Abstract
Myofascial pain syndrome (MPS) is a common chronic pain condition characterized primarily by the presence of myofascial trigger points (MTrPs). While the insulin-like growth factor-1 receptor (IGF-1R) is known to be upregulated in injured muscles and implicated in orofacial neuropathic pain, its role in the peripheral mechanisms underlying MTrPs remains poorly understood. In this study, we aimed to investigate the expression of IGF-1R in MTrPs and explore the molecular mechanisms by which IGF-1R activation induces pain-like behavior in a rat model of MTrPs. We used Sprague-Dawley rats to assess IGF-1R signaling, measuring pain-like behaviors with the Randall-Selitto test. Muscle tissue morphology was examined using hematoxylin and eosin (HE) staining, while IGF-1R-related proteins were quantified through immunohistochemistry (IHC) and Western blot (WB) analysis. Our results showed that IGF-1R expression is significantly elevated in the muscle tissue of rats with MTrPs, with this upregulation correlating positively with the hyperalgesia. Furthermore, activation of IGF-1R was found to induce pain-like behavior via the phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway. Notably, inhibition of PI3K/AKT reversed the pain-like behaviors triggered by IGF-1R activation. These findings suggest that the increased expression of IGF-1R and subsequent activation of the PI3K/AKT/mTOR pathway may contribute to heightened nociception in MTrPs, offering new insights into the molecular mechanisms underlying MPS and potential targets for therapeutic intervention.
Similar content being viewed by others
Introduction
Myofascial pain syndrome (MPS) is a localized chronic pain condition caused by myofascial trigger points (MTrPs) within the patient’s muscles1. Previous study has shown that MTrPs is the main cause of musculoskeletal pain, with its prevalence ranging up to 85% across the entire lifespan2. MTrPs are defined as palpable taut bands (TBs) within skeletal muscle that are sensitive to pain3. MTrPs are classified into two types based on whether they cause spontaneous pain: latent and active. It has been reported that latent MTrPs can become active if the muscle remains contracted for an extended period4,5. Myofascial trigger points not only directly induce pain but also cause autonomic responses such as changes in skin temperature, sweating, and tearing6. Many confounding factors are related to the presence of MTrPs, including hypovitaminosis, posture, fatigue, sleep disorders, and trauma7. MTrPs are prone to occur in the scalene muscles, scapular muscles, infraspinatus, and levator scapulae of the upper body8,9. MTrPs in taut bands can be identified using palpation, seen as a local twitch response or other specific types of pain10,11.
Receptor tyrosine kinases (RTKs) are implicated in signal transduction underlying central mechanisms of chronic pain, yet their role in peripheral mechanisms of myofascial trigger points (MTrPs) remains elusive. In our subsequent investigation, we identified that the RTK family potentiates nociceptive sensitization and sarcomere contraction in MTrPs, demonstrating its pivotal role in MTrPs pathogenesis12,13,14. The insulin-like growth factor 1 receptor (IGF-1R) is a membrane protein with tyrosine kinase activity. Its structure includes an extracellular region, which consists of an α-subunit responsible for recognizing and binding specific ligands, and an intracellular region with a β-subunit that possesses tyrosine kinase activity15. IGF-1, is mainly produced by the liver, but other organs such as muscles, bones, gonads, and the intestines can also synthesize this compound in large quantities16,17. Insulin-like growth factor-1 (IGF-1) binds to IGF-1R with high specificity. Current research on IGF-1/IGF-1R primarily focuses on their relationship with cancer development18,19. Few studies suggest that IGF-1 is upregulated in damaged muscle tissue and is associated with chronic pain conditions such as endometriosis, bone cancer, and orofacial pain20,21. Previous studies have shown that IGF-1 and IGF-1R are linked to both hypertrophy and atrophy in skeletal muscle through the PI3K/AKT signaling pathway, but their role in the pathogenesis of MTrPs remains unclear22,23.
The PI3K/AKT pathway is essential for signal transduction, playing a crucial role not only in cell proliferation and apoptosis but also in regulating skeletal muscle metabolism24,25,26. There is little literature regarding the pathway’s additional involvement in pain transmission14,27. AKT, is a serine/threonine-specific protein kinase activated by PI3K, and then phosphorylated to mediate a broad spectrum of downstream responses28,29. The mTOR and RhoA proteins are downstream targets of the PI3K/AKT pathway. The mTOR protein is a serine/threonine kinase that exists as two complexes: mTOR complex1 (mTORC1) and mTOR complex2 (mTORC2)30. Numerous studies have shown that mTOR is associated with hyperalgesia31. These findings suggest that the mTOR pathway plays a key role in peripheral sensitization. RhoA is a small GTPase protein of the Rho family. These proteins activate downstream pathways by binding to GTP32. As reported previously, RhoA is an important signaling effector for actin cytoskeletal rearrangement and has an important role in smooth muscle contraction. As stated earlier, MTrPs is characterized by chronic pain and taut bands of contractile myofibers. We hypothesize that the PI3K/AKT pathway and its downstream proteins may play important roles in the MTrPs.
Based on these findings, we hypothesize that IGF-1R mediates nociceptive sensitization in myofascial trigger points (MTrPs) via activation of the PI3K/AKT/mTOR pathway. To test this hypothesis, we employed a rat model of MTrPs to investigate the role of IGF-1R and its downstream signaling cascades in MTrPs pathophysiology.
Results
Assessment of mechanical pain withdrawal threshold
Starting from the second week, the mechanical withdrawal threshold in the MTrPs group (n = 21) was significantly lower compared to the control group (n = 21)(*P < 0.05, **P < 0.01, ***P < 0.001). As the experiment progressed, a decrease in mechanical withdrawal threshold was observed in the MTrPs group, whereas the control group showed no significant difference in mechanical withdrawal threshold compared to baseline (Fig. 1A).
