Abstract
This report presents the development of Pd-BPA/Fe3O4, a cutting-edge magnetic nanocatalyst. The synthesis process employed (2,2′-Bipyridin-5-yl) alanine groups as linkers to immobilize palladium nanoparticles (Pd NPs) on the Fe3O4 surface, effectively reducing particle agglomeration. This innovative surface functionalization of magnetic particles serves as a promising approach to merge the benefits of heterogeneous and homogeneous catalysis. The catalyst underwent thorough characterization using various analytical techniques such as SEM, XRD, FTIR, ICP, EDS, and VSM. Its performance was assessed through catalytic applications in the synthesis of aromatic sulfides via C-S coupling reactions, achieving derivative yields between 85 and 98%. A leaching test was conducted to confirm the catalyst’s heterogeneous nature. Impressively, Pd-BPA/Fe3O4 demonstrated excellent reusability, maintaining catalytic efficiency over five cycles of recovery and reuse without notable loss in activity.
Similar content being viewed by others
Introduction
In modern catalysis, supported heterogeneous catalysts have emerged as transformative tools with substantial environmental and industrial relevance1,2. By contrast, the application of homogeneous catalysts has been significantly limited, largely to avoid the cumbersome processes of their separation and regeneration from reaction mixtures3,4. The strategic design and reproducible synthesis of supported catalytic systems hold immense importance in enabling catalytic organic transformations5,6,7,8. Among the various approaches explored, nanomaterial-supported catalysts have gained considerable attention9. Recent advancements have seen extensive research on metal and metal oxide nanoparticle catalysts, which are supported on inert solid platforms such as organic polymers, activated carbon, boehmite, carbon nanotubes, clays, iron oxides, alumina, or molecular sieves (e.g., MCM-41, SBA-15)10,11,12. Among these options, economical iron oxides specifically ferrites stand out due to their distinct advantages, including ease of magnetic separation from reaction mixtures, high surface-to-volume ratio, and the ability to functionalize their surfaces through available hydroxyl groups13. Further exploration into this area has brought spinel ferrites (Fe3O4) into prominence as versatile materials14,15. These ferrites exhibit advanced magnetic and electronic properties alongside unique catalytic potential, attributed to their specialized structural designs16,17. Nanomaterials offer promising potential as effective supports for immobilizing homogeneous catalysts, allowing the benefits of both homogeneous and heterogeneous catalysis to be combined18,19,20. One key advantage in catalytic reactions is the ease and speed with which catalysts can be separated from the reaction mixture21,22,23. Currently, the recycling of heterogeneous catalysts often involves labor-intensive filtration processes and unavoidable material loss during recovery. Magnetic nanoparticles have emerged as highly suitable alternatives in catalytic applications24,25. Over the past few years, these nanoparticles have been studied extensively, as their ability to be separated using an external magnet greatly simplifies catalyst recovery compared to conventional separation methods5,26,27.
Sulfides and sulfones serve as crucial intermediates in the production of high-value fine chemicals and biologically active molecules, including pharmaceuticals, agrochemicals, and chiral auxiliaries28,29,30,31. As a result, the selective oxidation of sulfides to sulfoxides or sulfones has gained considerable interest, being one of the cornerstone transformations in organic synthesis32. Additionally, diaryl sulfides represent a significant class of therapeutic agents in medicinal chemistry33,34. Consequently, there is a growing emphasis on developing efficient and mild approaches for synthesizing these compounds, marking an expansive field in organic chemistry21,35,36. In recent years, C–S cross-coupling reactions have garnered substantial attention for their versatility in synthesizing a wide range of bioactive natural products and pharmaceutical agents37,38. However, C–S bond formation reactions remain relatively underexplored compared to C–N and C–O coupling processes. This gap is partly attributable to the proclivity of organic sulfur compounds to bind with metals, thereby functioning as metal deactivators and posing challenges in reaction optimization38,39.
This report details the synthesis of a highly effective heterogeneous catalyst, Pd-BPA coated on Fe3O4 magnetic nanoparticles (MNPs), and its successful application in facilitating high-yield C-S coupling reactions under mild conditions. The findings demonstrate that the catalytic activity of this heterogeneous system closely matches that of a homogeneous catalyst. Furthermore, the catalyst proved to be easily separable from the reaction mixture with minimal loss of activity.
