Abstract
Immunocompromised patients were excluded most of the time from trials testing corticosteroids in COVID-19. This study aimed to assess the associations between early corticosteroid use and (1) mortality at day 60, and (2) the occurrence of nosocomial infections in immunocompromised patients with severe COVID-19 admitted to the ICU. It was a multicentre retrospective study, achieved in French ICUs of the Outcomerea™ network and medical ICUs of 4 other hospitals in France. This study included immunocompromised patients admitted to an ICU between January 1, 2020, and August 31, 2022, for severe COVID-19, with an ICU stay of more than 2 days. Patients were classified as receiving early corticosteroid therapy if they were given steroids within the first 5 days following ICU admission. Each patient was categorized into one of four immunosuppression subgroups: ‘corticosteroid therapy,’ ‘monocytic alteration,’ ‘cellular immunosuppression,’ or ‘humoral immunosuppression.’ Survival analyses were performed, and confounding by indication was addressed using propensity score weighting with overlap. 383 patients were included, 50 were into the no-early-corticosteroids group and 333 in the early-corticosteroids group. In the overlap cohort, 118 were included (46 in the non-early-corticosteroids and 72 in the early-corticosteroids group). There was no association with day-60 mortality (IPTWoverlapHR = 0.97, 95% CI [0.51; 1.85], p = 0.92). There was also no association with the occurrence of nosocomial infections (IPTWoverlapSubHR = 2.59, CI 95% [0.77; 8.7], p = 0.13). We report that steroids had no benefit on mortality in immunocompromised patients admitted to ICU for severe COVID-19.
Similar content being viewed by others
Background
Corticosteroids are widely used to modulate inflammation in various conditions1. Early in the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, the RECOVERY trial demonstrated that dexamethasone reduced mortality in patients with severe COVID-192, and a meta-analysis3 confirmed the beneficial effects of steroids, leading the WHO to recommend corticosteroid administration in severe COVID-19. However, these recommendations may not apply to all subgroups of patients with severe COVID-19 pneumonia due to patient heterogeneity4. Additionally, corticosteroid therapy has been associated with an increased risk of nosocomial infections in COVID-19 patients5,6,7,8.
Immunocompromised patients constitute a subgroup of patients with their own characteristics9. They were most of the time excluded from therapeutic trials investigating corticosteroids in COVID-1910 (Table S1). There are no specific recommendations for immunocompromised patients admitted to the ICU11, particularly those with COVID-19 pneumonia12.
Early data in the pandemic suggested a high mortality among immunocompromised patients13, who are more prone to severe forms of the disease14, and therefore more likely to undergo intensive care15. Several reports have described the persistence of SARS-CoV-2 replication with severe symptoms for immunocompromised patients, including those with lymphoma16,17. In general, this group of patients also has no SARS-CoV-2 seroconversion18. Because of pre-existing immunosuppression, the effect of corticosteroids could be more harmful. Immunocompromised patients may respond differently to steroids compared to the general population due to: (1) an altered immune response, characterized by a reduced cytokine storm; (2) an increased risk of secondary infections, including opportunistic infections, reactivation of latent viral infections (e.g., cytomegalovirus [CMV], herpes simplex virus), and prolonged viral shedding; (3) interactions with immunosuppressive therapies; (4) a blunted vaccine response; (5) metabolic and systemic effects, such as uncontrolled hyperglycemia and an increased risk of adrenal insufficiency; and (6) musculoskeletal complications, including muscle loss and ICU-acquired weakness, particularly in patients already at high risk due to pre-existing sarcopenia. It therefore seems essential to determine the effect of corticosteroids in this patient population.
Against this background, the aim of our study was to evaluate the association between early corticosteroid therapy (early-CS) initiated as treatment for COVID-19 and prognosis in immunocompromised adults admitted to intensive care for COVID-19.
Materials and methods
Type of study and population
We conducted a multicentre retrospective study involving patients admitted to one of the French ICU (N = 11) of the Outcomerea™ network or to one of the medical ICUs of the university teaching hospitals of Créteil, Nantes, Strasbourg, and Montpellier.
The OUTCOMEREA™ cohort has already been described in details19. During the pandemic, patients over 16 years of age admitted to 11 French ICUs were included in this prospective, multicenter observational cohort. Their clinical and biological data were recorded in the database daily throughout their ICU stay. Additionally, during the pandemic, specific COVID-19-related data—such as the type and date of the first symptoms—were incorporated from the onset of the outbreak.
Population
Patients were eligible if they were admitted between January, 1 2020 and August 31, 2022 for severe COVID-19. They were included if they were immunocompromised, had a ICU length of stay (LOS) of more than 48 h, and were not transferred from another ICU. Patients were excluded if their immunosuppression type could not be identified retrospectively, and if they objected to the use of data from their clinical records.
Objectives of the study
The main objective was to assess the association between early-CS and mortality at day 60 in immunocompromised patients with a severe form of COVID-19 admitted to the ICU. The secondary objectives were to assess the association between early-CS and the duration of invasive mechanical ventilation and the occurrence of nosocomial infections in the same population. Subgroup analyses were performed according to the type of immunosuppression and period of admission.