A rat model of taut band-associated persistent muscle pain. (A) Mechanical withdrawal thresholds change over time. The mechanical withdrawal thresholds decreased significantly from week 2 to week 12 in MTrPs group compared with the control group (**P < 0.01), n = 21 rats per group. The data expressed as mean ± standard deviation (mean ± SD). (B) Light microscopy images of gastrocnemius muscle fibers with H&E staining. Cross section: The size of muscle fibers increased and became rounder in MTrPs group. Longitudinal section: muscle fibers contracted and formed tapered ends in MTrPs group, along with larger gaps between fibers.
Histological appearance of the gastrocnemius muscle
Microscopic analysis of HE staining in the gastrocnemius muscle showed that the fibers in the control group were arranged regularly, with uniform size and polygonal shape. In contrast, in the MTrPs group, the muscle fibers were larger and rounder in cross-sectional slides. Longitudinal sections of the control group showed that the muscle cells were arranged tightly in a regular pattern, whereas in the MTrPs group, enlarged pyramidal myofibers were observed and the spaces between the muscle fibers were clearly increased (Fig. 1B).
Expression of IGF-1 and IGF-1R was increased in the MTrPs group
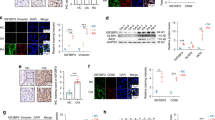
IHC showed that the expressions of IGF-1 and IGF-1R were higher in skeletal muscle tissues from MTrPs group than those in the control group (Fig. 2A)(The arrows indicate expression of IGF-1 and IGF-1R). Further analysis by WB suggested that IGF-1 and IGF-1R expression levels were increased significantly in the MTrPs group compared to that in the control group (Fig. 2B)(*P < 0.05, **P < 0.01, ***P < 0.001).
IGF-1 and IGF-1R expression was increased in MTrPs group. (A) IGF-1 and IGF-1R was detected in gastrocnemius muscle tissue (The arrows indicate expression of IGF-1 and IGF-1R.). The mean optical densities of IGF-1 and IGF-1R were significantly higher in MTrPs group compared to control group (**P < 0.01). The data expressed as mean ± standard deviation (mean ± SD). (B) IGF-1 and IGF-1R protein was analyzed by Western blot. The fold changes of IGF-1 and IGF-1R were remarkedly higher in MTrPs group than that in control group (**P < 0.01). The data expressed as mean ± standard deviation (mean ± SD).
The IGF-1R inhibitor (picropodophyllin) and PI3K inhibitor (LY294002) reversed mechanical pain withdrawal threshold in the MTrPs model
The results of IHC and WB suggested that IGF-1R expression was significantly higher in the MTrPs group, leading us to hypothesize that the hyperalgesia in MTrPs may be associated with upregulation of this protein (*P < 0.05, **P < 0.01, ***P < 0.001). To confirm this possibility, we injected IGF-1R inhibitor, picropodophyllin, into the MTrPs rats. The results of several continuous observation experiments showed that the hyperalgesia in the MTrPs group increased significantly at 0.5, 1, 2 and 4 h after intramuscular injection, with the maximal effect achieved at 1 h (Fig. 3A). This result indicates that IGF-1R contributes to the production of hyperalgesia in MTrPs. As the PI3K/AKT pathway is one of the most important intracellular signaling pathway, we investigated whether it is also involved in the pathological processes. Injection of the PI3K inhibitor, LY294002, into the local muscle tissue caused marked changes in mechanical withdrawal thresholds at 0.5,1, 2, and 4 h, with the highest mechanical pain withdrawal threshold at 2 h indicating maximum analgesic effect (Fig. 3B).
Several related proteins were detected by WB. After injection of picropodophyllin, the expression level of IGF-1R decreased significantly (*P < 0.05, **P < 0.01, ***P < 0.001). Concurrently, in both the picropodophyllin and LY294002 injection groups, the expression levels of PI3K, p-AKT, mTOR, RhoA, and p-MLC were decreased markedly compared with those measured in the unmedicated MTrPs group (Fig. 3C)(*P < 0.05, **P < 0.01, ***P < 0.001). This result indicates that IGF-1R may activate the expression of downstream proteins mTOR and RhoA via the PI3K/AKT pathway, leading to hyperalgesia.
IGF-1R inhibitor (picropodophyllin) and PI3K inhibitor (LY294002) reversed mechanical pain in the MTrPs model. (A) The mechanical withdrawal thresholds of MTrPs rats increased significantly at 0.5, 1, 2, and 4 h after intramuscular injection of picropodophyllin (**P< 0.01), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD). (B) The mechanical withdrawal thresholds of MTrPs rats increased significantly at 0.5, 1, 2, and 4 h after intramuscular injection of LY294002 (**P< 0.01), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD). (C) After intramuscular injection of picropodophyllin to MTrPs rats, protein expression levels of IGF-1R, PI3K, p-AKT, mTOR, RhoA, and p-MLC were significantly decreased compared to the MTrPs group (*P < 0.05, **P< 0.01). And when LY294002 was used, the protein expression levels of PI3K, p-AKT, mTOR, RhoA, and p-MLC were significantly decreased (**P < 0.01), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD).
