Abstract
This study aimed to identify prognostic indicators and develop a nomogram to predict the overall survival (OS) of widowed bladder cancer (WBCa) patients. WBCa patients between 2004 and 2015 were identified using the Surveillance, Epidemiology, and End Results (SEER) database. The patients were randomly divided into the training and validation sets at a 7:3 ratio. Independent prognostic factors were determined using univariate and multivariate Cox analyses. We constructed a nomogram to predict 3- and 5-year for WBCa patients based on the results of multivariate Cox regression analysis. Consistency index (C-index), receiving operating characteristic (ROC), and calibration curve were used to assess the predictive accuracy of the nomogram. Gender, age at diagnosis, ethnicity, histologic type, histologic grade, tumor‑node metastasis (TNM) stage, and surgery were identified as independent predictors of OS. The C-index value of the nomogram for predicting OS was 0.704 and 0.701 in the training and validation cohorts, respectively. The ROC curves and calibration plots indicated that the model was relatively accurate. Independent prognostic factors for WBCa patients were identified, and a nomogram was constructed to predict the 3- and 5-year OS. The model enables clinicians to determine cancer patients’ survival prognosis and formulate personalized treatment plans.
Similar content being viewed by others
Introduction
Globally, cancer represents a major health challenge, and it is the second leading cause of death in the United States. It was estimated that in 2023, the United States had witness 1,958,310 new cancer cases and 609,820 cancer deaths. According to a report, there was an estimated 82,290 new cases of urinary bladder carcinoma in 2023, and 16,710 deaths related to it1. Bladder cancer is characterized by high recurrence, rapid progression, and high mortality rates. Recent advances in surgical treatments, radiotherapy, and chemotherapy have led to improved 5-year survival rates in bladder cancer patients. Trimodality treatment and radical cystectomy have similar oncological outcomes in patients with muscle-invasive bladder cancer, as evidenced by a multi-institutional propensity score matched and weighted analysis2. Specifically, the study reported a 5-year OS rate of 66%~77%, a 5-year cancer-specific survival of 81%~85%, a 5-year metastasis-free survival of 74%~75%, and a 5-year disease-free survival of 73%~76%2. Nevertheless, identifying prognostic factors for bladder cancer is vital.
The most commonly accepted prognostic factor for bladder cancer at present is the clinical stage; however, the presence of other prognostic factors cannot be ruled out. In the process of carcinogenesis and tumor progression, cytogenetic changes play an essential role in tumor prognosis. Loss of the Y chromosome was observed in 10–40% of tumor patients, which indicated a more advanced phase of the tumor3. Besides, tumors exhibiting the loss of the Y chromosome might be more aggressive and may weaken the tumor response of the immune system. Studies have also reported poorer outcomes in patients who lost the Y chromosome compared with bladder cancer patients who retained the Y chromosome3. With the deeper understanding of human disease and health, sociopsychological factors, increasing attention is being directed toward sociopsychological factors. Fang et al.4 analyzed big data and reported that age, ethnicity, sex, surgery type, and histologic type were also prognostic factors of bladder cancer OS. Many studies5,6,7,8 revealed that marital status was a prognostic factor for bladder cancer patients. A previous study9 has indicated that bladder cancer patients who were married have a better prognosis, showing higher survival rates. Datta et al.10 reported that married patients have a higher survival rate compared to unmarried patients. However, Nelles et al.11 presented opposing viewpoints, reporting that married bladder cancer patients have no distinct survival advantage compared to their unmarried counterparts. In contrast, they found that widowed men, especially widowed men over 70 years of age, have a poorer prognosis than married men. According to a recent study6, marital status has a gender-specific effect on urothelial bladder cancer. Compared to married men, unmarried men tend to present with a more advanced tumor stage at radical cystectomy and poorer cancer-specific mortality. Nonetheless, marriage status showed no significant impact on oncological outcomes in women. While the effects of marital status on the outcomes of bladder cancer patients remain controversial, married patients are hypothesized to benefit from a more favorable prognosis than others. Still, few studies have investigated the prognostic factors and outcomes in widowed bladder cancer patients. Widowed patients often represent underprivileged sections of society due to distinct psychosocial vulnerabilities or treatment disparities. Besides, they may receive less social support and lower protective health-promoting factors, thereby having poorer survival chances. Social support and health education may emerge as important factors limiting the treatment. Therefore, widowed patients should be investigated separately, rather than grouping them with all unmarried individuals.
Around 30% of the US population is included in the SEER program, which collects data on cancer diagnosis, treatment, and survival from 18 registries12. Therefore, the effects of marital status on bladder cancer can be analyzed. Using nomograms, clinicians can predict the occurrence and progression of diseases based on meaningful clinical indicators. In the medical field, nomograms have been widely applied to predict the prognosis of individuals with diverse cancer types, including breast cancer, gastric cancer, and lung cancer13,14,15. The present study developed and validated a nomogram model to estimate the 3-year and 5-year OS rates for widowed bladder cancer patients. The results provide clinicians with more accurate and personalized prognosis estimates, facilitating the formulation of clinical treatment plans for widowed bladder cancer patients.
