Abstract
This cross-sectional study was to investigate the associations of self-reported oral health, tooth loss, interdental cleaning and dental visits with serum C-reactive protein (CRP). Data regarding oral health questionnaires, dentition examination and serum high-sensitivity CRP from 6358 participants aged 30 − 80 years were analyzed in US National Health and Nutrition Examination Survey 2015 − 2020. Weighted linear regression analyses were performed. In the fully adjusted model, very good or excellent self-reported oral health was associated with lower ln-transformed serum CRP compared to poor or fair self-reported oral health (regression estimates (β) and 95% Confidence interval (CI): -0.106(-0.196, -0.016), P = 0.037). Further multivariate analyses demonstrated a positive association between the number of missing teeth and ln-transformed serum CRP (β and 95% CI: 0.010(0.001, 0.019), P = 0.047), while interdental cleaning frequency was inversely associated with ln-transformed serum CRP (day/week of performing interdental cleaning β and 95% CI: -0.024(-0.041, -0.008), P = 0.010). No significant association was observed between time since last dental visit and ln-transformed serum CRP in the multivariate linear regression model. In conclusion, better self-reported oral health, fewer missing teeth and higher interdental cleaning frequency were associated with lower serum CRP in US population.
Similar content being viewed by others
Introduction
Dental caries and periodontitis are the most prevalent oral diseases and primary causes of tooth loss1,2,3,4. Chronic periodontitis and apical periodontitis may compromise the epithelial barrier, facilitating systemic dissemination of oral microbiota5,6,7. Porphyromonas gingivalis, one of the important pathogens in these conditions, has been detected in the brains of Alzheimer’s disease patients6 and carotid plaques of periodontitis patients8. An animal study further confirmed the translocation of Porphyromonas gingivalis from infected pulp to the liver, contributing to steatohepatitis7. Consequently, recent investigations revealed that periodontitis and apical periodontitis were associated with elevated systemic inflammation and cardiovascular diseases9,10,11,12,13,14,15,16,17,18,19,20. Zhang et al. found that periodontal disease was related with elevated serum C-reactive protein (CRP)21, an important predictor of future cardiovascular diseases22. Conversely, successful periodontal and endodontic therapies reduce systemic CRP23,24,25,26,27. Recently, Haworth et al. reported that tooth loss could reflect the cumulative oral disease burden and socioeconomic status28. Tooth loss was therefore proposed as a reliable measure of dental disease and oral health. Additionally, world-wide studies showed that tooth loss was also a possible risk factor for systemic inflammation, cardiovascular and cerebrovascular diseases and all-cause mortality29,30,31,32,33,34,35,36,37,38,39,40. A recent study reported that significant tooth loss was associated with serum CRP > 1 mg/dL41. Interestingly, Yu et al. further reported that self-rated oral health status predicted systemic comorbidities as effectively as periodontitis42. Counting missing teeth and self-reported oral health questionnaire are straightforward means to measure dental status for large population studies. However, the dose-dependent response of tooth loss and self-rated oral health status on serum CRP has not been sufficiently reported.
Moreover, recent investigations found that dental flossing and frequent dental visits reduced oral diseases and may be associated with better systemic health. Population-based studies reported that inter-dental cleaning was a possible protective factor for caries and periodontitis43,44,45,46,47. Some recent studies further found that dental flossing was associated with lower systemic inflammation, lower prevalence of metabolic syndrome and decreased mortality41,42,48,49. Additionally, several reports demonstrated that infrequent dental visit was related with all-cause mortality42,50,51. Despite the positive effects of interdental cleaning on oral health, prevalence of daily interdental cleaning remains low (about 30%) in developed countries, and this number did not increase in the past 30 years52,53,54,55. However, the dose-dependent response of interdental cleaning and dental visits on serum CRP has not been deeply studied.
The present study therefore aimed to investigate the associations of self-rated oral health status, tooth loss, inter-dental cleaning and dental visits with serum high-sensitivity CRP in a nationally representative sample by analyzing data from the National Health and Nutrition Examination Survey (NHANES) 2015 − 2020 cycles. The hypotheses were that better self-rated oral health status, fewer teeth missing, more frequent inter-dental cleaning and shorter time to visit dentists would be associated with lower serum CRP.