Definitions
Diagnosis of SARS-CoV-2 infection
The diagnosis of SARS-CoV-2 pneumonia was confirmed by a positive PCR test, regardless of the patient’s serological status. Severe COVID-19 was defined according to WHO criteria as an oxygen saturation of less than 90% on room air (and by extension, the need for oxygen therapy).
Early-CS therapy for COVID-19 pneumonia
Patients were considered to have received early corticosteroid (early-CS) therapy if they were administered at least 6 mg of dexamethasone daily (or an equivalent dose: 160 mg of hydrocortisone hemi-succinate or 40 mg of prednisone) for two or more days within the first 5 days of their ICU stay. Patients in the “no early-CS group” received steroids for at most 1 day during the first 5 days after ICU admission, regardless of whether they had received steroids before ICU admission. Some patients in the “no-early-CS group” received steroids after day 5, but they remained classified in the “no-early-CS group”. Steroid administration occurring after day 5 was considered late steroid administration.
Immunosuppression
Each patient was classified by three independent physicians (BS, JD, LC) into one of the four subgroups of immunosuppression: “corticosteroid therapy” (CORTICO), “monocytic alteration” (MONO), “cellular immunosuppression” (CELL) and “humoral immunosuppression” (HUM). Agreement between at least two of the three was required (Table 1). This Classification has been proposed by specialists in critically ill immunocompromised patients and already used in several studies9. Patients who fulfilled criteria for several subgroups of immunosuppression were only included in one of the four. In addition, complementary analyses were performed by classifying patients according to the origin of immunosuppression: solid organ transplantation (SOT), hematologic malignancy, solid tumor, auto-immune diseases (AutoID) and human immune immunodeficiency virus (HIV).
Nosocomial infections
The presence or absence of hospital-acquired pneumonia (HAP), ventilator-associated pneumonia (VAP), and ICU-bloodstream infections (ICU-BSI) was documented following the standard definitions set by the Centers for Disease Control and Prevention (CDC)20. To diagnose HAP-VAP or ICU-BSI, quantitative cultures of specimens were required. Nosocomial infections, including HAP-VAP and ICU-BSI, were included in the analysis. Early-onset VAP was defined as occurring within the first 7 days after mechanical ventilation initiation, while late-onset VAP occurred after this period21. The at-risk period for VAP was considered from 48 h after intubation until the removal of the tracheal tube and weaning from invasive ventilation.
The occurrence of viral reactivations, such as herpes simplex virus (HSV) and cytomegalovirus (CMV), was also reported. In immunocompromised patients during the pandemic, all collections testing positive for CMV and/or HSV were treated22. Additionally, the occurrence of COVID-19-associated pulmonary aspergillosis (CAPA) was documented. CAPA was classified as possible or probable according to established guidelines23. During the pandemic in France, infection monitoring was conducted via weekly serum PCR tests for HSV and CMV, Beta-D-glucan, and galactomannan, as well as in bronchoalveolar lavage samples when available.
Throughout the COVID-19 pandemic in France, different SARS-CoV-2 variants have predominated over time: (1) in early 2020, the original SARS-CoV-2 strain was dominant; (2) from late 2020 to early 2021, the Alpha variant (B.1.1.7) became predominant; (3) from mid to late 2021, the Delta variant (B.1.617.2) took over; (4) from late 2021 to the present, the Omicron variant (B.1.1.529) and its sub-lineages have remained dominant; and (5) currently, the KP.3.1.1 sub-lineage of Omicron is the most prevalent strain.
Collection
Patient data was collected either on admission comprising basic patient characteristics such as age, sex, chronic diseases, relevant comorbidities as defined by the Knaus Scale24, usual treatment, elements characterizing their COVID-19, i.e. date of onset of symptoms, initial management (corticosteroids, other immunosuppressants, antivirals and oxygen support), illness severity on admission as assessed by simplified acute physiology score II (SAPS II)25, sequential organ failure assessment score (SOFA)26, organ support, and biological parameters including, when available, data characterizing the patient’s immune status (leukocytes, neutrophils, lymphocytes, monocytes, C-reactive protein, ferritin, procalcitonin, and hemostasis [D-dimer, fibrinogen]), or collected during the patient’s stay in the ICU, including treatment with corticosteroids, organ support, the occurrence of nosocomial infections and patient outcome based on ICU and hospital LOS, and mortality at ICU and hospital discharge, and at day 60 (D60).
Statistical analyses
Patient characteristics were expressed as n (%) for categorical variables and median (interquartile range [IQR]) for continuous variables. Comparisons were made with exact Fisher tests for categorical variables and Wilcoxon tests for continuous variables.
The primary outcome measure was Day-60 mortality. We employed a Cox model with propensity score weighting with overlap weights to account for potential confounding by indication27. Overlap weights target the population which mimics the characteristics of a pragmatic randomized trial. Moreover, they have been shown to overcome some of the limitations of other weighting formulas such as Inverse Probability of Treatment Weighting (IPTW), and optimize precision of the treatment effect estimate among propensity score weighting methods28. Henceforth, we will refer to this adjustment methodology of propensity score weighting with overlap weights simply as “overlap weighting”. The propensity score was estimated using a logistic regression model that included covariates that could influence the decision to use early steroids and affect prognosis. These covariates were selected through a dual approach, combining clinical expertise with statistical analysis to ensure comprehensive confounding control29. Finally, we included the following covariates in the weight model: period of inclusion, age, gender, body mass index (BMI), comorbidities, organ supports including invasive mechanical ventilation, laboratory features, including PaO2/FiO2 ratio, type of immunosuppression and other immunomodulatory treatments on inclusion30,31,32. To avoid extreme weights, weights were truncated at the 10-90th percentile33.
