Abstract
Osteoporosis and osteopenia, collectively referred to as Low Bone Mass Disorders (LBMD), are characterized by decreased bone density and increased fracture risk. Studies have identified a potential link between cardiovascular disease and LBMD, and the role of lipid metabolism in this has attracted widespread attention. We analyzed participant information collected from multiple NHANES cycles using weighted multivariable logistic regression, subgroup analysis, restricted cubic splines (RCS) curve analysis, and causal mediation analysis. Non-high-density lipoprotein to high-density lipoprotein ratio (NHHR) was significantly negatively associated with low bone mass disorders in a fully adjusted model. We found that NHHR exhibited an L-shaped relationship with the prevalence of LBMD, with a negative association when NHHR < 3.38 and no significant association when NHHR > 3.38. So we could manage both disorders through NHHR. Causal mediation analysis demonstrated that NHHR was a partial mediator of the effect of body mass index (BMI) on low bone mass disorders (LBMD), accounting for 2.3% of the total effect. This suggests that part of the effect in weight affecting low bone mass disorders is mediated by the NHHR. This inspires us to utilize NHHR to guide weight management.
Similar content being viewed by others
Introduction
Low Bone Mass Disorders (LBMD), including osteopenia and osteoporosis, refer to a group of disorders characterized by diminished levels of bone mineral density. This reduction makes bones more susceptible to weakness and increases the potential for fractures. As the world’s population ages, the prevalence of LBMD continues to rise in the elderly population1,2, which is prone to fragility fractures, resulting in limited mobility, deformity, chronic pain and even disability3,4. In particular, femoral fractures and fragility hip fractures due to osteoporosis in the elderly may lead to a variety of complications characterized by poor prognosis and high mortality3,5. Therefore, femoral BMD is widely used as an indicator for the diagnosis of osteopenia and osteoporosis and is an efficient tool for detecting LBMD6,7. Due to the current increase in the number of patients with LBMD and the severe impact of associated fractures on patients’ quality of life and finances8,9, it is essential for early LBMD diagnosis and intervention.
New research has revealed an association between obesity and LBMD, noting that populations with lower weight tend to have lower bone density and higher prevalence of LBMD10,11. Body mass index (BMI), a ratio of weight to height squared (kg/m²), is the standard metric for weight status classification in epidemiological research. Previous studies have shown that body mass index (BMI)-mediated obesity has a protective effect on bone mineral density (BMD) and that optimal benefit is observed above a specific BMI threshold12,13. However, whether obesity directly affects or mediates the effects of BMD remains controversial. It is abundantly clear that obesity may predispose to a number of underlying diseases and increase the risk of cardiovascular disease (CVD). Therefore, non-strategic weight gain is not recommended to reduce LBMD risk. Further research should focus on safety thresholds to scientifically guide weight management.
To explore the relationship between lipid metabolism and osteoporosis, numerous studies have attempted to reveal a potential link between the two by analyzing various lipoprotein14,15,16. Some studies have suggested that high-density lipoprotein cholesterol (HDL-C) may be protective against osteoporosis, whereas low-density lipoprotein cholesterol (LDL-C) may increase the risk of this disease14,15. However, other studies have suggested the opposite, indicating that HDL may be a risk factor for osteoporosis, whereas LDL may be protective16,17. At present, the specific link between lipoproteins and osteoporosis remains unclear. Some studies have confirmed a significant association between cardiovascular disease and osteoporosis, particularly in the elderly population, suggesting that there may be a common pathophysiological mechanism or common risk factors for both18. As the population aged, the incidence of both cardiovascular diseases and osteoporosis has been increasing19,20. Studies have revealed that patients with cardiovascular diseases are more likely to develop osteoporosis and vice versa21,22, prompting an increasing number of scholars to focus on clinical biomarkers with the aim of guiding the prevention and treatment of both diseases together19,20,23. NHHR is a composite indicator for cardiovascular diseases, particularly atherosclerosis, which integrates information on all lipid particles that both promote and inhibit atherosclerosis24. NHHR demonstrates superior predictive capacity for cardiovascular disease (CVD) risk stratification compared to conventional single-parameter biomarkers25,26. Therefore, in this study, we chose to use NHHR to comprehensively assess the effects of HDL cholesterol and non-HDL cholesterol lipoprotein to explore their relationship with LBMD, providing a new perspective for the combined management of cardiovascular disease (CVD) and LBMD, and offering scientific guidance for weight management in populations at risk of osteoporosis.
Methodology of the study
Study design and population
The data for this study were obtained from the National Health and Nutrition Examination Survey (NHANES), a survey conducted by the National Center for Health Sciences (NCHS) of the Centers for Disease Control and Prevention (CDC). The health and nutritional status of the U.S. population is assessed by means of questionnaires, physical examinations, laboratory tests, and other records, and rigorous sampling and validation are used to ensure the accuracy of the data27,28,29,30. This survey was conducted with documented consent from every participant, and ethical authorization was secured through the Ethics Review Board of the U.S. Centers for Health Statistics.