Methods
Data source and patients
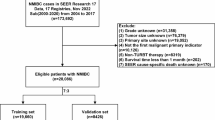
The procedures in this study strictly adhered to the principles laid out in the Helsinki Declaration. The data were obtained from the SEER database of the National Cancer Institute using SEER*Stat software (http://seer.cancer.gov/). Proper access was obtained to collect and analyze the data from the SEER database. Since the SEER database does not contain any personal patient information, ethics approval was not needed. Inclusion Criteria: (1) year of diagnosis 2004 ~ 2015; (2) primary site C67; (3) diagnostic confirmed by positive histology; (4) first malignant primary indicator; (5) marital status widowed. Exclusion criteria: (1) unknown or 0 survival time; (2) unknown race or histologic grade; (3) age < 20 years; (4) unclear surgery information; (5) unknown TNM stage. The study flowchart is shown in Fig. 1.
Study flowchart.
Study variables
The SEER database includes the demographic characteristics, clinicopathological features, treatment, and prognosis status of cancer patients. The study variables included demographic characteristics (sex, age at diagnosis, and race), clinicopathological features (histologic grade, histologic type, T staging, N staging, M staging, and distant organ metastasis), treatment modalities, and survival situation. Age was divided into three groups (40 ~ 59 years, 60 ~ 79 years, and 80 years and older). Moreover, the patients were divided into the well-differentiated (Grade I ~ II) and poorly differentiated groups (Grade III ~ IV) based on the histologic grade. Histologic type included transitional cell carcinoma (TCC), squamous cell carcinoma (SCC), adenocarcinoma, and others. Medullary bladder cancer TNM status was determined by using the sixth edition of TNM classification for the tumor (T), node (N), and metastasis (M) status. T staging was categorized into non-muscle invasive (Ta/T1/Tis) and muscle invasive (T2 ~ T4), while N (M) staging was categorized into metastasis (N1 ~ N3 and M1) and without metastasis (N0 and M0). Surgery was classified into four groups: none, transurethral resection of bladder tumor (TURBT), radical cystectomy (RC), and others. Due to the lack of precise treatment timing in the SEER records, chemotherapy and radiation therapy were modeled as time-fixed covariates (yes/no). The primary outcome measure in this study was OS, which was defined as the survival time from diagnosis until death.
Statistical analysis
Patient data from the SEER database were randomly divided into the training and validation sets at a 7:3 ratio. Categorical variables were expressed as frequencies (n) and percentages (%). Chi-square tests were performed to compare groups. A univariate Cox analysis was performed to screen prognostic factors. Subsequently, independent risk factors were evaluated using a multivariate Cox analysis. In both univariate and multivariate analyses, variables with p values less than 0.05 were considered statistically significant. Then, a nomogram for 3- and 5-year OS was constructed based on the independent prognostic factors (P < 0.05) identified in the multivariate Cox analysis. In order to evaluate the model’s ability to predict outcomes, the C-index and calibration curve were utilized. Furthermore, the receiver operating characteristic (ROC) curve was used to assess the predictive performance of the 3- and 5-year OS nomograms. Additionally, Kaplan-Meier curves were plotted, and log-rank tests were performed to compare survival curves. R software (www.rproject.org) was used for statistical analysis. In this study, p values less than 0.05 were considered statistically significant.
Results
Patients baseline characteristics
A total of 15,509 WBCa patients were enrolled in the present study, with a male-to-female ratio of almost 1:1. The elderly showed a high incidence of WBCa, while people under 60 accounted for only 2.5% of the study population. In terms of race, 13,944 (89.9%) patients were white, 896 (5.8%) patients were black, and 669 (4.3%) patients were of other races. In terms of tumor characteristics, urothelial carcinoma (n = 14,907, 96.1%), non-muscle-invasive bladder cancer (n = 11,020, 71.1%), without lymph node metastasis (n = 14,911, 96.1%), and distant metastasis (n = 15,021, 96.9%) were the most common. The total number of patients undergoing no surgery, TURBT, RC, and others was 530 (3.4%), 11,011 (71.0%), 1,498 (9.7%), and 2,470 (15.9%), respectively. Notably, 1,188 (7.7%) patients received radiotherapy, while 2,815 (18.2%) patients received chemotherapy. Among all the patients, 12,132 (78.2%) eventually died. The patients were randomized into the training (n = 10,857) and validation (n = 4,652) sets according to a 7:3 ratio. Table 1 shows the baseline characteristics of the patients.