Experimental
Materials
The key materials needed for synthesizing the nanocatalyst, as well as the coupling derivatives, reagents, and solvents, were obtained from Merck, Fluka, and various other chemical suppliers.
Preparation of Pd-BPA/Fe3 O 4
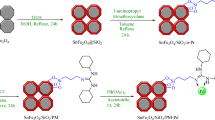
Fe₃O₄ was initially synthesized using the co-precipitation method27,40. The preparation began with the modification of Fe₃O₄: 1 g of Fe₃O₄ was refluxed with 2.5 mL of 3-chloropropyltrimethoxysilane (CPTMS) in 30 mL of anhydrous toluene for 24 h, resulting in Cl/Fe₃O₄. In the next step, 1 g of Cl/Fe₃O₄ was combined with 2.5 mmol of (2,2′-Bipyridin-5-yl)alanine dihydrochloride (BPA) in 25 mL of ethanol. This mixture was also subjected to reflux conditions for 24 h, producing a BPA-modified Schiff base/Fe₃O₄ material. Lastly, palladium nanoparticles were deposited onto the BPA/Fe₃O₄ substrate. For this, 1 g of BPA/Fe₃O₄ was mixed with 3 mmol of PdCl₂ in 25 mL of ethanol and stirred under reflux conditions for another 24 h. After completion, the palladium nanocatalyst (Pd-BPA/Fe₃O₄) was filtered, washed with ethanol, and dried for further use (Fig. 1).
Synthesis of Pd-BPA/Fe3O4.
General procedure for aromatic sulfide
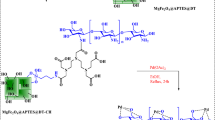
A reaction mixture composed of aryl halide (2 mmol), sulfur (S8, 1 mmol), potassium hydroxide (KOH, 0.7 mmol), and Pd-BPA/Fe3O4 nanocatalyst (0.02 g) was stirred under solvent-free conditions, with progress tracked using thin-layer chromatography (TLC). Upon completing the reaction, the mixture was cooled to room temperature before being diluted by adding water. The magnetic nanocatalyst was removed via magnetic separation and thoroughly washed with ethyl acetate. The remaining solution underwent extraction using a combination of ethyl acetate and water, followed by drying over sodium sulfate (Na2SO4, 0.9 g). The solvent was then evaporated, yielding pure ether derivatives as illustrated in Fig. 2.
Synthesis of diaryl sulfide derivatives using Pd-BPA/Fe3O4.
Selected NMR data
See supplementary figures S1 to S7.
Catalyst characterizations
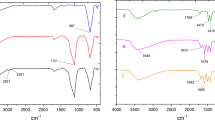
FT-IR analysis (Fig. 3) illustrates the spectra of Fe3O4, Cl/Fe3O4, BPA/Fe3O4, and Pd-BPA/Fe3O4 magnetic nanoparticles (MNPs). The absorption bands observed for Fe3O4 (Fig. 3a) align with previously reported studies. In Fig. 3b, the Cl/Fe3O4 spectrum displays bands at 2905 and 2837 cm−1, corresponding to the bending vibrations of CH2, thereby confirming the attachment of BPA chain molecules. New bands at 1458 and 1561 cm−1 in the BPA/Fe3O4 spectrum (Fig. 3c) further confirm the presence of BPA within the sample, providing evidence that BPA is successfully stabilized. The presence of BPA groups is validated by distinct bands at 3010 cm−1 (O–H stretching) and 1643 cm−1 (C-N stretching), indicating the successful functionalization of BPA/Fe3O4 with melamine-based BPA groups (Fig. 3c). Additionally, the peak at 1595 cm−1 in Fig. 1d signifies metal–ligand coordination, as evidenced by the shift of this band from 1643 cm−1 to a lower frequency range (1595 cm−1). This shift becomes evident when comparing Fig. 3c and Fig. 3d, confirming the effective attachment of BPA organic ligands and the subsequent coordination of Pd ions within the hybrid material. These interactions are clearly reflected in the spectra of both curves.
Comparative study of FTIR spectra of (a) Fe3O4, (b) Cl/Fe3O4, (c) BPA/Fe3O4 (d) Pd-BPA/Fe3O4.