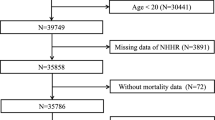
A total of 56,769 individuals from NHANES 2005–2010, 2013–2014, and 2017–2020 were used in this study; the remaining years were not uploaded for femoral BMD and were therefore not included in this study. Initially, participants who had not undergone laboratory testing (and therefore lacked NHHR data) were excluded. Subsequently, those lacking data on femoral bone density, body mass index, smoking status, blood pressure status, kidney disease, diabetes or other diseases were also excluded. It should be noted that due to limitations in the availability of NHANES blood data, our study population only included adults aged 20 years and older. The study ultimately included a sample of 18,283 participants. Figure 1 illustrates the specific inclusion and exclusion process. All data were obtained from NHANES, and the study dataset, documents, and protocols are available free of charge on the NHANES website31.
Flowchart for inclusion and exclusion of NHANES 2005–2010, 2013–2014, and 2017–2020 participants.
Definition of low bone mass disorders
LBMD include osteopenia and osteoporosis and are diagnosed on the basis of bone mineral density (BMD). The BMD data in the database is checked with a dual-energy X-ray absorptiometry instrument (Hologic, Bedford, MA, USA) for femoral neck, trochanter, intertrochanter and total femoral BMD. Based on the World Health Organization criteria32, participants with BMD T-score between − 1.0 and − 2.5 standard deviation (SD) at any femoral measurement site were classified as having osteopenia, whereas those with T-score ≤ −2.5 SD were classified as having osteoporosis. Both conditions can be collectively referred to as LBMD, and if all the values of BMD in all regions are greater than or equal to the mean of the young population minus 1 SD, they are considered normal. The reference group for the young population in this study was people aged 20–29 years, and the corresponding thresholds for LBMD in each femoral region are shown in Supplementary Table 1.
Definition of NHHR and BMI measurement
Serum samples were analyzed to determine levels of high-density lipoprotein cholesterol (HDL-C) and total cholesterol. Total cholesterol (TC) was determined via the Wahlefeld method, HDL was determined via the magnesium sulfate/dextran method and non-HDL was obtained by subtracting HDL from total cholesterol. The non-high-density lipoprotein to high-density lipoprotein ratio (NHHR) was calculated as non-HDL divided by HDL, and the values were retained to two decimal places. Additionally, NHHR was considered as a meaningful mediator variable in this study. The body mass index (BMI) is determined by using a formula where weight in kilograms is divided by height in meters squared, expressed as BMI = weight (kg)/height^2 (m^2).
Ascertainment of covariates
The selection of covariates was based on previous studies15,33 and clinical observations. This study incorporated a range of variables, including demographic risk factors, associated disease factors. Regarding demographic risk factors, factors like age, sex, education level, and racial/ethnic background were included. Education level was categorized as less than 9th grade, 9th-12th grade, high school grade, AA degree, college graduate or above, other grade. Race was categorized as Mexican American, Other Hispanic, Non-Hispanic White, Non-Hispanic Black, Other Race. In addition, age was classified into two groups according to previous studies in subgroup analysis: those younger than 60 and those older than 60. The inclusion criteria for diabetes, hypertension and kidney disease in terms of underlying disease were considered. Additionally, the presence of smoking history was considered. To estimate the presence of smoking, the self-reported criterion was ‘whether you have smoked at least 100 cigarettes in your lifetime’. Due to the limitations of the 2-hour oral glucose tolerance test measurement data, three complementary criteria were used to diagnose diabetes: physician diagnosis, fasting blood glucose ≥ 7.0 mmol/L, or glycated hemoglobin (HbA1c) ≥ 6.5%. Hypertension was determined by self-reported physician notification of hypertension or by three standardized measurements of arterial blood pressure of ≥ 140 mmHg systolic or ≥ 90 mmHg diastolic. Chronic kidney disease (CKD) was determined is determined by the eGFR value (eGFR < 60 mL/min/1.73 m²). Self-reported information on the above diseases was collected through standardised NHANES interview questionnaires. For example, the diagnosis of hypertension was based on participants’ responses to the question, ‘Have you ever been told by a doctor or other health professional that you have high blood pressure?’ Other diseases covered in the study (e.g., kidney disease, arthritis, diabetes, etc.) were defined in a similar way, and all NHANES survey interviews were conducted by trained health interviewers using scientifically validated questionnaires under strict quality control protocols.