Independent prognosis‑predictive factors of OS
Univariate Cox regression analysis suggested that sex, age at diagnosis, race, histologic grade, histological type, T stage, N stage, M stage, surgery, and radiotherapy were factors related to OS in WBCa. Multivariate Cox regression analysis suggested that sex, age at diagnosis, sex, race, histologic grade, histological type, T stage, N stage, M stage, and surgery were prognostic factors for OS. Supplementary Table 1 shows the results of the univariate and multivariate analyses. In the multivariate analysis, male (HR:1.239, 95%CI:1.187 ~ 1.294, P < 0.001), higher age (60 ~ 79 years vs. 40 ~ 59 years, HR:1.962, 95%CI:1.615 ~ 2.382, P < 0.001; 80 ~ years vs. 40 ~ 59 years, HR:4.172, 95%CI: 3.438 ~ 5.063, P < 0.001), black race (HR:1.196, 95%CI: 1.093 ~ 1.309, P < 0.001), higher histologic grade (Grade III ~ IV vs. Grade I ~ II, HR:1.347, 95%CI:1.282 ~ 1.415, P < 0.001), non-urothelial carcinoma (SCC vs. TCC, HR:2.176, 95%CI: 1.831 ~ 2.585, P < 0.001; adenocarcinoma vs. TCC, HR:1.602,95%CI:1.243 ~ 2.065, P < 0.001), and invasive carcinoma (T2 ~ T4 vs. Ta/T1/Tis, HR:2.537, 95%CI:2.381 ~ 2.702, P < 0.001) were significantly associated with a poor OS. In addition, lymph node metastasis (N1 ~ N3 vs. N0, HR:1.732, 95%CI:1.545 ~ 1.941, P < 0.001) and distant metastasis (M1 vs. M0, HR:2.340, 95%CI:2.069 ~ 2.646, P < 0.001) also predicted a poorer OS. Moreover, treatment modalities (TURBT vs. None, HR:0.813, 95%CI:0.724 ~ 0.913, P < 0.001; RC vs. None, HR:0.418, 95%CI:0.363 ~ 0.482, P < 0.001; Others vs. None, HR:0.807, 95%CI:0.713 ~ 0.915, P = 0.001) were identified as prognostic protective factors for OS. Figure 2 shows the Kaplan–Meier curves for age at diagnosis, sex, T stage, N stage, M stage, and surgery.
Kaplan–Meier estimated OS for WBCa patients stratified by age (a), sex (b), stage T (c), stage N (d), stage M (e), and surgery (f). (All P < 0.001).
Development of a prognostic nomogram for OS
All factors showing a P value less than 0.05 in the multivariate Cox regression analysis were used to construct the predictive model to predict 3- and 5-year overall survival (Fig. 3). Each variable is predicted by its corresponding point, and the total score is calculated by adding the scores of all the variables. Individual survival axes are drawn between the “total point” and the “survival axes” to estimate the probability of 3- and 5-year OS. For example, a 70 years (47) white (11) female (0) patient with histologic grade II (0), TCC (0), and T2N0M0 (63), underwent RC (0), yielding a total score of 121, corresponding to a predicted 80% 3-year survival rate and 70% 5-year survival rate.
Prognostic nomogram predicting 3- and 5-year OS rates for WBCa patients.
Validation and calibration of the nomogram
The C-index was used to evaluate the discrimination ability of the nomogram, yielding values of 0.704 and 0.701 in the training and validation sets, respectively. These results indicated that the nomogram is an effective tool for predicting OS. The ROC curve of the nomogram also showed acceptable accuracy. The area under the curve (AUC) of the 3- and 5-year OS was 0.762 and 0.757 in the training set (Fig. 4a), while 0.757 and 0.753 in the validation set, respectively (Fig. 4b). Meanwhile, the training and validation sets were calibrated for the 3- and 5-year OS nomogram. According to the calibration plots, the nomogram demonstrated a favorable predictive accuracy in both sets (Fig. 4c and d).
The ROC curve for predicting the 3- and 5-year OS in the training (a) and validation (b) set for WBCa patients; the calibration curve for predicting 3- and 5-year OS in the training (c) and validation (d) set for WBCa patients.
Discussion
Bladder cancer is the second most common malignant carcinoma of the genitourinary tract; in addition, the tumor ranks 10th in terms of incidence among both sexes, and 7th among men16. Generally, bladder cancer can be classified as muscle-invasive bladder cancers or non-muscle-invasive bladder cancers. Although 75% of patients are diagnosed at the non-muscle-invasive stage, 78% of patients relapse within 5 years. Once the cancer progresses to invasive stage, bladder cancer becomes more malignant with poor prognostics. Notably, untreated patients are exposed to a significantly higher mortality risk. The therapeutic options include TURBT, partial cystectomy, RC, radiation therapy, and chemical therapy. TURBT remains the therapy of choice for non-muscle-invasive bladder cancer, while muscle-invasive bladder cancer is primarily treated with RC. Despite remarkable improvements in treatment, the prognosis of bladder cancer remains unsatisfactory.
Previous studies [6–7,9−11] have reported that unmarried (including never married, divorced, and widowed) bladder cancer patients showed a poor prognosis. However, the prediction model of marital status in bladder cancer was lacking. Besides, further research is warranted to focus on WBCa patients as they are often underprivileged in society. Notably, social support and health education may be important factors limiting the treatment. Hence, the present study focused on this group of patients and developed a nomogram model to predict the 3- and 5-year OS in WBCa patients.