We then used a weighted Cox proportional-hazard model into the population that overlapped their PS, to estimate the risk of death within the first 60 days of ICU stay of early-CS. The proportionality of hazard risk for early-CS was tested using martingale residuals. All the analyses were stratified according to centers.
The analyses were carried out similarly for the risks of nosocomial infection, however, because of the competing risks of living alive from ICU and /or death, we used a Fine and Gray sub-distribution hazard models34 with ICU discharge and death as competing risk instead of Cox models35.
We assessed the robustness of our findings using multiple sensitivity analyses, into the whole initial population: (1) a simple matching based on covariates at admission; (2) matching on propensity score and (3) a raw multivariable Cox proportional-hazard model. Similar analyses were performed in subgroups defined by the type of immunosuppression. Subgroup analyses were also performed on the entire cohort after excluding patients who received only a single day of steroids at ICU admission. Interactions between subgroups were tested.
For all tests, the level of significance was set at 5%. Missing data were imputed by multiple imputation with a dataset, using PROC MI in SAS. All statistical analyses were performed using SAS software, version 9.4 (SAS Institute, Cary, NC) and R (Version 3.4.0; R Core Team, Wien, Austria).
Results
Population characteristics
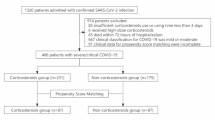
The study involved 383 immunosuppressed patients with an ICU LOS of more than 48 h (Fig. S1) and distributed as follows: CELL (N = 237), CORTICO (N = 29), HUM (N = 107), MONO (N = 10). Overall, 110 patients had SOT, 73 a solid tumor, 111 hematologic malignancy, 75 AutoID, and 14 HIV. The distribution and characteristics of the patients by centers are given in Fig. S2 and Table S2.
Comparison according to steroid administration for severe COVID-19
In our cohort, 333 patients were included in the early-CS subgroup. Their characteristics are reported in Table 2. Most of them received only DXM (Fig. S3), for 10 days (Figs. S4–S6). The patients from the early-CS group presented a higher incidence of cardio-vascular disease (early-CS: 59.5% vs. 36%, p < 0.01), were less severely ill (SAPS II: early-CS: 38 [31; 48] versus 43.5 [29; 60], p = 0.04; SOFA : early-CS: 4 [3; 6] versus 5 [4; 9], p < 0.01)), were more hypoxemic (PaO2/FiO2: Early-CS: 112.9 [83.1; 176.7] versus no-early-CS group: 144 [87; 309.5], p < 0.01), but were more often under high flow nasal canula (HFNC) on admission (early-CS: 248 (78.9%) vs 29 (52%), p < 0.01). The patients of the no-early-CS group could receive steroids (16% with DXM) but for less than 2 days. Patients of the early-CS developed more often VAP (24.7% vs 4%, p < 0.01), had a longer ICU LOS (11 [6; 20] vs 6 [4; 13], p < 0.01) but similar ICU and day-60 mortality. The distribution of the patients according to steroid treatment duration is shown in Figs. S4 and S5. Table S3 reports the description of the 11 patients of the non-early-CS subgroup that received only 1 day of steroids.
Comparison according to the subgroups of immunosuppression
The details of steroid duration by subgroup of immunosuppression are given in Fig. S6. The groups had quite similar characteristics but differed on some points. For instance, patients of the MONO subgroup were older (p = 0.01), those in the HUM group had a lower BMI, and the CELL group patients had a higher incidence of chronic kidney disease (p < 0.01). Patients in the MONO subgroup were more severely ill according to SOFA score (p < 0.01), and in the HUM and MONO groups more often under HFNC on admission. However, the admission PaO2/FiO2 ratio did not differ between groups. Convalescent plasma therapy was mostly given to patients from the HUM and MONO subgroups, but in less than 25% of the cases. None of the main outcomes including occurrence of nosocomial infections, ICU and hospital LOS, and mortality at any study endpoint differed between subgroups (Table 3). The characteristics of the patients according to the origin of the immunosuppression are given in Table S4.
Clinical outcomes
Mortality at day 60 before propensity score weighting
Patients not alive until D60 were older (69.6 years [63.2; 75] vs. 62.6 [55.6; 70], p < 0.01), had a higher incidence of comorbidities including chronic cardiovascular disease (89 [63.1%] vs. 127 [52.5%], p = 0.04) and chronic kidney disease (57 [40.4%] vs. 63 [26%], p < 0.01). They were also more severely ill on admission and required more often invasive mechanical ventilation and vasopressors. They had lymphopenia more often, and had higher plasma D-dimer, and serum ferritin values. They also developed nosocomial infections more often during their ICU stay (Table 4).