Figure 4 presents the X-ray diffraction (XRD) patterns of Pd-BPA/Fe3O4. The synthesis of Fe3O4 magnetic nanoparticles (MNPs) is confirmed through distinct XRD peak positions observed at 2θ = 30°, 37°, 43°, 54°, 57°, and 64°, which correspond to the (220), (311), (400), (422), (511), and (440) crystal planes, respectively. Furthermore, the XRD pattern of the catalyst reveals that the Fe3O4 phase remains intact following modification with a different organic functional group, as highlighted in Fig. 4.
XRD spectrum of Pd-BPA/Fe3O4.
Thermogravimetric analysis (TGA) was performed to determine the percentage of chemisorbed Pd-BPA on the surface of Fe3O4 magnetic nanoparticles (MNPs). The TGA curves for Pd-BPA/Fe3O4 are illustrated in Fig. 3. The data show a minor weight loss of 7% below 200 °C, attributed to the removal of surface hydroxyl groups and adsorbed water. A more significant weight loss of approximately 24% occurs between 200 and 600 °C, corresponding to the decomposition of the Pd-BPA organometallic complex attached to the Fe3O4 MNP surface. These results confirm the successful chemisorption of the Pd-BPA organometallic complex onto the Fe3O4 MNPs (Fig. 5).
TGA curve of Pd-BPA/Fe3O4.
The elemental composition of Pd-BPA/Fe3O4 was analyzed using the EDX spectrum (Fig. 6). The results confirmed the presence of Nitrogen, Silicon, Iron, Oxygen, Carbon, and Palladium within the catalyst, validating the successful synthesis of nanoparticles. Additionally, the analysis demonstrated the effective immobilization of Pd-BPA onto the surface of Fe3O4 magnetic nanoparticles (MNPs). To further quantify the palladium content in Pd-BPA/Fe3O4, an ICP-OES analysis was performed. The results indicated that the Pd concentration in the catalyst was 1.1 × 10⁻3 mol g⁻1. Furthermore, ICP analysis was utilized to assess the extent of Pd leaching after catalyst recycling. It was observed that the reused catalysts retained a Pd concentration of 1.0 × 10⁻3 mol g⁻1, suggesting minimal leaching of Pd from the Pd-BPA/Fe3O4 framework.
EDS analysis of Pd-BPA/Fe3O4.
To assess the morphology and particle size of the Pd-BPA/Fe3O4 hybrid material, scanning electron microscopy (SEM) was employed, as illustrated in Fig. 7. Based on the SEM images, the catalytic particles within the Pd-BPA/Fe3O4 sample exhibit a spherical shape. Notably, this particular morphology contributes to optimizing the catalyst’s performance by simultaneously enhancing its efficiency, increasing the frequency of molecular interactions, or reducing the activation energy of the rate-determining step (Fig. 7).
SEM images of Pd-BPA/Fe3O4.
The surface area of the Pd-BPA/Fe3O4 nanomaterial was examined using a fully automated BET surface area analyzer, which measures nitrogen multilayer adsorption as a function of relative pressure (Ps/P0). As illustrated in Fig. 8, the prepared nanostructure exhibited a BET surface area of 6.7 m2/g. Additionally, the Barrett-Joyner-Halenda (BJH) method offered insights into the pore size distribution and volume, revealing a pore size of 8.3 nm and a pore volume of 0.05 cm3/g.
N2-adsorption isotherms of Pd-BPA/Fe3O4.
The magnetic characteristics of uncoated magnetic spinel ferrite (Fe3O4) and Pd-BPA/Fe3O4 magnetic nanoparticles (MNPs) were analyzed using VSM within an applied magnetic field range of − 10,000 to + 10,000 Oe at room temperature, as illustrated in Fig. 9. The results indicate a reduction in saturation magnetization from approximately 47 emu/g to around 23 emu/g, which can be attributed to the addition of the newly coated layer, confirming the successful synthesis of the catalyst. Although the Pd-BPA/Fe3O4 demonstrates a lower saturation magnetization compared to Fe3O4 nanoparticles, its magnetic responsiveness remains adequate for efficient separation from various reaction environments.