Material and methods
Study design and population
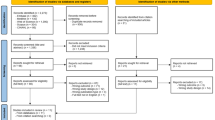
The Centers for Disease Control and Prevention examined the health and nutrition condition of a representative United States population in 2-year cycles using a complex multistage sampling design. As the NHANES 2015 − 2020 cycles included oral health questionnaire, dentition examination and serum high-sensitivity CRP test, the data of NHANES 2015 − 2020 cycles were extracted for the current cross-sectional study. The study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines of the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) network. Written informed consent was obtained from all NHANES participants, and all the data collection and procedures were approved by the National Center for Health Statistics (NCHS) Research Ethics Review Board. In the present study, data collection included questionnaires, interviews, physical examinations, and laboratory tests. In general, questionnaire and interview were carried out in participant’ home, while physical examination and blood specimen collection were performed at mobile examination center (MEC). The original data and more details could be accessed on the NHANES website (https://wwwn.cdc.gov/nchs/nhanes/Default.aspx). Figure 1 shows the screening process of this study population. The inclusion criteria were as follows: (1) participants aged ≥ 30 years with teeth requiring interdental cleaning and (2) participants with complete data regarding serum high-sensitivity CRP, the frequency of performing interdental cleaning, dentition status, and self-rated oral health status. The exclusion criteria were as follows: (1) participants aged < 30 years; (2) participants with missing/incomplete data regarding the frequency of performing interdental cleaning, dentition status, self-rated oral health status, and serum high-sensitivity CRP; (3) participants with serum high-sensitivity CRP > 100 mg/L; (4) participants with current antibiotic use, history of cancer or malignancy, positive hepatitis B surface antigen, chronic kidney disease (estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73 m2 or the urinary albumin-to-creatinine ratio ≥ 30 mg/g56) or history of rheumatoid arthritis; and (5) pregnant females (Fig. 1).
Flow diagram of the screening of study participants.
Serum high-sensitivity CRP measurement
Serum specimens were stored and tested in Advanced Research Diagnostics Laboratory (ARDL), University of Minnesota, Minneapolis. The measurement used the two-reagent, immunoturbidimetric system. The detailed laboratory method could refer to https://wwwn.cdc.gov/Nchs/Nhanes/2017-2018/P_HSCRP.htm. In the present study, serum CRP > 3.0 mg/L was defined as elevated serum CRP15,22. Serum high-sensitivity CRP was further ln-transformed to improve the adherence to normal distribution in the regression analyses57.
Dentition examination
All the oral examinations were conducted by trained dentists in the MEC. The dentition data excluded third molars in the present study. The total number of missing teeth for a participant was defined as the sum of unreplaced teeth lost due to dental diseases, replaced teeth lost due to dental diseases and permanent root tips58.
Oral health questionnaire
As participants with ≥ 30 years of age were eligible for the oral health questionnaire including self-reported oral health status, interdental cleaning frequency, and time since the last dental visit in the NHANES 2015–2020 cycles42,50,54, so participants with < 30 years of age were excluded in the current study. Participants who declined or missed the three questions or did not know the answer to the three questions were further excluded in the present study (Fig. 1).The questionnaire was performed by trained interviewers using Computer-Assisted Personal Interview (CAPI) system. Self-reported oral health status was extracted from the answer to the question: “Overall, how would you rate the health of your teeth and gums?” Interdental cleaning data were extracted from the answer to the question: “Aside from brushing our teeth with a toothbrush in the last seven days, how many days did you use dental floss or any other device to clean between your teeth?” Time since the last dental visit was obtained from the response to the question “About how long has it been since you last visited a dentist? Include all types of dentists, such as, orthodontists, oral surgeons, and all other dental specialists, as well as dental hygienists”. In addition, history of scaling and root planing treatment was extracted from the response to the question “Have you ever had treatment for gum disease such as scaling and root planing, sometimes called deep cleaning?”.
Covariate assessments
Participants’ demographic data including age, gender, race, family Poverty income ratio (PIR), marital status and education levels were obtained by standardized questionnaires. Current smokers were defined as smoking cigarettes now. Former smokers were defined as having smoked at least 100 cigarettes in life but not smoking cigarettes now. Never smokers were defined as having not smoked at least 100 cigarettes in life. Drinking status was extracted from the question: “During the past 12 months, about how often did you drink any type of alcoholic beverage?” Weight statuses were divided into three categories: BMI < 25.0 kg/m2, BMI 25.0–30.0 kg/m2, and BMI > 30.0 kg/m2. Decreased high-density lipoprotein cholesterol (HDL) was defined as HDL < 50 mg/dL for female participants and HDL < 40 mg/dL for male participants or the history of taking lipid-lowering medication. Elevated fasting blood glucose (FPG) was defined as FPG ≥ 100 mg/dL or the history of taking antidiabetic medication. High blood pressure was defined as systolic diastolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 85 mmHg or the history of taking antihypertensive medication. Short sleep duration was defined as self-reported sleep duration < 6 h/day. Depression status was defined as the Patient Health Questionnaire (PHQ-9) score ≥ 5.
The assessment of dietary quality adopted Healthy Eating Index-2020 (HEI-2020) and the two sets of 24-h dietary questionnaire data in NHANES. In the present study, high dietary quality was defined as respondent’s Healthy Eating Index-2020 score greater than the 60th percentile59. Metabolic equivalent minutes of moderate to vigorous physical activity per week (MET) was calculated from the Global Physical Activity Questionnaire. Participants were then classified into ≥ 600 MET-minutes/week group and < 600 MET-minutes/week group60.