Statistical analysis
The data collected were analyzed according to the NHANES analytical guidelines, and continuous variables in the study were expressed as the mean ± standard deviation for those that conformed to a normal distribution and as the median and interquartile range for those that did not conform to normal distribution. Categorical variables are expressed as number and percentage. To identify whether there was a potential nonlinear relationship between NHHR, BMI and LBMD, as well as to describe the difference in the effect relationship as the values of the two scales increased, we divided NHHR and BMI into four groups according to their respective interquartile sizes. Given the complex multistage probability sampling design used in NHANES, all analyses included sample weights (WTMEC2YR), clustering variables (SDMVPSU), and stratification variables (SDMVSTRA) to ensure that the estimated results were nationally representative.The association among NHHR, BMI and LBMD were analyzed via multifactorial logistic regression, which revealed the effect value and whether it was statistically significant according to the OR and 95% CI, with the lowest value group, Quartile 1, as a reference, in order to describe the associations between the different value groups. Based on the adjusted covariates, it was divided into three models. Model 1 was performed for univariate analysis without adjustment. Model 2 was adjusted for demographic variables to include age, sex, education, and racial/ethnicity. Model 3 further included smoking, hypertension, diabetes, and chronic kidney disease, and multiple study models were compared to test the associations and their stability. We also conducted subgroup analyses to investigate heterogeneity and potential association between NHHR and LBMD in different populations. In order to analyze the correlation between NHHR and LBMD prevalence, we performed smoothed curve fitting to investigate whether there was a linear or non-linear relationship between the two. All statistical analyses were performed with R software (version 4.4.0) and EmpowerStats (version 6.0). The main R packages used include: survey package, mediation package, rms package, and mgcv package.
Result
Baseline features of the subjects
This study utilized complex sampling weight analysis from the NHANES to include 18,283 adults aged ≥ 20 years with complete blood and bone density data, representing a weighted population of 140,351,693 in the United States. Characteristics of the weighted population showed: mean age of 51 years, 49.80% male, a smoking rate of 46.99%, and a prevalence of low bone mass disorder (LBMD) of 52.14%. Weighted comparisons revealed that the prevalence of LBMD was significantly lower in the fourth quartile of NHHR compared to the first quartile (45.99% vs. 59.48%, p < 0.001), while BMI was significantly higher (29.7 vs. 24.4 kg/m², p < 0.001); As detailed in Table 1 weighted results.
Logistic regression analysis
According to the analysis in Table 2, the weighted results indicate that there is a negative correlation between NHHR and LBMD prevalence regardless of whether confounding variables are adjusted for. In Model 1 (without adjustment for covariates), a negative correlation was found between NHHR levels and the prevalence of LBMD, and each unit increase in NHHR reduced the prevalence of LBMD by 13% (0.87 (0.84, 0.90), p < 0.001). In Model 3 (incorporating age, sex, education, race, hypertension, diabetes, smoking, and kidney disease covariates into the regression model), NHHR was negatively associated with LBMD, with each unit increase in NHHR decreasing the prevalence of LBMD by 13% (0.87, (0.85, 0.90), p < 0.001). Additionally, in Model 3, the NHHR and LBMD prevalence rates in the highest NHHR group (fourth quartile) were 0.55 times those in the lowest NHHR group (first quartile) (0.55, (0.49, 0.63), p < 0.001), and the difference between the fourth and first quartiles was statistically significant. In the NHHR quartile analysis, the p for trend was < 0.001.
When covariates are not considered, the weighted results indicate a significant correlation between BMI and LBMD, and for every unit increase in BMI, the prevalence of LBMD decreased by 10% (0.90, (0.80, 0.91), p < 0.001). When age, sex, education, race, hypertension, diabetes, smoking, and kidney disease covariates were included in the regression model, BMI and LBMD remained negatively and statistically significant (0.88, (0.87, 0.88), p < 0.001), with a 12% decrease in LBMD prevalence for each unit increase in BMI. In Model 3, the highest BMI group had an 84% decrease in LBMD prevalence compared with the lowest BMI group (Q1). The specific data are shown in Table 3.
Subgroup analysis
Weighting subgroup analysis was performed using model 3 to explore the association between NHHR and LBMD in different populations. Among the subgroups according to age (grouped with 60 as the cutoff), race, smoking status, and hypertension status, NHHR and LBMD were negatively correlated, and the correlation were statistically significant (p < 0.001). However, Fig. 2 shows that there was no significant interaction between the above subgroups (p for interaction > 0.05). In the diabetes subgroup, a significant negative correlation between NHHR and LBMD was observed in individuals without diabetes, whereas in diabetic patients, NHHR showed a non-significant negative correlation with LBMD. This phenomenon was also observed in the chronic kidney disease subgroup. There were significant interactions between the diabetes subgroup and the chronic kidney disease subgroup. In both sex subgroups, NHHR and LBMD showed significant negative correlation (0.83, (0.79, 0.87), 0.92, (0.89, 0.95)), and the p for interaction < 0.001, the correlation between gender and direction is negative, but there are significant differences in effect size (the protective effect is stronger in females), and there is a risk of false positives. Specific data are shown in Fig. 2.
Weighted Subgroup analysis of the association between NHHR and Low bone mass disorders.
Weighted restricted cubic splines (RCS) curve fitting
Based on the weighted RCS curve fitting, we revealed an L-shaped association between NHHR and LBMD (see Fig. 3). There is an inflection point of 3.38 (log-likelihood ratio test p < 0.001) in the trend of the association between NHHR and LBMD. At < 3.38, the effect value is 0.77, indicating a significant negative correlation ((0.73, 0.81), p < 0.001). For every 1 unit increase in NHHR, the prevalence of LBMD was reduced by 23%. At > 3.5, the effect value was 1.00 ((0.96, 1.04), p = 0.830), NHHR was not associated with LBMD. Specific data from the threshold effect analysis are shown in Table 4.