VSM curves of (a) Fe3O4 (b) Pd-BPA/Fe3O4.
Catalytic studies
As part of this research, the catalytic effect of Pd-BPA/Fe3O4 was investigated in organic reactions, including the oxidation of sulfides and C–S coupling reactions. The study then shifted its focus to evaluating the catalyst’s utility in S-arylation reactions. To determine the optimal reaction conditions for C–S arylation using the Pd-BPA/Fe3O4 catalyst, the coupling of thiourea with iodobenzene was selected as the model reaction. The collected data is summarized in Table 1. To optimize the reaction parameters, several factors were examined, such as solvent type, catalyst quantity, and the impact of base and temperature on reaction efficiency. Initially, the catalyst amount was optimized, with 0.02 g of Pd-BPA/Fe3O4 found to yield the best results. Notably, no reaction was observed in the absence of the catalyst, even after 12 h. Next, various solvents were tested, with ethanol (EtOH) emerging as the most effective solvent, resulting in the highest product yield. The effect of different bases was also assessed, including Et3N, Na2CO3, K2CO3, KOH, and NaOH. Among these, KOH delivered the highest yield of the desired product. Finally, temperature effects were explored, revealing that reflux conditions offered the optimal outcome for the reaction.
Once the optimized reaction conditions were established, a selection of commercially available thiourea and phenol was tested with various aryl halides, as illustrated in Scheme 2. The corresponding outcomes are presented in Table 2. As indicated in the table, the desired products were obtained in yields ranging from moderate to excellent. The findings clearly revealed that the S-arylation reactions involving thiourea and para-substituted aryl halides proceeded more efficiently compared to those with ortho- or meta-substituted aryl halides.
Figure 10 illustrates the catalytic cycle for synthesizing sulfides using Pd-BPA/Fe3O4. The cycle starts with the reaction between S8 and KOH, which produces potassium disulfide as the initial intermediate. This intermediate subsequently interacts with Pd-BPA/Fe3O4 to form palladium disulfide. The next step involves an oxidative addition of an aryl halide with palladium disulfide, leading to intermediate 1. Reductive elimination follows, generating intermediate 2. The process continues with intermediate 2 undergoing another oxidative addition with a second aryl halide, resulting in compound 3. In the final stage, reductive elimination occurs, yielding diphenyl sulfide and simultaneously regenerating the palladium catalyst, as depicted in Fig. 10.
Possible mechanism for the synthesis of sulfides.
Hot filtration
The heterogeneity of Pd-BPA/Fe3O4 MNPs in the reaction mixture was evaluated using a hot filtration test conducted during the synthesis of diphenyl sulfide under optimal reaction conditions. At half the reaction time, the process was stopped, and the product was obtained with a 51% yield. Subsequently, the reaction was conducted again, and at the midpoint, the catalyst was removed from the mixture using magnetic separation, allowing the reaction to proceed further without it. The results revealed that in the absence of the catalyst, only minimal conversion (< 3%) of the coupling reaction occurred during the remaining reaction period, even under heating. This confirms that the nanocatalyst operates entirely as a heterogeneous catalyst within the reaction medium.
Catalyst recyclability
The recyclability of Pd-BPA/Fe3O4 in biphenyl production was assessed under optimal conditions. Remarkably, the catalyst retained consistent activity over five successive cycles, achieving a reaction efficiency of 96% in the fifth cycle (Fig. 11).
Recyclability of Pd-BPA/Fe3O4 in the synthesis of C-S coupling reactions.
To highlight the advantages of Pd-BPA/Fe3O4, we compared its performance in sulfide synthesis with previously reported catalysts in the literature. The superior catalytic activity of Pd-BPA/Fe3O4 resulted in higher product yields and shorter reaction times compared to other documented catalysts. Additionally, this novel catalyst demonstrates comparable performance in terms of stability, ease of separation, and cost-effectiveness. Furthermore, its recyclability process is notably faster than that of other catalysts, as shown in Table 3.