Statistics
MEC exam weights were used to consider the planned oversampling of specific groups according to NHANES. The descriptive data were presented as mean ± standard deviations for continuous variables and weighted percentages for categorical variables. Weighted chi-square tests and weighted linear regression models were used to compare the categorical and continuous variables of the baseline clinical data respectively. The missForest package was used to perform multiple imputation for missing data61. Weighted multivariable linear regression analyses were then used to analyze the associations of self-reported oral health status, the number of missing teeth, interdental cleaning frequency, and the time since the last dental visit with ln-transformed serum high-sensitivity CRP. The associations were presented as regression estimates β and 95% Confidence interval (CI). Confounding variables were considered from three aspects: clinical relevance, P < 0.05 in the univariate analysis, and the sufficient event data to perform a regression model. Statistical software programs (RStudio 2023.12.1+402; Posit Software and IBM SPSS Statistics for Windows, v21.0; IBM Corp) were used for statistical analyses (α = 0.05).
Results
Baseline data
A total of 6358 participants aged 30 − 80 years were included in the present study (Fig. 1). Table1 demonstrated the comparison of demographic, clinical and dental characteristics of the control participants and the participants with elevated serum high-sensitivity CRP (> 3.0 mg/L). 32.61% of the participants had elevated levels of serum CRP (Table1). The average age of the participants was 49.3 ± 12.8 years. When compared to the control participants with serum CRP ≤ 3.0 mg/L, the elevated serum CRP group had significantly higher percentages of females, Black participants, participants with a less than > high school education, participants with family PIR ≤ 3.5, participants with low physical activity and low dietary quality, BMI ≥ 30 kg/m2, elevated FPG, low HDL, high blood pressure, short sleep, and depression (Table1). Moreover, higher prevalence of very poor or fair self-reported oral health status, more than 6 months since the last dental visit, more missing teeth and less frequent interdental cleaning were found in the elevated serum CRP participants (Table1).
Association of self-reported oral health status with serum high-sensitivity CRP
Figure 2 presented the weighted multivariate linear regression analyses for the association between self-reported oral health status and ln-transformed serum high-sensitivity CRP. In the fully adjusted model, very good or excellent self-reported oral health was associated with lower ln-transformed serum high-sensitivity CRP when the participants with poor or fair self-reported oral health were regarded as the reference group (regression estimates (β) and 95% CI: −0.106(−0.196, −0.016), P = 0.037, Fig. 2).
Weighted multivariate linear regression analyses for the association between self-reported oral health status and ln-transformed serum high-sensitivity CRP. Model 1: adjusting self-reported oral health status, age, sex and race. Model 2: adjusting self-reported oral health status, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, and dietary quality. Model 3: adjusting self-reported oral health status, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Data are shown as regression estimates (β) and 95% CI. Abbreviations: CI, Confidence interval; HDL, High-density lipoprotein cholesterol; FPG, Fasting Plasma Glucose; CRP, C-reactive protein.
Association of the number of missing teeth with serum high-sensitivity CRP
Figure 3 illustrated the weighted linear regression analyses for the association between the number of missing teeth and ln-transformed serum high-sensitivity CRP. The multivariate linear analyses found that the number of missing teeth was positively associated with ln-transformed serum high-sensitivity CRP after adjustment (regression estimates (β) and 95% CI: 0.010(0.001, 0.019), P = 0.047, Fig. 3).
Weighted multivariate linear regression analyses for the association between the number of missing teeth and ln-transformed serum high-sensitivity CRP. Model 1: adjusting the number of missing teeth, age, sex and race. Model 2: adjusting the number of missing teeth, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, and dietary quality. Model 3: adjusting the number of missing teeth, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Data are shown as regression estimates (β) and 95% CI. Abbreviations: CI, Confidence interval; HDL, High-density lipoprotein cholesterol; FPG, Fasting Plasma Glucose; CRP, C-reactive protein.
Associations of interdental cleaning frequency and dental visiting with serum high-sensitivity CRP
Figure 4 and 5 showed the weighted linear regression analyses for the associations of interdental cleaning frequency and dental visiting with ln-transformed serum high-sensitivity CRP. The weighted multivariable linear analyses demonstrated that interdental cleaning frequency (days/week) was significantly and inversely associated with ln-transformed serum high-sensitivity CRP (regression estimates (β) and 95% CI: −0.024(−0.041, −0.008), P = 0.010, Fig. 4). However, the weighted multivariable linear analyses did not find a significant association between the time since the last dental visit and ln-transformed serum high-sensitivity CRP (Fig. 5).