Weighted Restricted Cubic Splines (RCS) Curve Fitting. Notes: Subfigure A: Association between NHHR and LBMD without adjusting for confounding factors; Subfigure B: Association between NHHR and LBMD after full adjustment for confounding factors.
Causal mediation analysis
Causal mediating effect analysis, revealed that the NHHR mediated the negative association of BMI with LBMD, with an indirect effect of −0.0007 ((−0.0011, −0.0001), p = 0.020). Figure 4 shows the multiple pathways by which BMI affects LBMD, as well as the influence of mediating factor. The direct effect of BMI on the prevalence of LBMD continued to be significantly negatively correlated after controlling for NHHR (−0.0273 (−0.0283, −0.0261), p < 0.001). According to Table 5, the effect of BMI on LBMD includes both direct and indirect effects, where the mediating effect is 2.3%.
Mediated analysis model path diagram.Notes: Body mass index was defined as the independent variable; Low bone mass disorders as the dependent variable; and NHHR as the mediating variable. Path a represents the regression coefficient of the association between BMI and NHHR. Path b represents the regression coefficient of the association between NHHR and Low bone mass disorders. Path c represents the simple total effect of NHHR on Low bone mass disorders. Path c’ represents the direct effect of BMI on Low bone mass disorders when controlling for NHHR.
Discussion
In recent years, a large number of studies have centered around biological and lipid markers associated with LBMD and have gradually reported the potential links between LBMD and various factors such as metabolism and the environment. This research explored the potential link between NHHR lipid markers and LBMD, revealing an L-shaped association between NHHR levels and LBMD prevalence. Multifactorial logistic analyses revealed that a negative correlation was observed between NHHR levels and the incidence of low bone mineral density among adult individuals, and the negative association remained stable after adjusting for multiple covariates, with Q4 showing a significant more negative association compared with Q1. An important interaction was observed between diabetes and gender categories, showing a strong negative correlation between NHHR and low bone mineral density (LBMD) in individuals without diabetes, a negative relationship was observed between NHHR and low bone mineral density (LBMD) in individuals with diabetes. This phenomenon was also observed in the chronic kidney disease subgroup. And a negative correlation between NHHR and LBMD was noted in both male and female subgroups, nevertheless, variations in effect magnitude or variance may induce statistical interactions despite consistent effect directions, potentially leading to false positives. Our study revealed a significant negative correlation between BMI and LBMD, which remained stable even after adjusting for multiple covariates. Specifically, the prevalence of LBMD was 84% lower in the high BMI group compared than in the low BMI group. To further explore this relationship, we conducted a mediation effect analysis, using NHHR as the mediating variable. The results revealed not only a direct effect between BMI and the prevalence of LBMD in adults, but also a partial indirect effect mediated through NHHR, with a mediated effect size of 2.3%. This finding provides a new perspective for an in-depth study of how BMI affects LBMD prevalence. Through the restricted cubic splines (RCS) curve fitting technique, we also observed a negative correlation between NHHR and adult LBMD prevalence, with an L-shaped distribution of the curve. When NHHR was less than 3.38, there was a significant negative correlation between NHHR and LBMD prevalence, with a 23% decrease in LBMD prevalence for each unit increase in NHHR. However, when NHHR exceeded 3.38, NHHR was not correlated with LBMD prevalence.
Several studies have revealed a strong link between obesity and bone density33,34,35. Specifically, obese individuals or those with a high body mass index (BMI) typically exhibit greater bone density and a lower risk of fracture. However, patients who are overly obese or have an abnormally high BMI may experience a decrease in BMD along with an increased risk of fragility fractures. This phenomenon may be associated with metabolic abnormalities and high risk of falling11. This suggests that BMI, as an indicator of body weight status, has a dual effect on BMD: within a certain range, an elevated BMI contributes to higher BMD and a lower incidence of LBMD; however, when the BMI is too high, it instead decreases BMD and contributes to fragility fractures36,37,38. The effect of BMI on BMD is exerted by both adipose tissue and muscle tissue, with muscle tissue affected mainly through mechanical loading. Adipose tissue, on the other hand, can affect bone density through lipid metabolism, and its effects have received increasing attention11,39. Previous studies have shown that HDL-C is positively correlated with bone density and is considered a protective factor, whereas LDL-C show a negative correlation with bone density and is considered a risk factor14,15,40. However, some studies have also shown a negative correlation between HDL-C and BMD16,17,41, there are conflicting conclusions about the effects of lipid metabolism on BMD, and no basic research has conducted to clarify the exact mechanisms of the effects of lipoproteins on BMD.