Conclusion
In summary, we have successfully developed an effective magnetic nanocatalyst for the selective synthesis of sulfides under eco-friendly conditions. This catalyst was engineered by anchoring a Pd-BPA complex onto Fe3O4 magnetic nanoparticles (MNPs) through covalent cross-linking. Its structure was extensively characterized with techniques such as ICP, TGA, XRD, EDS, VSM, SEM, FT-IR, and BET. This method offers several advantages: straightforward operation, high efficiency, cost-effectiveness, clean reactions with excellent yields, and relatively short reaction times. The catalyst boasts a high surface area, easy recovery and reusability for multiple cycles without notable activity loss, eco-friendly production using readily available materials, straightforward filtration separation, affordability, chemical stability, practical reaction times, and a user-friendly methodology. These attributes underscore its potential for application in various organic reactions. Moreover, recovery tests demonstrate its reusability with consistent performance over multiple uses.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Veisi, H. et al. Biosynthesis of CuO nanoparticles using aqueous extract of herbal tea (Stachys lavandulifolia) flowers and evaluation of its catalytic activity. Sci. Rep. 11, 1983 (2021).
Liu, M., Liu, Z. & Qiao, L. Preparation of a magnetic solid acid FeO@PS-SOH and its use in hydrolysis of cellulose. J. Chem. Technol. Biotechnol. 99, 1914–1920 (2024).
Veisi, H. et al. Pd immobilization biguanidine modified Zr-UiO-66 MOF as a reusable heterogeneous catalyst in Suzuki-Miyaura coupling. Sci. Rep. 11, 21883 (2021).
Hu, C., Yang, Z., Yan, F. & Sun, B. Extraction of the toluene exposure biomarkers hippuric acid and methylhippuric acid using a magnetic molecularly imprinted polymer, and their quantitation by LC-MS/MS. Microchim. Acta 186, 135 (2019).
Zhang, H., Zhang, L.-L., Tan, X., Li, H. & Yang, S. Catalytic high-yield biodiesel production from fatty acids and non-food oils over a magnetically separable acid nanosphere. Ind. Crops Prod. 173, 114126 (2021).
Sahoo, J. K., Rath, J., Dash, P. & Sahoo, H. EDTA functionalized magnetic nanoparticle as a multifunctional adsorbent for Congo red dye from contaminated water. AIP Conf. Proc. 1832, 1–4 (2017).
Dehghani, F., Hashemian, S. & Shibani, A. Effect of calcination temperature for capability of MFe2O4 (M=Co, Ni and Zn) ferrite spinel for adsorption of bromophenol red. J. Ind. Eng. Chem. 48, 36–42 (2017).
Rafiq, M., Javed, A., Nasir, M., Khan, M. & Hussain, D. A. Understanding the structural, electronic, magnetic and optical properties of spinel MFe2O4 (M=Mn, Co, Ni) ferrites. Ceram. Int. 46, (2019).
Veisi, H. et al. Bio-inspired synthesis of palladium nanoparticles fabricated magnetic Fe3O4 nanocomposite over Fritillaria imperialis flower extract as an efficient recyclable catalyst for the reduction of nitroarenes. Sci. Rep. 11, 4515 (2021).
Tamoradi, T., Zarei-Ahmady, A., Karmakar, B. & Nabizade, N. Response surface method (RSM) process optimization of sulfides oxidation in the presence of orange peel extract immobilized magnetic hybrid nanocomposite catalyst. Appl. Organomet. Chem. 39, e7958 (2025).
Vallet-Regí, M., Balas, F. & Arcos, D. Mesoporous materials for drug delivery. Angew. Chem. Int. Ed. 46, 7548–7558 (2007).
Morales, G. et al. Aldol condensation of furfural and methyl isobutyl ketone over Zr-MOF-808/silica hybrid catalysts. Fuel 339, 127465 (2023).
Veisi, H., Mohammadi, L., Hemmati, S., Tamoradi, T. & Mohammadi, P. In situ immobilized silver nanoparticles on rubia tinctorum extract-coated ultrasmall iron oxide nanoparticles: An efficient nanocatalyst with magnetic recyclability for synthesis of propargylamines by A3 coupling reaction. ACS Omega 4, 13991–14003 (2019).
Gallo-Cordova, Á. et al. New insights into the structural analysis of maghemite and (MFe2O4, M=Co, Zn) ferrite nanoparticles synthesized by a microwave-assisted polyol process. Mater. Chem. Front. 4, 3063–3073 (2020).