Weighted multivariate linear regression analyses for the association between the interdental cleaning frequency and ln-transformed serum high-sensitivity CRP. Model 1: adjusting interdental cleaning frequency (number of days per week), age, sex and race. Model 2: adjusting interdental cleaning frequency (number of days per week), self-reported oral health status, history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Model 3: adjusting interdental cleaning frequency (number of days per week), the number of missing teeth, history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Data are shown as regression estimates (β) and 95% CI. Abbreviations: CI, Confidence interval; HDL, High-density lipoprotein cholesterol; FPG, Fasting Plasma Glucose; CRP, C-reactive protein.
Weighted multivariate linear regression analyses for the association between time since the last dental visit and ln-transformed serum high-sensitivity CRP. Model 1: adjusting time since the last dental visit, age, sex and race. Model 2: adjusting time since the last dental visit, self-reported oral health status, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Model 3: adjusting time since the last dental visit, the number of missing teeth, interdental cleaning frequency (number of days per week), history of scaling and root planing treatment, age, sex, race, family income, education levels, marital status, smoking status, drinking status, dietary quality, overweight status, decreased HDL status, elevated FPG status, and high blood pressure status. Data are shown as regression estimates (β) and 95% CI. Abbreviations: CI, Confidence interval; HDL, High-density lipoprotein cholesterol; FPG, Fasting Plasma Glucose; CRP, C-reactive protein.
Discussion
In this study, very good or excellent self-rated oral health and increased interdental cleaning frequency were inversely associated with serum CRP but the number of missing teeth was positively associated with serum CRP, while time interval since the last dental visit was not significantly associated with serum CRP in US population. The hypotheses were partly accepted.
Serum CRP has been reported to be an important marker for future cardiovascular diseases22. In the present study, we only used the simple and straightforward indicators to investigate the association of oral health with systemic health. Missing teeth counting and oral health questionnaire could be simply performed without specific dental instruments by any medical professionals, so they could be conveniently used in the daily practice and population based investigations. These indicators combined objective examination of missing tooth and subjective assessment of self-rated oral health status. Meanwhile, the indicators considered daily oral hygiene and professional dental visit assessments. The strict sampling strategy and the large sample size further ensured the validity of the results. This study also took the extensive cardiovascular disease risk factors into consideration and thus supported that very good or excellent self-reported oral health, more frequent interdental cleaning and fewer teeth missing were associated with lower serum CRP.
In the current study, very good or excellent self-reported oral health status was found to be related with lower serum CRP when compared with poor or fair self-reported oral health status. Meanwhile, the number of missing teeth was demonstrated to be positively related with serum CRP levels. Endodontic lesions and periodontal disease are the most common causes of tooth loss1,2,3,4. The oral microbiome could spread to blood and distant organs due to the damage of epithelial barrier5,6,7,8, which may bring about systemic inflammation changes. Recently, Yu et al. reported that self–rated oral health status had the similar predicting effects for systemic comorbidities with periodontal disease42. They further found that fair or poor self-reported oral health was related with increased cardiovascular diseases42. These results were consistent with our current and previous findings62, as we found that population with fair or poor self-reported oral health status had a significantly higher level of serum CRP and a significantly higher prevalence of circadian syndrome62 when compared with population with very good or excellent self-reported oral health status. Self-reported oral health could reflect the periodontal status62 and was associated with systemic inflammation levels, so self-rated oral health status is a simple and convenient indicator for the evaluation of the link between oral and systemic health. In addition, we found a positive association between the number of missing natural teeth and serum CRP. Interestingly, the meta-analysis showed that a 2-tooth increase in missing teeth was associated with a 3% increment of coronary heart disease and stroke risk29. These results supported that the number of missing teeth is a straightforward and reliable indicator of dental status and could be used in the investigation about oral and systemic health. In contrast, Hiratsuka et al. demonstrated that it was nutritional status other than systemic inflammation that linked tooth missing with mortality in the population with 1–9 teeth36. The study is a small sample investigation. The extraction of most pathological teeth may result in inflammation resolution for the 1–9 teeth population. So large sample investigations may be needed to further validate the mediating role of systemic inflammation in tooth loss and mortality.
The present study also found that interdental cleaning frequency was inversely correlated with serum CRP. Recent investigations demonstrated that dental flossing was a possible protective factor for oral and systemic health42,48,49,58. Kim et al. reported that not using dental flossing was associated with obesity, hypertension and metabolic syndrome in the South Korean population48. Yu et al. further found the negative association of flossing with mortality42. Luo et al. also reported that inter-dental cleaning was related with lower serum CRP41. Despite the positive effects of interdental cleaning on oral and systemic health, the prevalence of performing daily interdental cleaning is only 1/3 in the US and South Korea53,54,55. In the present study, NHANES did not distinguish the detailed inter-dental cleaning methods such as flossing and interdental brushing. Previous meta-analyses suggested that the use of dental floss, wood sticks, and oral irrigators, in addition to tooth brushing, had an unclear or small effect on decreasing the prevalence of gingivitis but the use of interdental brushes in combination with tooth brushing had a moderate effect on the prevention of plaque and gingivitis63. However, the association of different interdental brushing methods with CRP was still unknown. In summary, we recommended that inter-dental cleaning should be propagated to more population as flossing may be beneficial both for oral and systemic health.