Firstly, prognostic factors for WBCa patients were identified by multivariate Cox analysis. The Cox analysis revealed that age at diagnosis and T stage are independent predictors of OS. These factors were also top contributors in the nomogram. The nomogram revealed that age is the most important factor in determining risk scores for 3- and 5-year OS (Fig. 3). The results of our study indicated that patients with a higher grade of tumor T stage and age at diagnosis exhibit poorer prognosis. Nonetheless, the impact of age on the prognosis of bladder cancer patients remains controversial. Several studies support the conclusion that aging increases the risk of poor survival outcomes4,17,18. Older patients, especially those aged 85 years and above, may not tolerate aggressive treatment. However, Luzzago et al.19 reported that older age is associated with a lower cancer-specific mortality for patients with regional and metastatic bladder cancer. In the present study, both 3- and 5-year OS decreased with age among WBCa patients (P < 0.001). This discrepancy may be attributed to a variety of causes. First, smokers exhibit a higher disease-specific mortality, suggesting that smoking may contribute to bladder cancer development. Specifically, the harm caused by smoking increases with the intensity and duration of smoking20,21,22. Smoking is more common in men than women, and the accumulation of chemical carcinogens in the bladder increases with age23,24. Secondly, malignant tumors occur more frequently in elderly people, showing an intimate association with age. The ability of neutrophils and macrophages to engulf pathogens decreases with age, resulting in immune disorders that promote tumor development25. Furthermore, telomeres shorten as cells age, impairing DNA mutation repair in cells26,27. Finally, older patients may not tolerate aggressive treatment, which may exacerbate the difference in outcomes between them and younger patients. Moreover, elderly patients may not be eligible for surgery due to cardiovascular disease, diabetes, and other conditions28. The situation is also more complicated in older people due to post-treatment complications, such as fever, infections, and blood clots29. Related complications greatly impact postoperative recovery and quality of life for surgical patients. Compared with open surgery, laparoscopic surgery could increase postoperative quality of life. Gavi et al.30 reported that robotic-assisted radical cystectomy has a lower minor complication (Clavien–Dindo I–II) rate compared to open radical cystectomy (51% vs. 86%, P = 0.01). No significant difference in major complication (Clavien–Dindo grade > = III) rate was observed at 90 days between robotic-assisted radical cystectomy and open radical cystectomy before and after propensity score-matched analysis (before: 85% vs. 86%, P = 0.36; after: 88% vs. 85%, P = 0.22). These results implied no evident advantages in terms of reducing major complications or enhancing quality of life with robotic-assisted radical cystectomy. However, Palermo et al.31 reported contrary results. The presence of liver and kidney disease may also affect the use of chemotherapy drugs and immune checkpoint inhibitors32,33. Therefore, the age should be considered to predict the prognosis of WBCa patients.
Some demographic characteristics of the WBCa patients, such as gender and ethnicity, were also identified as independent predictors of OS. Previous studies34,35,36 on bladder cancer outcomes have shown that Black patients have a worse outcome than White patients. Notably, Black patients may be less likely to receive treatment, especially RC and pelvic lymph node dissection, which are associated with improved outcomes in the entire cohort. In addition, a review of the SEER database by Hollenbeck et al.37 showed that Black patients received fewer endoscopic evaluations than White patients, despite a similar rate of cystectomy. In addition, the mortality rate in Black patients was still higher than in White patients. However, the Florida Cancer Data System review found that Black patients were more commonly treated with RC and showed better OS than White patients despite presenting with higher stage tumors, lymph node involvement, and metastasis38. Our study showed poorer outcomes in Black WBCa patients (Black vs. White, HR:1.96, 95%CI: 1.093 ~ 1.309, P < 0.001). Furthermore, the majority of previous studies39,40,41 revealed that women with bladder cancer have a poorer prognosis compared to men. Only one study42 suggested that gender was not an independent predictor of recurrence-free survival, OS, and cancer-specific survival. However, no study has demonstrated that women have better prognoses. Unlike previous studies, our study found a higher OS in WBCa women compared to males (male vs. female, HR:1.239, 95%CI: 1.187 ~ 1.294, P < 0.001).