Suresh Kumar, G. et al. Microwave-assisted combustion synthesis of soft ferromagnetic spinel MFe2O4 (M=Ni, Mg, Zn) nanoparticles using citrus limon fruit extract as a fuel. Appl. Phys. A Mater. Sci. Process. 127, (2021).
Zhao, H. et al. In vitro toxicity evaluation of ultra-small MFe2O4 (M=Fe, Mn, Co) nanoparticles using A549 cells. RSC Adv. 5, 68454–68460 (2015).
Sha, O. et al. Solvent-free mechanochemical preparation of graphene oxide–Fe3O4 and its application in magnetic dispersive solid-phase extraction of illegal dyes in food samples. J. Sep. Sci. 44, 2290–2300 (2021).
Nciri, R., Ali Rothan, Y., Nasri, F. & Ali, C. Fe3o4-water nanofluid free convection within an inclined 2d rectangular enclosure heated by solar energy using finned absorber plate. Appl. Sci. 11, 1–16 (2021).
Wang, Q., Chen, T., Bai, P., Lyu, J. & Guo, X. Fe3O4-loaded ion exchange resin for chromatographic separation of boron isotopes: Experiment and numerical simulation. Chem. Eng. Res. Des. 171, 358–366 (2021).
Liu, X. et al. Enhanced electromagnetic wave absorption performance of core-shell Fe3O4@poly(3,4-ethylenedioxythiophene) microspheres/reduced graphene oxide composite. Carbon 178, 273–284 (2021).
Hadrup, N. et al. Pulmonary toxicity of Fe2O3, ZnFe2O4, NiFe2O4 and NiZnFe4O8 nanomaterials: Inflammation and DNA strand breaks. Environ. Toxicol. Pharmacol. 74, 103303 (2020).
Dippong, T. & Levei, E. A. Chapter III Synthesis and characterization of MFe2O4(M : Zn , Ca , Mg) photocatalysts. 4, 1–12 (2021).
Sun, H. & Li, D. Recyclable MFe2O4(M=Mn, Zn, Cu, Ni, Co) coupled micro-nano bubbles for simultaneous catalytic oxidation to remove NO: X and SO2 in flue gas. RSC Adv. 10, 25155–25164 (2020).
Zhang, M. et al. Synthesis, characterization and catalytic behavior of MFe2O4 (M=Ni, Zn and Co) nanoparticles on the thermal decomposition of TKX-50. J. Therm. Anal. Calorim. 141, 1413–1423 (2020).
Abbas, M., Zhang, J., Mansour, T. S. & Chen, J. Hierarchical porous spinel MFe2O4 (M=Fe, Zn, Ni and Co) nanoparticles: Facile synthesis approach and their superb stability and catalytic performance in Fischer-Tropsch synthesis. Int. J. Hydrog. Energy 45, 10754–10763 (2020).
Cui, X. et al. Synergistic catalysis by Fe3O4-biochar/peroxymonosulfate system for the removal of bisphenol a. Sep. Purif. Technol. 276, 119351 (2021).
Ahmadi, S., Habibi, D., Heydari, S. & Roshani Asl, E. The capable xanthine-based adsorbent for removal of the Cd(II), Ni(II) and Pb(II) ions from aqueous solution via their complexation and the use of its corresponding Cd complex for the green synthesis of indazolophthalazinetriones. Res. Chem. Intermed. 49, 763–789 (2023).
Wang, L., Zhang, Y., Yao, J. & Li, H. Metal-free synthesis of sulfones and sulfoxides through aldehyde-promoted aerobic oxidation of sulfides. Catal. Letters https://doi.org/10.1007/s10562-021-03706-5 (2021).
Mirfakhraei, S., Hekmati, M., Eshbala, F. H. & Veisi, H. Fe3O4/PEG-SO3H as a heterogeneous and magnetically-recyclable nanocatalyst for the oxidation of sulfides to sulfones or sulfoxides. New J. Chem. 42, 1757–1761 (2018).
Anumol, C. N., Chithra, M., Govindaraj Shalini, M. & Sahoo, S. C. Effect of annealing on structural and magnetic properties of NiFe2O4/ZnFe2O4 nanocomposites. J. Magn. Magn. Mater. 469, 81–88 (2019).