This study did not find a significant association of time interval since the last dental visit with serum CRP. Recent studies suggested that successful endodontic and periodontal therapies could decrease systemic CRP23,24,25,26,27. Some investigations further found that infrequent dental visit was related with increased mortality42,50,51. But in this cross-sectional study, we did not observe significant differences in serum CRP for the different time intervals between dental visits. However, when interpreting the null association observed in the present study, the"time since last dental visit"variable may not accurately reflect the frequency or quality of professional dental care, as it does not distinguish between preventive check-ups and therapeutic procedures. Because NHANES did not include detailed dental treatment history, whether dental treatment or regular dental examination were beneficial for systemic health needs further prospective investigations.
Limitations of the present study should be noted. Because the nature of self-reported oral health status and behaviour, reliance on self-reported oral health may introduce recall and reporting biases. As NHANES did not evaluate the exact reason for tooth extraction, we could not differentiate the specific effects of apical periodontitis and periodontal disease on serum CRP. In addition, this was a cross-sectional study, which could not permit causal inferences. The dose-dependent effects of inter-dental cleaning on systemic health still need to be validated by long-term prospective studies. Even though the study excluded the biased populations and comprehensive covariates were adjusted, there were some unknown factors. The study also did not collect the use of other oral health care products. The present study aggregated different oral hygiene behaviors, such as flossing, interdental brushing, and use of oral irrigators because NHANES did not collect the detailed inter-dental cleaning methods63. The association of different interdental brushing methods with CRP needs further investigations. Some unmeasured factors such as anti-inflammatory medication use, chronic inflammatory conditions, or psychosocial stress may still influence serum CRP levels. Because NHANES dataset is only representative of the US population, the findings may not translate to populations with different healthcare access or oral hygiene practices.
Conclusions
In summary, poor or fair self-reported oral health, tooth loss and infrequent interdental cleaning were associated with higher levels of serum CRP in the US population. Maintaining optimal oral health, preserving more natural teeth and daily interdental cleaning may be associated with lower systemic inflammation.
Data availability
The original data are available on the website of NHANES (https://www.cdc.gov/nchs/nhanes/index.htm). The processed data in this study are available from the corresponding author on reasonable request.
Abbreviations
- NHANES:
-
National health and nutrition examination survey
- CRP:
-
C-reactive protein
References
Petersen, P. E. & Ogawa, H. The global burden of periodontal disease: towards integration with chronic disease prevention and control. Periodontol 2000 60, 15–39. https://doi.org/10.1111/j.1600-0757.2011.00425.x (2012).
Sun, R. et al. Global and regional trends in prevalence of untreated caries in permanent teeth: Age-period-cohort analysis from 1990 to 2019 and projections until 2049. J. Dent. 147, 105122. https://doi.org/10.1016/j.jdent.2024.105122 (2024).
Aida, J. et al. Reasons for permanent tooth extractions in Japan. J. Epidemiol 16, 214–219. https://doi.org/10.2188/jea.16.214 (2006).
Chestnutt, I. G., Binnie, V. I. & Taylor, M. M. Reasons for tooth extraction in Scotland. J. Dent. 28, 295–297. https://doi.org/10.1016/s0300-5712(99)00069-x (2000).
Zhang, J., Huang, X., Lu, B., Zhang, C. & Cai, Z. Can apical periodontitis affect serum levels of CRP, IL-2, and IL-6 as well as induce pathological changes in remote organs?. Clin. Oral Investig. 20, 1617–1624. https://doi.org/10.1007/s00784-015-1646-6 (2016).
Dominy, S. S. et al. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5, eaau3333. https://doi.org/10.1126/sciadv.aau3333 (2019).
Furusho, H. et al. Dental infection of Porphyromonas gingivalis exacerbates high fat diet-induced steatohepatitis in mice. J. Gastroenterol 48, 1259–1270. https://doi.org/10.1007/s00535-012-0738-1 (2013).
Brun, A. et al. Oral microbiota and atherothrombotic carotid plaque vulnerability in periodontitis patients. A cross-sectional study. J. Periodontal Res. 56, 339–350. https://doi.org/10.1111/jre.12826 (2021).
Berlin-Broner, Y., Febbraio, M. & Levin, L. Association between apical periodontitis and cardiovascular diseases: a systematic review of the literature. Int. Endod J. 50, 847–859. https://doi.org/10.1111/iej.12710 (2017).
Liljestrand, J. M. et al. Association of Endodontic Lesions with Coronary Artery Disease. J. Dent Res. 95, 1358–1365. https://doi.org/10.1177/0022034516660509 (2016).
Garrido, M. et al. Elevated Systemic Inflammatory Burden and Cardiovascular Risk in Young Adults with Endodontic Apical Lesions. J. Endod 45, 111–115. https://doi.org/10.1016/j.joen.2018.11.014 (2019).