Similar to other malignancies, histologic grade, histologic type, and American Joint Commission on Cancer (AJCC) stage were important prognostic factors. Additionally, surgery was identified as an independent protective factor for WBCa patients, as in previous studies. In the present study, 4,489 (28.9%) patients had infiltration of the muscle layer, and 11,020 (71.1%) had no myometrial invasion. Consequently, 11,011 (71.0%) patients underwent TURBT and 1,498 (9.7%) underwent RC. At present, TURBT remains the preferred surgical procedure for treating non-muscle invasive bladder cancer, while the gold standard for muscle invasive bladder cancer is open radical cystectomy with pelvic lymph node dissection. Bladder cancer patients with distant metastases may receive comprehensive treatment measures. In recent years, the use of robot-assisted laparoscopy has increased in urology, gynecology, and general surgery. Some studies43,44 revealed that robot-assisted radical cystectomy also has technical feasibility and safety. Another multinational multi-institutional study45 reported long-term oncological outcomes associated with robot-assisted radical cystectomy. According to their findings, robot-assisted radical cystectomy with intracorporeal urinary diversion yields similar long-term oncologic outcomes to open surgery. Interestingly, radiotherapy and chemotherapy were not identified as independent prognostic factors in our study, which might be due to missing variables or selection bias. Besides, radiotherapy and chemotherapy are limited by their toxic effects, chemoresistance, and treatment interruption, thereby worsening prognosis. Radiotherapy and chemotherapy provided effective systemic therapy for micrometastases, enhanced the delivery of drugs directly into the bladder, and improved the performance status of naive patients following major surgery46. However, high-grade toxicity was observed in more than one out of three patients, which yielded no significant improvement in OS. Therefore, selecting patients for neoadjuvant chemotherapy while preventing drug toxicity is challenging47. Bladder cancer treatment options have increased following the introduction of immune checkpoint blockade and targeted therapy in recent years. Pembrolizmab has been approved as a second-line therapy, and avelumab for maintenance therapy48. A multi-center randomized49 phase 3 trial investigated the use of atezolizumab, either as monotherapy or alongside platinum-based chemotherapy, compared to chemotherapy alone for first-line treatment of patients with locally advanced or metastatic bladder carcinoma. The results indicated that combined treatment with atezolizumab and platinum-based chemotherapy could significantly increase survival. Other multi-center real-world retrospective studies suggested that neoadjuvant immunotherapy showed the highest complete response rate and pathological de-escalation rate50,51. In contrast, another randomized phase 3 trial study52 reported that combined treatment with pembrolizumab and platinum-based chemotherapy did not significantly improve efficacy and prolong survival. The studies mentioned above indicate that immunotherapy and targeted therapy should not be broadly implemented for advanced muscle-invasive bladder cancer.
Based on the above results, a prognostic nomogram-based model was constructed to predict the 3- and 5-year OS of WBCa patients. Incorporating multiple independent prognostic factors could improve the model’s accuracy to assess an individual’s survival probability. In the past few years, several nomograms have been developed for different tumor types and have shown a strong predictive ability surpassing that of the TNM system. In addition, clinicians can intuitively evaluate patients’ physical condition and offer individual predictions by using nomograms53. Meanwhile, the C-index, ROC curves, and calibration plots indicated that the model constructed for WBCa patients was relatively accurate. Our prediction model is simple and easy to use. Bladder cancer is divided into non-muscular invasive bladder cancer (Ta/T1/Tis) and high-grade muscular invasive bladder cancer (T2 ~ T4) according to the European Association of Urology guidelines. Patients with distant metastasis were diagnosed at an advanced stage, demonstrating poor prognosis and short survival. For example, a male patient with distant metastasis has a total score of (distant metastasis [M1]: 60; T2 ~ T4: 63). According to our prediction model, the 3-year survival rate is no more than 80%, while the 5-year survival rate is no more than 70%. Besides, bladder cancer is mostly observed in older individuals, typically affecting those over 60 years of age. A 65-year-patient with distant metastasis has a total score of more than 170 (distant metastasis [M1]: 60; T2 ~ T4: 63; 65 years: 47). Such a patient has a 3-year survival rate of no more than 60%, and a 5-year survival rate of no more than 45%. If the patient undergoes the TURBT or RC, the 3-year survival rate is no more than 40% or 60%, and the 5-year survival rate is no more than 25% or 45%. Patients with distant metastasis or older individuals typically do not benefit significantly from surgery, as evidenced by the above postoperative survival rates. Meanwhile, patients face a substantial surgical risk for the chance of a cure. Surgery should not be performed in patients with distant metastasis or age > = 85 years. The survival and surgical risk should be evaluated with caution, and treatment should be individualized for WBCa patients according to our prediction model.
To the extent of our knowledge, this is the first cohort study to examine risk factors for WBCa prognosis and develop a standardized nomogram. Nevertheless, the limitations of this study should be acknowledged. First, it was a retrospective study based on the SEER database, and may involve selection bias. Therefore, large-scale prospective studies should be conducted to verify and explore possible prognostic factors. Secondly, only general treatment information is available in the SEER database, and the absence of detailed clinical information, such as specific chemotherapy and radiotherapy regimens, targeted therapy, and immunotherapy, may have introduced biases. Besides, the SEER database only classifies “widowed” as a single marital status category without distinguishing the duration of widowhood, which poses challenges in the clinical translation of the model. Furthermore, the nomogram model has not been externally validated. Therefore, we plan to analyze additional data in the United States and our country to enhance the model during subsequent analysis.