Mortality at day 60 after propensity score weighting
In that analysis, 118 patients were included, 72 in the early-CS and 46 in the no-early-CS (Tables 5 and S5). All the descriptions of the propensity score are reported in Figs. S7–S10, including standardized Mean Differences (SMDs), before and after weighting (Fig. S10), and weights with and without truncation (Table S6).
After propensity score weighting, early CS administration was not associated with an increased risk of death (IPTWoverlapHR = 0.97, 95% CI [0.51; 1.85], p = 0.92) (Figs. 1 and 2; Table S7). No association was found in all subgroups (Cell, Humoral, Solid Organ Transplantation, before or after august 2020, ICU LOS > 5 days) (Table S7, Fig. 1). No association was found into the sensitive analyses (Tables S8–S10).
Association between early-CS and (A) day-60 mortality*, (B) nosocomial infections**, in the whole cohort and in the different subgroups. CELL cellular immunodepression, SOT solid organ transplantation, ICU intensive care unit. *Cox model using propensity score weighting with overlap weights. **Fine Gray sub-distribution models using propensity score weighting with overlap weights.
Association between early-CS and (A) day-60 mortality*, (B) nosocomial infections**, in the whole cohort. HR hazard ratio. * Cox model using propensity score weighting with overlap weights. ** Fine Gray sub-distribution models using propensity score weighting with overlap weights.
Nosocomial infection occurrence before propensity score weighting
The distribution of the microorganisms identified during the first nosocomial infection are reported in Table S11. Most of the nosocomial infections were secondary to VAP. In our cohort, the only reported fungal infections were cases of CAPA (N = 30). The distribution of CAPA cases was similar between patients who did and did not receive corticosteroids during their ICU stay.
Patients who acquired nosocomial infections during their ICU stay had more chronic cardiovascular disease, were more often on invasive mechanical ventilation and vasopressors on ICU admission, received more steroids, and experienced worse outcomes including ICU LOS and mortality (Table 4).
Nosocomial infection occurrence after propensity score weighting
After propensity score weighting, early CS was not associated with an increased risk of nosocomial infection during ICU stay (IPTWoverlapSubHR = 2.59, CI 95% [0.77; 8.7], p = 0.13) (Fig. 1 and 2; Tables S5–S7). No association was found in subgroups nor into the sensitive analyses (Fig. 1, Tables S7–S10), Only trends in favor of increased risk of nosocomial infection due to early-CS could be observed.
Discussion
This study is one of the first to investigate the impact of steroids on immunocompromised patients with severe COVID-19 who were admitted to the ICU. Our cohort included 62% of patients with cellular immunodeficiency, 28% with humoral immunodeficiency, 7% who were receiving steroids, and 3% with altered monocyte levels. At the time of ICU admission, most patients were on steroids, primarily dexamethasone. Steroid use did not reduce mortality, nor was associated with an increased risk of nosocomial infections.
These findings warrant several important considerations.
First, it is one the first study to include only immunocompromised critically ill patients. Other studies included hospitalized COVID-19 immunocompromised patients and mainly focused on the consequence of immunodepression itself36,37. For instance, Turtle et al.15, based on the WHO ISARIC CCP-UK prospective cohort, reported higher mortality among immunocompromised individuals (29% vs. 21%) and identified an increased mortality risk (aOR 1.44, p < 0.01), but a decreased likelihood of ICU admission and intubation.
Then, our study provides a snapshot of how immunocompromised patients were treated in France between January 2020 and August 2022. In our cohort, 87% of patients received steroids, while 18% received anti-Il6 therapies. No patients received anti-JAK 2 inhibitors, nor did any benefit from adsorption therapy. The high use of steroids likely reflects adherence to guidelines, whereas the lower use of anti-Il6 therapies may suggest caution due to the potential risks of further immune suppression. Only 10% of patients received convalescent plasma, primarily those with humoral deficiencies, despite studies showing benefits of convalescent plasma for immunosuppressed patients38, particularly those with B-cell malignancies39. Similarly, only 6% received remdesivir, despite evidence demonstrating its benefit40,41. This may be explained by contraindications in patients with renal failure, as well as delayed recommendations for its use42,43. Finally, less than 1% of our patients received a combination of antivirals, steroids, and passive immunotherapy with hyperimmune convalescent plasma. This combination of treatments has shown promising results and has now been adopted for most immunocompromised patients44.
In our study, steroid treatment for severe COVID-19 in immunocompromised patients did not improve vital outcomes. Until nowadays, few studies have evaluated the impact of immunomodulators in immunocompromised patients, mainly focusing on anti-Il1 and anti-Il6 therapies45,46,47, but not steroids. For example, a cohort of non-ICU hospitalized solid organ transplant patients receiving tocilizumab (24.8%) showed no increased mortality (41% vs. 28%, p = 0.27) or secondary infections (34% vs. 24%, p = 0.55)47. More recently, a meta-analysis was achieved10. Ultimately, 11 trials with 397 immunocompromised patients were included, showing no significant difference in mortality, secondary infections, or change in outcomes between those treated with immunomodulators and controls. Notably, all patients in these trials received steroids regardless of immunomodulator use. The lack of benefit from immunomodulatory treatment may be due, in part, to delayed adaptive responses, prolonged viral replication43,44, and the occurrence of nosocomial infections, including reactivation of latent infections. Additionally, factors such as worsening adrenal insufficiency, exacerbation of pre-existing sarcopenia with muscle loss, and the development of ICU-acquired weakness may have contributed. Co-infections and secondary infections are a significant issue for patients with severe COVID-195,7,8. Immunocompromised patients are particularly susceptible to such complications48,49. Such a risk was well illustrated, in India, by the increase mucormycosis rates among kidney transplant recipients with COVID-19 surged, with incidences ranging from 4.4% to 10%44,49,50.