IGF-1 induced pain-like behaviors and reversal by the PI3K inhibitor LY294002 in control group
To verify the role of the PI3K/AKT pathway, we administered an IGF-1R specific agonist IGF-1 factor as a local injection in the control group. These rats exhibited obvious hyperalgesia 1 h after the injection, which peaked around 2 h, indicating that upregulation of IGF-1R was associated with hyperalgesia (Fig. 4A). The activated PI3K/AKT pathway was also considered to be involved in this process. After pretreating a group of rats with the PI3K inhibitor, LY294002, and subsequently injecting IGF-1, the results showed that there was no significant change in the mechanical withdrawal threshold in this group compared to the control group (Fig. 4B).
The WB results were used to aid interpretation and confirmed that the expression levels of IGF-1, IGF-1R, PI3K, p-AKT, mTOR, and RhoA in the control rats were significantly higher after injection of IGF-1 protein than that observed in the control group (*P < 0.05, **P < 0.01, ***P < 0.001). When LY294002 was injected as a pretreatment prior to IGF-1 injection, the expression levels of IGF-1 and IGF-1R proteins remained unchanged compared to those observed following a single intramuscular injection of IGF-1, whereas the expression levels of PI3K, p-AKT, mTOR, and RhoA were significantly decreased (Fig. 4C)(*P < 0.05, **P < 0.01, ***P < 0.001). Taken together, these results indicate that IGF-1R leads to hyperalgesia in MTrPs mainly through the PI3K/AKT pathway. When IGF-1R levels were upregulated, the pathway proteins PI3K, p-AKT, the downstream proteins mTOR and RhoA also increased, while the rats exhibited a significant increase in hyperalgesia. While applying the PI3K inhibitor, downstream proteins of the PI3K/AKT pathway didn’t present the same changes (Fig. 5).
IGF-1 induced pain behaviors and reversed by PI3K inhibitor LY294002 in control rats. (A) The mechanical withdrawal thresholds of control rats decreased significantly at 1, 2, and 4 h after intramuscular injection of IGF-1 (*P< 0.05, **P < 0.01), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD). (B) The mechanical withdrawal thresholds of control rats showed no significant differences after intramuscular injection of IGF-1 and LY294002 compared with the control group (P > 0.05), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD). (C) After intramuscular injection of IGF-1 to control rats, protein expression levels of IGF-1, IGF-1R, PI3K, p-AKT, mTOR, and RhoA were significantly increased compared to the control group (*P < 0.05, **P < 0.01). And when LY294002 was added, the protein expression levels of IGF-1 and IGF-1R remained significantly different (*P < 0.05, **P < 0.01), while the expression levels of PI3K, p-AKT, mTOR, and RhoA showed no significant differences compared to the control group (P > 0.05), n = 6 rats per group. The data expressed as mean ± standard deviation (mean ± SD).
Discussion
This study demonstrates the crucial role of IGF-1R in the pathogenesis of MTrPs using a well-established rat model. We found that IGF-1 and IGF-1R were significantly upregulated in the gastrocnemius muscle of MTrPs rats. Inhibition of IGF-1R significantly alleviated hyperalgesia, while activation with IGF-1 mimicked the pain phenotype in normal rats. These effects were mediated primarily through activation of the PI3K/AKT pathway, which subsequently upregulated mTOR and RhoA/p-MLC. These results highlight the IGF-1R/PI3K/AKT as a critical contributor to hyperalgesia and may lead the muscle contracture in MTrPs.
Previous research on the IGF-1R protein has focused mainly on its cellular proliferation and anti-apoptotic function which are related mainly to tumor growth33. Only a few studies have investigated a potential role for IGF-1R and its ligand (IGF-1) in the generation of peripheral pain34. There is evidence that IGF-1 is present in knees with circumscribed cartilage lesions and is involved in pain perception in a concentration-dependent manner35. IGF-1 is also upregulated in nerve injuries and may induce orofacial neuropathic pain sensitization36. However, no relevant study has been reported on muscle pain. Chronic muscle pain remains a significant issue for the global population, with myofascial pain syndrome being a major type of this condition37. Currently, there are several hypotheses on a possible mechanism for MPS38,39with the most prominent being the integrated hypothesis40,41. This theory postulates that end plate damage results in excessive acetylcholine (ACh) release, leading to sustained depolarization of muscle fibers, muscle contraction, and raised energy demands. At the same time, nearby blood vessels are compressed, causing ischemia and hypoxia. This situation creates an energy crisis, which in turn, impedes the calcium pump that transfers excess calcium from the cell cytosol to the endoplasmic reticulum, promoting the release of some allogenic substances. The release of neuroactive substances can further promote the formation of Ach to form a vicious cycle42. In this classic theory, the interaction between Ach and calcium plays a substantial role, with Ach thought to promote intracellular calcium release and mediate calcium influx by activating G protein-coupled receptors, resulting in an increase in intracellular calcium concentration. However, the results of our experiment provide an alternate explanation to the pathophysiological process, as we confirm that the expression of IGF-1 and its receptor IGF-1R are markedly increased in MTrPs. Published literature suggests that IGF-1R activation by IGF-1 induces translocation of the transient receptor potential vanilloid 2 (TRPV2) channel to the cell membrane43. TRPV2 belongs to the TRPV subfamily of transient receptor potential (TRP) channel families and plays a critical role in promoting calcium influx. For nociceptive neurons, calcium influx leads to activation of calcium/calmodulin-dependent kinase II, causing neuronal hyperexcitability and neuropathic pain36. MPS is closely associated with sustained muscle contraction, and the contractile properties of skeletal muscles are also closely related to intracellular calcium levels. Calcium ions also bind to troponin and alter its spatial conformation, permitting tropomyosin movement on the thin filament to expose the myosin-binding site on actin, resulting in actin-myosin interaction and contraction44.