Gomes, M. S. et al. Can apical periodontitis modify systemic levels of inflammatory markers? A systematic review and meta-analysis. J. Endod 39, 1205–1217. https://doi.org/10.1016/j.joen.2013.06.014 (2013).
Sun, J. et al. Association between C-Reactive protein and periodontitis in an obese population from the NHANES 2009–2010. BMC Oral Health 23, 512. https://doi.org/10.1186/s12903-023-03189-3 (2023).
Meisel, P. et al. Competing interplay between systemic and periodontal inflammation: obesity overrides the impact of oral periphery. Clin. Oral Investig. 25, 2045–2053. https://doi.org/10.1007/s00784-020-03514-y (2021).
Ioannidou, E., Swede, H. & Dongari-Bagtzoglou, A. Periodontitis predicts elevated C-reactive protein levels in chronic kidney disease. J. Dent. Res. 90, 1411–1415. https://doi.org/10.1177/0022034511423394 (2011).
Lee, J. H. & Mun, S. J. Relationship between C-reactive protein level and periodontitis and systemic diseases. J. Periodontol 95, 494–501. https://doi.org/10.1002/JPER.23-0008 (2024).
Slade, G. D. et al. Relationship between periodontal disease and C-reactive protein among adults in the Atherosclerosis Risk in Communities study. Arch. Intern. Med. 163, 1172–1179. https://doi.org/10.1001/archinte.163.10.1172 (2003).
Bretz, W. A. et al. Systemic inflammatory markers, periodontal diseases, and periodontal infections in an elderly population. J. Am. Geriatr. Soc. 53, 1532–1537. https://doi.org/10.1111/j.1532-5415.2005.53468.x (2005).
Paraskevas, S., Huizinga, J. D. & Loos, B. G. A systematic review and meta-analyses on C-reactive protein in relation to periodontitis. J. Clin. Periodontol 35, 277–290. https://doi.org/10.1111/j.1600-051X.2007.01173.x (2008).
Garrido, M. et al. C-reactive protein expression is up-regulated in apical lesions of endodontic origin in association with interleukin-6. J. Endod 41, 464–469. https://doi.org/10.1016/j.joen.2014.12.021 (2015).
Zhang, Y., Leveille, S. G. & Edward, J. Wisdom teeth, periodontal disease, and C-reactive protein in US adults. Public Health 187, 97–102. https://doi.org/10.1016/j.puhe.2020.07.039 (2020).
Casagrande, S. S. & Lawrence, J. M. Cardiovascular disease risk factors and their associations with inflammation among US adolescents: NHANES, 2015 to March 2020. BMJ Open Diabetes Res. Care 12, e004148. https://doi.org/10.1136/bmjdrc-2024-004148 (2024).
Alim, B. A., Canturk, E. & Koksal, C. The effect of treated apical periodontitis before heart valve surgery on C-reactive protein levels. Oral Dis. 27, 632–638. https://doi.org/10.1111/odi.13554 (2021).
Garrido, M. et al. Reduced C-reactive protein levels after root canal treatment in clinically healthy young apical periodontitis individuals at cardiovascular risk. A prospective study. Int. Endod J. 57, 406–415. https://doi.org/10.1111/iej.14029 (2024).
Al-Abdulla, N. et al. Successful endodontic treatment reduces serum levels of cardiovascular disease risk biomarkers-high-sensitivity C-reactive protein, asymmetric dimethylarginine, and matrix metalloprotease-2. Int. Endod J. 56, 1499–1516. https://doi.org/10.1111/iej.13979 (2023).
Bakhsh, A., Moyes, D., Proctor, G., Mannocci, F. & Niazi, S. A. The impact of apical periodontitis, non-surgical root canal retreatment and periapical surgery on serum inflammatory biomarkers. Int. Endod J. 55, 923–937. https://doi.org/10.1111/iej.13786 (2022).
Caribe, P. M. V. et al. Influence of the treatment of periodontal disease in serum concentration of sirtuin 1 and mannose-binding lectin. J. Periodontol 91, 900–905. https://doi.org/10.1002/JPER.19-0236 (2020).
Haworth, S. et al. Tooth loss is a complex measure of oral disease: Determinants and methodological considerations. Community Dent. Oral Epidemiol 46, 555–562. https://doi.org/10.1111/cdoe.12391 (2018).
Cheng, F. et al. Tooth loss and risk of cardiovascular disease and stroke: A dose-response meta analysis of prospective cohort studies. PLoS ONE 13, e0194563. https://doi.org/10.1371/journal.pone.0194563 (2018).
Abnet, C. C. et al. Tooth loss is associated with increased risk of total death and death from upper gastrointestinal cancer, heart disease, and stroke in a Chinese population-based cohort. Int. J. Epidemiol 34, 467–474. https://doi.org/10.1093/ije/dyh375 (2005).
