. R., Fraceto, L. F. & Ávila, D. S. Safety assessment of nanopesticides using the roundworm Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 139, 245–253. https://doi.org/10.1016/j.ecoenv.2017.01.045 (2017). Epub 2017 Feb 3.
Oliveria, H. C. et al. Evaluation of the side effects of Poly (epsiloncaprolactone) nanocapsules containing atrazine toward maize plants. Front. Chem. 3, 61. https://doi.org/10.3389/fchem.2015.00061 (2015).
Pontes, M. S. et al. Chitosan/tripolyphosphate nanoformulation carrying paraquat: insights of its enhanced herbicidal activity. Environ. Sci. Nano. 8, 1336–1351. https://doi.org/10.1039/D0EN01128B (2021).
Xiang, Y., Zhang, G., Chi, Y., Cai, D. & Wu, Z. Fabrication of a controllable nanopesticide system with magnetic collectability. J. Chem. Eng. 328, 320–330. https://doi.org/10.1016/j.cej.2017.07.046 (2017).
Sipponen, M. H., Lang, H., Crestini, C., Henn, A. & Österberg, M. Lignin for Nano- and microscaled carrier systems: applications, trends, and challenges. ChemSusChem 12, 2039–2054. https://doi.org/10.1002/cssc.201900480 (2019).
Moreno, A. & Sipponen, M. H. Overcoming challenges of lignin nanoparticles: expanding opportunities for scalable and multifunctional nanomaterials. Acc. Chem. Res. 57, 1918–1930. https://doi.org/10.1021/acs.accounts.4c00206 (2024).
Abbas, A., Wang, Z., Zhang, Y., Peng, P. & she, D. Lignin-based controlled release fertilizers: A review. J. Biol. Macromol. 222, 1801–1817. https://doi.org/10.1016/j.ijbiomac.2022.09.265 (2022).
Gigli, M. et al. Lignin-based nano-enabled agriculture: A mini-review. Front. Plant. Sci. 13, 976410. https://doi.org/10.3389/fpls.2022.976410 (2022).
Author information
Authors and Affiliations
Contributions
A.R.Y. and A. R. methodology, A.R. and M.M; investigation, S.G.G, writing—original draft preparation, S.G.G, writing-review and editing, A.R.Y., A.R. and M.M.; All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ganzagh, S.G., Yousefi, A.R., Rahdar, A. et al. Lignin-based nanoencapsulation for sustainable herbicide delivery: controlled release and bioactivity of 2,4-D and MCPA compared to commercial formulations. Sci Rep 15, 24532 (2025). https://doi.org/10.1038/s41598-025-09467-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09467-0
Keywords
This article is cited by
-
Sustainable nanomaterials for precision dental medicine: green synthesis, therapeutic applications, and future directions
Journal of Nanobiotechnology (2026)