The PI3K/AKT signaling pathway is widely recognized for its involvement in cellular metabolism, growth, and survival45. In the pain field, most literature has addressed its roles in central sensitization or inflammation-related neuropathic pain27,46. Studies in rats suggested that PI3K mediates pain behavior by activating the peripheral EphrinBs/EphBs signaling pathway47while AKT is involved in phosphorylation of Thr308 and Ser473 to regulate diverse biological processes48. Several studies have demonstrated that the PI3K/AKT pathway plays an important role in these disease processes49while in our experiment, also showed that the hyperalgesia of MTrPs was associated strongly with this pathway.
We observed that the expression of mTOR protein, an important downstream protein of the PI3K/AKT pathway, was significantly elevated in MTrPs rats. The majority of studies on the role of mTOR in pain have focused at the spinal cord level, with evidence suggesting that mTOR also participates in cerebral processing of pain31,50. Our experimental data indicate that mTOR is also significantly elevated in animal models of peripheral pain. Previous studies have found that under normal conditions, the expression level of activated mTOR protein in the dorsal root ganglion (DRG) and dorsal horn is very low, and intrathecal injection of the mTOR inhibitor rapamycin does not affect pain perception51. In chronic pain conditions like bone cancer and inflammatory pain, the level of mTOR protein has been shown to be increased at the specific location, with pain hypersensitivity being attenuated after an intrathecal injection of rapamycin. Current literature indicates that mTOR mainly affects chronic pain instead of acute pain51,52,53which is consistent with the findings of our study. RhoA is another downstream target protein of PI3K/AKT, and is associated with smooth muscle contraction54,55. However, no study has addressed whether the expression of RhoA protein correlates with skeletal muscle contraction. In our experiments, we found that RhoA expression was substantially higher in rats in the MTrPs group, and at the same time, expression of its downstream protein p-MLC, which has been described as related to contractions of skeletal muscle, was increased to the same degree56,57. These results may suggest that RhoA is also involved in regulating skeletal muscle contraction via the RhoA/MLC signaling pathway.
We present evidence that IGF-1R mediates hyperalgesia in the MTrPs rat model. In addition, our data confirm that IGF-1R regulates myofascial pain mainly through the PI3K/AKT pathway. Administration of the IGF-1R inhibitor (picropodophyllin) or the PI3K inhibitor (LY 294002) significantly alleviated hyperalgesia in the MTrPs model. This provides a new direction for the future development of clinical treatments for MTrPs. While the research holds certain theoretical and practical significance, it also has some limitations. We were unable to precisely acquire the trigger point tissue samples due to a limitation of current techniques. This resulted in a small amount of surrounding tissue being taken at the same time. Two mTOR complexes (mTORC1 and mTORC2) are involved in pain formation. We only examined the total mTOR expression to explore the correlation between this protein and the pain process. The p-AKT-S473 has a more significant impact on mTORC2, and the expression of mTORC2 warrants further investigation. Regarding RhoA, there is no direct evidence proving its role in skeletal muscle contraction. Our experiments focusing on RhoA are already underway.
In conclusion, our study provides novel evidence that IGF-1R contributes to hyperalgesia in MTrPs in a rat model, primarily through the PI3K/AKT pathway. While these findings offer preliminary insights into the role of the IGF-1R/PI3K/AKT axis in the pathophysiology of MTrPs, certain limitations should be acknowledged, including the inability to isolate pure trigger point tissue and the need for further characterization of additional signaling components such as mTORC1/2 and RhoA. Moreover, it is important to emphasize that this study was conducted exclusively in animals. Therefore, future studies involving human muscle-related models are needed to further validate the role of IGF-1R in the pathophysiology of MTrPs.
IGF-1R regulates the expression of downstream proteins relating to generation of hyperalgesia and muscle contraction in myocyte.
Materials and methods
Animals
The animal experiments were approved by the Animal Care and Use Committee of Shandong University (Approval ID: KYLL-2022ZM-175). All procedures involving animals were conducted in strict accordance with the guidelines and regulations set forth by the Animal Care and Use Committee of Shandong University. The study adhered to all relevant ethical guidelines and regulations, in compliance with national and international animal welfare standards. Additionally, the study followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals throughout the entire process. Care and handling were performed in accordance with the institution’s guidelines and the ARRIVE guidelines. Six-week-old Sprague-Dawley (SD) rats (male: female = 1:1), weighing 180–220 g, were provided by the Experimental Animal Center of Qilu Hospital, Shandong University, and were housed throughout the experiment at the same center. The animals were housed, four per cage, in a temperature-controlled environment (24 °C) with a light-dark cycle (12:12 h) and a relative humidity of 20–30%. Food and water were available continuously.
Model of MTrPs
We used the same animal model as that reported by Zhang58. The MTrPs rat model was established by blunt striking on the gastrocnemius muscle of the left hindlimb and eccentric exercise for 8 weeks, followed by a recovery period of 4 weeks. All animal samples involved in the experiment were obtained from the left hindlimb gastrocnemius muscle of rats. Before striking, the rats were anesthetized with 3% pentobarbital sodium (30 mg/kg, intraperitoneal). Then the left gastrocnemius muscle in the MTrPs group was marked, with this site then hit weekly at a fixed time by a hand-made stick dropped freely from a height of 20 cm. The following day, the rats ran on a treadmill (Sans, Nanjing, China) at a downward angle of −16°for 90 min. During the 10-min warm-up period, the speed was increased gradually from 12 to 16 m/min. The condition of the limbs, movement ability, and health of the rats were examined weekly during the experimental procedures. Three consecutive steps were used to confirm the presence of active MTrPs: palpation of the contracture nodule in the gastrocnemius muscle, a decrease in the mechanical withdrawal threshold, and a local twitch response (LTR) during a needling procedure (Supplementary Fig. 1).