Conclusion
Independent prognostic factors for WBCa patients were identified from data from the SEER database, including gender, age at diagnosis, ethnicity, histologic type, histologic grade, TNM stage, and surgery. Then, a nomogram was constructed to predict the 3- and 5-year OS. This model facilitates the prediction of survival and the formulation of personalized treatment plans. However, the model requires further validation.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- OS:
-
Overall survival
- AUC:
-
Area under the curve
- SEER:
-
Surveillance, Epidemiology, and End Results
- TNM:
-
Tumor‑node metastasis
- AJCC:
-
American Joint Commission on Cancer
- HR:
-
Hazard rations
- C‑index:
-
Consistency index
- ROC:
-
Receiver operating characteristic
- TCC:
-
Transitional cell carcinoma
- SCC:
-
Squamous cell carcinoma
- TURBT:
-
Transurethral resection of bladder tumour
- RC:
-
Radical cystectomy
- WBCa:
-
Widowed bladder cancer
References
Siegel, R. L. et al. Cancer statistics, 2023. CA-CANCER J. CLIN. 73 (1), 17–48. https://doi.org/10.3322/caac.21763 (2023).
Zlotta, A. R. et al. Radical cystectomy versus trimodality therapy for muscle-invasive bladder cancer: a multi-institutional propensity score matched and weighted analysis. LANCET ONCOL. 24 (6), 669–681. https://doi.org/10.1016/S1470-2045(23)00170-5 (2023).
Russo, P. et al. Relationship between loss of Y chromosome and urologic cancers: New future perspectives. Cancers (Basel)https://doi.org/10.3390/cancers16223766 (2024).
Fang, W. et al. Ethnicity and survival in bladder cancer: A population-based study based on the SEER database. J. Transl. Med.18(1), 145. https://doi.org/10.1186/s12967-020-02308-w (2020).
Li, S. et al. Nomogram to predict overall survival in patients with primary bladder neuroendocrine carcinoma: A population-based study. Future Oncol.18(38), 4171–4181. https://doi.org/10.2217/fon-2022-0843 (2023).
Rosiello, G. et al. Unmarried men have worse oncologic outcomes after radical cystectomy for nonmetastatic urothelial bladder cancer. UROL. ONCOL-SEMIN ORI. 38 (3), 76e1–76e9. https://doi.org/10.1016/j.urolonc.2019.10.018 (2019).
Niu, Q. et al. The effect of marital status on the survival of patients with bladder urothelial carcinoma: A SEER database analysis. Medicine97(29), e11378. https://doi.org/10.1097/MD.0000000000011378 (2018).
Klaassen, Z. et al. Female, black, and unmarried patients are more likely to present with metastatic bladder urothelial carcinoma. CLIN. GENITOURIN. CANC. 14 (5), e489–e492. https://doi.org/10.1016/j.clgc.2016.04.006 (2016).
Gore, J. L. et al. Marriage and mortality in bladder carcinoma. Cancer104(6), 1188–1194. https://doi.org/10.1002/cncr.21295 (2005).
Datta, G. D. et al. Marital status and survival following bladder cancer. J. EPIDEMIOL. COMMUN. H. 63 (10), 807–813. https://doi.org/10.1136/jech.2008.082438 (2009).
Nelles, J. L., Joseph, S. A. & Konety, B. R. The impact of marriage on bladder cancer mortality. UROL. ONCOL-SEMIN ORI. 27 (3), 263–267. https://doi.org/10.1016/j.urolonc.2008.04.016 (2008).
Hankey, B. F., Ries, L. A. & Edwards, B. K. The surveillance, epidemiology, and end results program: a national resource. CANCER EPIDEM BIOMAR. ; 8 (12): 1117-21. PMID: 10613347. (1999).
Pan, X. et al. Nomogram for predicting the overall survival of patients with inflammatory breast cancer: A SEER-based study. BREAST 47, 56–61. https://doi.org/10.1016/j.breast.2019.05.015 (2019).
Yang, Y. et al. Based on the development and verification of a risk stratification nomogram: Predicting the risk of lung cancer-specific mortality in stage IIIA-N2 unresectable large cell lung neuroendocrine cancer compared with lung squamous cell cancer and lung adenocarcinoma. Front. Oncol.12, 825598. https://doi.org/10.3389/fonc.2022.825598 (2022).
Yu, C. & Zhang, Y. Development and validation of prognostic nomogram for young patients with gastric cancer. Ann. Transl. Med.7(22), 641. https://doi.org/10.21037/atm.2019.10.77 (2019).
Liu, L. et al. A model for predicting overall survival in bladder cancer patients with signet ring cell carcinoma: A population-based study. Eur. J. Med. Res.28(1), 61. https://doi.org/10.1186/s40001-022-00970-y (2023).
Lin, W. et al. Impact of age at diagnosis of bladder cancer on survival: A surveillance, epidemiology, and end results-based study 2004–2015. Cancer Control30, 10732748231152322. https://doi.org/10.1177/10732748231152322 (2023).