Zhang, W., Zhou, P., Liu, W., Wang, H. & Wang, X. Enhanced adsorption/extraction of five typical polycyclic aromatic hydrocarbons from meat samples using magnetic effervescent tablets composed of dicationic ionic liquids and NiFe2O4 nanoparticles. J. Mol. Liq. 315, 113682 (2020).
Zhang, Q., Wang, J.-X., Yu, G.-F., Xue, J.-L. & Xiao, X.-F. Treatment of oily sludge by persulfate activated in different ways and mechanism research. Xiandai Huagong/Modern Chem. Ind. 40, 120–123 (2020).
Sun, N. et al. Cu-catalysed Chan-Lam synthesis of unsymmetrical aryl chalcogenides under aqueous micellar conditions. Green Chem. 25, 2782–2789 (2023).
Adam, M. S. S., Makhlouf, M. M., Alharbi, A. & El-Metwaly, N. M. Novel isatin-based complexes of Mn(II) and Cu(II) ions: Characterization, homogeneous catalysts for sulfides oxidation, bioactivity screening and theoretical implementations via DFT and pharmacokinetic studies. J. Mol. Liq. 351, 118620 (2022).
Zhang, H.-Y. et al. A magnetic metal–organic framework as a highly active heterogeneous catalyst for one-pot synthesis of 2-substituted alkyl and aryl(indolyl)kojic acid derivatives. New J. Chem. 41, 7108–7115 (2017).
Virumbrales, M., Saez-Puche, R., Torralvo, M. J. & Blanco-Gutierrez, V. Mesoporous silica matrix as a tool for minimizing dipolar interactions in NiFe2O4 and ZnFe2O4 nanoparticles. Nanomaterials 7, 151 (2017).
Smith, A. L. et al. Thermodynamic modelling assessment of the ternary system Cs-Mo-O. Calphad Comput. Coupling Phase Diagrams Thermochem. 75, 102350 (2021).
Batmani, H., Noroozi Pesyan, N. & Havasi, F. Synthesis and characterization of MCM-41-Biurea-Pd as a heterogeneous nanocatalyst and using its catalytic efficacy in C–C, C–N and C–O coupling reactions. Appl. Organomet. Chem. 32, e4419 (2018).
Sedighipoor, M., Kianfar, A. H., Mohammadnezhad, G., Görls, H. & Plass, W. Unsymmetrical palladium(II) N, N, O, O-Schiff base complexes: Efficient catalysts for suzuki coupling reactions. Inorganica Chim. Acta 476, 20–26 (2018).
Zhang, F., Shen, B., Jiang, W., Yuan, H. & Zhou, H. Hydrolysis extraction of diosgenin from Dioscorea nipponica Makino by sulfonated magnetic solid composites. J. Nanoparticle Res. 21, 269 (2019).
Wang, L. et al. Catalytic aza-Wittig reaction of acid anhydride for the synthesis of 4H-benzo[d][1,3]oxazin-4-ones and 4-benzylidene-2-aryloxazol-5(4H)-ones. ACS Catal. 6, 4010–4016 (2016).
Xu, H. J., Zhao, X. Y., Deng, J., Fu, Y. & Feng, Y. S. Efficient C-S cross coupling catalyzed by Cu2O. Tetrahedron Lett. 50, 434–437 (2009).
Wang, L. et al. Ir-catalyzed C-S coupling of quinones with sulfonyl chloride. J. Iran. Chem. Soc. 13, 1797–1803 (2016).
Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Group Project under grant number (RGP2/179/46).
Author information
Authors and Affiliations
Contributions
Mohammed Asiri. Shelesh krishna saraswat. Vicky Jain. Suhas Ballal. Amit Sharma. Munthar Kadhim Abosaoda. Abhayveer Singh. Parminder Singh. Subhashree Ray. and Aashna Sinha. Funding acquisition, Supervision, Conceptualization, Resources, Writing-review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Asiri, M., Saraswat, S.k., Jain, V. et al. Palladium nanoparticles stabilized on the BPA-functionalized Fe3O4 as the recoverable catalysts for synthesis of aromatic sulfide by C–S coupling reactions. Sci Rep 15, 34019 (2025). https://doi.org/10.1038/s41598-025-13144-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13144-7