Assessment of mechanical withdrawal threshold
Mechanical withdrawal threshold in the rats was measured using a Randall-Selitto apparatus (Shandong Provincial Institute of Science and Technology, Jinan, Shandong, China). This apparatus was equipped with a round head probe (tip diameter, 8 mm) that set the extraction threshold of the left hindlimb when subjected to mechanical stimulation of the muscle. All the rats were acclimated to the laboratory environment for 7 days before the experiments, and then in a quiet environment, were restrained in a plastic cylinder with their hind limbs exposed. A Randall-Selitto probe was applied to the left gastrocnemius muscle to generate an automatic increase in pressure, with the mechanical withdrawal threshold being measured when the rat withdrew its limb. The stimulation was repeated five times at one-min intervals and the average value calculated after removing the minimum and maximum values.
Drugs and injection procedures
These procedures were performed at week 13 after modeling. IGF-1, picropodophyllin, LY294002, and the solvent DMSO were applied to the left hind limbs of the experimental animals. The drug dose and regimen were based on those of previously published studies14,59. The rats were divided randomly into 6 groups with 6 animals in each group: Control + DMSO (1%), MTrPs + DMSO (1%), MTrPs + IGF-1R inhibitor (picropodophyllin, MCE, 1 mg/ml), MTrPs + PI3K inhibitor (LY294002, MCE, 1 mg/ml), control + IGF-1R agonist IGF-1 (Abcam, 10 µg/ml), and control + IGF-1R agonist IGF-1 (Abcam, 10 µg/ml) + PI3K inhibitor (LY294002, MCE, 1 mg/ml). The drugs were injected intramuscularly at marked sites of the gastrocnemius muscle (30 µL × 3 points per rat) based on the aforementioned grouping. The mechanical withdrawal threshold was measured continuously at 8 time points 1 day after the injection (0, 0.5, 1, 2, 4, 6, 8, and 24 h). The threshold trend and the time point at which maximal change in the withdrawal threshold occurred were then recorded for each group. The previous steps were repeated the next day until the recording time point was reached. The rats were then sacrificed by cervical dislocation under deep anesthesia, followed by immediate excision of the muscle tissues from the injection sites for HE staining, IHC and WB.
Anesthesia and euthanasia protocol
Each rat was anesthetized using 2–3% isoflurane in oxygen. After successful modeling or drug intervention, the rats were euthanized in the laboratory using an overdose of isoflurane. The isoflurane flow concentration was adjusted to 5%, and the rats were kept exposed to isoflurane for an additional 2 min after the cessation of breathing.
Immunohistochemistry
The HE staining slides were screened for representative areas of MTrPs. The immunohistochemistry experiments were performed on active MTrPs sections. The paraffin embedded sections were baked at 65 °C for 2 h, separated in xylene and rehydrated in an ethanol gradient, followed by heating in ethylene-diaminetetraacetic acid (EDTA) buffer in a microwave to retrieve the antigen. Endogenous peroxidase was blocked with 3% H2O2 for 10 min, followed by incubation with normal goat serum for 60 min at 37℃. The muscle sections were then incubated overnight at 4 °C in a humidified chamber with either IGF-1 antibody (1:200, Abcam, ab36532) or IGF-1R antibody (1:100, Beyotime, AF7182). The slides were washed and then incubated with biotin-labeled goat anti-rabbit secondary antibody at 37 °C for 30 min. After rinsing three times with phosphate-buffered saline (PBS), the staining was visualized by Diaminobenzidine (DAB) using hematoxylin as the counterstain, with the images obtained by light microscopy.
Western blotting
The animals were treated according to the aforementioned experimental groupings. Samples of the injected muscle tissues of the left gastrocnemius were collected and placed immediately into liquid nitrogen and then stored in a −80 °C refrigerator. The tissue samples were lysed on ice in RIPA lysis buffer, with protease and phosphatase inhibitors being added prior to the procedure. The lysate was centrifuged for 15 min at 4 °C, followed by collection of the supernatants and measurement of protein concentrations using the BCA protein concentration assay kit. After mixing with loading buffer, the samples were heated at 100 °C for 10 min. The total proteins were separated by 10% and 12.5% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and then transferred to polyvinylidene fluoride (PVDF) membranes. After treatment with blocking solution for 1.5 h at room temperature, the membranes were incubated at 4 °C overnight with the following primary antibodies: IGF-1 antibody (1:5000, Abcam, ab36532), IGF-1R antibody (1:1000, Beyotime, AF7182), PI3K p110 alpha antibody (1:1200, Affinity, AF5112), p-AKT antibody (1:1000, Affinity, AF0016), AKT antibody (1:1000, Affinity, AF6261), mTOR antibody (1:1000, Beyotime, AF1648), RhoA (1:5000, Abcam, ab187027), and p-MLC (1:1000, Affinity, AF8618). The membranes were washed with tris-buffered saline Tween-20 (TBST) and then incubated with secondary antibody (1:5000, Beyotime) at room temperature. Finally, the blots were visualized using a chemiluminescent reagent. GAPDH was selected as the reference control.
Statistical analysis
The results were analyzed using SPSS 28.0 software, with the data expressed as mean ± standard deviation (mean ± SD). The rat mechanical withdrawal threshold data at different time points were analyzed by multivariate repeated measures ANOVA. For the protein expression data, Student’s t-test was used to compare data between two groups, one-way ANOVA to compare more than two groups, and the Tukey post hoc method for multiple comparisons. The results were considered significant when the P < 0.05 (*P < 0.05; **P < 0.01).