Amiri, M. et al. Survival rate of patients with bladder cancer and its related factors in Kurdistan Province (2013–2018): A population-based study. BMC Urol.20(1), 195. https://doi.org/10.1186/s12894-020-00769-1 (2020).
Luzzago, S. et al. Effect of age on Cancer-specific mortality in patients with urothelial carcinoma of the urinary bladder: A Population-based Competing-risks analysis across disease stages. AM. J. CLIN. ONCOL-CANC. 43 (12), 880–888. https://doi.org/10.1097/COC.0000000000000766 (2020).
Cumberbatch, M. G. et al. The role of tobacco smoke in bladder and kidney carcinogenesis: A comparison of exposures and Meta-analysis of incidence and mortality risks. EUR. UROL. 70 (3), 458–466. https://doi.org/10.1016/j.eururo.2015.06.042 (2015).
Stern, M. C. et al. Polymorphisms in DNA repair genes, smoking, and bladder cancer risk: findings from the international consortium of bladder cancer. CANCER RES. 69 (17), 6857–6864. https://doi.org/10.1158/0008-5472.CAN-09-1091 (2009).
Teoh, J. Y. et al. Global trends of bladder cancer incidence and mortality, and their associations with tobacco use and gross domestic product per capita. EUR. UROL. 78 (6), 893–906. https://doi.org/10.1016/j.eururo.2020.09.006 (2020).
Freedman, N. D. Association between smoking and risk of bladder cancer among men and women. JAMA306(7), 737–745. https://doi.org/10.1001/jama.2011.1142 (2011).
Samanic, C. et al. Smoking and bladder cancer in spain: effects of tobacco type, timing, environmental tobacco smoke, and gender. CANCER EPIDEM BIOMAR. 15 (7), 1348–1354. https://doi.org/10.1158/1055-9965.EPI-06-0021 (2006).
Jackaman, C. et al. Aging and cancer: the role of macrophages and neutrophils. AGEING RES. REV. 36, 105–116. https://doi.org/10.1016/j.arr.2017.03.008 (2017).
Campisi, J. & Yaswen, P. Aging and cancer cell biology, AGING CELL. 2009; 8 (3): 221-5. (2009). https://doi.org/10.1111/j.1474-9726.2009.00475.x
Shay, J. W. Role of telomeres and telomerase in aging and cancer. CANCER DISCOV. 6 (6), 584–593. https://doi.org/10.1158/2159-8290.CD-16-0062 (2016).
Turrentine, F. E. et al. Surgical risk factors, morbidity, and mortality in elderly patients. J. AM. COLL. Surg. 203 (6), 865–877. https://doi.org/10.1016/j.jamcollsurg.2006.08.026 (2006).
Lin, H. S. et al. Frailty and post-operative outcomes in older surgical patients: A systematic review. BMC Geriatr.16(1), 157. https://doi.org/10.1186/s12877-016-0329-8 (2016).
Gavi, F. et al. Assessing trifecta and pentafecta success rates between robot-assisted vs. open radical cystectomy: A propensity score-matched analysis. Cancers (Basel)https://doi.org/10.3390/cancers16071270 (2024).
Palermo, G. et al. The mental and emotional status after radical cystectomy and different urinary diversion orthotopic bladder substitution versus external urinary diversion after radical cystectomy: A propensity score-matched study. INT. J. UROL. 31 (12), 1423–1428. https://doi.org/10.1111/iju.15586 (2024).
Lichtman, S. M. et al. International society of geriatric oncology chemotherapy taskforce: evaluation of chemotherapy in older patients–an analysis of the medical literature. J. CLIN. ONCOL. 25 (14), 1832–1843. https://doi.org/10.1200/JCO.2007.10.6583 (2007).
Martini, A. et al. Adjuvant immunotherapy in patients with high-risk muscle-invasive urothelial carcinoma: The potential impact of informative censoring. Cancer128(15), 2892–2897. https://doi.org/10.1002/cncr.34255 (2022).
Gild, P. et al. Racial disparity in quality of care and overall survival among black vs. white patients with muscle-invasive bladder cancer treated with radical cystectomy: A National cancer database analysis. UROL. ONCOL-SEMIN ORI. 36 (10), 469. https://doi.org/10.1016/j.urolonc.2018.07.012 (2018).
Kaye, D. R. et al. Do African American patients treated with radical cystectomy for bladder cancer have worse overall survival?? Accounting for pathologic staging and patient demographics beyond race makes a difference. Bladder Cancer2(2), 225–234. https://doi.org/10.3233/BLC-150041 (2016).
Williams, S. B. et al. Population-based assessment of racial/ethnic differences in utilization of radical cystectomy for patients diagnosed with bladder cancer. CANCER CAUSE CONTROL. 28 (7), 755–766. https://doi.org/10.1007/s10552-017-0902-2 (2017).
Hollenbeck, B. K. et al. Racial differences in treatment and outcomes among patients with early stage bladder cancer. Cancer116(1), 50–56. https://doi.org/10.1002/cncr.24701 (2010).