Due to the inconsistent results and limited quality of studies on steroid use, there is no strong recommendation for the use of immunomodulatory therapies in immunocompromised patients with severe COVID-19 admitted to the ICU. However, the Centers for Disease Control and Prevention (CDC) guidelines for immunocompromised COVID-19 patients recommend using immunomodulatory therapies at the same doses and durations as for the general population43.
Immunocompromised patients remain at high risk for severe COVID-19, despite widespread vaccination, natural immunity, and the emergence of new viral variants. The primary reasons include a reduced ability to mount an effective immune response51, decreased vaccine efficacy44,52,53, and prolonged viral shedding.
In this context, the use of steroids in immunocompromised COVID-19 patients—as well as in future viral pandemics, particularly those caused by respiratory viruses—requires caution54. Contemporary COVID-19 and other viral infections may trigger varying immune responses, and not all involve a similar hyperinflammatory phase. As seen with influenza, steroids may be associated with prolonged viral shedding and an increased risk of secondary infections.
The use of steroids in immunocompromised patients with septic acute respiratory failure remains a complex issue. It is well established that low-dose corticosteroids reduce mortality in critically ill, non-immunocompromised patients with severe community-acquired bacterial pneumonia (CAP)55. Steroids have also demonstrated benefits in critically ill patients with respiratory infections, septic shock, or acute respiratory distress syndrome (ARDS)56. However, in most randomized controlled trials assessing steroid use, immunocompromised patients represent only a small proportion or are entirely excluded. Notably, a study by Meduri et al.57 included 20% immunocompromised patients but did not find a protective effect, unlike other studies that reported benefits from steroids but included fewer than 6% immunocompromised patients55,58,59.
This study has both strengths and limitations.
Among its strengths, it is a multicenter study with a large cohort of immunocompromised ICU patients. Although the analysis was retrospective, it relied on prospectively collected data. We accounted for multiple confounding factors, including the period of admission, applied propensity score methods to minimize bias, and conducted sensitivity analyses to ensure robustness.
However, the study also has several limitations. The primary concern is the small sample size of the non-steroid group (only 50 patients), which may lead to a type I error. Additionally, there were baseline differences between the steroid and non-steroid groups, including the inclusion period and the severity of hypoxemia at ICU admission. To mitigate these biases, we used inverse probability of treatment weighting (IPTW) with overlap, which is particularly robust when handling imbalanced sample sizes and reduces information loss. Second, our analysis accounted for admission period, IMV, PaO₂/FiO₂ ratio, and anti-interleukin treatments, including anti-IL-6 and anti-IL-1 therapies. However, some potential confounders—such as viral variants, vaccination status, and other treatments including anti-SARS-CoV-2 antibodies, plasmapheresis, C5 inhibitors, Janus kinase inhibitors, and antiviral therapies—were not directly included. Nonetheless, these factors are closely associated with the admission period. Third, some key outcomes were not collected, such as longitudinal viral load measurements. Fourth, the classification of immuno-compromised status may be debated18; however, we also provided results based on more conventional classifications, including solid organ transplantation and hematologic malignancies. Other subgroup analyses were not feasible due to sample size limitations.
Fifth, immortality bias may have occurred. Patients in the early corticosteroid (CS) group may have had a survival advantage, as they had to survive at least 2 days to receive treatment (defined as at least two doses of steroids within the first 5 days). However, only a small proportion of patients in the early-CS group did not receive steroids on days 1 and 2 (N = 8/333; 2.5%). To address this, we conducted a sensitivity analysis including only patients who survived beyond day 5, which yielded consistent results. Finally, we did not perform a survival analysis differentiating between the types of nosocomial infections, specifically bacterial versus fungal.
Given these limitations, our retrospective design restricts causal inference, meaning we can only report associations, and all findings should be interpreted with caution.