The preponderance of evidence suggests that there may be a complex link between CVD and LBMD22,42, and the mechanisms and indicators of the link between the two diseases are of particular importance for their joint prevention and management. NHHR, the ratio of non-HDL-C to HDL-C, is a combined indicator of CVD, particularly atherosclerosis, and collects information about all atherogenic and antiatherogenic lipid particles25,26, with high early warning value in CVD. This indicator collects information on all atherogenic and anti-atherogenic lipid particles and has high early warning value in CVD. In this study, the non-high-density lipoprotein cholesterol ratio (NHHR) was used as a combined index to comprehensively assess the effects of including LDL-C and HDL-C on the prevalence of LBMD and bone density. We hope to explore the potential clinical link between CVD and LBMD by studying the association between NHHR and LBMD, and to provide safe and effective strategies for the prevention and treatment of both diseases.
To our knowledge, this study represents the first cross-sectional study to examine the relationship between NHHR and LBMD, and the mediating effects of NHHR, which have not been explored in depth in large-scale epidemiological studies before. We found a negative correlation between NHHR and the prevalence of LBMD, suggesting that elevated NHHR decreases the prevalence of LBMD. A possible mechanism for this is, on the one hand, through the influence of HDL-C on BMD, which can be influenced by estrogens that have a significant biological effect on lipid metabolism in the blood and bone43. Moreover, HDL and BMD may have a greater genetic influence. Several animal experiments have shown that HDL-C and BMD are simultaneously influenced by the Apoe gene, with gene expression being reflected in a decrease in HDL-C levels and an increase in BMD44,45. On the other hand, BMD is affected by LDL-C, which can affect osteoclast activity by preventing apoptosis and mediating cholesterol transfer, thus affecting BMD46,47,48. To further investigate the association between NHHR and LBMD prevalence, we performed restricted cubic splines (RCS) curve fitting to investigate linear or nonlinear associations between the two, and the graphs showed an L-shaped association between the two. Threshold effect analysis revealed an inflection point at 3.38, and NHHR at < 3.38, NHHR and LBMD were significantly negatively correlated, with a 23% decrease in LBMD for every 1 unit increase in NHHR. At NHHR > 3.38 NHHR was not correlated with LBMD. That is, the prevalence decreased with increasing in NHHR, and there was no further decrease in the prevalence beyond a certain value. As an early warning indicator, elevated NHHR is often elevated NHHR serves as an early warning sign and is frequently linked to a higher likelihood of developing conditions such as CVD and diabetes25,26. However, the results of this study suggest that elevated NHHR was negatively associated with the prevalence of LBMD, but its effect on the prevalence of LBMD was no longer significant when NHHR exceeded a specific threshold. This finding provides important guidelines for the prevention and treatment of both CVD and LBMD in the clinic. We can adjust dietary and pharmacological strategies based on the basis of the values of NHHR within the threshold range of NHHR to modulate lipid metabolism to achieve the joint prevention and treatment of these two diseases. In addition, chronic metabolic diseases such as cardiovascular disease may negatively affect BMD when NHHR exceeds the threshold, which may be related to the role of various components of the bone matrix, like type I collagen, proteoglycans, in osteogenesis and atherosclerosis49. Nonetheless, the relationship between NHHR and the prevalence of LBMD needs to be validated by further basic studies and the underlying mechanisms thoroughly explored.
In our study of the association between BMI and LBMD, the mediation effect analysis showed that there were direct and indirect effects in the pathway of BMI affecting LBMD, in which NHHR, as a mediator, mediated the indirect effect of BMI on LBMD, with a mediating effect of 2.3%. These findings suggest that some of the effects of BMI on LBMD and BMD indirectly affect the prevalence of BMD and LBMD by influencing lipid metabolism, and several studies have validated the results we derived50,51,52. This result has clinical and dietary recommendations, and when considering BMD or LBMD control through BMI, the NHHR index can be derived from laboratory blood biochemistry tests and used to guide clinical management of BMD and LBMD. In addition to this, previous studies have demonstrated a similar inflection point in the association between BMI and BMD36, so we can hypothesize that the saturation effect between BMI and BMD is mediated by NHHR in the BMI and LBMD association. This may be because after the BMI threshold is exceeded, the effects of other underlying diseases37,38,53,54,55, excessive mechanical loading, and lipid metabolism due to high BMI lead to a decrease in BMD, increased occurrence of LBMD, and an increase in fragility fractures. This finding has important clinical implications for LBMD and CVD in the clinic, and while individuals with low weight and BMI are at high risk for low BMD and LBMD, blindly increasing weight and BMI poses a risk to dietary health and underlying disease. From our results, it can be concluded that the inflection point of NHHR can be referred to control BMI and lipid metabolism levels to reduce the risk of LBMD while considering CVD and other underlying diseases, providing reference for clinical nutritional diet and scientific prevention and treatment.
We further conducted subgroup analyses to investigate the potential relationship between NHHR and LBMD in different populations, and to determine whether the association between NHHR and LBMD was stable in different populations. The results of the analyses showed that, in the diabetes group, NHHR showed a significant negative correlation with LBMD among non-diabetics, and a non-significant negative correlation was observed among diabetics and there was a significant interaction between suffering from diabetes and not suffering from diabetes. There was a significant interaction between diabetics and non-diabetics; diabetes altered the association between NHHR and LBMD. This result may be related to the fact that insulin signaling can activate osteoblasts56,57, and microangiopathy in diabetes also leads to changes in bone mineral density. In addition, insulin and insulin-like growth factors affect bone metabolism itself, which may influence bone metabolism in diabetic patients and may determine changes in bone metabolism in diabetic patients58.