Vedin, O. et al. Tooth loss is independently associated with poor outcomes in stable coronary heart disease. Eur. J. Prev. Cardiol. 23, 839–846. https://doi.org/10.1177/2047487315621978 (2016).
Lee, H. J., Choi, E. K., Park, J. B., Han, K. D. & Oh, S. Tooth Loss Predicts Myocardial Infarction, Heart Failure, Stroke, and Death. J. Dent. Res. 98, 164–170. https://doi.org/10.1177/0022034518814829 (2019).
Kotronia, E. et al. Poor Oral Health and Inflammatory, Hemostatic, and Cardiac Biomarkers in Older Age: Results From Two Studies in the UK and USA. J. Gerontol A Biol. Sci. Med. Sci. 76, 346–351. https://doi.org/10.1093/gerona/glaa096 (2021).
Hung, H. C. et al. The association between tooth loss and coronary heart disease in men and women. J. Public Health Dent. 64, 209–215. https://doi.org/10.1111/j.1752-7325.2004.tb02755.x (2004).
Tu, Y. K. et al. Associations between tooth loss and mortality patterns in the Glasgow Alumni Cohort. Heart 93, 1098–1103. https://doi.org/10.1136/hrt.2006.097410 (2007).
Hiratsuka, T. et al. Contribution of systemic inflammation and nutritional status to the relationship between tooth loss and mortality in a community-dwelling older Japanese population: a mediation analysis of data from the Tsurugaya project. Clin. Oral Investig. 24, 2071–2077. https://doi.org/10.1007/s00784-019-03072-y (2020).
Meurman, J. H., Janket, S. J., Qvarnstrom, M. & Nuutinen, P. Dental infections and serum inflammatory markers in patients with and without severe heart disease. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 96, 695–700. https://doi.org/10.1016/j.tripleo.2003.08.017 (2003).
Liljestrand, J. M. et al. Missing Teeth Predict Incident Cardiovascular Events, Diabetes, and Death. J. Dent. Res. 94, 1055–1062. https://doi.org/10.1177/0022034515586352 (2015).
Schwahn, C. et al. Missing, unreplaced teeth and risk of all-cause and cardiovascular mortality. Int. J. Cardiol. 167, 1430–1437. https://doi.org/10.1016/j.ijcard.2012.04.061 (2013).
Holmlund, A., Holm, G. & Lind, L. Number of teeth as a predictor of cardiovascular mortality in a cohort of 7,674 subjects followed for 12 years. J. Periodontol. 81, 870–876. https://doi.org/10.1902/jop.2010.090680 (2010).
Luo, H. et al. Oral Health, Diabetes, and Inflammation: Effects of Oral Hygiene Behaviour. Int. Dent. J. 72, 484–490. https://doi.org/10.1016/j.identj.2021.10.001 (2022).
Yu, Y. H., Steffensen, B., Chasman, D. I. & Buring, J. E. Self-reported oral health is associated with systemic health outcomes and all-cause mortality. J. Am. Dent. Assoc. 155, 233–243. https://doi.org/10.1016/j.adaj.2023.11.006 (2024).
Marchesan, J. T. et al. Flossing Is Associated with Improved Oral Health in Older Adults. J. Dent. Res. 99, 1047–1053. https://doi.org/10.1177/0022034520916151 (2020).
Chaffee, B. W., Persai, D. & Vora, M. V. Interdental Cleaning and Oral Health Status in an Adult Cohort, 2015 to 2018. J. Dent. Res. 99, 1150–1156. https://doi.org/10.1177/0022034520926139 (2020).
Marchesan, J. T. et al. Interdental Cleaning Is Associated with Decreased Oral Disease Prevalence. J. Dent. Res. 97, 773–778. https://doi.org/10.1177/0022034518759915 (2018).
Cepeda, M. S., Weinstein, R., Blacketer, C. & Lynch, M. C. Association of flossing/inter-dental cleaning and periodontitis in adults. J. Clin. Periodontol 44, 866–871. https://doi.org/10.1111/jcpe.12765 (2017).
Axelsson, P., Nystrom, B. & Lindhe, J. The long-term effect of a plaque control program on tooth mortality, caries and periodontal disease in adults. Results after 30 years of maintenance. J. Clin. Periodontol. 31, 749–757. https://doi.org/10.1111/j.1600-051X.2004.00563.x (2004).
Kim, Y. H. et al. Oral health behaviors and metabolic syndrome: the 2008–2010 Korean National Health and Nutrition Examination Survey. Clin. Oral Investig. 18, 1517–1524. https://doi.org/10.1007/s00784-013-1112-2 (2014).
Moradpour, F. et al. Prevalence of metabolic syndrome and its association with oral health: First results from the Kurdish cohort study. Health Sci. Rep. 6, e1602. https://doi.org/10.1002/hsr2.1602 (2023).