Conclusion
Our findings showed that steroids had no beneficial effect on mortality in immunocompromised patients admitted to the ICU for severe COVID-19. At the present time we would advise to pursue steroids for the patients on long term steroids but not for the other. Large, randomized control studies, including immunocompromised patients, should be performed to better identify the indications and dose of corticosteroids not only for the patients with COVID-19, but also for the patients with CAP and/or ARDS.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ARDS:
-
Acute respiratory distress syndrome
- AutoID:
-
Auto-immune diseases
- CAP:
-
Community-acquired bacterial pneumonia
- CAPA:
-
COVID-19-associated pulmonary aspergillosis
- CDC:
-
Centers for Disease Control and Prevention
- CELL:
-
Cellular immunosuppression
- CMV:
-
Cytomegalovirus
- CORTICO:
-
Corticosteroid therapy
- D60:
-
Day 60
- DXM:
-
Dexamethasone
- Early-CS:
-
Early corticosteroid therapy
- HAP:
-
Hospital-acquired pneumonia
- HFNC:
-
High flow nasal canula
- HIV:
-
Human immune immunodeficiency virus
- HSV:
-
Herpes simplex virus
- HUM:
-
Humoral immunosuppression
- ICU:
-
Intensive care unit
- ICU-BSI:
-
ICU-bloodstream infections
- LOS:
-
Length of stay
- MONO:
-
Monocytic alteration
- SAPS II:
-
Simplified acute physiology score II
- SARS-CoV2:
-
Severe acute respiratory syndrome coronavirus 2
- SOFA:
-
Sequential organ failure assessment score
- SOT:
-
Solid organ transplantation
- SRLF:
-
Société Réanimation Langue Française
- VAP:
-
Ventilator-associated pneumonia
References
Rhen, T. & Cidlowski, J. A. Antiinflammatory action of glucocorticoids–new mechanisms for old drugs. N. Engl. J. Med. 353, 1711–1723 (2005).
ECOVERY Collaborative Group et al. Dexamethasone in hospitalized patients with Covid-19. N. Engl. J. Med. 384, 693–704 (2021).
The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: A meta-analysis. JAMA 324, 1330 (2020).
Bruse, N. et al. Clinical phenotyping uncovers heterogeneous associations between corticosteroid treatment and survival in critically ill COVID-19 patients. Intensive Care Med. 50, 1884–1896 (2024).
Wicky, P.-H. et al. Ventilator-associated pneumonia in COVID-19 patients admitted in intensive care units: relapse, therapeutic failure and attributable mortality-A multicentric observational study from the OutcomeRea network. J. Clin. Med. 12, 1298 (2023).
Laurichesse, G. et al. Mortality, incidence, and microbiological documentation of ventilated acquired pneumonia (VAP) in critically ill patients with COVID-19 or influenza. Ann. Intensive Care 13, 108 (2023).
Buetti, N. et al. COVID-19 increased the risk of ICU-acquired bloodstream infections: A case-cohort study from the multicentric OUTCOMEREA network. Intensive Care Med. 47, 180–187 (2021).
Rouzé, A. et al. Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: A European multicenter cohort study. Intensive Care Med. 47, 188–198 (2021).
Azoulay, E. et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensive Care Med. 46, 298–314 (2020).
Siempos, I. I. et al. Immunomodulators for immunocompromised patients hospitalized for COVID-19: A meta-analysis of randomized controlled trials. eClinicalMedicine 69, 102472 (2024).
Chaudhuri, D. et al. 2024 focused update: Guidelines on use of corticosteroids in sepsis, acute respiratory distress syndrome, and community-acquired pneumonia. Crit. Care Med. 52, e219–e233 (2024).
Agarwal, A. et al. A living WHO guideline on drugs for covid-19. BMJ 370, m3379 (2020).
Pereira, M. R. et al. COVID-19 in solid organ transplant recipients: Initial report from the US epicenter. Am. J. Transplant. 20, 1800–1808 (2020).
Passamonti, F. et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: A retrospective, multicentre, cohort study. Lancet Haematol. 7, e737–e745 (2020).
Turtle, L. et al. Outcome of COVID-19 in hospitalised immunocompromised patients: An analysis of the WHO ISARIC CCP-UK prospective cohort study. PLoS Med 20, e1004086 (2023).
Duléry, R. et al. Prolonged in-hospital stay and higher mortality after Covid-19 among patients with non-Hodgkin lymphoma treated with B-cell depleting immunotherapy. Am. J. Hematol. 96, 934–944 (2021).
Nakajima, Y. et al. Prolonged viral shedding of SARS-CoV-2 in an immunocompromised patient. J. Infect. Chemother. 27, 387–389 (2021).
Kim, C. S. et al. Nosocomial outbreak of COVID-19 from a Kidney transplant patient: Necessity of a longer isolation period in immunocompromised patients. Infect. Chemother. 55, 42–49 (2023).
Planquette, B. et al. Pseudomonas aeruginosa ventilator-associated pneumonia. Predictive factors of treatment failure. Am. J. Respir. Crit. Care Med. 188, 69–76 (2013).
Calandra, T., Cohen, J., International Sepsis Forum Definition of Infection in the ICU Consensus Conference. The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit. Care Med. 33, 1538–1548 (2005).
Vacheron, C.-H. et al. Increased incidence of ventilator-acquired pneumonia in coronavirus disease 2019 patients: A multicentric cohort study. Crit. Care Med. 50, 449–459 (2022).
Boers, L. S. et al. Pulmonary herpes simplex virus and cytomegalovirus in patients with acute respiratory distress syndrome related to COVID-19. Intensive Care Med. 50, 1251–1264 (2024).
Koehler, P. et al. Defining and managing COVID-19-associated pulmonary aspergillosis: The 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. Lancet Infect. Dis. 21, e149–e162 (2021).