Data availability
The data presented in this study are available from the corresponding author on reasonable request.
Abbreviations
- MPS:
-
Myofascial pain syndrome
- MTrPs:
-
Myofascial trigger points
- SD:
-
Sprague-dawley
- IGF-1:
-
Insulin-like growth factor-1
- IGF-1R:
-
Insulin-like growth factor-1 receptor
- TBs:
-
Taut bands
- WB:
-
Western blotting
- HE:
-
Hematoxylin-eosin staining
- IHC:
-
Immunohistochemistry
- IOD:
-
Integrated optical density
- PI3K:
-
Phosphatidylinositol 3-kinase
- AKT:
-
Protein kinase B
- mTOR:
-
Mammalian target of rapamycin
- MLCK:
-
Myosin light chain kinase
- EDTA:
-
Ethylene-diaminetetraacetic acid
- PBS:
-
Phosphate-buffered saline
- DAB:
-
Diaminobenzidine
- PVDF:
-
Polyvinylidene fluoride
- p-MLC:
-
Phosphorylation of myosin light chain
- ACh:
-
Acetylcholine
- TRPV2:
-
Transient receptor potential vanilloid 2
- TRP:
-
Transient receptor potential
References
袁仕国 et al. 化学性交感神经切除对肌筋膜激痛点炎症和肌卫星细胞成肌分化的影响 实用医学杂志 36, 7 (2020).
Fischer, M. J. et al. Evaluation of mitochondrial function in chronic myofascial trigger points - A prospective cohort pilot study using high-resolution respirometry. BMC Musculoskelet. Disord.19, 388. https://doi.org/10.1186/s12891-018-2307-0 (2018).
Lavelle, E. D., Lavelle, W. & Smith, H. S. Myofascial trigger points. Anesthesiol. Clin.25, 841–851. https://doi.org/10.1016/j.anclin.2007.07.003 (2007).
Simons, D. G. Review of enigmatic MTrPs as a common cause of enigmatic musculoskeletal pain and dysfunction. J. Electromyogr. Kinesiol.14, 95–107. https://doi.org/10.1016/j.jelekin.2003.09.018 (2004).
Shah, J. P. et al. Myofascial trigger points then and now: A historical and scientific perspective. Pm R. 7, 746–761. https://doi.org/10.1016/j.pmrj.2015.01.024 (2015).
Lavelle, E. D., Lavelle, W. & Smith, H. S. Myofascial trigger points. Med. Clin. North. Am. 91, 229–239. https://doi.org/10.1016/j.mcna.2006.12.004 (2007).
Thorp, S. L. et al. Functional Connectivity Alterations: Novel Therapy and Future Implications in Chronic Pain Management. Pain Physician 21, E207-e214 (2018).
Soares, A., Andriolo, R. B., Atallah, A. N. & da Silva E. M. Botulinum toxin for myofascial pain syndromes in adults. Cochrane Database Syst. Rev. CD007533 https://doi.org/10.1002/14651858.CD007533.pub3 (2014).
Barros-Neto, J. A. et al. Selenium and zinc status in chronic myofascial pain: Serum and erythrocyte concentrations and food intake. PLoS One11, e0164302. https://doi.org/10.1371/journal.pone.0164302 (2016).
Alvarez, D. J. & Rockwell, P. G. Trigger points: Diagnosis and management. Am. Fam. Physician65, 653–660 (2002).
Jin, F. et al. The pathophysiological nature of sarcomeres in trigger points in patients with myofascial pain syndrome: A preliminary study. Eur. J. Pain24, 1968–1978. https://doi.org/10.1002/ejp.1647 (2020).
Liu, Y. et al. Platelet-derived growth factor receptor-α induces contraction knots and inflammatory pain-like behavior in a rat model of myofascial trigger points. Anesthesiology https://doi.org/10.1097/aln.0000000000005167 (2024).
Jin, F., Zhao, L., Hu, Q. & Qi, F. Peripheral EphrinB1/EphB1 signalling attenuates muscle hyperalgesia in MPS patients and a rat model of taut band-associated persistent muscle pain. Mol. Pain16, 1744806920984079. https://doi.org/10.1177/1744806920984079 (2020).
Zhang, M., Zin, F., Zhu, Y. & Qi, F. Peripheral FGFR1regulates myofascial pain in rats via the PI3K/AKT pathway. Neuroscience 436, 1–10. https://doi.org/10.1016/j.neuroscience.2020.04.002 (2020).
Yuan, J., Yin, Z., Tao, K., Wang, G. & Gao, J. Function of insulin-like growth factor 1 receptor in cancer resistance to chemotherapy. Oncol. Lett. 15, 41–47. https://doi.org/10.3892/ol.2017.7276 (2018).
Molagoda, I. M. N. et al. Fermented oyster extract promotes Insulin-Like growth Factor-1-Mediated osteogenesis and growth rate. Mar. Drugs. 18 https://doi.org/10.3390/md18090472 (2020).
Labandeira-Garcia, J. L., Costa-Besada, M. A., Labandeira, C. M., Villar-Cheda, B. & Rodríguez-Perez, A. I. Insulin-like growth factor-1 and neuroinflammation. Front. Aging Neurosci.9, 365. https://doi.org/10.3389/fnagi.2017.00365 (2017).
Rigiracciolo, D. C. et al. IGF-1/IGF-1R/FAK/YAP transduction signaling prompts growth effects in triple-negative breast cancer (TNBC) cells. Cellshttps://doi.org/10.3390/cells9041010 (2020).