This study has several significant strengths. Compared with other cross-sectional studies, this study not only has a larger sample size but also has a greater degree of accuracy in representing U.S. national characteristics. We have further enhanced the credibility of the findings by including multiple covariates that account for a wide range of potential confounders. For the first time to our knowledge, this study synthesized the correlation between NHHR and LBMD, and the mediating effect of NHHR was also exhaustively explored in this study, which provides new research perspectives on preventive strategies and the clinical diagnosis and treatment of LBMD.
There are several limitations to this study. First, given its cross-sectional study design, we were unable to establish an exact causal relationship between NHHR and LBMD; therefore, the association needs to be verified by subsequent prospective studies or basic experiments. In assessing LBMD, this study was based only on BMD data in the femoral region and did not include sites such as the lumbar spine and forearm, which may have led to some omissions in LBMD. Second, although multiple covariates were included to ensure the stability of the association between NHHR and LBMD, it was not possible to exclude the effects of all confounding factors. In addition, as the study sample was limited to U.S. adults, it lacks representation of the underage population and other countries and ethnicities, necessitating larger population data to further test these associations.
Conclusion
In conclusion, our study revealed that NHHR was significantly negatively associated with the prevalence of low bone mass disorders, and there was an L-shaped relationship with an inflection point of 3.38, which facilitates our simultaneous intervention and early warning of low bone mass disorders and CVD. Moreover, we found that NHHR mediated the negative association between BMI and low bone mass disorders. This provides valuable suggestions for the prevention and diagnosis of low bone mass disorders and suggest that healthcare professionals should pay attention to lipid metabolism factors for the management of low bone mass disorders.
Data availability
All data for this study was obtained from the official NHANES website (https://www.cdc.gov/nchs/nhanes/).
Abbreviations
- NHANES:
-
National Health and Nutrition Examination Survey
- BMI:
-
Body mass index
- NCHS:
-
The National Center for Health Statistics
- CDC:
-
Centers for Disease Control and Prevention
- CI:
-
Confidence interval
- SD:
-
Standard deviation
- OR:
-
Odds ratio
- TC:
-
Total cholesterol
- HDL-C:
-
High-density lipoprotein cholesterol
- LDL-C:
-
Low-density lipoprotein cholesterol
- NHHR:
-
The non-high-density lipoprotein to high-density lipoprotein ratio
- CKD:
-
Chronic Kidney Disease
- CVD:
-
Cardiovascular disease
- BMD:
-
Bone mineral density
- LBMD:
-
Low bone mass disorders
References
Alejandro, P. & Constantinescu, F. A review of osteoporosis in the older adult: an update. Rheum. Dis. Clin. North. Am. 44 (3), 437–451 (2018).
Shevroja, E. et al. Update on the clinical use of trabecular bone score (TBS) in the management of osteoporosis: results of an expert group meeting organized by the European society for clinical and economic aspects of osteoporosis, osteoarthritis and musculoskeletal diseases (ESCEO), and the international osteoporosis foundation (IOF) under the auspices of WHO collaborating center for epidemiology of musculoskeletal health and aging. Osteoporos. Int. 34 (9), 1501–1529 (2023).
Black, D. M. & Rosen, C. J. Clinical practice. Postmenopausal osteoporosis. N Engl. J. Med. 374 (3), 254–262 (2016).
Camacho, P. M. et al. AMERICAN ASSOCIATION -2020 UPDATE. Endocr Pract 26(Suppl 1):1–46. (2020).
Cai, S. et al. Bone mineral density and osteoporosis in relation to all-cause and cause-specific mortality in NHANES: A population-based cohort study. Bone 141, 115597 (2020).
Kendler, D. L. et al. Repeating measurement of bone mineral density when monitoring with Dual-energy X-ray absorptiometry: 2019 ISCD official position. J. Clin. Densitom. 22 (4), 489–500 (2019).
Hans, D. et al. Skeletal sites for osteoporosis diagnosis: the 2005 ISCD official positions. J. Clin. Densitom. 9 (1), 15–21 (2006).
LeBoff, M. S. et al. The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos. Int. 33 (10), 2049–2102 (2022).
LeBoff, M. S. et al. Supplemental vitamin D and incident fractures in midlife and older adults. N Engl. J. Med. 387 (4), 299–309 (2022).
Zhao, C. et al. Higher BMI and lower femoral neck strength in males with type 2 diabetes mellitus and normal bone mineral density. Am. J. Med. Sci. 364 (5), 631–637 (2022).
Glauber, H. S., Vollmer, W. M., Nevitt, M. C., Ensrud, K. E. & Orwoll, E. S. Body weight versus body fat distribution, adiposity, and frame size as predictors of bone density. J. Clin. Endocrinol. Metab. 80 (4), 1118–1123 (1995).