Knaus, W. A. et al. The APACHE III prognostic system. Chest 100, 1619–1636 (1991).
A New Simplified Acute Physiology Score (SAPS II) Based on a European/North American Multicenter Study |JAMA| JAMA Network. https://jamanetwork-com.ezproxy.u-paris.fr/journals/jama/article-abstract/409979.
Vincent, J.-L., de Mendonca, A., Cantraine, F., Moreno, R., Takala, J., Suter, P. M., Sprung, C. L., Colardyn, F., Blecher, S. Ovid: Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: Results of a multicenter, prospective study. https://ovidsp-dc1-ovid-com.ezproxy.u-paris.fr/ovid-b/ovidweb.cgi?&S=OKIAFPDKLAACMGAGKPMJJHPMBIPFAA00&Link+Set=S.sh.22.23.27.39%7c16%7csl_10.
Li, F., Thomas, L. E. & Li, F. Addressing Extreme propensity scores via the overlap weights. Am. J. Epidemiol. 188, 250–257 (2019).
Thomas, L. E., Li, F. & Pencina, M. J. Overlap weighting: A propensity score method that mimics attributes of a randomized clinical trial. JAMA 323, 2417–2418 (2020).
Austin, P. C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav. Res. 46, 399–424 (2011).
Austin, P. C. & Stuart, E. A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med. 34, 3661–3679 (2015).
Moore, K. L., Neugebauer, R., Laan, M. J. & Tager, I. B. Causal inference in epidemiological studies with strong confounding. Stat. Med. 31, 1380–1404 (2012).
Austin, P. C., Grootendorst, P. & Anderson, G. M. A comparison of the ability of different propensity score models to balance measured variables between treated and untreated subjects: A Monte Carlo study. Stat. Med. 26, 734–753 (2007).
Hernán, M. A. & Robins, J. M. Causal Inference. (2016).
Fine, J. P. & Gray, R. J. A Proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc. 94, 496–509 (1999).
Wolkewitz, M., Cooper, B. S., Bonten, M. J. M., Barnett, A. G. & Schumacher, M. Interpreting and comparing risks in the presence of competing events. BMJ 349, g5060 (2014).
García-Suárez, J. et al. Impact of hematologic malignancy and type of cancer therapy on COVID-19 severity and mortality: Lessons from a large population-based registry study. J. Hematol. Oncol. 13, 133 (2020).
Baek, M. S., Lee, M.-T., Kim, W.-Y., Choi, J. C. & Jung, S.-Y. COVID-19-related outcomes in immunocompromised patients: A nationwide study in Korea. PLoS ONE 16, e0257641 (2021).
Hueso, T. et al. Convalescent plasma improves overall survival in patients with B-cell lymphoid malignancy and COVID-19: A longitudinal cohort and propensity score analysis. Leukemia 36, 1025–1034 (2022).
Lacombe, K. et al. Use of covid-19 convalescent plasma to treat patients admitted to hospital for covid-19 with or without underlying immunodeficiency: Open label, randomised clinical trial. BMJ Med. 2, e000427 (2023).
Gottlieb, R. L. et al. Early remdesivir to prevent progression to severe Covid-19 in outpatients. N. Engl. J. Med. 386, 305–315 (2022).
Mozaffari, E. et al. Remdesivir reduced mortality in immunocompromised patients hospitalized for COVID-19 across variant waves: findings from routine clinical practice. Clin. Infect. Dis. 77, 1626–1634 (2023).
Wu, M. Y. et al. WHO’s therapeutics and COVID-19 living guideline on mAbs needs to be reassessed. The Lancet 400, 2193–2196 (2022).
Immunocompromised. COVID-19 Treatment Guidelines https://www.covid19treatmentguidelines.nih.gov/special-populations/immunocompromised/.
Shoham, S. et al. Vaccines and therapeutics for immunocompromised patients with COVID-19. EClinicalMedicine 59, 101965 (2023).
Yamani, A. H. et al. Early use of tocilizumab in solid organ transplant recipients with COVID-19: A retrospective cohort study in Saudi Arabia. Immun. Inflamm. Dis. 10, e587 (2022).
Bodro, M. et al. Use of anti-cytokine therapy in kidney transplant recipients with COVID-19. J. Clin. Med. 10, 1551 (2021).
Pereira, M. R. et al. Tocilizumab for severe COVID-19 in solid organ transplant recipients: A matched cohort study. Am. J. Transplant. 20, 3198–3205 (2020).
Gudiol, C. et al. Co-infections and superinfections complicating COVID-19 in cancer patients: A multicentre, international study. J. Infect. 83, 306–313 (2021).
Bansal, S. B. et al. Risk factors and outcomes of COVID associated mucormycosis in kidney transplant recipients. Transpl. Infect. Dis. 24, e13777 (2022).
Meshram, H. S., Kumar, D. & Kute, V. B. Rare and unusual follow-up sequelae of coronavirus disease 2019: Splenic Mucormycosis in a renal transplant recipient. Transplant. Proc. 54, 1554–1556 (2022).
Antinori, A. & Bausch-Jurken, M. The burden of COVID-19 in the immunocompromised patient: Implications for vaccination and needs for the future. J. Infect. Dis. 228, S4–S12 (2023).