Chan, Y. T. et al. The interplay between IGF-1R signaling and Hippo-YAP in breast cancer stem cells. Cell. Commun. Signal. 21 https://doi.org/10.1186/s12964-023-01088-2 (2023).
Ma, Y., Guo, Z. & Wang, X. Tribulus terrestris extracts alleviate muscle damage and promote anaerobic performance of trained male boxers and its mechanisms: Roles of androgen, IGF-1, and IGF binding protein-3. J. Sport Health Sci.6, 474–481. https://doi.org/10.1016/j.jshs.2015.12.003 (2017).
Ma, L. et al. IGF/IGF-1R signal pathway in pain: A promising therapeutic target. Int. J. Biol. Sci.19, 3472–3482. https://doi.org/10.7150/ijbs.84353 (2023).
Feng, L., Li, B., Xi, Y., Cai, M. & Tian, Z. Aerobic exercise and resistance exercise alleviate skeletal muscle atrophy through IGF-1/IGF-1R-PI3K/Akt pathway in mice with myocardial infarction. Am. J. Physiol. Cell Physiol.322, C164-c176. https://doi.org/10.1152/ajpcell.00344.2021 (2022).
Yoshida, T. & Delafontaine, P. Mechanisms of IGF-1-Mediated regulation of skeletal muscle hypertrophy and atrophy. Cells 9 https://doi.org/10.3390/cells9091970 (2020).
Mi, X. J. et al. The protective effects of maltol on cisplatin-induced nephrotoxicity through the AMPK-mediated PI3K/Akt and p53 signaling pathways. Sci. Rep.8, 15922. https://doi.org/10.1038/s41598-018-34156-6 (2018).
Wan, X. et al. A novel miR-490-3p/hnRNPA1-b/PKM2 axis intermediates the Warburg effect and proliferation of colon cancer via PI3K/AKT pathway. https://doi.org/10.21203/rs.3.rs-3574711/v1(2023).
Ke, C. et al. Inhibition of infantile hemangioma growth and promotion of apoptosis via VEGF/PI3K/Akt axis by 755-nm long-pulse alexandrite laser. Biomed. J. 100675 https://doi.org/10.1016/j.bj.2023.100675 (2023).
Zhang, J., Wang, L., Wang, H., Su, Z. & Pang, X. Neuroinflammation and central PI3K/Akt/mTOR signal pathway contribute to bone cancer pain. Mol. Pain. 15, 1744806919830240. https://doi.org/10.1177/1744806919830240 (2019).
Li, J., Lang, J., Zeng, Z. & McCullough, L. D. Akt1 gene deletion and stroke. J. Neurol. Sci.269, 105–112. https://doi.org/10.1016/j.jns.2007.12.034 (2008).
He, A. et al. CDKL3 promotes osteosarcoma progression by activating akt/pkb. Life Sci. Alliance. 3 https://doi.org/10.26508/lsa.202000648 (2020).
Tan, S. H. et al. Critical role of SCD1 in autophagy regulation via lipogenesis and lipid rafts-coupled AKT-FOXO1 signaling pathway. Autophagy 10, 226–242. https://doi.org/10.4161/auto.27003 (2014).
Izumi, Y., Sasaki, M., Hashimoto, S., Sawa, T. & Amaya, F. Mtor signaling controls VGLUT2 expression to maintain pain hypersensitivity after tissue injury. Neuroscience308, 169–179. https://doi.org/10.1016/j.neuroscience.2015.09.013 (2015).
Ji, Y. et al. Rock signaling control PPARgamma expression and actin polymerization during adipogenesis. Saudi J. Biol. Sci. 24, 1866–1870. https://doi.org/10.1016/j.sjbs.2017.11.029 (2017).
Iyer, G. et al. Insulin-like growth factor 1 receptor mediated tyrosine 845 phosphorylation of epidermal growth factor receptor in the presence of monoclonal antibody cetuximab. BMC Cancer. 16, 773. https://doi.org/10.1186/s12885-016-2796-x (2016).
Zhang, Y. et al. Peripheral pain is enhanced by insulin-like growth factor 1 through a G protein–mediated stimulation of T-type calcium channels. Sci. Signal.7, ra94–ra94 (2014).
Schmal, H. et al. Pain perception in knees with circumscribed cartilage lesions is associated with intra-articular IGF-1 expression. Am. J. Sports Med.39, 1989–1996. https://doi.org/10.1177/0363546511406851 (2011).
Sugawara, S. et al. Increase in IGF-1 expression in the injured infraorbital nerve and possible implications for orofacial neuropathic pain. Int. J. Mol. Sci. 20 https://doi.org/10.3390/ijms20246360 (2019).
Gregory, N. S. & Sluka, K. A. Anatomical and physiological factors contributing to chronic muscle pain. Curr. Top. Behav. Neurosci. 20, 327–348. https://doi.org/10.1007/7854_2014_294 (2014).
DG, S. Myofascial trigger points, a possible explanation. Pain 10, 106–109 (1981).
Hubbard, D. R. & Berkoff, G. M. Myofascial trigger points show spontaneous needle EMG activity. Spine 18, 1803–1807 (1993).
Gerwin, R. D., Dommerholt, J. & Shah, J. P. An expansion of Simons’ integrated hypothesis of trigger point formation. Curr. Pain Headache Rep.8, 468–475 (2004).
Kuan, T. S. et al. The myofascial trigger point region: correlation between the degree of irritability and the prevalence of endplate noise. Am. J. Phys. Med. Rehabil. 86, 183–189 (2007).
Gerwin, R. D. The taut band and other mysteries of the trigger point: An examination of the mechanisms relevant to the development and maintenance of the trigger point. J. Musculoskelet. Pain.16, 115–121. https://doi.org/10.1080/10582450801960081 (2009).