Xu, K. et al. Association between dental visit behavior and mortality: a nationwide longitudinal cohort study from NHANES. Clin. Oral Investig. 28, 37. https://doi.org/10.1007/s00784-023-05471-8 (2023).
Park, S. Y. et al. Improved oral hygiene care attenuates the cardiovascular risk of oral health disease: a population-based study from Korea. Eur. Heart J. 40, 1138–1145. https://doi.org/10.1093/eurheartj/ehy836 (2019).
Han, S. J. The Use of Interdental Care Products in Korean Adults Aged 30 Years and Older and Factors Affecting Their Use: 4th to 7th Korean National Health and Nutrition Examination Survey. Int. J. Environ. Res. Public Health 19, 8639. https://doi.org/10.3390/ijerph19148639 (2022).
Fleming, E. B., Nguyen, D., Afful, J., Carroll, M. D. & Woods, P. D. Prevalence of daily flossing among adults by selected risk factors for periodontal disease-United States, 2011–2014. J. Periodontol 89, 933–939. https://doi.org/10.1002/JPER.17-0572 (2018).
Liang, L. & Aris, I. M. Minimal changes in daily flossing behavior among US adults from 2009 through 2020. J. Am. Dent. Assoc 155, 587-596.e2. https://doi.org/10.1016/j.adaj.2024.04.001 (2024).
Ronis, D. L., Lang, W. P., Farghaly, M. M. & Passow, E. Tooth brushing, flossing, and preventive dental visits by Detroit-area residents in relation to demographic and socioeconomic factors. J. Public Health Dent 53, 138–145. https://doi.org/10.1111/j.1752-7325.1993.tb02692.x (1993).
Inker, L. A. et al. New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. N. Engl. J. Med. 385, 1737–1749. https://doi.org/10.1056/NEJMoa2102953 (2021).
Nanri, H. et al. Association between dietary pattern and serum C-reactive protein in Japanese men and women. J. Epidemiol 21, 122–131. https://doi.org/10.2188/jea.je20100110 (2011).
Zhou, W. et al. Association of Interdental Cleaning Frequency With Tooth Loss and Self-Rated Oral Health: A Cross-Sectional Study. Int. Dent. J. 75, 100803. https://doi.org/10.1016/j.identj.2025.03.012 (2025).
Arabi, A. et al. Association between Serum Vitamin D Status and Circadian Syndrome: A Cross-Sectional Study. Nutrients 16, 2111. https://doi.org/10.3390/nu16132111 (2024).
Liu, C., Hua, L. & Xin, Z. Synergistic impact of 25-hydroxyvitamin D concentrations and physical activity on delaying aging. Redox Biol. 73, 103188. https://doi.org/10.1016/j.redox.2024.103188 (2024).
Stekhoven, D. J. & Buhlmann, P. MissForest–non-parametric missing value imputation for mixed-type data. Bioinformatics 28, 112–118. https://doi.org/10.1093/bioinformatics/btr597 (2012).
Li, Y. et al. Association of periodontitis, tooth loss, and self-rated oral health with circadian syndrome in US adults: a cross-sectional population study. BMC Oral Health 25, 713. https://doi.org/10.1186/s12903-025-06078-z (2025).
van der Weijden, F. & Slot, D. E. Oral hygiene in the prevention of periodontal diseases: the evidence. Periodontol 2000 55, 104–123. https://doi.org/10.1111/j.1600-0757.2009.00337.x (2011).
Acknowledgements
The authors thank all the staff, investigators and participants involved in the National Health and Nutrition Examination Survey for their contributions.
Funding
This work was supported by the National Natural Science Foundation of China (No. 8170041519), Fundamental Research Funds for the Central Universities of Central South University (No. 2021zzts1035), and Natural Science Foundation of Hunan Province (No. 2025JJ80535). The authors deny any conflicts of interest related to this study.
Author information
Authors and Affiliations
Contributions
Pengfei Liu: Investigation, Formal analysis and Validation; Yibo Li: Investigation, Formal analysis and Funding acquisition; Yuhao Liu: Investigation and Formal analysis; Tao Yin: Investigation, Formal analysis and Validation; Mi He: Formal analysis and Funding acquisition; Changyun Fang: Methodology; Shifang Peng: Conceptualization and Methodology; Yundong Liu: Conceptualization, Methodology, Investigation, Formal analysis, Funding acquisition, Writing- Original draft preparation, Writing- Reviewing and Editing, and Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The data and information used in this study are publicly available on the NHANES website (https://www.cdc.gov/nchs/nhanes/index.htm). All investigations and study procedures were approved by the NCHS Research Ethics Review Board. Written informed consent was obtained from all participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, P., Li, Y., Liu, Y. et al. Association of participant reported oral health, tooth loss, interdental cleaning and dental visits with serum CRP. Sci Rep 15, 27770 (2025). https://doi.org/10.1038/s41598-025-13163-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13163-4