DeDeugd, C. et al. The influence of race on overall survival in patients with newly diagnosed bladder cancer. J. RACIAL ETHN. HEALTH. 2 (1), 124–131. https://doi.org/10.1007/s40615-014-0055-x (2014).
Williams, S. B. et al. Survival differences among patients with bladder cancer according to sex: critical evaluation of radical cystectomy use and delay to treatment. UROL. ONCOL-SEMIN ORI. 35 (10), 602. .e1-602.e9 (2017).
Bryan, R. T. et al. A comparative analysis of the influence of gender, pathway delays, and risk factor exposures on the Long-term outcomes of bladder cancer. Eur. Urol. Focus. 1 (1), 82–89. https://doi.org/10.1016/j.euf.2015.01.001 (2015).
Thorstenson, A. et al. Gender-related differences in urothelial carcinoma of the bladder: a population-based study from the Swedish National registry of urinary bladder cancer. SCAND. J. UROL. 50 (4), 292–297. https://doi.org/10.3109/21681805.2016.1158207 (2016).
Pichler, R. et al. Gender-related outcome in bladder cancer patients undergoing radical cystectomy. J. Cancer.8(17), 3567–3574. https://doi.org/10.7150/jca.21130 (2017).
Liu, L. et al. Effects of different organ metastases on the prognosis of stage IV urothelial carcinoma of the bladder. J. ONCOL. 2022 (8594022). https://doi.org/10.1155/2022/8594022 (2022).
Mastroianni, R. et al. Open radical cystectomy versus Robot-Assisted radical cystectomy with intracorporeal urinary diversion: early outcomes of a Single-Center randomized controlled trial. J. Urol. 207 (5), 982–992. https://doi.org/10.1097/JU.0000000000002422 (2022).
Brassetti, A. et al. Long-term oncologic outcomes of robot-assisted radical cystectomy (RARC) with totally intracorporeal urinary diversion (ICUD): A multi-center study. World J. Urol.38(4), 837–843. https://doi.org/10.1007/s00345-019-02842-3 (2019).
Schag, C. C., Heinrich, R. L. & Ganz, P. A. Karnofsky performance status revisited: reliability, validity, and guidelines. J. CLIN. ONCOL. 2 (3), 187–193. https://doi.org/10.1200/JCO.1984.2.3.187 (1984).
Iacovino, M. L. et al. Novel therapeutic opportunities in neoadjuvant setting in urothelial cancers: A new horizon opened by molecular classification and immune checkpoint inhibitors. Int. J. Mol. Sci.https://doi.org/10.3390/ijms23031133 (2022).
Kokura, K. et al. A case of avelumab response to multiple bone and lymph node metastases of plasmacytoid variant bladder cancer. Urol. Case Rep. 47, 102358. https://doi.org/10.1016/j.eucr.2023.102358 (2023).
Galsky, M. D. et al. Atezolizumab with or without chemotherapy in metastatic urothelial cancer (IMvigor130): a multicentre, randomised, placebo-controlled phase 3 trial. LANCET 395 (10236), 1547–1557. https://doi.org/10.1016/S0140-6736(20)30230-0 (2020).
Hu, J. et al. Efficacy and biomarker analysis of neoadjuvant disitamab vedotin (RC48-ADC) combined immunotherapy in patients with muscle-invasive bladder cancer: A multi-center real-world study. Imeta4(3), e70033. https://doi.org/10.1002/imt2.70033 (2025).
Hu, J. et al. Neoadjuvant immunotherapy, chemotherapy, and combination therapy in muscle-invasive bladder cancer: A multi-center real-world retrospective study. Cell. Rep. Med. 3 (11), 100785. https://doi.org/10.1016/j.xcrm.2022.100785 (2022).
Powles, T. et al. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. LANCET ONCOL. 22 (7), 931–945. https://doi.org/10.1016/S1470-2045(21)00152-2 (2021).
Liu, L. et al. Development and validation a model for predicting overall survival of bladder cancer with lung metastasis: A population-based study. Eur. J. Med. Res.28(1), 279. https://doi.org/10.1186/s40001-023-01261-w (2023).
Acknowledgements
We acknowledge the efforts of the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries for creating the SEER database.
Funding
None.
Author information
Authors and Affiliations
Contributions
FS: Conceptualization, methodology, software, writing—original draft. YL: Data curation, methodology, review editing. QL: Data curation, visualization, methodology. LL: Methodology, software, writing—original draft. LG: Conceptualization, validation, writing—review & editing. PY: Data curation, writing draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval statement
The data of this study were obtained from the SEER database. The patients’ data were public and anonymous, so this study does not require ethical approval and informed consent.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, FZ., Liu, YX., Xu, QL. et al. Oncological outcome and survival predictors of widowed patients with bladder cancer in special populations. Sci Rep 15, 30638 (2025). https://doi.org/10.1038/s41598-025-13033-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13033-z