Gailly, P. TRP channels in normal and dystrophic skeletal muscle. Curr. Opin. Pharmacol. 12, 326–334. https://doi.org/10.1016/j.coph.2012.01.018 (2012).
Tanner, B. C., Daniel, T. L. & Regnier, M. Filament compliance influences cooperative activation of thin filaments and the dynamics of force production in skeletal muscle. PLoS Comput. Biol.8, e1002506. https://doi.org/10.1371/journal.pcbi.1002506 (2012).
Yu, L., Wei, J. & Liu, P. Attacking the PI3K/Akt/mTOR signaling pathway for targeted therapeutic treatment in human cancer. Semin. Cancer Biol.85, 69–94. https://doi.org/10.1016/j.semcancer.2021.06.019 (2022).
Liu, Y., Qin, X., Lu, X. & Jiang, J. Effects of inhibiting the PI3K/Akt/mTOR signaling pathway on the pain of sciatic endometriosis in a rat model. Can. J. Physiol. Pharmacol. 97, 963–970 (2019).
Guan, X. H. et al. Phosphatidylinositol 3-kinase mediates pain behaviors induced by activation of peripheral ephrinbs/ephbs signaling in mice. Pharmacol. Biochem. Behav. 95, 315–324 (2010).
Wei, Y., Zhou, J., Yu, H. & Jin, X. AKT phosphorylation sites of Ser473 and Thr308 regulate AKT degradation. Biosci. Biotechnol. Biochem. 83, 429–435. https://doi.org/10.1080/09168451.2018.1549974 (2019).
Liu, S. P. C. Y. Q. Z. D.-Q. PI3KAkt pathway - a potential therapeutic target for chronic pain. Curr. Pharm. Design. 23, 1860–1868 (2017).
Cho, C., Michailidis, V. & Martin, L. J. Revealing brain mechanisms of mTOR-mediated translational regulation: implications for chronic pain. Neurobiol. Pain. 4, 27–34. https://doi.org/10.1016/j.ynpai.2018.03.002 (2018).
Lutz, B. M., Nia, S., Xiong, M., Tao, Y. X. & Bekker, A. mTOR, a new potential target for chronic pain and opioid-induced tolerance and hyperalgesia. Mol. Pain. 11, 32. https://doi.org/10.1186/s12990-015-0030-5 (2015).
Lisi, L., Aceto,, P., Navarra, P. & Dello Russo, C. mTOR kinase: a possible Pharmacological target in the management of chronic pain. Biomed. Res. Int. (394257). https://doi.org/10.1155/2015/394257 (2015).
Wong, C. et al. mTORC2 mediates structural plasticity in distal nociceptive endings that contributes to pain hypersensitivity following inflammation. J. Clin. Invest. 132 https://doi.org/10.1172/JCI152635 (2022).
Al-Shboul, O. The role of the RhoA/ROCK pathway in gender-dependent differences in gastric smooth muscle contraction. J. Physiol. Sci.66, 85–92. https://doi.org/10.1007/s12576-015-0400-9 (2016).
Sauzeau, V. et al. Cyclic GMP-dependent protein kinase signaling pathway inhibits RhoA-induced Ca2 + sensitization of contraction in vascular smooth muscle. J. Biol. Chem.275, 21722–21729. https://doi.org/10.1074/jbc.M000753200 (2000).
Sitbon, Y. H., Yadav, S., Kazmierczak, K. & Szczesna-Cordary, D. Insights into myosin regulatory and essential light chains: A focus on their roles in cardiac and skeletal muscle function, development and disease. J. Muscle Res. Cell Motil.41, 313–327. https://doi.org/10.1007/s10974-019-09517-x (2020).
Hortemo, K. H., Munkvik, M., Lunde, P. K. & Sejersted, O. M. Multiple causes of fatigue during shortening contractions in rat slow twitch skeletal muscle. PLoS One. 8, e71700. https://doi.org/10.1371/journal.pone.0071700 (2013).
Histopathological nature. Of myofascial trigger points at different stages of recovery from injury in a rat model. Acupunct. Med. 35, 445–451. https://doi.org/10.1136/acupmed-2016-011212 (2017).
Miura, M. et al. Peripheral sensitization caused by insulin-like growth factor 1 contributes to pain hypersensitivity after tissue injury. Pain 152, 888–895. https://doi.org/10.1016/j.pain.2011.01.004 (2011).
Acknowledgements
We thank the Laboratory of Basic Medical Sciences of Qilu Hospital for its help.
Funding
This study was supported by the National Natural Science Foundation of China (No. 82272600), Natural Science Foundation of Shandong Province Grant (No. ZR2023MH207).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study. Material preparation, data collection and analysis were performed by Lingwei Zhou, Miao Pei, Tian Qi, Yu Liu, Xiaoyue Li, Xuan Li, Qinghe Chen, Shaozhong Yang and Feihong Jin. Feng Qi, Lingwei Zhou, Miao Pei conceived and designed the experiments. The first draft of the manuscript was written by Lingwei Zhou and Miao Pei. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animal experiments were approved by the Animal Care and Use Committee of Shandong University (Approval ID: KYLL-2022ZM-175). National Institutes of Health Guide for the Care and Use of Laboratory Animals were followed in the entire process.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, L., Pei, M., Qi, T. et al. IGF-1R promotes pain-like behavior in rats with myofascial pain syndrome via the PI3K/AKT pathway. Sci Rep 15, 29050 (2025). https://doi.org/10.1038/s41598-025-12552-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12552-z