Lin, Y. H. et al. Role of body fat and metabolic rate in Site-Specific fracture risk: A 20-Year Taiwanese cohort study. Med. Sci. Monit. 31, e947660 (2025).
Luo, Q. et al. The obesity paradox in osteoporosis risk among older adults is mostly driven by women: a population-based prospective study. BMC Geriatr. 25 (1), 58 (2025).
Jiang, Z. et al. Association of HDL and LDL levels with osteoporosis in rheumatoid arthritis: a retrospective cohort study. Eur. J. Med. Res. 29 (1), 439 (2024).
Xie, R., Huang, X., Liu, Q. & Liu, M. Positive association between high-density lipoprotein cholesterol and bone mineral density in U.S. Adults: the NHANES 2011–2018. J. Orthop. Surg. Res. 17 (1), 92 (2022).
Ersoy, G. S. et al. Lipid profile and plasma atherogenic index in postmenopausal osteoporosis. North. Clin. Istanb. 4 (3), 237–241 (2017).
Zeng, T., Tan, L., Yu, J. & Wu, Y. High density lipoprotein in rheumatoid arthritis: emerging role in predicting inflammation level and osteoporosis occurrence. Scand. J. Clin. Lab. Invest. 80 (5), 375–380 (2020).
Omura, Y., Nishio, Y. & Kashiwagi, A. [Osteoporosis and atherosclerosis]. Clin. Calcium. 17 (3), 346–353 (2007).
Farhat, G. N. & Cauley, J. A. The link between osteoporosis and cardiovascular disease. Clin. Cases Min. Bone Metab. 5 (1), 19–34 (2008).
Azeez, T. A. Osteoporosis and cardiovascular disease: a review. Mol. Biol. Rep. 50 (2), 1753–1763 (2023).
Marcovitz, P. A. et al. Usefulness of bone mineral density to predict significant coronary artery disease. Am. J. Cardiol. 96 (8), 1059–1063 (2005).
Hamerman, D. Osteoporosis and atherosclerosis: biological linkages and the emergence of dual-purpose therapies. Qjm 98 (7), 467–484 (2005).
Ahmadi, N. et al. The relation of low levels of bone mineral density with coronary artery calcium and mortality. Osteoporos. Int. 29 (7), 1609–1616 (2018).
Mao, Q., Zhao, J. & Zhao, X. Association of non-HDL-C-to-HDL-C ratio with coronary lesions and its prognostic performance in first-onset NSTEMI. Biomark. Med. 17 (1), 29–39 (2023).
Sheng, G. et al. Utility of Non-High-Density lipoprotein cholesterol to High-Density lipoprotein cholesterol ratio in evaluating incident diabetes risk. Diabetes Metab. Syndr. Obes. 15, 1677–1686 (2022).
Zhu, L. et al. Lipoprotein ratios are better than conventional lipid parameters in predicting coronary heart disease in Chinese Han people. Kardiol Pol. 73 (10), 931–938 (2015).
Chen, T. C. et al. National health and nutrition examination survey: Estimation procedures, 2011–2014. Vital Health Stat. 2, 1–26 (2018).
Akinbami, L. J. et al. National health and nutrition examination survey, 2017-March 2020 prepandemic file: Sample design, estimation, and analytic guidelines. Vital Health Stat. 1, 1–36 (2022).
Curtin, L. R. et al. The national health and nutrition examination survey: sample design, 1999–2006. Vital Health Stat. 2, 1–39 (2012).
Curtin, L. R. et al. National health and nutrition examination survey: sample design, 2007–2010. Vital Health Stat. 2, 1–23 (2013).
Skrivankova, V. W. et al. Strengthening the reporting of observational studies in epidemiology using Mendelian randomization: the STROBE-MR statement. Jama 326 (16), 1614–1621 (2021).
Kanis, J. A. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. WHO study group. Osteoporos. Int. 4 (6), 368–381 (1994).
Sun, A., Hu, J., Wang, S., Yin, F. & Liu, Z. Association of the visceral adiposity index with femur bone mineral density and osteoporosis among the U.S. Older adults from NHANES 2005–2020: a cross-sectional study. Front. Endocrinol. (Lausanne). 14, 1231527 (2023).
Russell, M. et al. Visceral fat is a negative predictor of bone density measures in obese adolescent girls. J. Clin. Endocrinol. Metab. 95 (3), 1247–1255 (2010).
Gilsanz, V. et al. Reciprocal relations of subcutaneous and visceral fat to bone structure and strength. J. Clin. Endocrinol. Metab. 94 (9), 3387–3393 (2009).
Ouyang, Y. et al. Saturation effect of body mass index on bone mineral density in adolescents of different ages: A Population-Based study. Front. Endocrinol. (Lausanne). 13, 922903 (2022).
Fassio, A. et al. The obesity paradox and osteoporosis. Eat. Weight Disord. 23 (3), 293–302 (2018).
Sadeghi, O., Saneei, P., Nasiri, M., Larijani, B. & Esmaillzadeh, A. Abdominal obesity and risk of hip fracture: A systematic review and Meta-Analysis of prospective studies. Adv. Nutr. 8 (5), 728–738 (2017).