Fung, M. & Babik, J. M. COVID-19 in immunocompromised hosts: What we know so far. Clin. Infect. Dis. 72, 340–350 (2021).
Galmiche, S. et al. Immunological and clinical efficacy of COVID-19 vaccines in immunocompromised populations: A systematic review. Clin. Microbiol. Infect. 28, 163–177 (2022).
Goldman, J. D., Robinson, P. C., Uldrick, T. S. & Ljungman, P. COVID-19 in immunocompromised populations: Implications for prognosis and repurposing of immunotherapies. J. Immunother. Cancer 9, e002630 (2021).
Dequin, P.-F. et al. Hydrocortisone in severe community-acquired pneumonia. N. Engl. J. Med. 388, 1931–1941 (2023).
Pirracchio, R., Venkatesh, B. & Legrand, M. Low-dose corticosteroids for critically ill adults with severe pulmonary infections: A review. JAMA 332, 318–328 (2024).
Meduri, G. U. et al. Low-dose methylprednisolone treatment in critically ill patients with severe community-acquired pneumonia. Intensive Care Med. 48, 1009–1023 (2022).
Torres, A. et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: A randomized clinical trial. JAMA 313, 677–686 (2015).
Blum, C. A. et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: a Multicentre, double-blind, randomised, placebo-controlled trial. Lancet 385, 1511–1518 (2015).
Acknowledgements
We thank Jeffrey WATTS for the medical writing. We thank all the members of the Outcomerea network. OUTCOMEREA Study Group: Jean-François Timsit (E mail: jean-francois.timsit@aphp.fr), Elie Azoulay, Maïté Garrouste-Orgeas, Jean-Ralph Zahar, Bruno Mourvillier, Michael Darmon, Christophe Clec’h, Corinne Alberti, Stephane Ruckly, Sébastien Bailly, Aurélien Vannieuwenhuyze, Romain Hernu, Christophe Adrie, Carole Agasse, Bernard Allaouchiche, Olivier Andremont, Pascal Andreu, Laurent Argaud, Claire Ara-Somohano, Elie Azoulay, Francois Barbier, Déborah Boyer, Jean-Pierre Bedos, Thomas Baudry, Jérome Bedel, Julien Bohé, Lila Bouadma, Jeremy Bourenne, Noel Brule, Cédric Brétonnière, Charles Cerf, Frank Chemouni, Christine Cheval, Julien Carvelli, Elisabeth Coupez, Martin Cour, Claire Dupuis, Etienne de Montmollin, Loa Dopeux, Anne-Sylvie Dumenil, Jean-Marc Forel, Marc Gainnier, Charlotte Garret, Dany Goldgran-Tonedano, Steven Grangé, Antoine Gros, Hédia Hammed, Akim Haouache, Romain Hernu, Tarik Hissem, Vivien Hong Tuan Ha, Sébastien Jochmans, Jean-Baptiste Joffredo, Hatem Kallel, Guillaume Lacave, Virgine Laurent, Alexandre Lautrette, Clément Le bihan Eric Magalhaes, Virgine Lemiale, Guillaume Marcotte, Jordane Lebut, Maxime Lugosi, Sibylle Merceron, Benoît Misset, Mathilde Neuville, Laurent Nicolet, Johanna Oziel, Laurent Papazian, Juliette Patrier, Benjamin Planquette, Aguila Radjou, Marie Simon, Romain Sonneville, Jean Reignier, Bertrand Souweine, Carole Schwebel, Shidasp Siami, Romain Sonneville, Nicolas Terzi, Gilles Troché, Marie Thuong, Guillaume Thierry, Marion Venot, Sondes Yaacoubi, Olivier Zambon, Julien Fournier, Stéphanie Bagur, Mireille Adda, Vanessa Vindrieux, Sylvie de la Salle, Pauline Enguerrand, Vincent Gobert, Stéphane Guessens, Helene Merle, Nadira Kaddour, Boris Berthe, Samir Bekkhouche, Kaouttar Mellouk, Mélaine Lebrazic, Carole Ouisse, Diane Maugars, Christelle Aparicio, Igor Theodose, Manal Nouacer, Veronique Deiler, Fariza Lamara, Myriam Moussa, Atika Mouaci and Nassima Viguier.
Funding
No funding to declare.
Author information
Authors and Affiliations
Contributions
CD, JFT, JD, BS conceived and designed the study. CD, JFT, KK, LC, JD, MN, SS, YC, VL, BM, DGT, CS, SR, FM, MR, AM collected data. CD, BS, JFT analyzed and interpreted data. CD, BS, JFT drafted the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the ethics committee of the Société de Réanimation de Langue Française (SRLF) (Institutional Review Board (IRB) 00014135) (Project number: 22-060) (Project number: 22-060) and complied with the Declaration of Helsinki and good clinical practice. Due to the retrospective nature of the study, the need for approval was waived by the IRB.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dupuis, C., Timsit, JF., Domitile, J. et al. Corticosteroids in immunocompromised ICU patients with severe COVID-19: a multicenter retrospective study. Sci Rep 15, 27252 (2025). https://doi.org/10.1038/s41598-025-10864-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-10864-8