Anagnostis, P., Florentin, M., Livadas, S., Lambrinoudaki, I. & Goulis, D. G. Bone health in patients with dyslipidemias: an underestimated aspect. Int J. Mol. Sci. 23(3), 1639 (2022).
Poli, A. et al. Plasma low-density lipoprotein cholesterol and bone mass densitometry in postmenopausal women. Obstet. Gynecol. 102 (5 Pt 1), 922–926 (2003).
Tang, Y., Wang, S., Yi, Q., Xia, Y. & Geng, B. High-density lipoprotein cholesterol is negatively correlated with bone mineral density and has potential predictive value for bone loss. Lipids Health Dis. 20 (1), 75 (2021).
Tankó, L. B. et al. Relationship between osteoporosis and cardiovascular disease in postmenopausal women. J. Bone Min. Res. 20 (11), 1912–1920 (2005).
Mendelsohn, M. E. & Karas, R. H. Molecular and cellular basis of cardiovascular gender differences. Science 308 (5728), 1583–1587 (2005).
Xiao, Q. et al. Plasminogen deficiency accelerates vessel wall disease in mice predisposed to atherosclerosis. Proc. Natl. Acad. Sci. U S A. 94 (19), 10335–10340 (1997).
Schilling, A. F. et al. Increased bone formation in mice lacking Apolipoprotein E. J. Bone Min. Res. 20 (2), 274–282 (2005).
Li, Z. et al. Cell death regulation: A new way for natural products to treat osteoporosis. Pharmacol. Res. 187, 106635 (2023).
Wang, B., Wang, H., Li, Y. & Song, L. Lipid metabolism within the bone micro-environment is closely associated with bone metabolism in physiological and pathophysiological stages. Lipids Health Dis. 21 (1), 5 (2022).
Cui, G. et al. TC and LDL-C are negatively correlated with bone mineral density in patients with osteoporosis. Am. J. Transl Res. 16 (1), 163–178 (2024).
Baldini, V., Mastropasqua, M., Francucci, C. M. & D’Erasmo, E. Cardiovascular disease and osteoporosis. J. Endocrinol. Invest. 28 (10 Suppl), 69–72 (2005).
Jia, L. & Cheng, M. Correlation analysis between risk factors, BMD and serum osteocalcin, cathek, PINP, β-crosslaps, TRAP, lipid metabolism and BMI in 128 patients with postmenopausal osteoporotic fractures. Eur. Rev. Med. Pharmacol. Sci. 26 (21), 7955–7959 (2022).
Han, H. et al. Correlation between bone density, bone metabolism markers with lipid metabolism markers and body mass index. BMC Musculoskelet. Disord. 25 (1), 162 (2024).
Nelson, L. R. & Bulun, S. E. Estrogen production and action. J. Am. Acad. Dermatol. 45 (3 Suppl), S116–124 (2001).
Yang, W. et al. Obesity and risk for respiratory diseases: a Mendelian randomization study. Front. Endocrinol. (Lausanne). 14, 1197730 (2023).
Patil, S. G., Arakeri, S. & Khode, V. Association of low BMI with aortic stiffness in young healthy individuals. Curr. Hypertens. Rev. 17 (3), 245–249 (2021).
Banack, H. R. et al. Relationship between BMI trajectories and cardiometabolic outcomes in postmenopausal women: a growth mixture modeling approach. Ann. Epidemiol. 72, 9–17 (2022).
Leidig-Bruckner, G. & Ziegler, R. Diabetes mellitus a risk for osteoporosis? Exp. Clin. Endocrinol. Diabetes. 109 (Suppl 2), S493–514 (2001).
Thrailkill, K. M., Lumpkin, C. K. Jr., Bunn, R. C., Kemp, S. F. & Fowlkes, J. L. Is insulin an anabolic agent in bone? Dissecting the diabetic bone for clues. Am. J. Physiol. Endocrinol. Metab. 289 (5), E735–745 (2005).
Moyer-Mileur, L. J., Slater, H., Jordan, K. C. & Murray, M. A. IGF-1 and IGF-binding proteins and bone mass, geometry, and strength: relation to metabolic control in adolescent girls with type 1 diabetes. J. Bone Min. Res. 23 (12), 1884–1891 (2008).
Acknowledgements
The authors thank all their colleagues in the Department of Orthopedic.
Funding
Inapplicable.
Author information
Authors and Affiliations
Contributions
XTC, WYD: Data collection, Investigation, Software, Design, Original manuscript. JW, HL, YKW: Conception, Graphing, Review, Data analysis. XTC: Wrote the paper. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Data for this study were obtained from the NHANES data builder and ethical approval was obtained from the Ethics Review Board of the Center for Health Statistics.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, X., Du, W., Li, H. et al. L-shaped association of the non-high-density lipoprotein to high-density lipoprotein ratio with low bone mass disorders. Sci Rep 15, 28583 (2025). https://doi.org/10.1038/s41598-025-12806-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12806-w